How Can You Mend A Broken Heart 2017

How Can You Mend A Broken Heart? 2017 Update on Heart Failure Gwen Bartlett BS Pharm, Pharm D, BCPS, BCCCP Assistant Professor of Pharmacy Practice Husson University October 15, 2017

Disclosure I have no relevant financial or non-financial conflicts of interest to disclose. 2

Learning Objectives l l l Summarize the evidence, utility, and limitations of established biomarkers in prevention, diagnosis, and prognosis of heart failure Discuss the mechanism of action, supporting clinical evidence, and appropriate use of the newer chronic heart failure therapies. Design an appropriate patient-specific evidencebased pharmacotherapy plan for heart failure management.

Impact of Heart Failure l Prevalence increased 5. 7 million to 6. 5 million l l l 5 -year survival HF s/p AMI: l l 1/3 of the US adult population stage A HF Projected increased prevalence of 46% by 2030 2001 – 2010: 61% (up from 54%) Hospitalized HF events l l HFr. EF: 53% (highest proportion: black males) HFp. EF: 47% (highest proportion: white females) Benjamin et al. Heart Disease and Stroke Statistics – 2017 Update: A Report From the American Heart Association. Circulation 2017; 135: e 1 -459

Key Guideline Changes l l l Biomarkers Management of stage C HFr. EF Prevention strategies Updates on HFp. EF Managing c. Comorbidities (i. e. , sleep apnea, anemia, HTN)

Stages in the Development of HF and Recommended Therapy by Stage. Clyde W. Yancy et al. Circulation. 2013; 128: e 240 -e 327 Copyright © American Heart Association, Inc. All rights reserved.

BIOMARKERS
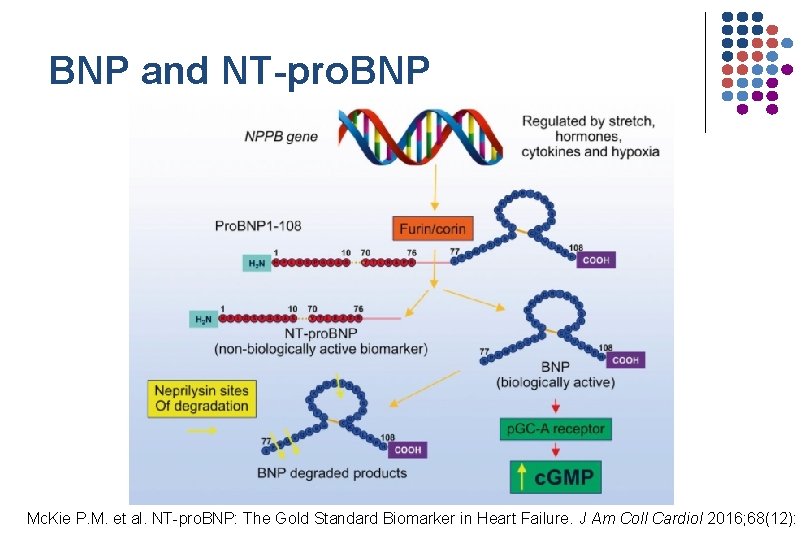
BNP and NT-pro. BNP Mc. Kie P. M. et al. NT-pro. BNP: The Gold Standard Biomarker in Heart Failure. J Am Coll Cardiol 2016; 68(12):

Level of Supporting Evidence for Use of Natriuretic Peptides in HF Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guidelines for the Management of Heart Failure. Circulation 2017; e 1 -75.
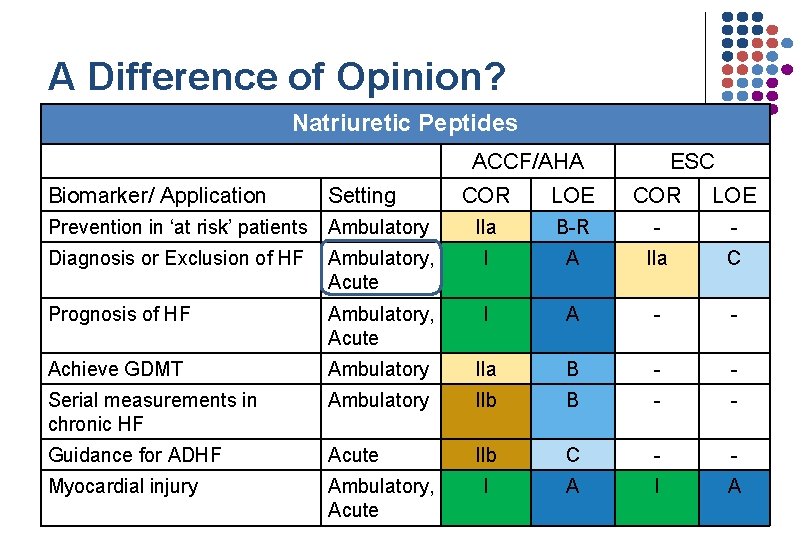
A Difference of Opinion? Natriuretic Peptides ACCF/AHA Biomarker/ Application Setting Prevention in ‘at risk’ patients ESC COR LOE Ambulatory IIa B-R - - Diagnosis or Exclusion of HF Ambulatory, Acute I A IIa C Prognosis of HF Ambulatory, Acute I A - - Achieve GDMT Ambulatory IIa B - - Serial measurements in chronic HF Ambulatory IIb B - - Guidance for ADHF Acute IIb C - - Myocardial injury Ambulatory, Acute I A
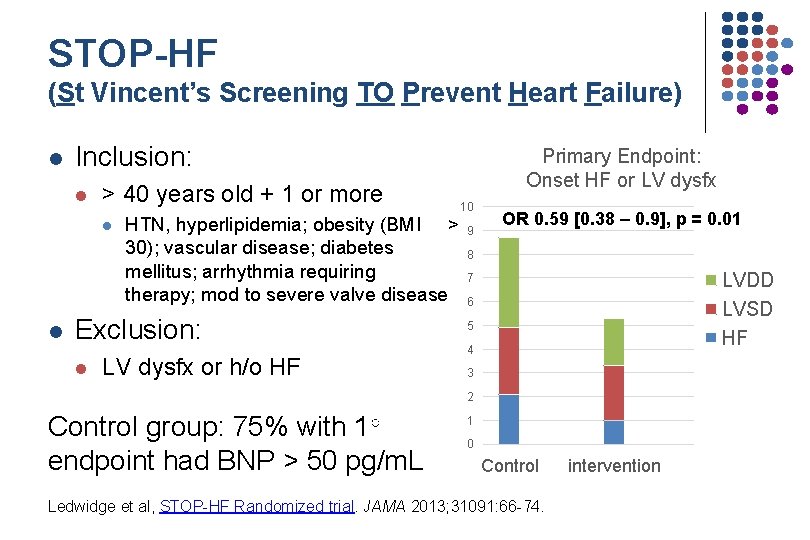
STOP-HF (St Vincent’s Screening TO Prevent Heart Failure) l Inclusion: l > 40 years old + 1 or more l l 10 HTN, hyperlipidemia; obesity (BMI > 9 30); vascular disease; diabetes 8 mellitus; arrhythmia requiring 7 therapy; mod to severe valve disease 6 Exclusion: l Primary Endpoint: Onset HF or LV dysfx or h/o HF OR 0. 59 [0. 38 – 0. 9], p = 0. 01 LVDD LVSD HF 5 4 3 2 Control group: 75% with 1○ endpoint had BNP > 50 pg/m. L 1 0 Control Ledwidge et al, STOP-HF Randomized trial. JAMA 2013; 31091: 66 -74. intervention

