Hormones Biochemistry Adrenal cortex Glucocorticoids Mineralocorticoids Androgens Gluconeogenesis

Hormones Biochemistry

Adrenal cortex • Glucocorticoids • Mineralocorticoids • Androgens • Gluconeogenesis • Na+ and K+ balance

• Zona glomerolosa – Mineralocorticoids • Zona fasciculata with zona reticularis – Glucocorticoids androgens • There is an overlap of biologic activity – Commonality of the hormone response elements

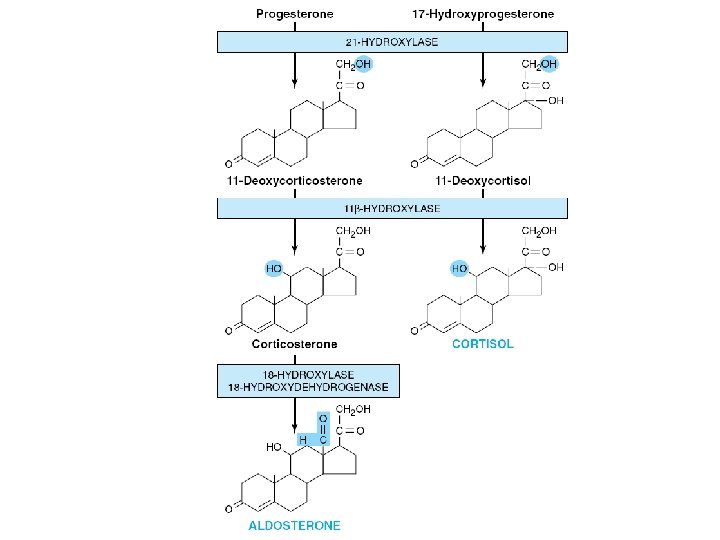
• Glucocorticoids are 21 -carbon steroids – Predominant glucocorticoid • Mineralocorticoids are also 21 –carbon steroids – Primary action • Retention of Na+ and excretion of K+ and H+ – 11 -deoxycorticosterone (DOC) – Aldosterone • Most potent Mineralocorticoid

• Androgens – Dehydroepiandrosterone • Major androgen precursor (DHEA) – Androstenedione – Certain cancers – Certain enzyme deficiencies • Extra-adrenal conversion – Peripheral aromatization • Androgens to estrogen – Postmenopausal women

