Hormones Biochemistry Classification of hormones Diversity in the

Hormones Biochemistry
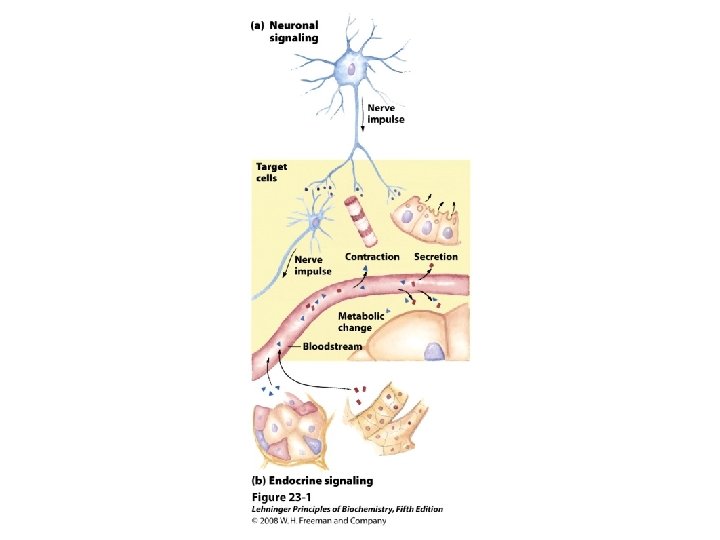
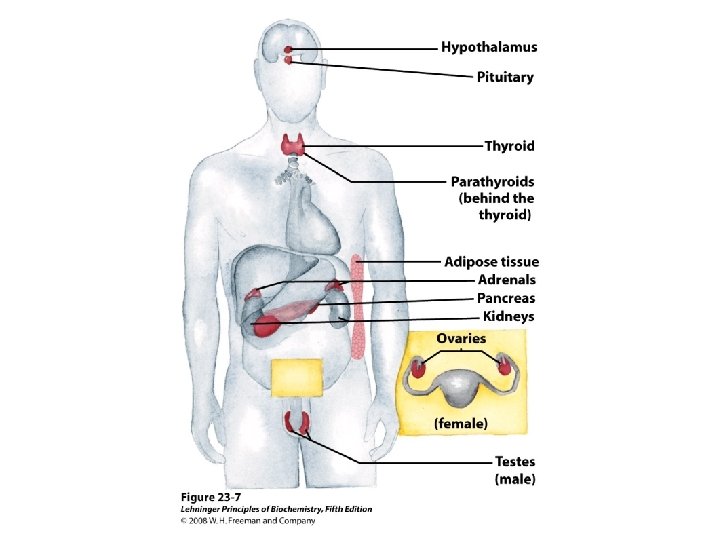
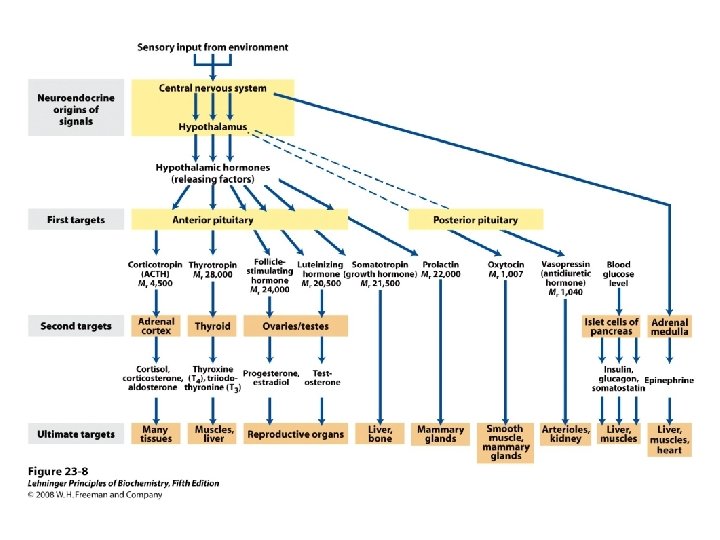
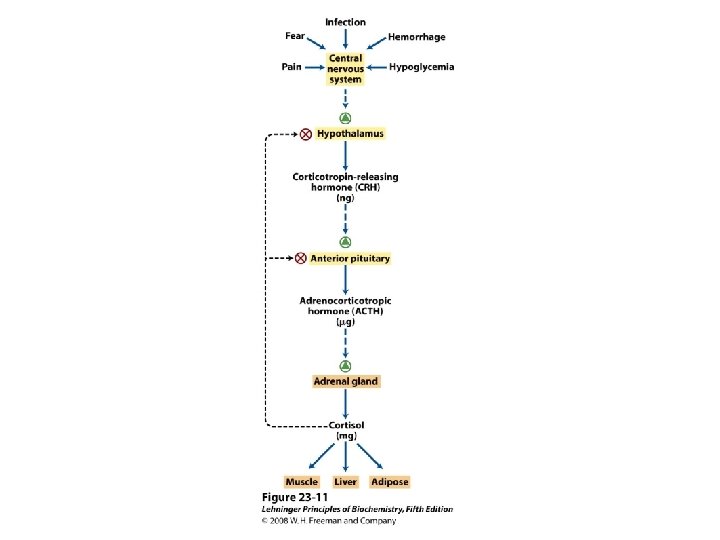

Classification of hormones


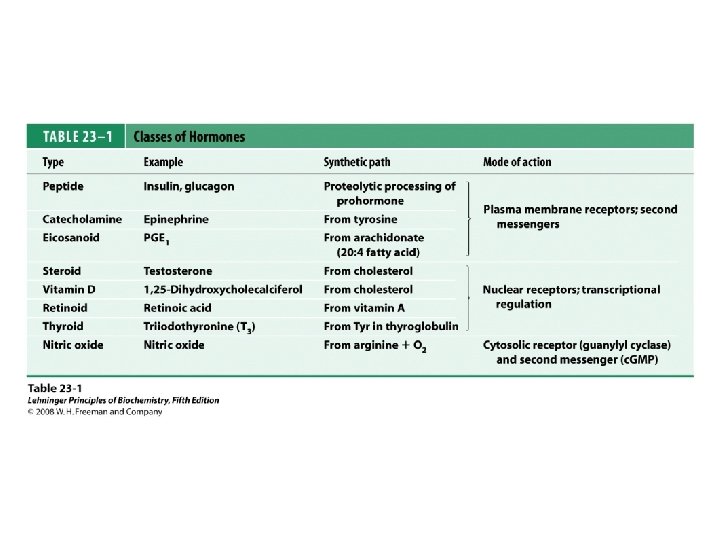

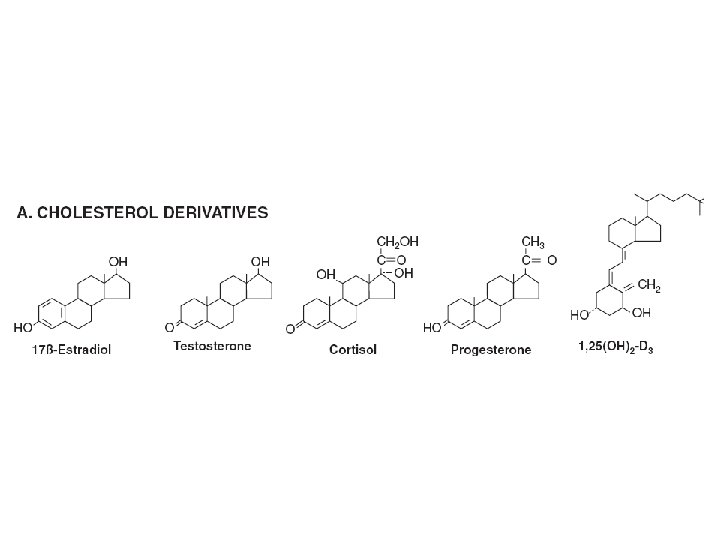
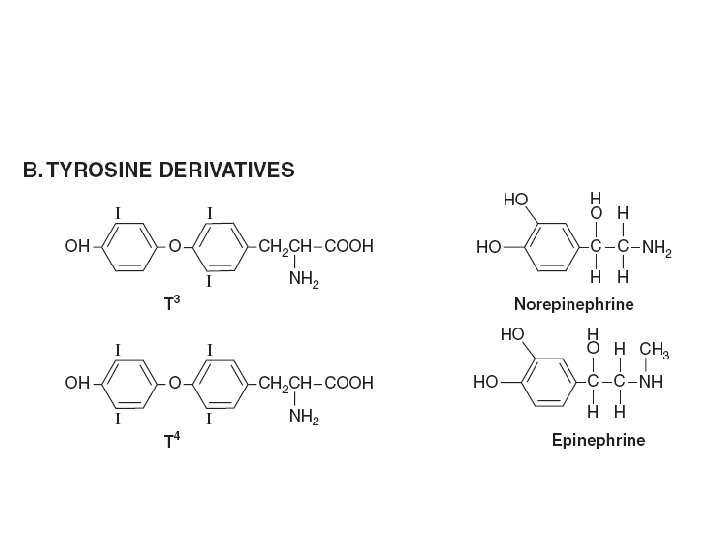
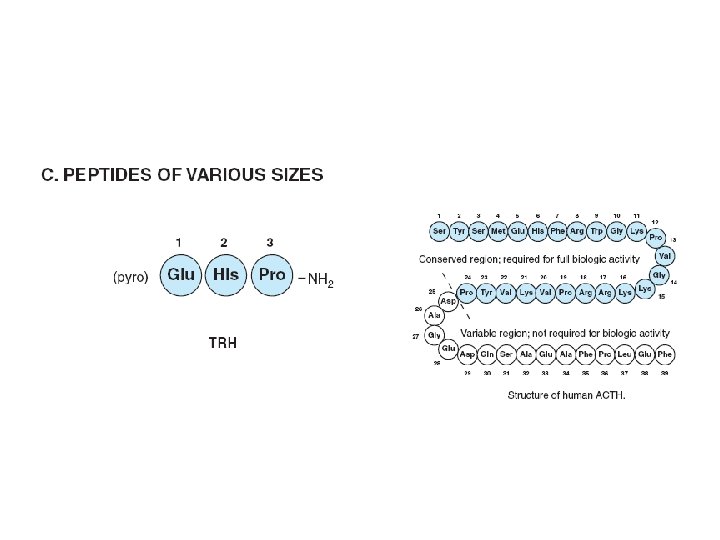
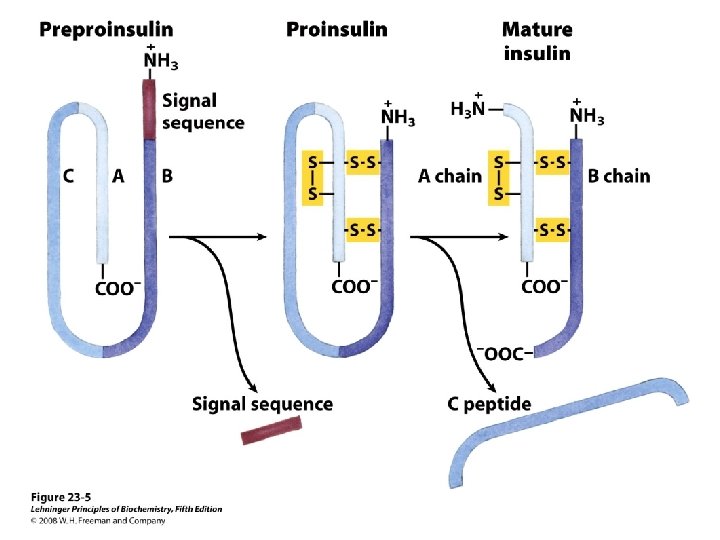