STOP-HF Secondary Outcomes (St Vincent’s Screening TO Prevent Heart Failure) All participants Participants with BNP ≥ 50 pg/m. L MACE = Stroke/TIA; PE/DVT; AMI; HF; or Arrhythmia Ledwidge et al, STOP-HF Randomized trial. JAMA 2013; 31091: 66 -74.

PONTIAC (NT-pro. BNP selected Preventi. On of cardiac eve. Nts in a popula. Tion of d. Iabetic patients without A history of Cardiac disease) l 300 patients: Inclusion: l l Type 2 DM for ≥ 6 months + > 18 years old + NT-pro. BNP > 125 pg/m. L Exclusion l h/o cardiac disease l l Primary Endpoint: Hospitalization or CV death ECG with a fib, ST-T-wave abnormalities or BBB, low LVEF, wall motion abnormalties, valve dysfx NT-pro. BNP identified high-risk and triggered up-titration of RAAS inhibitor + β-blocker Huelsmann M et al. PONTIAC Trial. J Am Coll Cardiol 2013; 62: 1365 -72.

Thresholds for BNP Are DIFFERENT! Ambulatory/Outpatient Acute/Inpatient BNP NT-pro. BNP 50 pg/m. L 100 pg/m. L 125 pg/m. L 300 pg/m. L Schreiber D et al. Natriuretic Peptides in Congestive Heart Failure. Medscape. Updated 9/2016 Ponikowski et al. 2016 ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure. Eur Heart J 2016: 37: 2129 -2200.

Limitations of BNP and NT-pro. BNP l Diagnosis: l l l Prognosis: l l Limited in HFp. EF Influenced by LVEF, age, sex, renal function, Na+ levels, BMI, and other comorbidities BNP elevation associated with increased mortality and hospital readmissions Guiding therapy: l l Limited evidence supporting Recent GUIDE-IT trial terminated due to futility Rubattu et al. Resetting the Neurohormonal Balance in Heart Failure: the Relevance of the Natriuretic Peptide System to the Clinical Management of Patients with HF. Heart Fail Rev 2017; 22; 279 -88

UPDATE IN MANAGING STAGE C HFr. EF Is It a “PARADIGM SHIFT”? ?

Stages in the Development of HF and Recommended Therapy by Stage. Clyde W. Yancy et al. Circulation. 2013; 128: e 240 -e 327 Copyright © American Heart Association, Inc. All rights reserved.
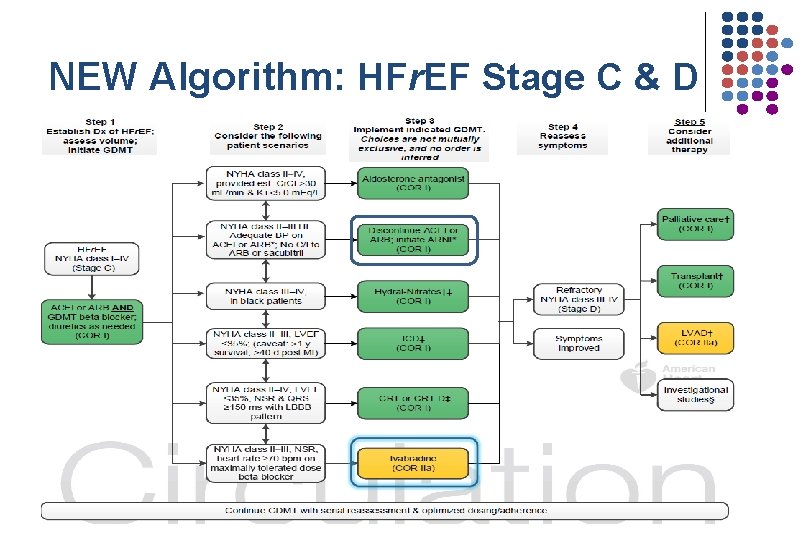
NEW Algorithm: HFr. EF Stage C & D

Reiterated from 2013: Stage C HFr. EF I A A IIa IIb III Recommendation The use of ACE inhibitors is beneficial for patients with prior or current symptoms of chronic HFr. EF to reduce morbidity and mortality The use of ARBs to reduce morbidity and mortality is recommended in patients with prior or current symptoms of chronic HFr. EF who are intolerant to ACE inhibitors because of cough or angioedema. Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation 2017; 136: e 137 -61.