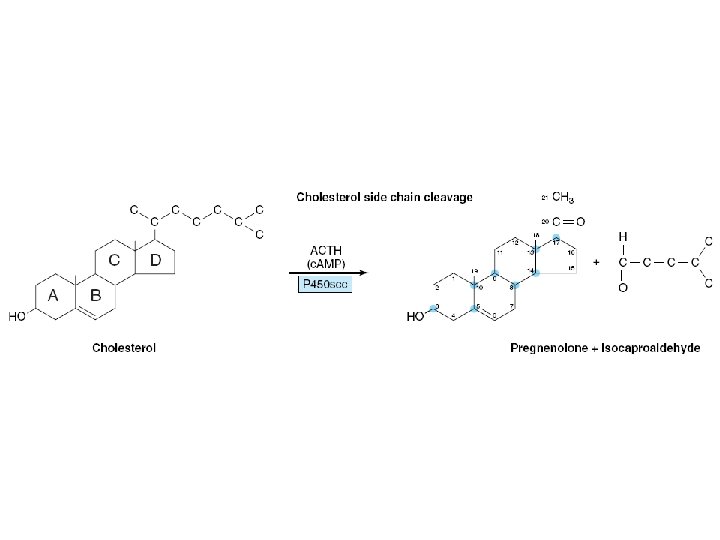
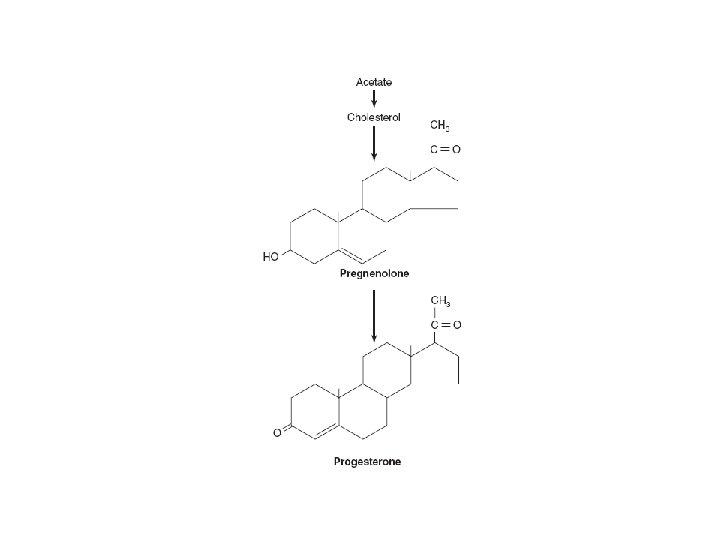
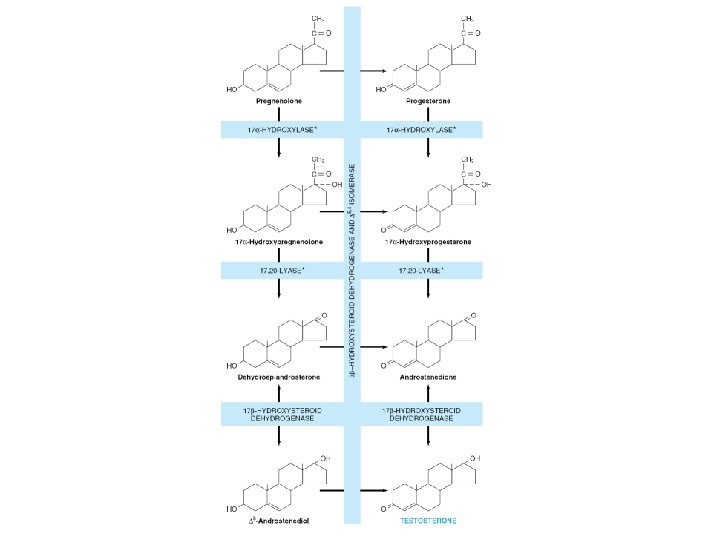
BIOSYNTHESIS OF ADRENAL STEROID HORMONES • From cholesterol • The steroid hormones – NOMENCLATURE • Common – – 17 -carbon cyclopentanoperhydrophenanthrene structure Estrane , 18 carbons Androstane 19 carbons Pregnane, 21 carbons
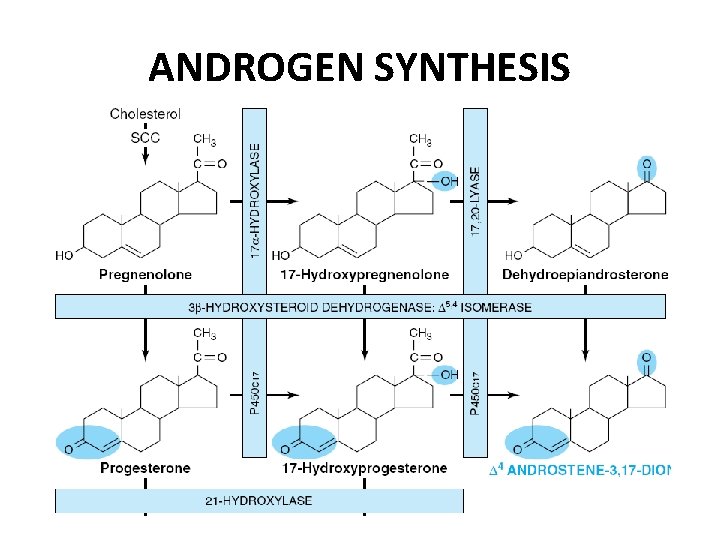
ANDROGEN SYNTHESIS

• Cortisol release occurs with a periodicity • Plasma Transport – Glucocorticoids • • Protein-bound and free forms Corticosteroid-binding globulin (CBG) Albumin Free fraction constitutes about 8% of the total – Mineralocorticoids • No transport protein • Weak association with albumin

Metabolism and Excretion • Glucocorticoids – In plasma • 80% of the 17 -hydroxycorticoids – Cortisol and its metabolites • About half – dihydro- and tetrahydro metabolites • Substantial amounts – Conjugation at the C 3 position with glucuronide » water soluble
Metabolism and Excretion • Excretion – Biliary – Urine (About 70% ) – Feces (About 20% ) – Skin

Metabolism and Excretion • Mineralocorticoids – Aldosterone is very rapidly cleared – tetrahydroaldosterone 3—glucuronide • Androgens – DHEA (sulfate) , androstenedione – Excreted as 17 - keto compounds – Testosterone • androsterone and etiocholanolone