Diversity in the storage of hormones
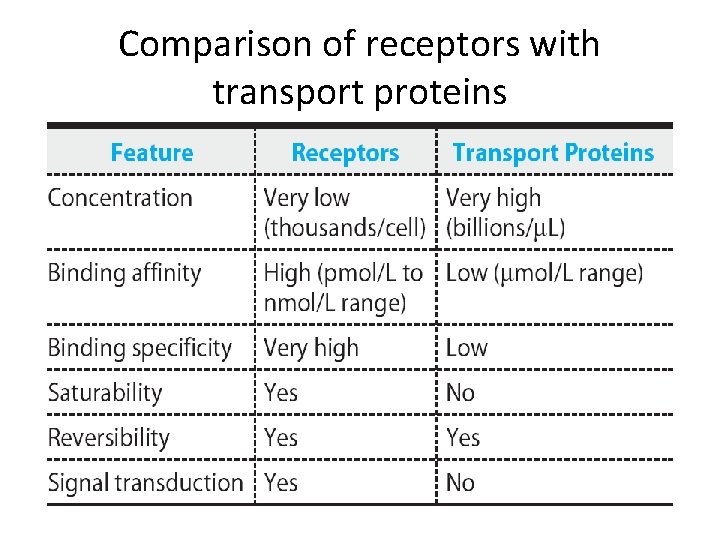
Comparison of receptors with transport proteins
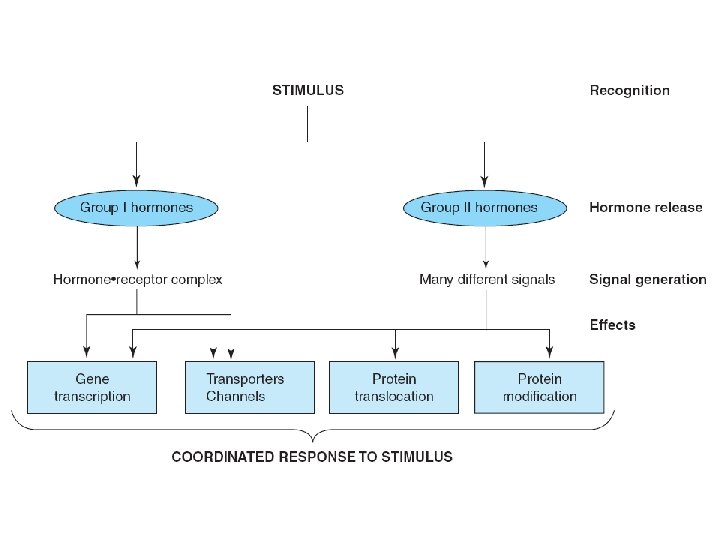
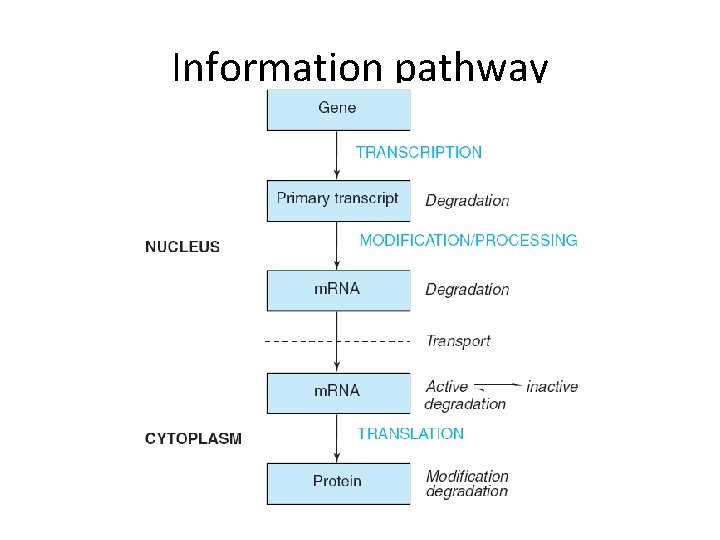
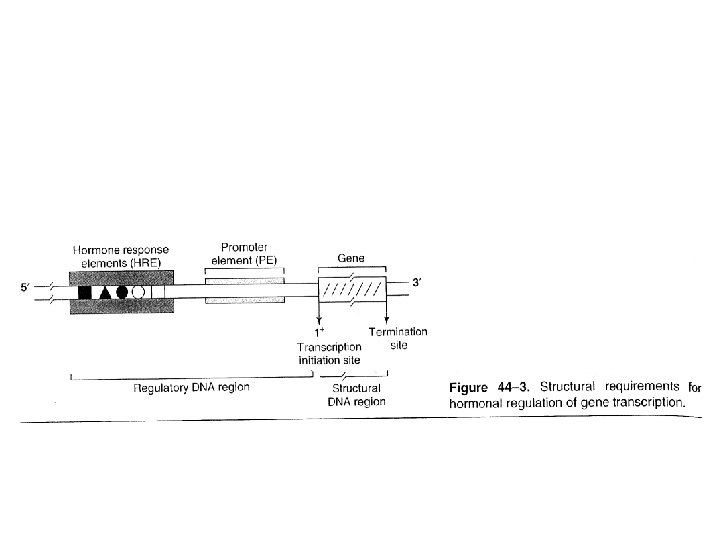
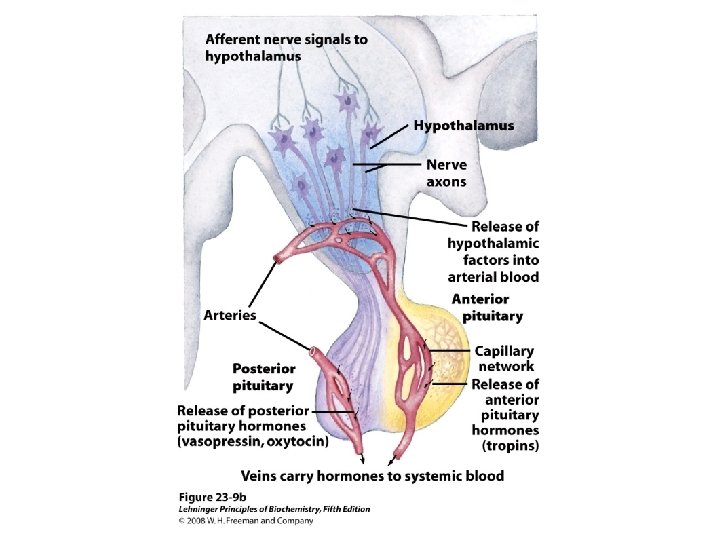
Information pathway
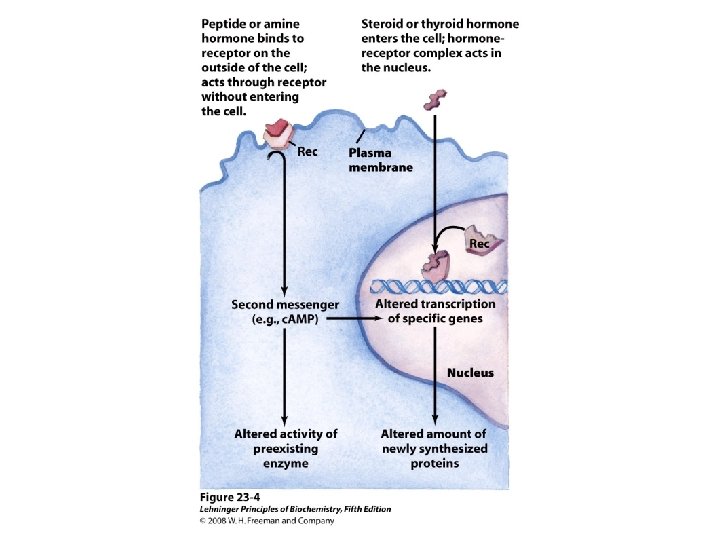
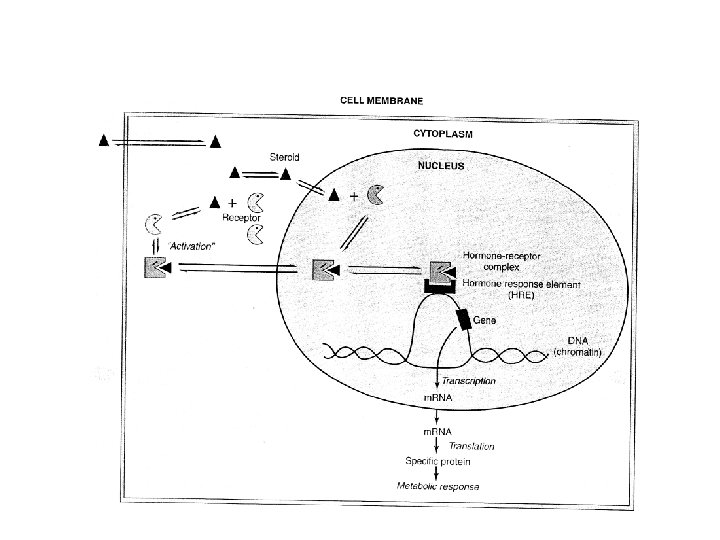