ARNI (ANGIOTENSIN RECEPTOR BLOCKER / NEPRILYSIN INHIBITOR)
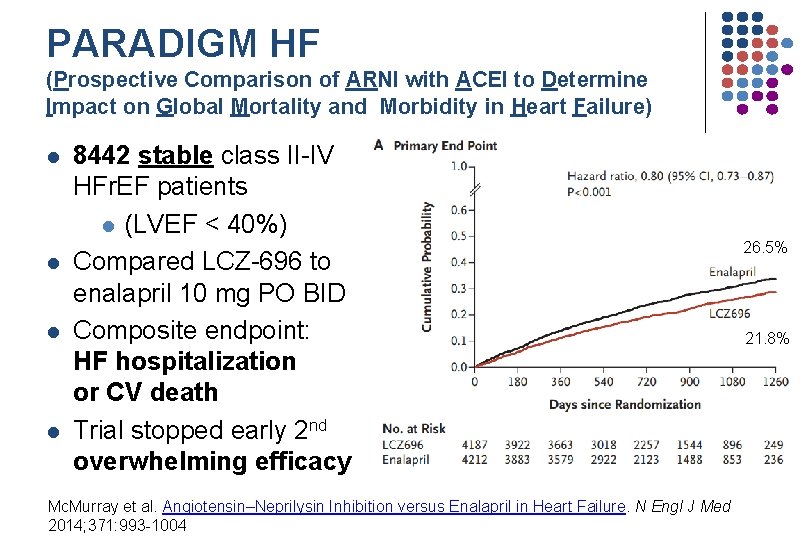
PARADIGM HF (Prospective Comparison of ARNI with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure) l l 8442 stable class II-IV HFr. EF patients l (LVEF < 40%) Compared LCZ-696 to enalapril 10 mg PO BID Composite endpoint: HF hospitalization or CV death Trial stopped early 2 nd overwhelming efficacy Mc. Murray et al. Angiotensin–Neprilysin Inhibition versus Enalapril in Heart Failure. N Engl J Med 2014; 371: 993 -1004 26. 5% 21. 8%

The BIG BANG!! Stage C HFr. EF I IIa IIb III Recommendation The clinical strategy of inhibition of the reninangiotensin system with A A B-R • ACE inhibitors OR • Angiotensin receptor blockers OR • Angiotensin receptor/neprilysin inhibitor in conjunction with evidence-based β-blockers and aldosterone antagonists in selected patients is recommended for patients with chronic HFr. EF to reduce morbidity and mortality. Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation 2017; 136: e 137 -61.
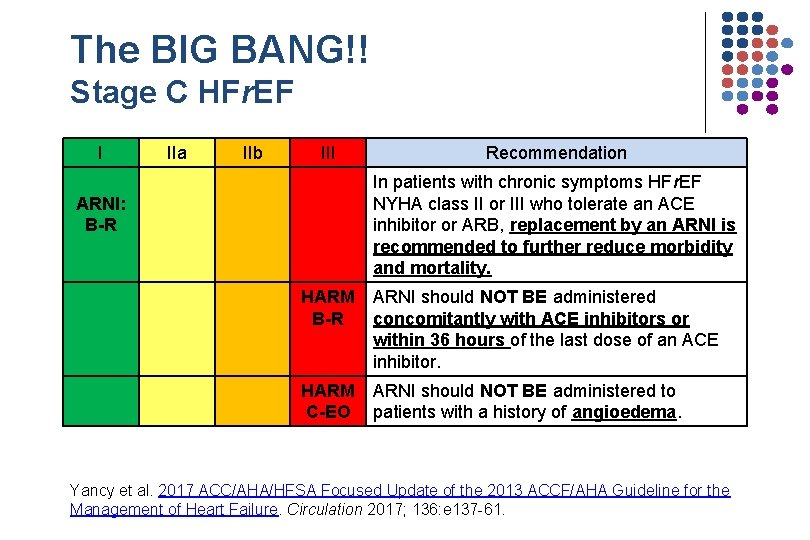
The BIG BANG!! Stage C HFr. EF I IIa IIb III Recommendation In patients with chronic symptoms HFr. EF NYHA class II or III who tolerate an ACE inhibitor or ARB, replacement by an ARNI is recommended to further reduce morbidity and mortality. ARNI: B-R HARM B-R ARNI should NOT BE administered concomitantly with ACE inhibitors or within 36 hours of the last dose of an ACE inhibitor. HARM C-EO ARNI should NOT BE administered to patients with a history of angioedema. Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation 2017; 136: e 137 -61.
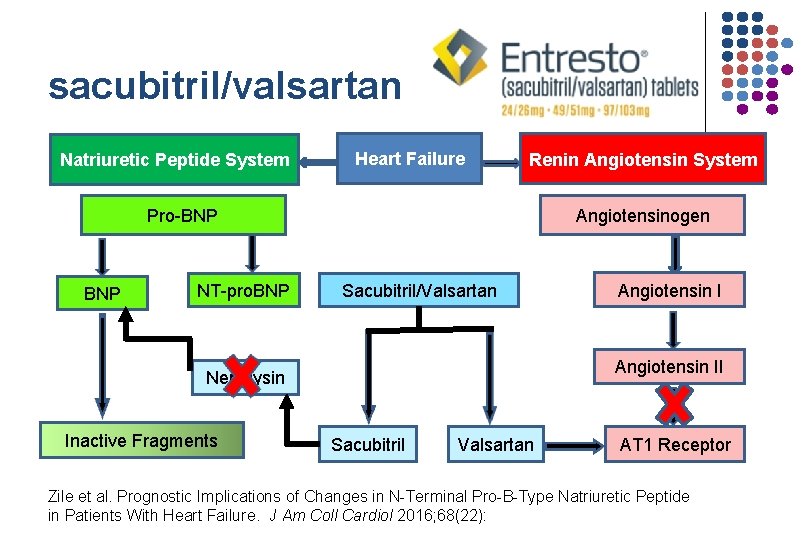
sacubitril/valsartan Natriuretic Peptide System Heart Failure Renin Angiotensin System Pro-BNP NT-pro. BNP Angiotensinogen Sacubitril/Valsartan Angiotensin II Neprilysin Inactive Fragments Angiotensin I Sacubitril Valsartan AT 1 Receptor Zile et al. Prognostic Implications of Changes in N-Terminal Pro-B-Type Natriuretic Peptide in Patients With Heart Failure. J Am Coll Cardiol 2016; 68(22):