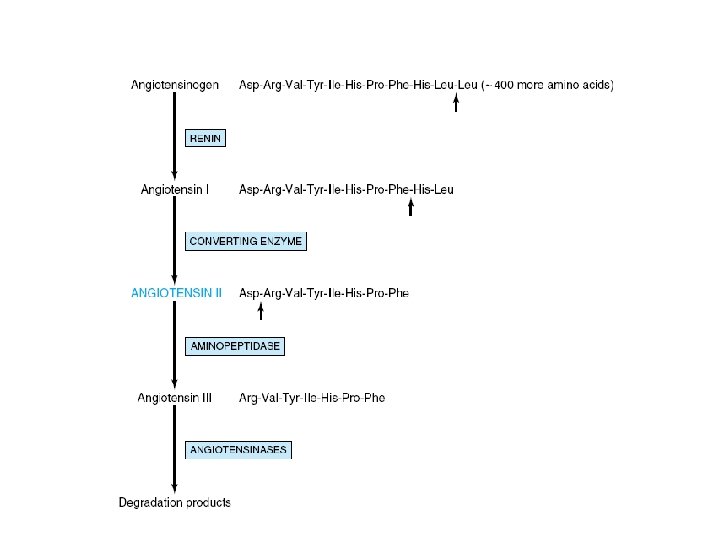
Regulation of synthesis • Glucocorticoid Hormones – Cortisol , ACTH, CRH • feedback loop • Mineralocorticoid Hormones – Primary regulators • renin-angiotensin system – Baroreceptor and salt effects , postural effects • Potassium – Sodium, ACTH, and neural mechanisms

Regulation of synthesis • Angiotensin II – Stimulate the conversion of • Cholesterol to pregnenolone • Corticosterone to 1 8 -hydroxycorticosterone and aldosterone – Mediator • Calcium and of phospholipid metabolites

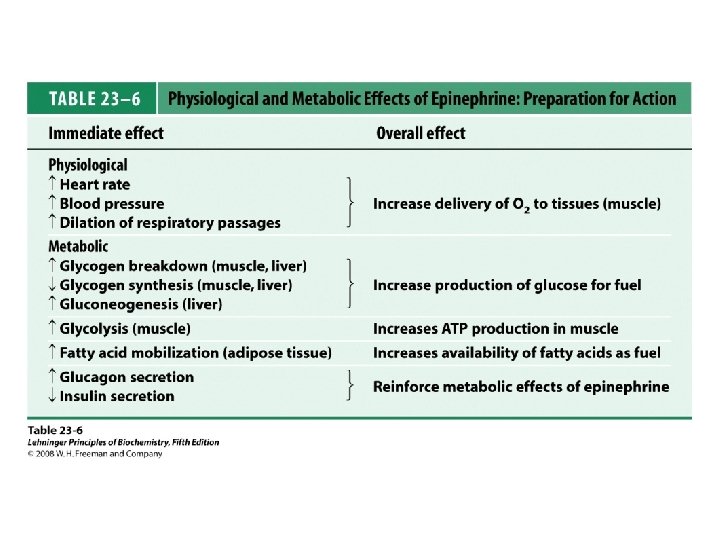
METABOLIC EFFECTS • Increase glucose production – Gluconeogenesis • Delivery of substrate • Amount (and activity) of key enzymes – Glycogenesis • Activation of glycogen synthetase – Lipid metabolism • Promote lipolysis – in extremities • Lipogenesis – face and trunk

METABOLIC EFFECTS • Protein and RNA metabolism – Anabolic effect at physiologic levels • Effects on host defense mechanisms – Suppress the immune response – Suppress the inflammatory response • Decrease circulating leukocytes • Decrease migration to the tissues • Blunt production of the potent inflammatory molecules – Prostaglandins and leukotrienes

METABOLIC EFFECTS • Normal water and electrolyte balance – Restraining ADH release – Increasing angiotensinogen • Maintenance of normal blood pressure and cardiac output

Mineralocorticoid Hormones • Electrolyte Balance and Ion Transport – Na+ retention – Secretion of K+, H+ and NH 4+ • Mineralocorticoid Hormones – Include • Aldosterone (the most potent) • 11 -deoxycorticosterone (DOC) • Cortisol or corticosterone

• The biologic effect depends on – Ability to bind to the receptor (affinity) – Concentration of free hormone

