How Atoms Combine 7 3 stable Atoms combine

How Atoms Combine (7. 3)

stable • Atoms combine to become more ____. • The most stable elements in the periodic table are the Noble gases __________ because they have the maximum electrons number of _______ in their outermost orbits. (they 8 valence electrons) have _____ • There are 3 ways in which an atom that does not have a full valence shell can fill it: gain 1. it can _____ electrons, Ionic bond lose 2. it can _____ electrons, covalent bond share 3. it can _____ electrons. bond All 3 methods result in the formation of a chemical ______. atoms A chemical bond is the attraction between 2 _____.

( IONIC COMPOUNDS & )

IONIC BONDING metal Ionic bonds are formed between _____ non-metal elements. Circle the elements and _______ compounds below that contain ionic bonds. KCl H 2 O O 2 Mg. O Na 2 S NH 3 Cl 2 Ionic bonds are formed by the attraction of 2 oppositely charged ions (an ion is a _____ charged ______ particle).
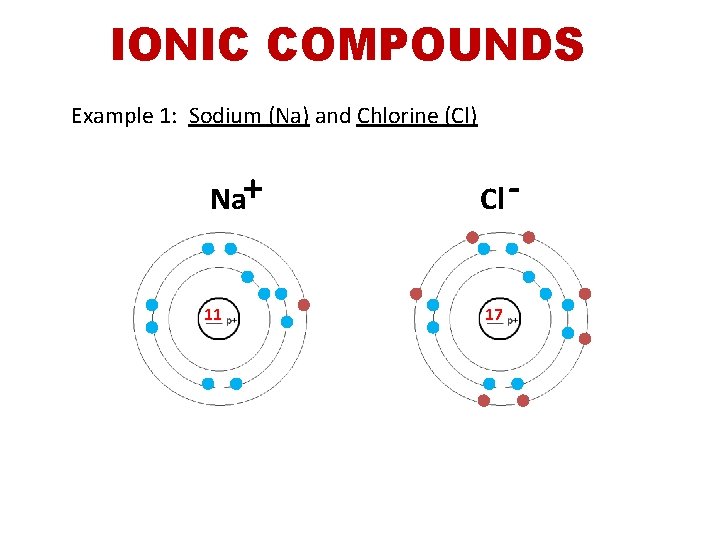
IONIC COMPOUNDS Example 1: Sodium (Na) and Chlorine (Cl) Na+ 11 Cl - 17

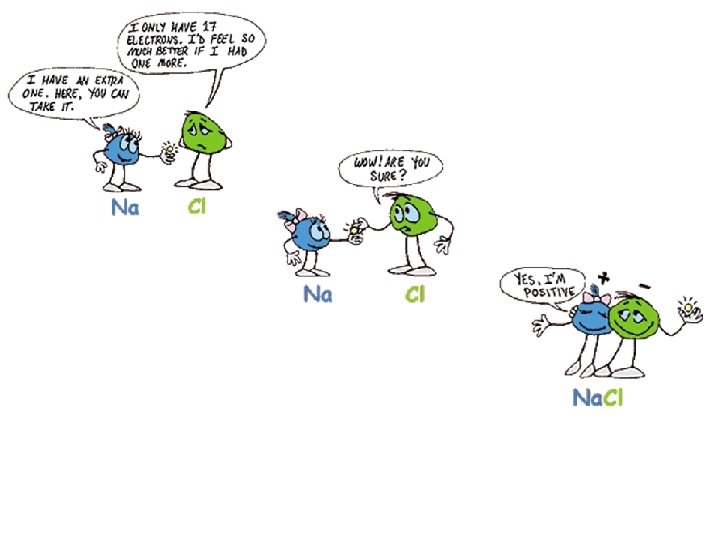
IONIC BONDING Example 1: Sodium (Na) and Chlorine (Cl) • the sodium atom loses its outermost electron to chlorine so that both atoms get a full outer shell (and stable become ______). Sodium has lost an electron and now has a _______ charge, while chlorine has positive negative gained an electron and now has a _______charge. oppositely attract • _______ charged atoms ______, forming ionic an _______ bond.
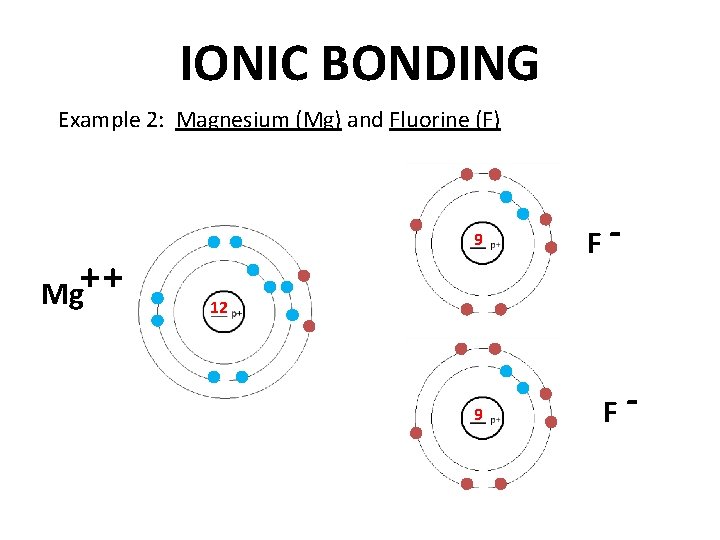
IONIC BONDING Example 2: Magnesium (Mg) and Fluorine (F) 9 Mg+ + F- 12 9 F-