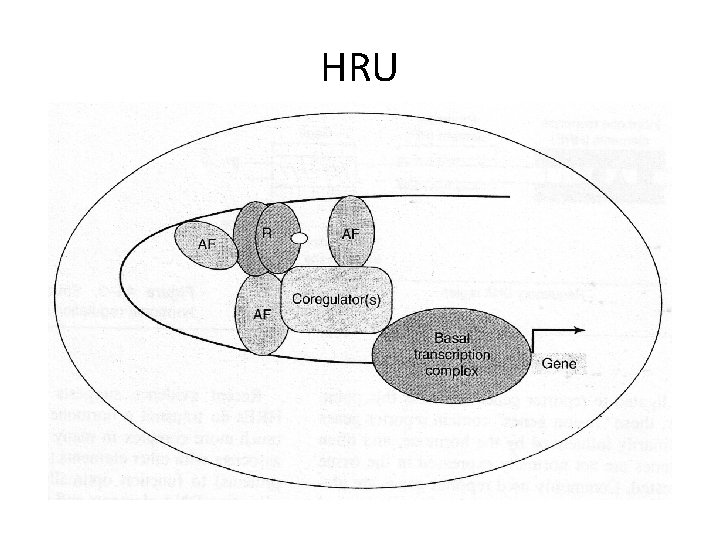
HRU
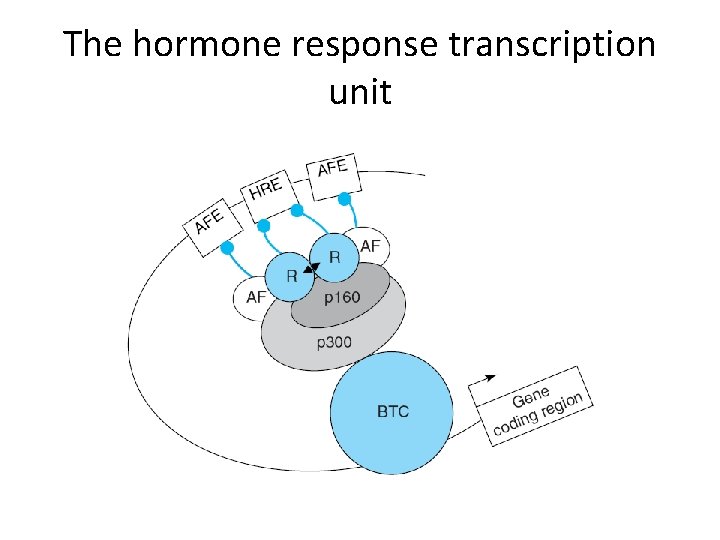
The hormone response transcription unit
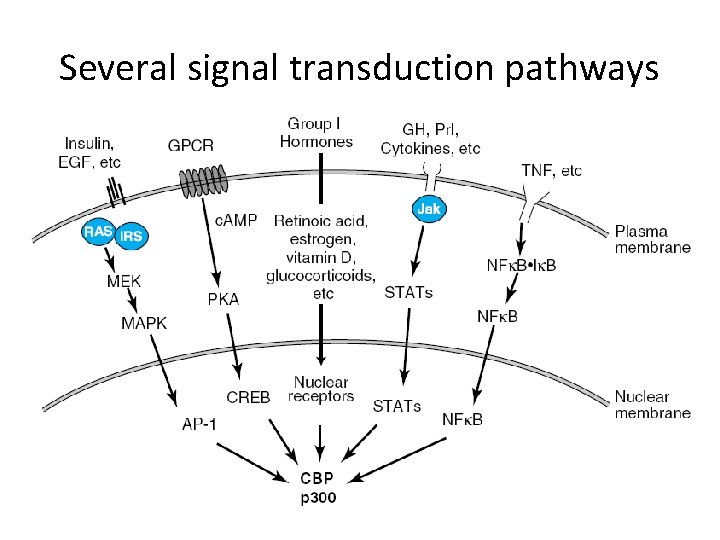
Several signal transduction pathways
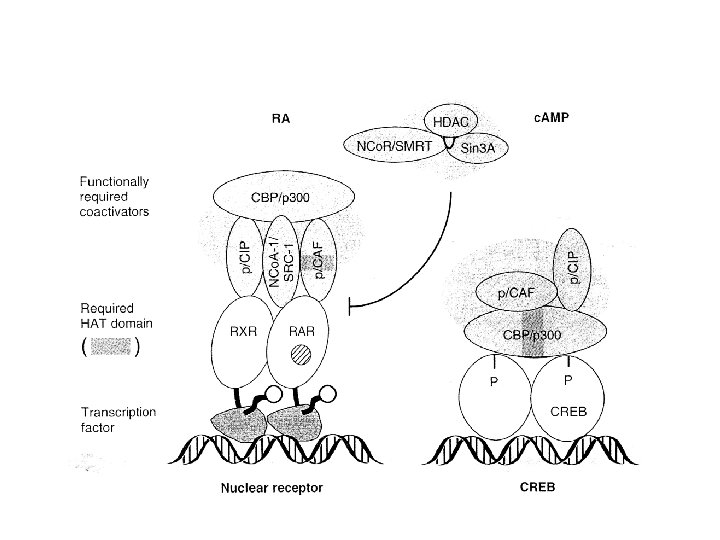
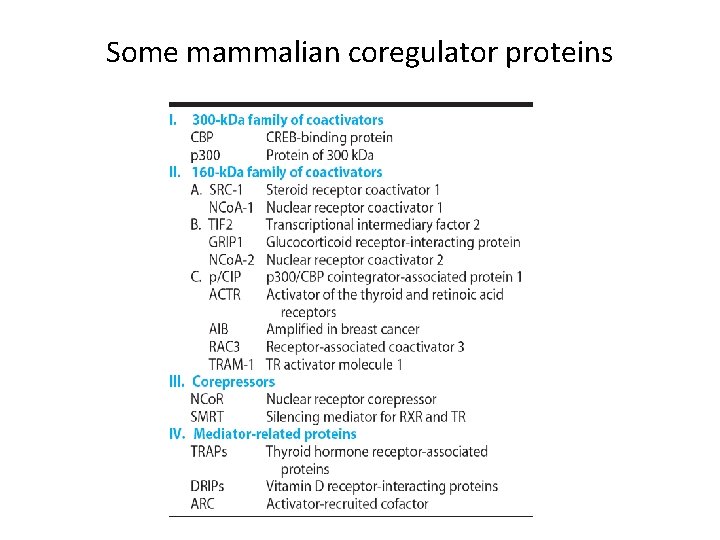
Some mammalian coregulator proteins