• Glucocorticoid Hormones Regulate Gene Expression – By binding to GRE & by forming HRU • Affect amount of critical proteins – Affect other steps in the “information flow” • Control of the rate of gene transcription • regulate the rate of degradation of specific m. RNAs – Growth hormone – Phosphoenolpyruvate carboxykinase) • Posttranslational processing

Aldosterone action • By protein and RNA synthesis – Ion transport • Target Cells – Kidney – Parotid, and colon and other organs • the effective “free” concentration – Greater than corticosterone or DOC • Presence of 11 β-hydroxy steroid dehydrogenase in target tissue

Aldosterone action • Aldosterone increases – Number of apical membrane Na+ channels – activity of several mitochondrial enzymes • Induction citrate synthase • Intracellular concentration of Na+ • Creates the energy source for removal of this ion through the serosal pump

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • Glucocorticoid Hormone – Insufficiency – Excess • Primary adrenal insufficiency (Addison’s disease) – Hypoglycemia – Low blood pressure – Decreased glomerular filtration rate – Plasma Na+ levels are low – K+ levels are high

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • blood lymphocyte and eosinophil counts are increased • Pigmentation of skin – Exaggerated ACTH • Secondary adrenal insufficiency – Deficiency of ACTH • Tumor, infarction, or infection • Like above , without hyperpigmentation

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • Glucocorticoid excess – Cushing’s syndrome • • • Pharmacologic use steroids ACTH—secreting pituitary adenoma adrenal adenomas ectopic production of ACTH Signs & symptoms – hyperglycemia or glucose intolerance – severe protein catabolic effects » thinning of the skin, muscle wasting » negative nitrogen balance » Osteoporosis

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • Truncal obesity • hypernatremia, hypokalemia, alkalosis, edema, and hypertension

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • Disorders of Mineralocorticoid Excess – Primary aldosteronism (Conn’s syndrome) • Manifestations – hypertension, hypokalemia, hypernatremia, and alkalosis – renin and angiotensin II levels are suppressed – Secondry aldosteronism • Renal artery stenosis – Decrease perfusion pressure » hyperplasia and hyperfunction of the juxtaglomerular cells • elevated levels of renin and angiotensin II

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • Congenital Adrenal Hyperplasia – Enzyme Deficiency • deficiency of end products • accumulation of intermediates • exaggerated production of steroids from alternative pathways – Common Features • develop in utero • deficient cortisol production – ACTH overproduction » adrenal hyperplasia • Overproduction of adrenal androgens – adrenogenital syndrome (alternative designation)

PATHOPHYSIOLOGY OF THE ADRENAL CORTEX • Causes – 21 -hydroxylase deficiency • account for more than 90% of cases – 11β-hydroxylase deficiency • Most of the rest – Other deficiencies • • 3β-hydroxysteroid dehydrogenase 17α-hydroxylase cholesterol desmolase 1 8 - hydroxylase, and 1 8 -dehydrogenase – affect only aldosterone biosynthesis » do not cause adrenal hyperplasia


Hormones of the gonads • The Testes cell types – Leydig cells (also called interstitial cells) • Produce testosterone in response to LH – Sertoli cells • Provide the environment – Seminiferous tubules • Produce Spermatozoa

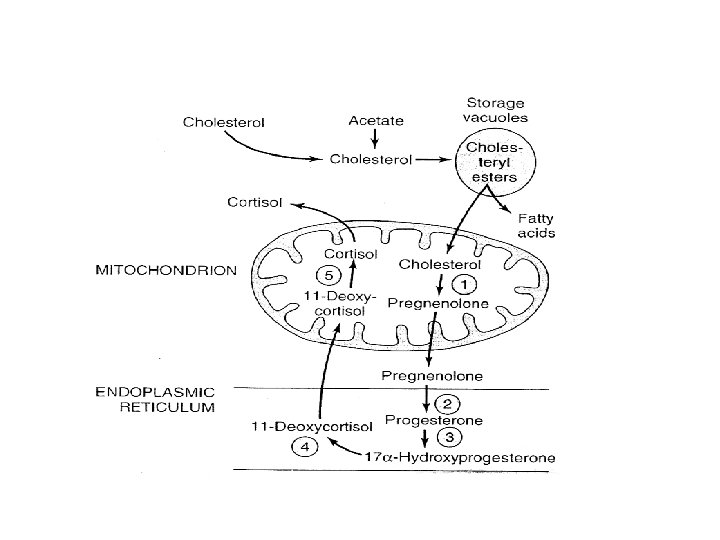
Synthesis of Gonadal Steroids • Critical Steps – Cholesterol Side-Chain Cleavage Enzyme – 3β-Hydroxysteroid Dehydrogenase • Precursor – Cholesterol • Rate—limiting step – Delivery of cholesterol to the mitochondria • by the transport protein (STAR)