sacubitril/valsartan Indication Fixed dose combination: sacubitril (neprilysin inhibitor) + valsartan (ARB) is indicated to reduce the risk of CV death and HF hospitalization in patients with HFr. EF Dosage Start with 49/51 mg two times daily. Double the dose after 2 – 4 weeks as tolerated to a maintenance dose of 97/103 mg two times daily. Renal/Hepatic Impairment For patients not currently taking an ACEI or ARB, or for those with severe renal impairment (i. e. , e. GFR < 30 m. L/min) or moderate hepatic impairment, begin with 24/26 mg two times daily Switching from an ACE inhibitor Stop ACE inhibitor fro 36 hours prior to starting treatment. Contraindications History of angioedema related to previous ACE inhibitor or ARB, concomitant use of ACE I, concomitant use of aliskiren in patient with diabetes. WARNING: hyperkalemia, pregnancy Adverse Effects Hypotension, hyperkalemia, cough, dizziness, renal failure, and angioedema (i. e. , 0. 5% sacubitril/valsartan versus 0. 2% enalapril) Entresto® package labeling: Last revised 8/2015
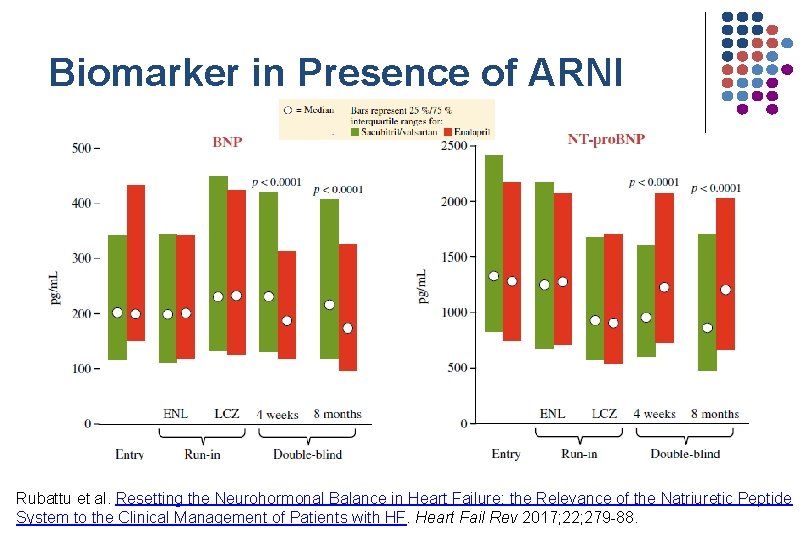
Biomarker in Presence of ARNI Rubattu et al. Resetting the Neurohormonal Balance in Heart Failure: the Relevance of the Natriuretic Peptide System to the Clinical Management of Patients with HF. Heart Fail Rev 2017; 22; 279 -88.

ARNI trials l PARAGON-HF l l l TRANSITION l l Evaluating sacubitril/valsartan in HFp. EF Anticipate publication in 2019 Evaluating in-hospital initiation when admission for ADHF PARADISE-MI l Evaluating effectiveness post MI to reduce HF events

IVABRADINE (HYPERPOLARIZATION-ACTIVATED CYCLIC NUCLEOTIDE GATED CHANNEL)
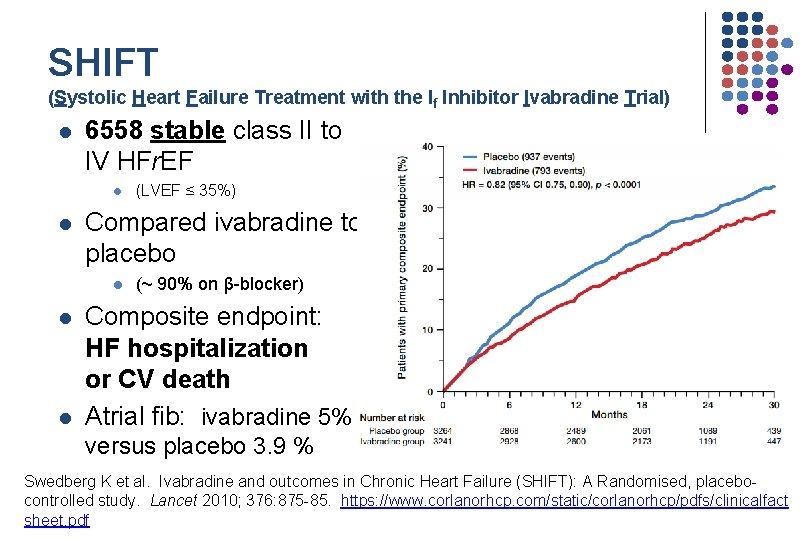
SHIFT (Systolic Heart Failure Treatment with the If Inhibitor Ivabradine Trial) l 6558 stable class II to IV HFr. EF l l Compared ivabradine to placebo l l l (LVEF ≤ 35%) (~ 90% on β-blocker) Composite endpoint: HF hospitalization or CV death Atrial fib: ivabradine 5% versus placebo 3. 9 % Swedberg K et al. Ivabradine and outcomes in Chronic Heart Failure (SHIFT): A Randomised, placebocontrolled study. Lancet 2010; 376: 875 -85. https: //www. corlanorhcp. com/static/corlanorhcp/pdfs/clinicalfact sheet. pdf