Subclassification of group II. A hormones
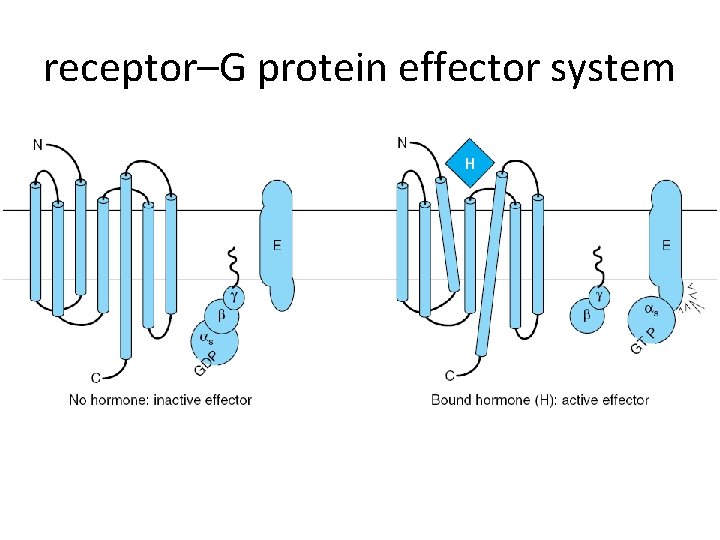
receptor–G protein effector system
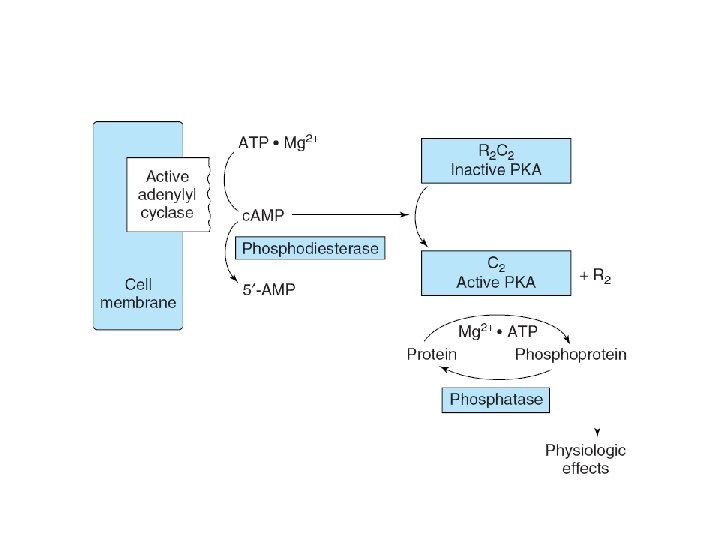
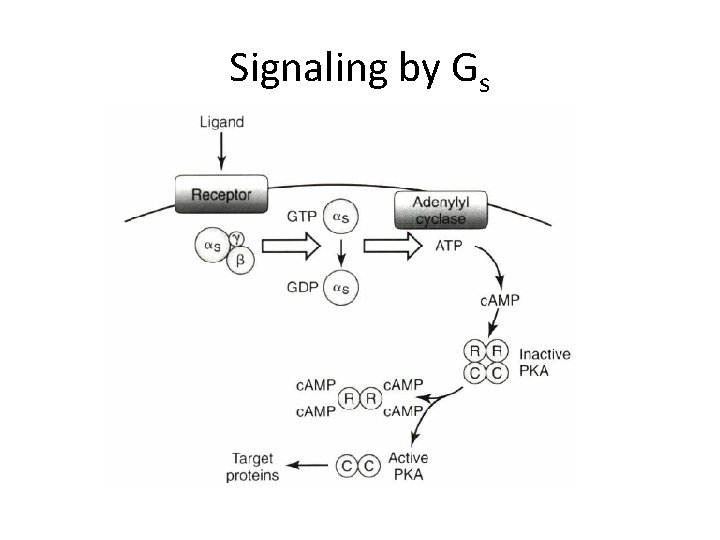
Signaling by Gs
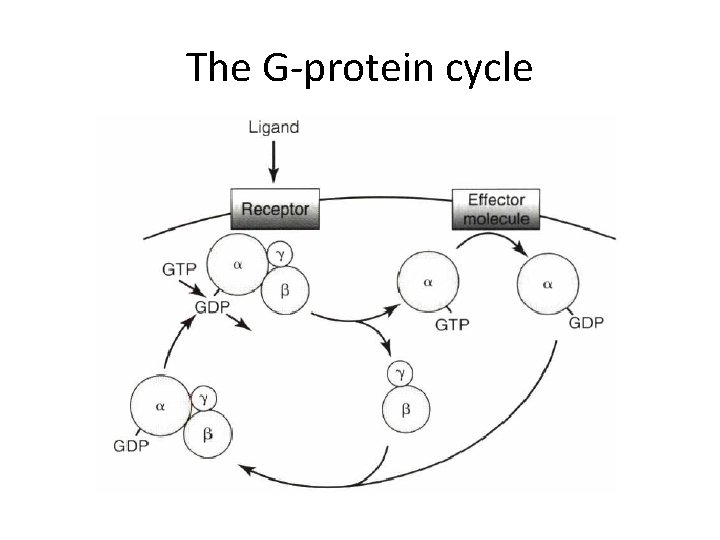
The G-protein cycle
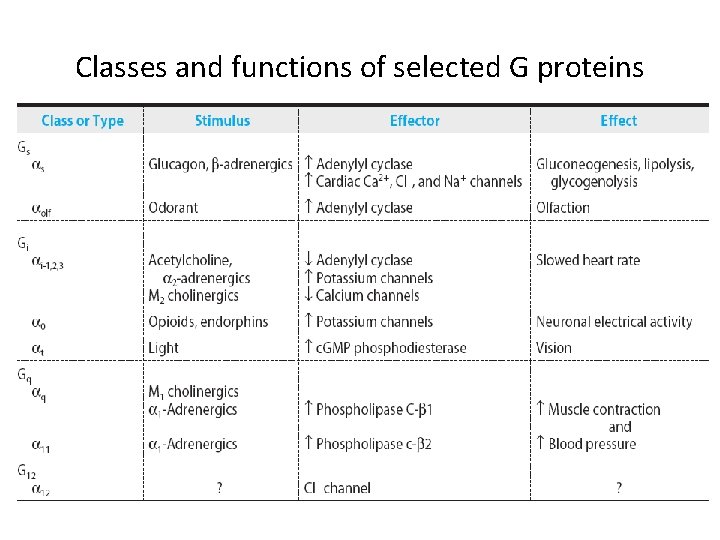
Classes and functions of selected G proteins
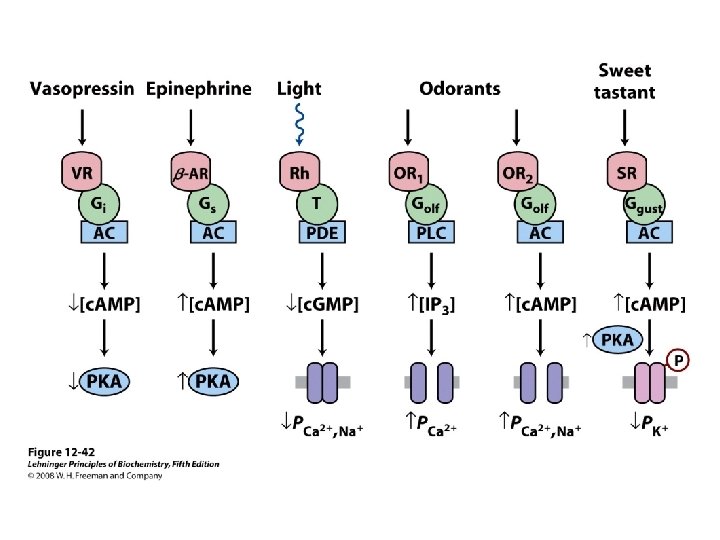
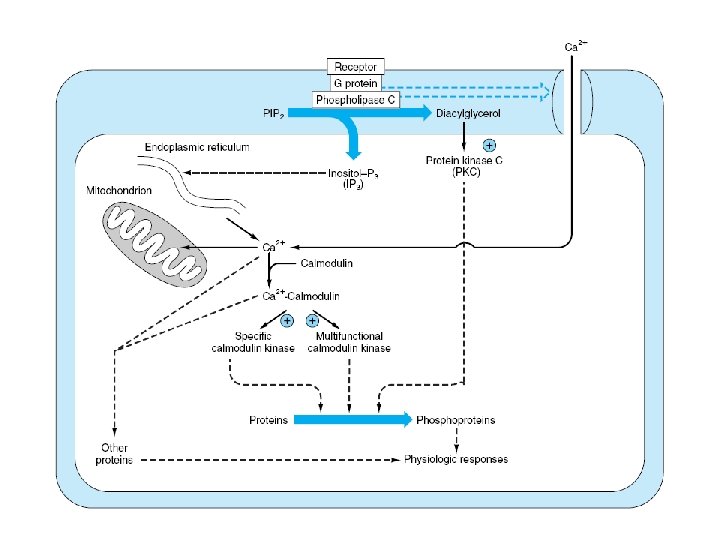
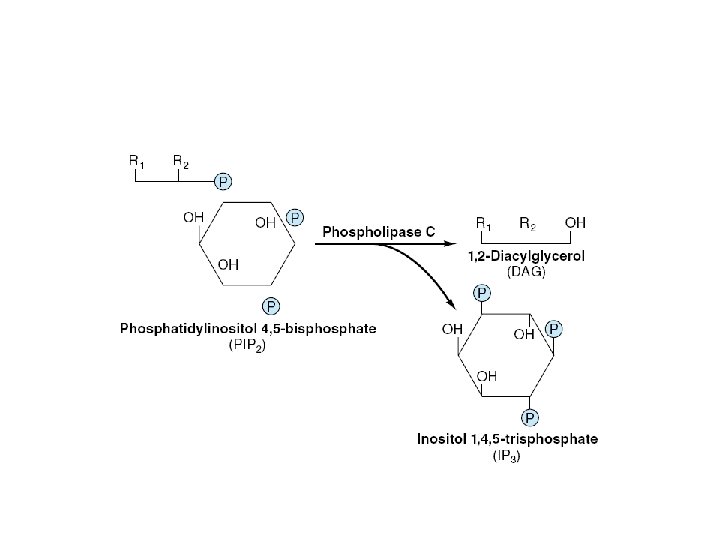
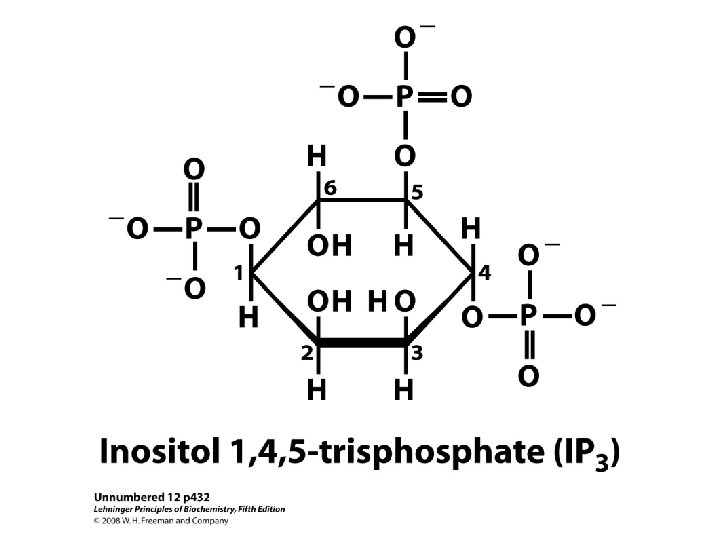