IONIC BONDING Example 2: Magnesium (Mg) and Fluorine (F) • Magnesium needs to lose 2 to gain 1 electrons. Fluorine needs electron. Magnesium can only lose two electrons if it reacts with 2 fluorine atoms. Mg. F 2 • The formula for this compound is ______
Molecules & Covalent Bonding

Molecules & Covalent Bonding non-metal • Covalent bonds are formed from ______ elements. • Unlike ionic bonds that are formed from ions that lost or ____ gained electrons, covalent bonds have ______ are formed from atoms that _______ share electrons. • The attraction of 2 atoms for the shared pair of covalent bond. electrons results in a _____ • Two or more atoms joined by covalent bonds form molecule a _____.

Molecules & Covalent Bonding Example #1: Hydrogen Molecule The hydrogen molecule consists of 2 hydrogen 1 valence atoms. Each hydrogen atom has _____ electron. If each atom shares its electron with the 2 electrons in other atom, then each atom has _____ their outer shell (1 st shell). The shell is now full and the atoms (and molecule) are _______. stable
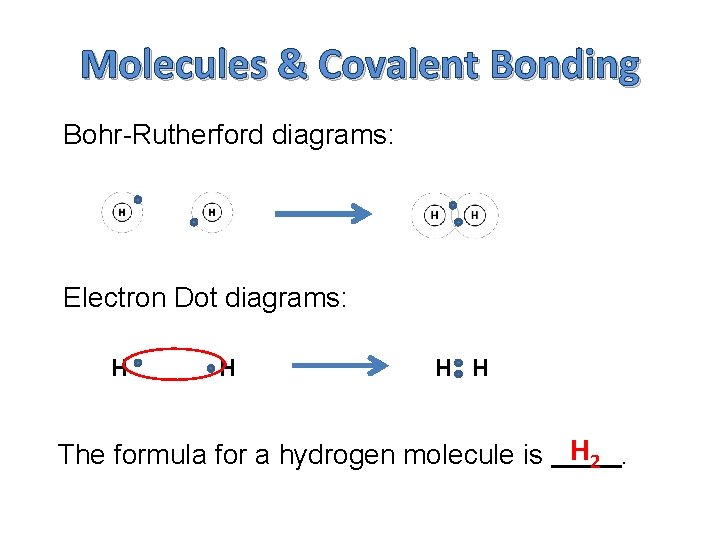
Molecules & Covalent Bonding Bohr-Rutherford diagrams: Electron Dot diagrams: H H The formula for a hydrogen molecule is H 2.
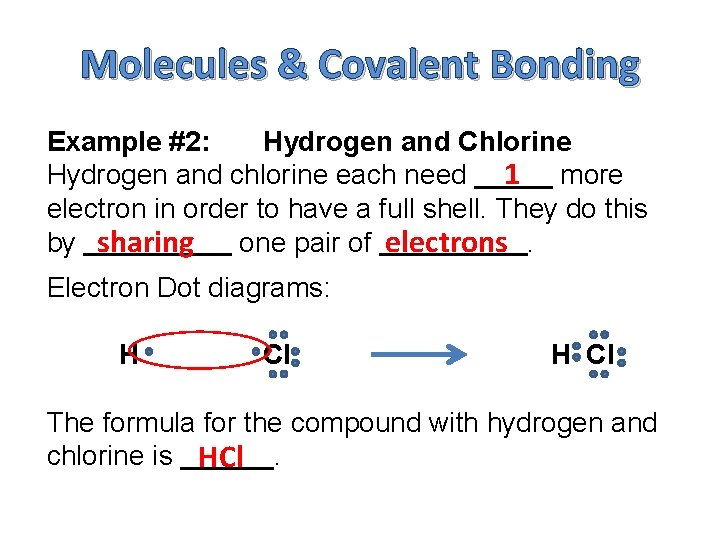
Molecules & Covalent Bonding Example #2: Hydrogen and Chlorine Hydrogen and chlorine each need 1 more electron in order to have a full shell. They do this by sharing one pair of electrons. Electron Dot diagrams: H Cl The formula for the compound with hydrogen and chlorine is HCl.