And the SHIFT: Role of Ivabradine Stage C HFr. EF I IIa B-R IIb III Recommendation Ivabradine can be beneficial to reduce HF hospitalization for patients with symptomatic (NYHA class II – III) stable chronic HFr. EF (LVEF ≤ 35%) who are receiving GDMT, including a beta-blocker at maximum tolerated dose, and who are in sinus rhythm with a heart rate of 70 bpm or greater at rest. GDMT: Guideline-direct management and therapy Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation 2017; 136: e 137 -61.
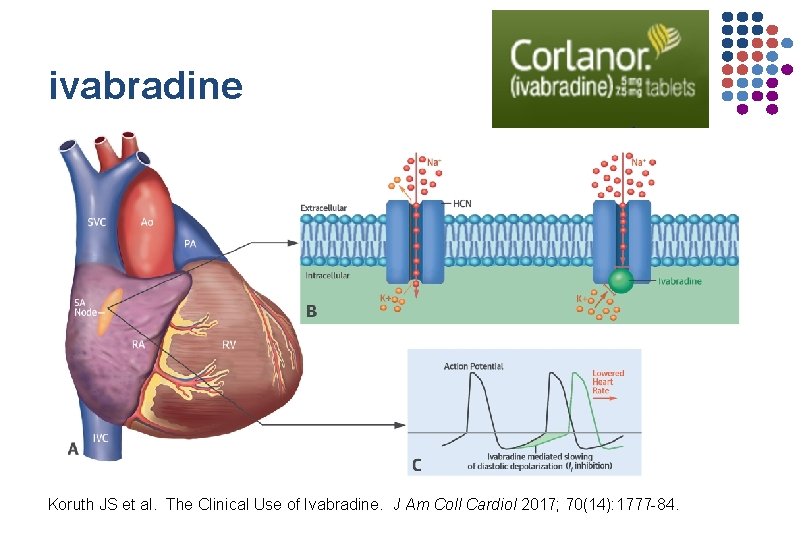
ivabradine Koruth JS et al. The Clinical Use of Ivabradine. J Am Coll Cardiol 2017; 70(14): 1777 -84.

ivabradine Indication Ivabradine is a HCN channel blocker indicated to reduce the risk of HF hospitalization in patients with stable, symptomatic chronic heart failure with LVEF ≤ 35%, who are in sinus rhythm with resting heart rate ≥ 70 bpm and either are on maximally tolerated doses of beta-blockers or have a contraindication to beta-blocker use. Dosage Start with 5 mg two times daily. After 2 weeks. Adjust dose based on heart rate. The maximum dose is 7. 5 mg two times daily. Conduction defects or risk of hemodynamic compromise with bradycardia: start at 2. 5 mg two time daily. HR > 60 bpm: ↑ by 2. 5 mg BID; HR 50 -60 bpm: continue; HR < 50 or signs and symptoms of bradycardia, ↓ by 2. 5 mg BID or if at 2. 5 mg BID, DC Renal/Hepatic CYP 3 A 4 substrate: avoid moderate and strong inhibitors or inducers. Contraindicated in severe hepatic impairment (Child-Pugh C). No data available for severe renal insufficiency (e. GFR < 15 m. L/min). Contraindications ADHF; BP < 90/50 mm Hg; SSS or 3 rd degree AV block without PPM; Resting HR < 60 bpm prior to initiation; Severe hepatic impairment; Pacemaker dependence. WARNING: pregnancy (Fetal toxicity) Adverse Effects Fetal toxicity; Atrial fibrillation; Bradycardia + conduction disturbances; Phosphenes (visual light disturbances) Corlanor® package labeling: Last revised 1/’ 2017
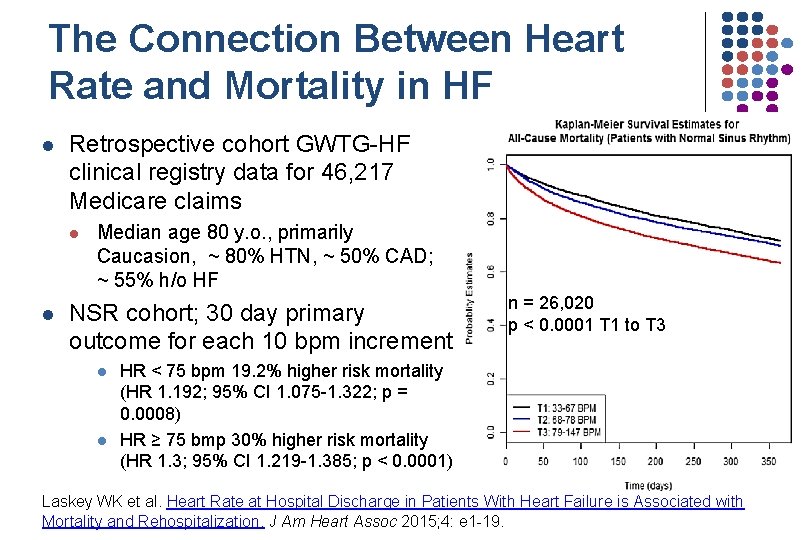
The Connection Between Heart Rate and Mortality in HF l Retrospective cohort GWTG-HF clinical registry data for 46, 217 Medicare claims l l Median age 80 y. o. , primarily Caucasion, ~ 80% HTN, ~ 50% CAD; ~ 55% h/o HF NSR cohort; 30 day primary outcome for each 10 bpm increment l l n = 26, 020 p < 0. 0001 T 1 to T 3 HR < 75 bpm 19. 2% higher risk mortality (HR 1. 192; 95% CI 1. 075 -1. 322; p = 0. 0008) HR ≥ 75 bmp 30% higher risk mortality (HR 1. 3; 95% CI 1. 219 -1. 385; p < 0. 0001) Laskey WK et al. Heart Rate at Hospital Discharge in Patients With Heart Failure is Associated with Mortality and Rehospitalization. J Am Heart Assoc 2015; 4: e 1 -19.
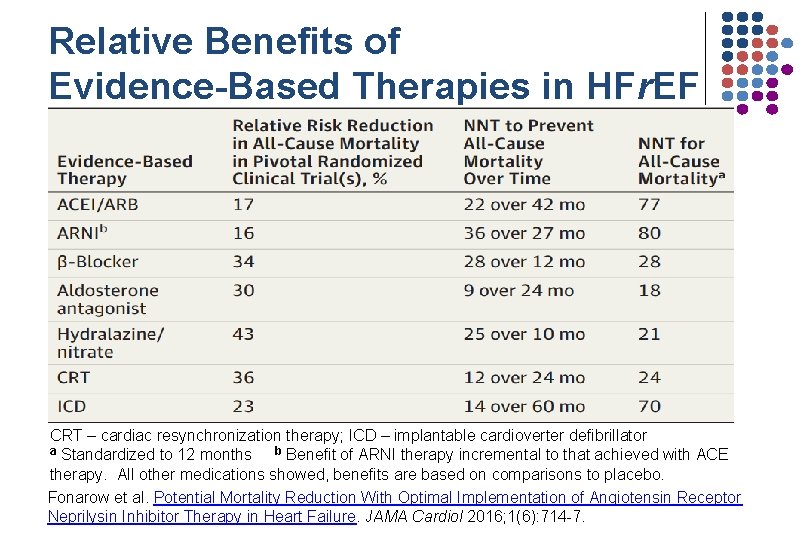
Relative Benefits of Evidence-Based Therapies in HFr. EF CRT – cardiac resynchronization therapy; ICD – implantable cardioverter defibrillator a Standardized to 12 months b Benefit of ARNI therapy incremental to that achieved with ACE therapy. All other medications showed, benefits are based on comparisons to placebo. Fonarow et al. Potential Mortality Reduction With Optimal Implementation of Angiotensin Receptor Neprilysin Inhibitor Therapy in Heart Failure. JAMA Cardiol 2016; 1(6): 714 -7.