Enzymes and proteins regulated by calcium or calmodulin
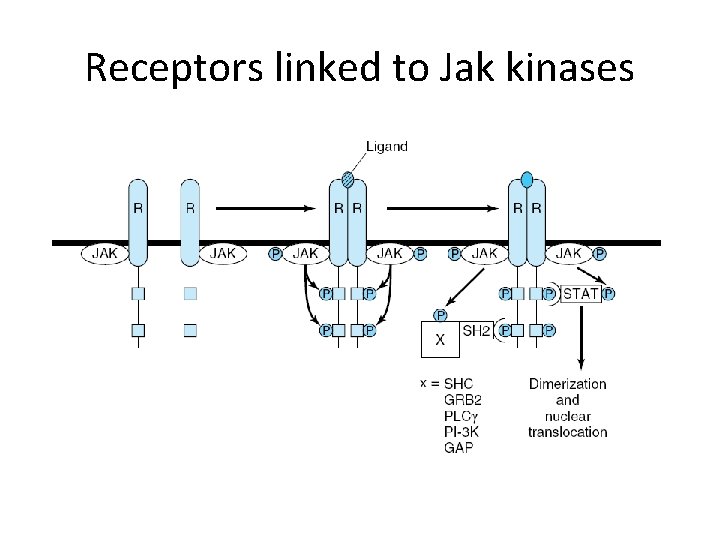
Receptors linked to Jak kinases

Anterior pituitary hormones • Growth hormone, prolactin, chorionic somatomammotropin group – Growth promoting and lactogenic activity – 190 -199 aa – h. GH vs h. CS 85% – h. GH vs h. PRL 35% – Common antigenic determinants • Glycoprotein hormone group • Pro-opiomelanocortin peptide family

Growth hormone (GH) • Synthesis – Somatotropes • • Single plypeptide 22 k. Da 191 aa Receptor – Cytokine– hematopoietin receptor super family – Single membrane spanning domain

Physiological & biochemical actions • Essential for – Postnatal growth – Normal carbohydrate, lipid , nitrogen, mineral metabolism • Growth related effects – By IGF-I

Physiological & biochemical actions • Protein synthesis – Increase transport of amino acids into muscle cells – Positive nitrogen balance • Generalized increase in protein synthesis • Decrease plasma and urinary amino acid and urea • Increase synthesis of RNA, DNA • Carbohydrate metabolism – Hyperglycemia • Decreased peripheral utilization of glucose • Hepatic gluconeogenesis • Diabetogen

Physiological & biochemical actions • Impairment of glycolysis • Increase liver glycogen • Lipid metabolism – Lipolysis • Ketogenesis • Mineral metabolism – GH or IGF-I • Growth of long bones at epiphysial plates (appositional or acral growth in adults) – Promote a positive Ca, Mg, P, balance • Retention Na, K, Cl

Physiological & biochemical actions • Prolactin-like effect – Stimulation of mammary glands – Lactogenesis •

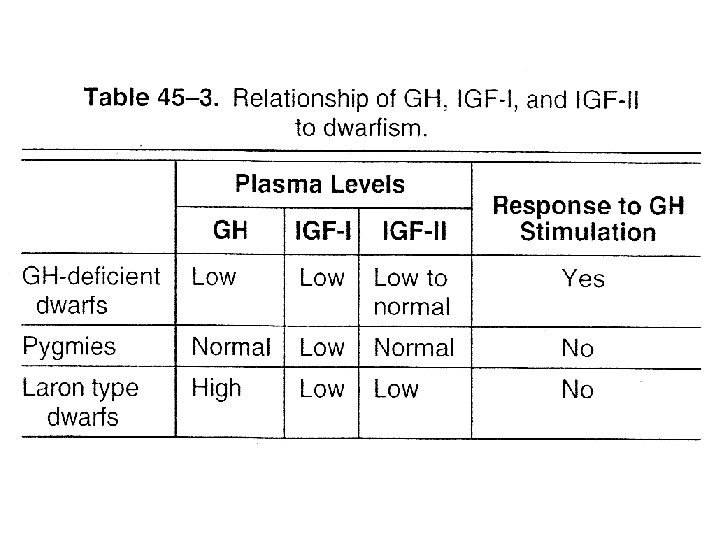
Pathophysiology • Deficient amounts of GH – Dwarfism • Infants fail to grow properly • Types of dwarfism (table) – GH deficient – Target organ resistance » Laron , Lack functional hepatic GH receptor » Pygmies, have post GH receptor defect • GH excess – Gigantism • Before epiphysial close

Pathophysiology • Acromegaly – GH excess after epiphysial closure – Metabolic problem • Diabetes mellitus

Prolactin (PRL, lactogenic hormone, mammotropin, luteotropic H) • • 23 k. Da Lactotropes Similar (structure) to GH & CS Receptor – Similar to GH • Single membrane spanning domain • Signaling like GH

Physiological & biochemical actions • Initiation & maintenance of lactation – Primed by sex hormones • Target tissue – Breast

Pathophysiology • Excess PRL – Amenorrhea (cessation of menses) – Galactorrhea (milk discharge) • In Men – Gynecomastia (breast enlargement) – Impotence

Chorionic somatomammotropin (CS) Placental lactogen • No definite function in human • In bioassay – Lactogenic – Luteotropic – Metabolic effects • • Inhibition of glucose uptake Lipolysis Ca & N retention Retention of phosphorus & K

Glycoprotein hormones • TSH, LH, FSH, CG • Two subunits – αβ • • α is Identical β Specific biologic activity Receptor recognition (both) Synthesize from unique m. RNA from separate genes Glycosylation may be necessary for αβ interaction β is limiting for the production of complete hormone Synthesizes as preprohormones


Glycoprotein hormones • FSH, LH, h. CG – gonadotropins • Gametogenesis • Steroidogenesis • 25 k. Da • FSH – Follicular cells (ovary) – Sertoli cells (testis) – Mediator • c. AMP

Glycoprotein hormones • LH (luteinizing hormone) – Corpus luteum • Progesterone – Leydig cells • Testosterone – Mediator • c. AMP – Acetate to squalene, – Cholesterol to pregnenolone – Prolonged exposure • Desensitization – Down regulation of receptor

Glycoprotein hormones • h. CG (human chorionic gonadotropin) – Syncytiotrophoblast cells of placenta – Closely resembles LH – Indicator of pregnancy • TSH – 30 k. Da – c. AMP – Acute effects (in minutes) • All phases of T 3 and T 4 synthesis

Glycoprotein hormones • TSH – Chronic effects (days) • Increase synthesis of – Proteins – Phospholipids – Nucleic acids • Size and number of thyroid cells – Metabolic effects • Due to thyroid hormones