Molecules & Covalent Bonding Example #3: Hydrogen and Oxygen Hydrogen needs 1 more electron in order to have a full outer shell. Oxygen needs ____ 2 more electrons to have a full outer shell. Oxygen can get a full shell if it shares its 2 unpaired electrons with 2 hydrogen atoms. This also gives each hydrogen atom a full outer shell.
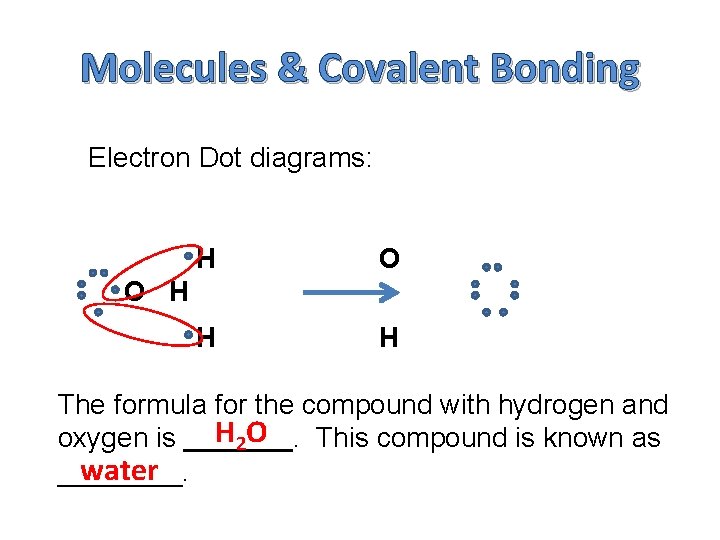
Molecules & Covalent Bonding Electron Dot diagrams: H O H The formula for the compound with hydrogen and oxygen is H 2 O. This compound is known as water ____.

Molecules & Covalent Bonding NOTE: When drawing electron dot diagrams for a molecule, use the stable octet rule. With the exception of hydrogen, each atom of the molecule should have 8 electrons (dots) surrounding it (including shared electrons).
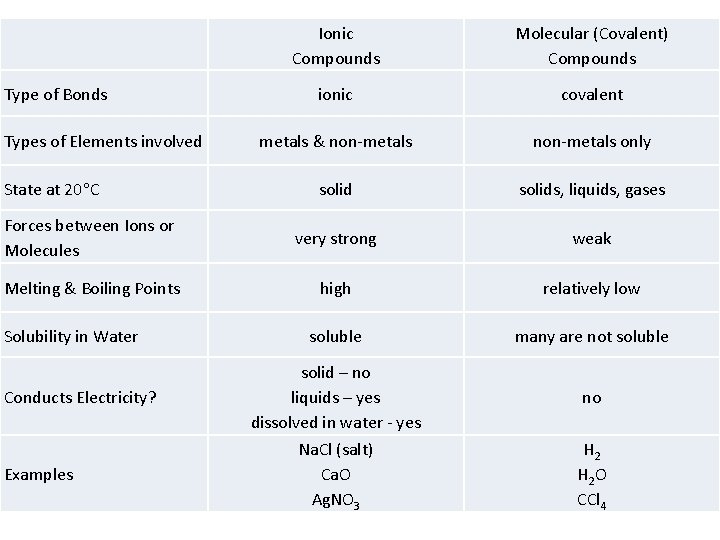
Ionic Compounds Molecular (Covalent) Compounds ionic covalent metals & non-metals only solids, liquids, gases Forces between Ions or Molecules very strong weak Melting & Boiling Points high relatively low soluble many are not soluble solid – no liquids – yes dissolved in water - yes no Na. Cl (salt) Ca. O Ag. NO 3 H 2 O CCl 4 Type of Bonds Types of Elements involved State at 20 C Solubility in Water Conducts Electricity? Examples
- Slides: 18