Patient Management Tools: 2017 HF GWTG l ACHIEVEMENT MEASURE l l QUALITY MEASURE l l ACE/ARB or ARNI at discharge (Updated) ARNI at discharge (New) Reporting Measures l Ivabradine at discharge (New)

PREVENTION

Stages in the Development of HF and Recommended Therapy by Stage. Clyde W. Yancy et al. Circulation. 2013; 128: e 240 -e 327 Copyright © American Heart Association, Inc. All rights reserved.
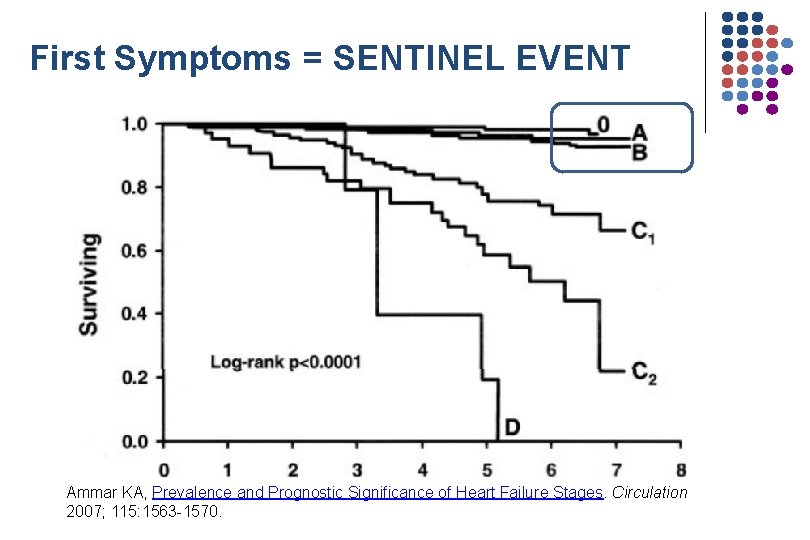
First Symptoms = SENTINEL EVENT Ammar KA, Prevalence and Prognostic Significance of Heart Failure Stages. Circulation 2007; 115: 1563 -1570.
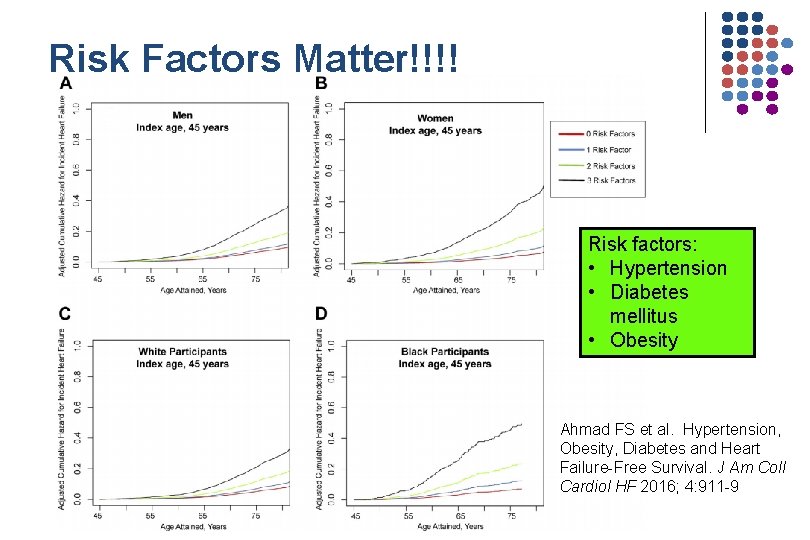
Risk Factors Matter!!!! Risk factors: • Hypertension • Diabetes mellitus • Obesity Ahmad FS et al. Hypertension, Obesity, Diabetes and Heart Failure-Free Survival. J Am Coll Cardiol HF 2016; 4: 911 -9