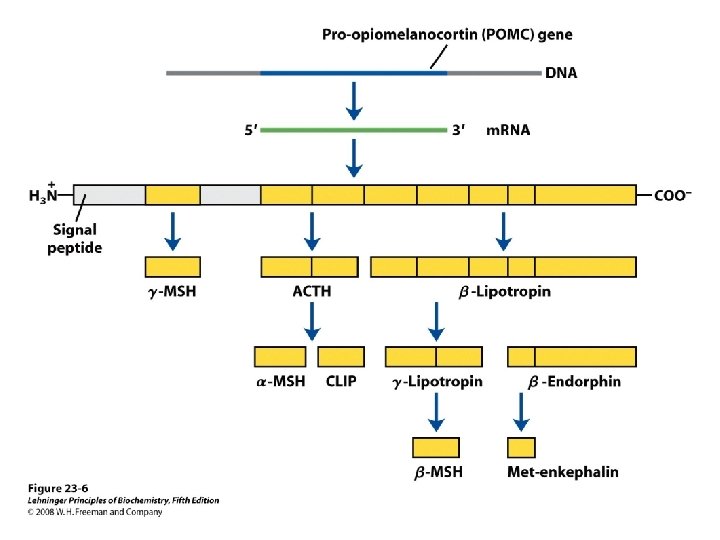
Pro-opiomelanocortin(POMC) • Consists of – ACTH, LPH, MSH • Synthesized as precursor 285 aa • Processed differently in various regions • The most conserved regions – Amino terminal • ACTH region – β- endorphin region

Pro-opiomelanocortin (POMC)

Pro-opiomelanocortin(POMC) • Anterior lobe – ACTH & β-LPH • Intermediate & other tissues (gut, placenta. . . ) – Further cleavages occur • ACTH – Single chain polypeptide • 39 aa – Regulates growth and function of adrenal cortex – 24 aa N full biologic activity • ACTH 1 -24, invariant between species

Pro-opiomelanocortin(POMC) • Synthesis of adrenal steroids – Conversion of cholesterol to pregnenolone • Adrenal cortical growth – Trophic effect • Protein & RNA synthesis • Mediator – c. AMP

Pathophysiology • Excessive production ACTH – Cushing’ syndrome – Hyperpigmentation • MSH- like activity • α or β MSH release – Metabolic manifestations • Negative balance – N, P, K • Na retention – Hypertension, edema

Pathophysiology Glucose intolerance Diabetes mellitus Increased plasma fatty acids Decreased circulating eosinophils & lymphocytes • Increased PMN leukocytes • •

• β- lipotropin(β-LPH) – Carboxyl terminal 91 aa of POMC – Lipolysis & fatty acid mobilization • Physiological role is minimal – Serve as precursor • for β-endorphin • Endorphins – β-endorphin • Precursor for other endorphins

Endorphins • Inactive in pituitary – Acetylated • Receptor – The same as opiates • Control of pain

MSH (melanocyte-stimulating hormone) • Stimulates melanogenesis – Darkening of the skin • Includes – α MSH, β MSH, γ MSH • Actual circulating MSH activity in human – Within β or γ LPH • Addison’s disease – Insufficient production of glucocorticoids • Hyper pigmentation • Plasma MSH activity – ACTH , more likely β or γ LPH

Posterior pituitary • Vasopressin – Increased blood pressure at pharmacologic dose • Antidiuretic hormone (ADH) – Promote reabsoption of water from distal renal tubules • Oxytocin – Acceleration of birth • Uterine smooth muscle contraction • Milk ejection

Posterior pituitary • ADH – Supraoptic nucleus • Oxytocin – Paraventricular nucleus • Axoplasmic flow – Neurophysins I • Oxytocin, 19 k. Da – Neurophysins II • ADH, 21 k. Da
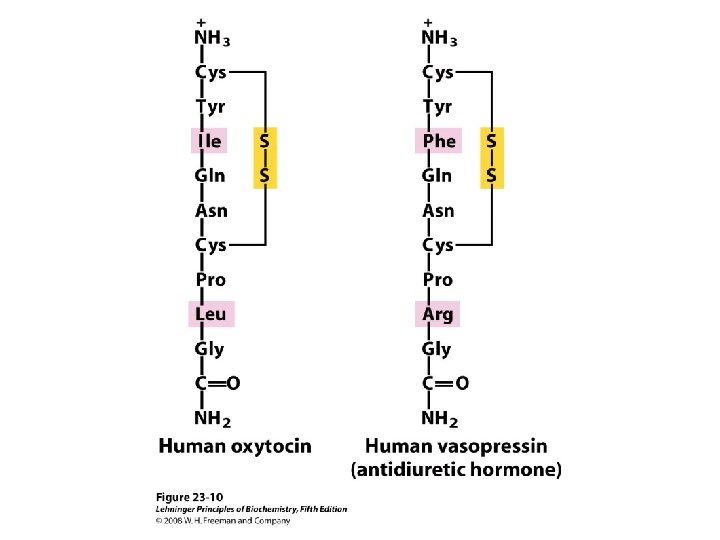

Posterior pituitary • Very short half life – 2 -4 min • Close structural similarity – Exhibit some of the effects of the other • Metabolism – Liver – Renal excretion of ADH

Oxytocin • Primary stimulus – Stimulation of nipple – Oxytocin fragment • As prolactin releasing factor • Estrogen – Production of Oxytocin & Neurophysins I • Progesterone – Inhibitor of production & Neurophysins I

Oxytocin • Mechanism of action – Unknown – Contraction of myoepithelial cells surrounding the mammary alveoli • Receptors – Uterine , mammary tissue – Estrogens + – Progesterone – • Oxytocin & Neurophysins I – Produce in ovary • Inhibit steroidogenes

ADH • Regulation of secretion – Increased plasma osmolality • • • Osmoreceptors , hypothalamus Baroreceptors , heart & vascular systems Epinephrine – Expanded plasma volume – Ethanol – • Mechanism of action – Target cell • Distal tubules & collecting

ADH • Increase permeability of cells to water – Results in volumes in the range of 0. 5 -1 L/d • Receptors – V 1 & V 2 – V 2 • Renal epithelial cells – c. AMP – Hypercalcemia » Inhibit adenylyl cyclase • Excessive volume of urine

ADH • V 1 – Extrarenal receptors • Activation of phospholipase C – IP 3 & DAG – Ca 2+ – PKC » Vasoconstriction

Pathophysiology • Diabetes insipidus – ADH secretion or action – Large volume of urine – Primary • Insufficient ADH – Hereditary nephrogenic Diabetes insipidus • ADH normal • Target cell – Receptor defect – Acquired nephrogenic Diabetes insipidus • Lithium administration

Pathophysiology • Inappropriate secretion of ADH – Ectopic production (by tumor, other diseases ) – ADH • Normal or increased • In the presence of hypoosmolality! – Dilutional hyponatremia – Hypertonic urine


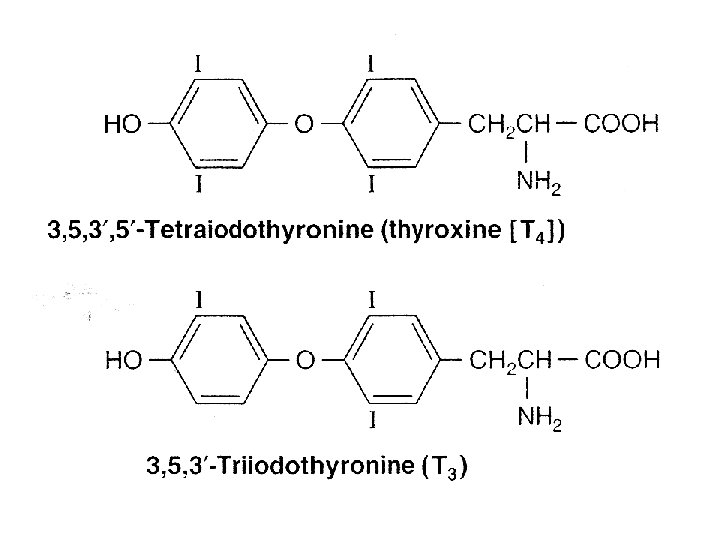
Thyroid hormones • Hormones – 3, 5, 3’-triiodothyronine (T 3) • Most of the biologic effect – 3, 5, 3’, 5’-tetra-lodothyronine (T 4, thyroxine) • Regulate – gene expression – Tissue differentiation – General development – General metabolism
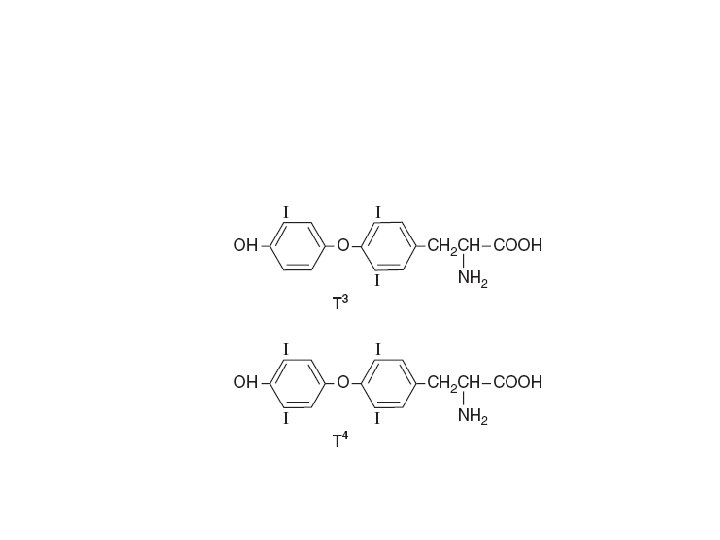

