How a Study Section works Robert Freund Ph

How a Study Section works Robert Freund, Ph. D. Scientific Review Administrator Virology B Study Section Center of Scientific Review Freundr@csr. nih. gov 301 -435 -1050

Subjects we will cover: • Submitting an application • Review of the application The study section • After the study section What next?

Dual Review System for Grant Applications First Level of Review Scientific Review Group · Provides Initial Scientific Merit Review of Grant Applications · Rates Applications and Makes Recommendations for Appropriate Level of Support and Duration of Award Second Level of Review Council/Institute · Assesses Quality of SRG Review of Grant Applications · Makes Recommendation to Institute Staff on Funding · Evaluates Program Priorities and Relevance · Advises on Policy

Center for Scientific Review · Division of Receipt and Referral: Central receipt point for most PHS grant applications · Institute assignment · Assignment to Scientific Review Group · Study Section: Conducts initial scientific merit review of most research applications submitted to the NIH

Applications Submitted to NIH Over 60, 000 grant applications are submitted to NIH each year.

CSR Study Sections · Standing Study Sections when the subject matter of the application matches the referral guidelines for the study section. Each study section has 20 - 30 members. Approximately 80 applications are reviewed at each study section meeting · Ad Hoc Special Emphasis Panels (SEPs) when the subject matter does not fit into any study section, or when assignment of an application to the most appropriate study section would create a conflict of interest.

Submit Cover Letter • Suggest appropriate Institute Multiple institute assignments • Suggest appropriate Study Section Go to web site: www. csr. nih. gov Description of Study Sections Study Section Rosters
How to choose a study section www. csr. nih. gov



Assignment Notification Letter Assignment Number: 2 R 01 HL 12345 - 12 A 1 Dual Assignment: NS Scientific Review Group: Virology B (Vir. B) Information about SRGs may be found on the CSR Home page (http: //www. csr. nih. gov) Scientific Review Administrator: DR. ROBERT FREUND , SRA CTR FOR SCIENTIFIC REV 6701 ROCKLEDGE DR RM 3202 MSC 7808 BETHESDA MD 20892 (301) 435 - 1050 Institute/Center: NATL HEART, LUNG, & BLOOD INST DIV/EXTRAMURAL AFFAIRS RK 2 7100 NATIONAL INSTITUTES OF HEALTH BETHESDA, MD 20892 (301) 480 -5295

CSR Study Sections • Administrative Review • Recruitment Standing Members Temporary Members • Assignments Match Expertise/Interests with Applications Conflict of Interest Issues Workload of Reviewers

Study Section Actions · Unscored (lower half) · Scored, Scientific Merit Rating (priority scores and percentiles) · Deferral (this cycle, next cycle)
Mock Study Section Video

Streamlining · Reviewers categorize applications in “lower half” in scientific merit using preliminary scores. · Lower half applications: · Not discussed or scored · Written critiques are provided to the applicant · Not taken to Advisory Council · In most cases, unscored applications have potential and are worth revising

Scored Applications • Reviewers (usually 3) present opinions • Discussion (approx. 15 min. per application) • Discuss other considerations (vertebrate animals, human subjects, bioharzards) • Score • Discuss Budget

Review Criteria · Significance: Does the study address an important problem? How will scientific knowledge be advanced? · Approach: Are design and methods well-developed and appropriate? Are problem areas addressed? · Innovation: Are there novel concepts or approaches? Are the aims original and innovative? · Investigator: Is the investigator appropriately trained? · Environment: Does the scientific environment contribute to the probability of success? Are there unique features of the scientific environment? Overall Evaluation & Score Reflects Impact on Field
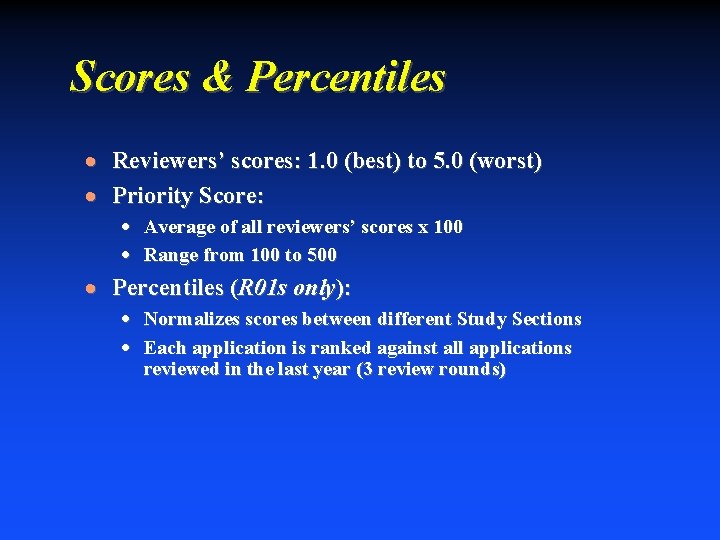
Scores & Percentiles · Reviewers’ scores: 1. 0 (best) to 5. 0 (worst) · Priority Score: · Average of all reviewers’ scores x 100 · Range from 100 to 500 · Percentiles (R 01 s only): · Normalizes scores between different Study Sections · Each application is ranked against all applications reviewed in the last year (3 review rounds)

Summary Statement Once applications are reviewed, the results are documented by the SRA in a summary statement and forwarded to the Institute (and the PI) where a funding decision is made: The summary statement contains: · Overall Resume and Summary of Review Discussion · Essentially Unedited Critiques · Priority Score and Percentile Ranking · Budget Recommendations · Administrative Notes

Getting the Results Mailer: score & percentile Program staff: Score & percentile Payline information Advice on revision The NIH Commons Summary Statement

After Review? • Contact Program Officer Funding Decisions, advise and interpretation • Revise Application Address everything in Introduction Don’t follow comments blindly Resubmit

NIH Grant Receipt, Review, and Award Schedule Feb-March June-July Oct-Nov Feb-Mar Sept Feb June Dec 1 Apr 1 July 1 Receipt Dates Review Dates National Advisory Council Board Dates Earliest Possible Beginning Date
- Slides: 22