Prevention of Incident HF: Control BP I B-R IIa IIb III Recommendation In patients at increased risk, stage A HF, the optimal blood pressure in those with hypertension should be less than 130/80 mm Hg * Will be consistent with new ACC/AHA Hypertension panel guidelines upcoming in 2017: SPRINT trial Class IA recommendation for Stage C HFr. EF and HFp. EF as well!! Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation 2017; 136: e 137 -61.

New Focus is PREVENTION! l Stage A (i. e. , asymptomatic without LV dysfx) l l l Hypertension Diabetes mellitus Coronary artery disease Obesity HF can be prevented and progression to HF interrupted BNP or NT-pro. BNP screening to refine risk prediction Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guidelines for the Management of Heart Failure. Circulation 2017; e 1 -75.

UPDATE IN MANAGING HFp. EF

Prevention of Incident HF: Control BP I IIa IIb B-R III Recommendation In appropriately selected patients with HFp. EF (with EF ≥ 435%, elevated BNP levels or HF admission within 1 year, e. GFR > 30 m. L/min, creatinine < 2. 5 mg/d. L, potassium < 5. 0 m. Eq/L), aldosterone receptor antagonists might be considered to decrease hospitalization. Yancy et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure. Circulation 2017; 136: e 137 -61.
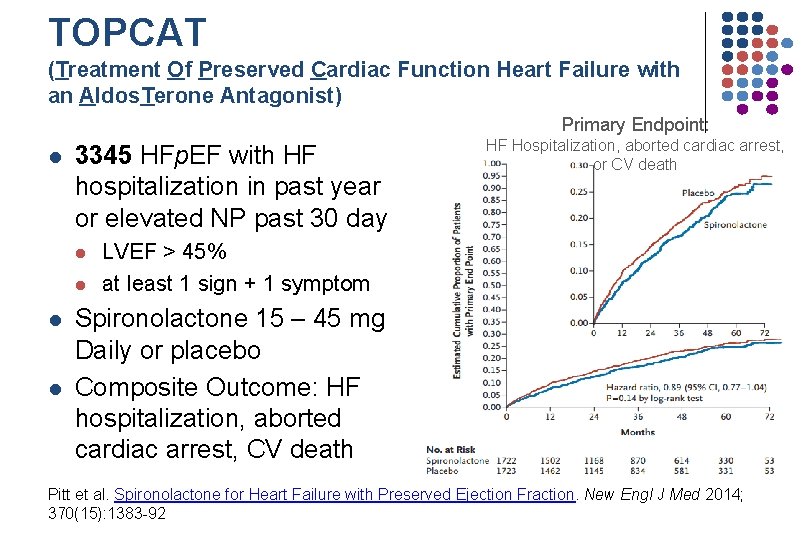
TOPCAT (Treatment Of Preserved Cardiac Function Heart Failure with an Aldos. Terone Antagonist) Primary Endpoint: l 3345 HFp. EF with HF hospitalization in past year or elevated NP past 30 day l l HF Hospitalization, aborted cardiac arrest, or CV death LVEF > 45% at least 1 sign + 1 symptom Spironolactone 15 – 45 mg Daily or placebo Composite Outcome: HF hospitalization, aborted cardiac arrest, CV death Pitt et al. Spironolactone for Heart Failure with Preserved Ejection Fraction. New Engl J Med 2014; 370(15): 1383 -92

TOPCAT Pfeffer et al. Regional Variation in Patients and Outcome in the TOPCAT trial. Circulation 2015; 131: 34 -42. de Denus S et al. Spironolactone Metabolites in TOPCAT – New Insights into Regional Variation. New Engl J Med 2017. ;

HFp. EF l No specific therapies available to reduce morbidity and mortality in HFp. EF l Aggressively manage cardiac comorbidities which exacerbate diastolic dysfx: HTN, CAD, and A fib l Treat non-cardiac comorbidities: obesity, CKD, sleep apnea, iron deficiency anemia, and encourage lifestyle modifications

IRON DEFICIENCY

IRONOUT-HF (IRON Repletion Effects on Oxygen Up. Take in Heart Failure Trial) l l 225 stable patients HFr. EF NYHA II – IV symptoms Iron polysaccharide 150 mg PO BID x 16 wks Primary endpoint: Change in peak oxygen uptake (VO 2) Outcome: High-dose ORAL iron did not improve exercise capacity: assessed by VO 2, 6 -minute walk distance, and QOL assessed by KCCQ score Lewis et al. Effect of Oral Iron Repletion on Exercise Capacity in Patients With Heart Failure With Reduced Ejection Fraction and Iron Deficiency. JAMA 2017; 317(19): 1958. 66

https: //www. heart. org/HEARTORG/Conditions/Heart. Failure/Heart-Failure-Guidelines-Toolkit_ UCM_491412_Sub. Home. Page. jsp

Conclusions l l l Data suggests biomarkers may be valuable in identify high-risk patients in order to PREVENT development of heart failure ARNI therapy is now considered a 1 st line therapy in selected patients and is a 2017 GWTG-HF Achievement measure Hypertension management remains the most important modifiable risk factor to PREVENT development of HF