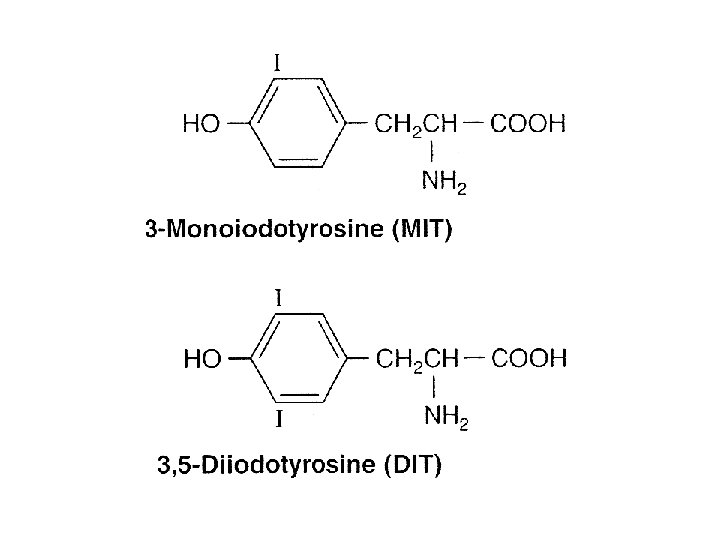
• MIT – Monoiodotyrosine • DIT – Diiodotyrosine • Iodine deficiency – Decrease • T 4: T 3 ratio • DIT: MIT ratio • Mechanism of action – Similar to steroid hormones

• Thyroid Hormone Biosynthesis – Precursor of T 4 and T 3 • Thyroglobulin – Storage form of T 3 & T 4 • TSH – Enhances transcription of the thyroglobulin gene – Thyroglobulin hydrolysis • Inhibited by I- • Thiourea drugs – Inhibit I- oxidation • Coupling – Two DIT molecules to form T 4 – MIT and DIT to form T 3
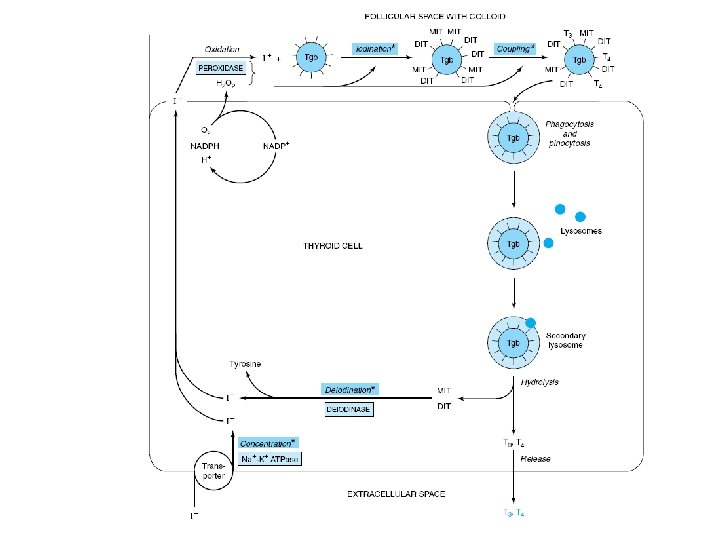
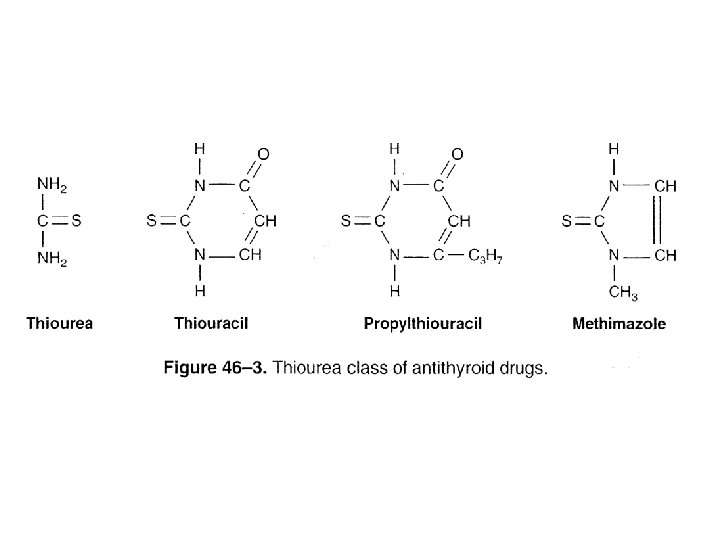


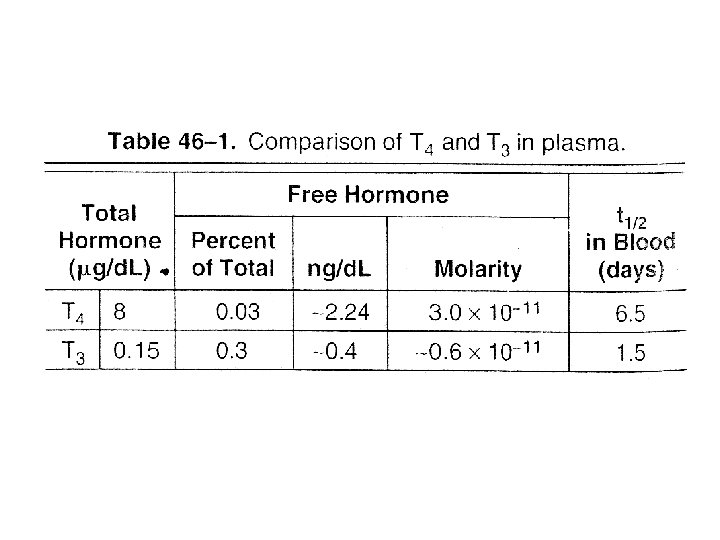
• TRANSPORTED – Thyroxine-binding globulin (TBG) – Thyroxine- binding prealbumin (TBPA) • Biologic activity – Free fraction • TBG is also subject to regulation – Increased • Estrogens – Decreased • Androgen or glucocorticoid therapy, liver diseases • Extrathyroidal deiodination – T 4 to T 3

• T 3 binds to receptor with 10 times the affinity of T 4 • T 4 – T 3 – Reverse T 3 (r. T 3) • Weak agonist • Larger amounts in chronic disease • Propylthiouracil and propranolol – Decrease conversion of T 4 to T 3

thyroid hormone metabolism • • • Total deiodination Deamination Decarboxylation Glucuronidation Sulfation • Excreted into – Bile, – Urine • Glucuronide conjugate

thyroid hormone action • Nuclear receptor • T 3 binds with higher affinity than T 4 • Effect – Enhance general protein synthesis • Cause positive nitrogen – By gene transcription – T 3 and glucocorticoids enhance transcription of the GH gene • Very high concentrations of T 3 – Inhibit protein synthesis • Negative nitrogen balance

thyroid hormone action • Required for normal development in humans – Cretinism • Intrauterine or neonatal hypothyroidism

PATHOPHYSIOLOGY • Goiter – Simple goiter • Decreased thyroid hormone production • Elevated TSH – Causes • Iodide deficiency • Iodide excess • Treated with – Exogenous thyroid hormone – Supplementation or restriction of iodide

PATHOPHYSIOLOGY • Hypothyroidism – Insufficient Free T 3 or T 4 – Thyroid failure – Disease of the pituitary or hypothalamus – Basal metabolic rate is decreased – Treated with exogenous thyroid hormone replacement

• Hyperthyroidism, Thyrotoxicosis – Excessive Production of Thyroid Hormone • Causes – Graves’ disease • Production of thyroid-stimulating Ig. G (TSI) – Activates the TSH receptor » Excessive T 3 and T 4 • Treated by – Blocking hormone production • Antithyroid drug


Parathyroid hormone (PTH) • Calcium ions – Neuromuscular excitability – Blood coagulation – Secretory processes – Enzyme reactions – Release of hormones – Release of neurotransmitters – Intracellular action of a number of hormones – Bone mineralization

• PTH, 1, 25(OH) 2 -D 3 – regulate the amount of calcium in the extracellular fluid • Influencing the transport of calcium across the membrane • Plasma calcium – Complexed with organic acids • 7% of total – Protein—bound (46%) • Primarily to albumin – Ionized (47%)

• Elevation of plasma calcium – Muscle paralysis and coma • Decrease plasma calcium – Tetanic convulsions • Hypoalbuminemia – Decrease of total plasma calcium • p. H-dependence • Acidosis favors the ionized form

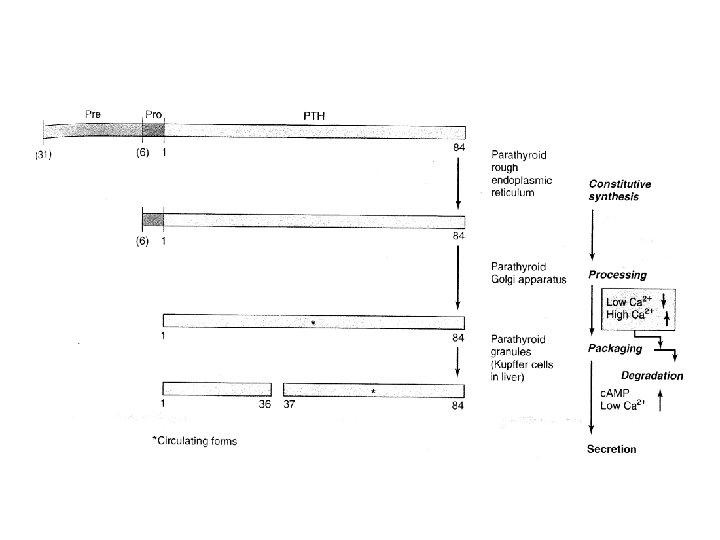
• PTH – 84 aa – Contains no carbohydrate – Biologic activity • PTH 1 -34 • Region 25— 34 is for receptor binding • PTH is synthesized as a 115 -amino-acid precursor – Immediate precursor • pro. PTH – Primary gene product • Prepro PTH
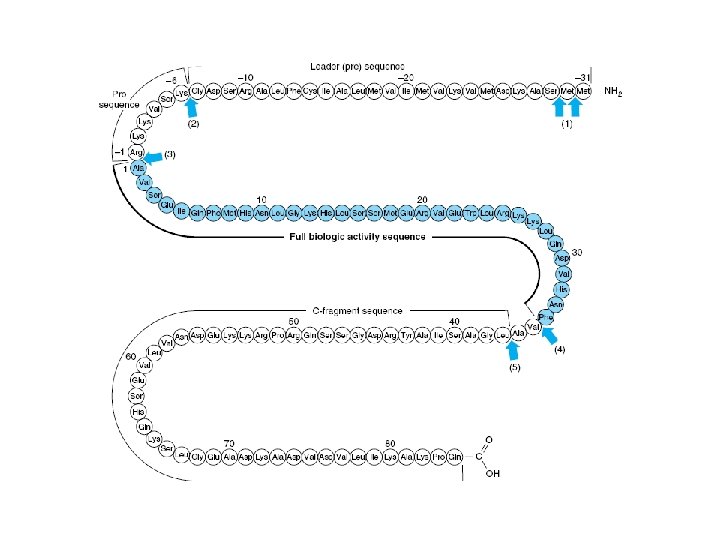

• Regulation of Synthesis – Plasma Ca 2+ • Decrease Ca 2+ – Increase of PTH m. RNA • Effects of Ca 2+ – Gene transcription, m. RNA stability, and m. RNA translation – 1, 25(OH) 2 -D 3 • Regulate the activity of the PTH gene – Inhibits transcription

• Regulation of Metabolism – Most of the newly synthesized PTH is degraded • Regulated by Ca 2+ • Within the gland – Most of the proteolysis • PTH is inactivated by cleavage between residues 36 and 37 • Peripheral metabolism (like above) – Liver and kidneys • Regulation of Secretion – Concentration of ionized calcium • Sensing – G protein-associated Ca 2+ receptor located on parathyroid cells

• Phosphate has no effect on PTH secretion • PTH Acts via a Membrane Receptor – c. AMP • The PTH response system – subject to down-regulation of receptor – and to desensitization • acting directly on kidney and bone • indirectly on the intestinal mucosa – Through stimulation of synthesis of 1. 25(OH)2 -D 3

• • reduces the renal clearance increases the rate of dissolution of bone calcium absorption from the intestine The most rapid changes – Action on the kidney • Largest effect – In bone

• PTH Affects Phosphate Homeostasis – PTH increases renal phosphate clearance • the net effect – Increase the ECF calcium – Decrease phosphate

• Hypoparathyroidism – Insufficient amounts of PTH • Biochemical hallmarks – Decreased serum ionized calcium – Elevated serum phosphate – Usual cause • Removal or damage of the glands – Secondary hypoparathyroidism • Autoimmune destruction – Primary hypoparathyroidism

• Pseudohypoparathyroidism – inherited disorder – active PTH – end-organ resistance – Several types • Partial deficiency of the Gs • Defective step beyond the formation of c. AMP

Hyperparathyroidism • Excessive production of PTH – Adenoma – Parathyroid hyperplasia – Ectopic production PTH or PTH-related peptide • Biochemical hallmarks – Hypercalcemia – Excessive PTH – Depressed phosphate • Secondary hyperparathyroidism – Renal failure (parenchyma loss) • Decreased conversion 25(OH)-D to 1, 25(OH) 2 -D 3 • Hyperplasia of the gland • Hypersecretion of PTH

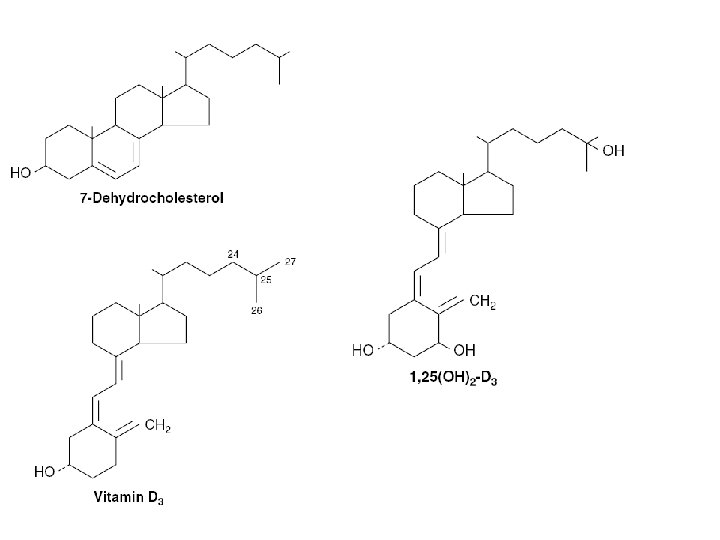
1 , 25(OH)2 -D 3 • Rickets – Deficient mineralization of the skeleton • Osteomalacia – adult equivalent to rickets – failure to mineralize bone • Active vitamin D metabolite – 1, 25(OH)2 -D 3 – Intestinal Absorption of Calcium and Phosphate

1 , 25(OH)2 -D 3 • Vitamin D deficiency – New bone formation slows (along with PTH) – Bone remodeling is also impaired (along with PTH) • augment the actions of PTH – on renal calcium reabsorption

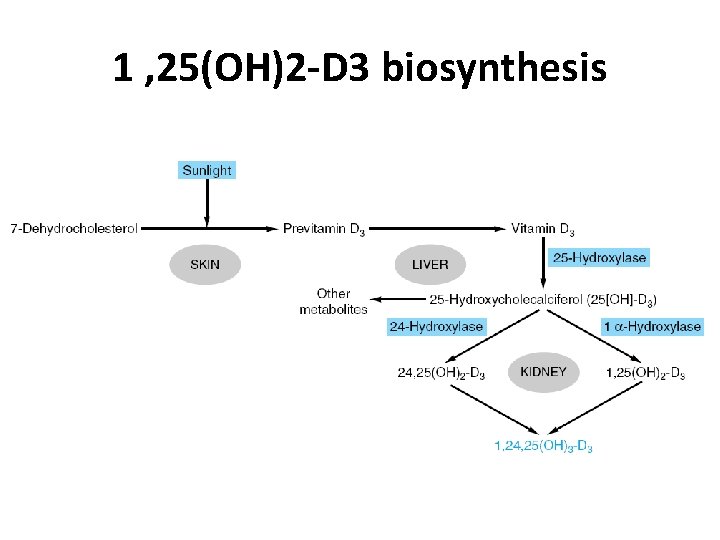
1 , 25(OH)2 -D 3 • Biosynthesis – Skin, liver , kidney • major form of vitamin D found in plasma – 25(OH)-D 3 • the most potent metabolite of vitamin D – 1, 25(OH)2 -D 3 • placenta has a 1 α-hydroxylase – extrarenal source of 1, 25(OH) 2 -D 3
1 , 25(OH)2 -D 3 biosynthesis

1 , 25(OH)2 -D 3 • Regulation of Metabolism and Synthesis – Feedback regulation – Hypocalcemia • increases of 1 α-hydroxylase activity – Requires PTH • hypophos. Phatemia – induce 1α-hydroxylase activity » a weaker stimulus • High levels of 1, 25(OH)2 -D 3 – inhibit renal 1 α-hydroxylase – Stimulate a 24 -hydroxylase » formation of 24, 25(OH)2 -D 3

1 , 25(OH)2 -D 3 • Action – Similar to Steroid Hormones – Localization in the nuclei of • Intestinal villus and crypt cells • Osteoblasts • Distal renal tubular cells

1, 25(OH)2 D 3 • Receptor – member of the steroid receptor family – down-regulates transcription PTH gene – induction of an m. RNA for calcium-binding protein (CBP) – CBP may bind Ca 2+ and protect the mucosa cell against the large fluxes of Ca 2+

Pathophysiology • Rickets – Low plasma calcium and phosphorus – Poorly mineralized bone – vitamin D-dependent rickets • Type I – Defect in the conversion of 25(OH)-D 3 to 1, 25(OH)2 D 3 • Type II – nonfunctional receptor » single amino acid change in one of the zinc fingers • Osteomalacia

Pathophysiology • Osteomalacia – Low plasma calcium and phosphorus • mineralization of osteoid to form bone is impaired – Diseased renal • formation of 1. 25(OH)2 -D 3 is reduced – Calcium absorption decreases » Hypocalcemia • Compensatory increase of PTH • extensive bone turnover • renal osteodystrophy (Associated bone outcomes)

Calcitonin (CT) • 32 -aa peptide • by the parafollicular C cells thyroid • Role – Inhibit PTH & Calcitriol action on bone & kidney • Decrease blood Calcium & Phosphate • Increase urinary excretion
- Slides: 129