Self-Assessment Question #1 J. J. , a 78 y. o. male, is seen in HF clinic for his monthly follow up appt for clinical reassessment and potential medication titration. J. J. has Stage C heart failure (LVEF 35%) with NYHA III symptoms. Due to ongoing symptoms, two weeks prior the decision was made to discontinue lisinopril and convert to an ARNI. Allergies: seasonal rhinitis Current HF meds include: l l l metoprolol succinate 100 mg orally Daily sacubitril/valsartan 49/51 mg orally BID furosemide 40 mg orally BID

Self-Assessment Question #1 Of the following biomarkers, which has the strongest supporting evidence and would be least impacted by the initiation of an ARNI? A. B. C. Pre-pro. BNP ANP NT-pro. BNP CNP D. Rationale: Answer C is correct. Both BNP and NT-pro. BNP are considered the gold standard biomarkers of HF. Pre-pro. BNP is an intracellular precursor peptide. As such, it would not likely be measurable in the blood (Answer A is incorrect). Both active natriuretic peptides, ANP and CNP are measurable in the plasma. Currently, however, there is limited data describing the utility of these biomarkers in this setting (Answer B and D are incorrect). Based on available data, the interpretation of NT-pro. BNP appears to be unaffected by neprilysin inhibition (Answer C is correct). Because BNP is metabolized by neprilysin, sacubitril inhibition of this enzyme results in a modest increase in serum BNP (i. e. , 200 pg/m. L), but less than that of acute cardiac decompensation. If only a BNP assay is available for disease monitoring in a given clinical setting, it can still be useful for disease monitoring. A new steady state BNP level should be achieved approximately 1 month after sacubitril initiation. This BNP serum level can be used as the new “higher” baseline level.

Self-Assessment Question #2 The following 4 patients with HF and comorbidities including type 2 DM and ASCVD have been stable for the past 6 months on ACEI and beta blocker therapy. For which patient does current evidence strongly support consideration of initiation of an ARNI (angiotensin receptor blocker/neprilysin inhibitor)? A. B. C. D. Stable Stage A HFr. EF with NYHA functional class I symptoms Stable Stage C HFr. EF with NYHA functional class II symptoms Stable HFp. EF with NYHA functional class III symptoms Hospitalized HFp. EF with NYHA functional class IV symptoms Rationale: Answer B is correct. The evidence for the recommendation to consider an ARNI is a result of the findings from the PARADIGM-HF trial which included OUTpatients with LVEF < 35% and NYHA functional classes II – IV; in fact, ~ 70% of patients enrolled were classified as NYHA functional class II (Answer B is correct). Although the PARAGON-HF trial is evaluating patients with HFp. EF, at this point there is insufficient data to support a recommendation in this population (Answers C and D are incorrect).

Self-Assessment Question #3 A. M. , a 69 y. o. female 5’ 6” 172 lbs (78 kg), is seen in HF clinic for follow up. A. M. continues to suffer symptoms of HF despite appropriate medical therapy. A. M. ’s current HF meds include: l l metoprolol succinate 200 mg orally Daily valsartan 40 mg orally BID furosemide 40 mg orally BID spironolactone 12. 5 mg orally Daily Allergies: lisinopril (angioedema) Chem panel: Na + 134 | K + 4. 8 | Cl - 102 | CO 2 - 26 | BUN 24 | SCr 1. 6 |Glucose 126

Self-Assessment Question #3 The nurse practitioner stops you in the hall and asks about conversion to ARNI. Which of the following is the most appropriate response? A. B. C. D. Based on the serum K + level, it would be appropriate to consider converting Based on the serum creatinine, it would be inappropriate to consider converting Based on the allergy history, it would be inappropriate to consider converting Hold the valsartan for 36 hours; then proceed with converting to the ARNI. Rationale: Answer C is correct. Although ANRI therapy can be considered in patients with renal impairment, hyperkalemia, and cough associated with ACEI therapy, it is contraindicated to utilize in patients with ACEI-associated or ARB-associated angioedema (Answer D is incorrect). Recognizing that ARBs do not inhibit kininase thus resulting in lower incidence of cough and angioedema, the new 2017 guidelines do provide a level I class of recommendation to use ARBs in the setting of ACEI-associated angioedema with the precaution that there is some potential for crossreactivity. Although AM does have renal impairment (est. Cl. Cr ~ 30 m. L/min) there are recommendations supporting initiating sacubitril/valsartan initiation, but at a lower dose of 24/26 mg (Answer B is incorrect)

Self-Assessment Question #3 Rationale: Answer C is correct. Although ANRI therapy can be considered in patients with renal impairment, hyperkalemia, and cough associated with ACEI therapy, it is contraindicated to utilize in patients with ACEIassociated or ARB-associated angioedema (Answer C is correct). Recognizing that ARBs do not inhibit kininase thus resulting in lower incidence of cough and angioedema, the new 2017 guidelines do provide a level I class of recommendation to use ARBs in the setting of ACEI-associated angioedema with the precaution that there is some potential for cross-reactivity. Although AM does have renal impairment (est. Cl. Cr ~ 30 m. L/min) there are recommendations supporting initiating sacubitril/valsartan initiation, but at a lower dose of 24/26 mg (Answer B is incorrect). Certainly drugs which impact the RAAS system all have the potential to augment serum K+. However, there is some suggestion that the incidence of hyperkalemia may, in fact, be lower with an ARNI than ACEI. It does merit monitoring of serum K+ levels, but this would not be justification not to initiate the drug (Answer A is incorrect). The recommendation and package labeling require ACEI to be held for 36 hours prior to initiating an ARNI, but this doesn’t apply to the ARBs (Answer D is incorrect).

Review of Classifications of Heart Failure
- Slides: 58