Regulation of synthesis • Through a feed back loop – Involves the pituitary and the hypothalamus

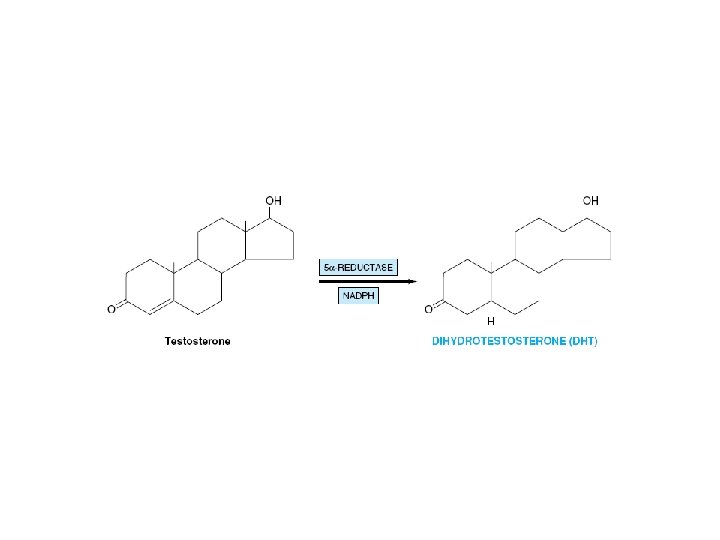
• Dihydrotestosterone (DHT) – Secretion from the testes • 50— 100 µg of DHT per day – Peripheral Conversion • Most DHT • 17β-estradiol (E 2) – By testes – Peripheral aromatization – From • Testosterone • Androstenedione

• The role of E 2 – FSH regulation • Testosterone Binds to a Specific Plasma Protein – Albumin – Sex hormone- binding globulin (SHBG) – Testosterone-estrogen-binding globulin (TEBG) – Production • increased by – Estrogens, certain types of liver disease, hyperthyroidism

• Decreased by – Androgens, hypothyroidism • Free (biologically active) form – 1 -3% • The primary function of SHBG – to restrict the free concentration • Increase of SHBG – aging, cirrhosis, and hyperthyroidism – Increased free E 2: testosteronc ratio • Estrogenization – Gynecomastia

• The major steroid secreted by the adult testes – Testosterone • Released as it is produced • Testosterone is metabolized by two pathways – Oxidation at the 17 -position • Produces 17 -ketosteroids – Reduction of the A ring • Double bond • The 3 -ketone • Produces DHT (target tissues) • Most significant metabolic product

• Active form of the hormone in – Prostate, external genitalia, and some areas of the skin – Plasma content of DHT • One-tenth that of testosterone • 400 µg of DHT is produced daily • 5 mg of testosterone produced daily

• Testosterone is a prohormone – Converted into • Dihydrotestosterone • Estradiol – in the brain » determine the sexual behavior • 17 -ketosteroid metabolites – Androsterone – Etiocholanolone – Conjugated with glucuronide and sulfate

• Testosterone is Converted into • Dihydrotestosterone • Estradiol • 17 -ketosteroid metabolites

Regulation of Testicular Steroidogenesis • Steroidogenesis Is Stimulated by LH – Leydig cells • • Membrane receptors Adenylyl cyclase c. AMP Cholesterol transport by STAR Side chain cleavage by P 450 scc Testosterone Feedback control Inhibition of Gn. RH release

Regulation of Testicular Steroidogenesis • FSH – Sertoli cells – Synthesis of androgen-binding protein (ABP) – Provides high concentration of testosterone to support spermatogenesis • FSH androgens – Sertoli cells • Produce inhibin – Negative feedback » Regulate FSH secretion

Androgens function • Major Androgens – Testosterone and DHT • Target cells for DHT – 5α-reductase activity • Prostate external genitalia, and genital skin • Targets for testosterone – Wolffian structures, spermatogonia, muscles, bone, kidney, and brain

Mechanism of action • Testosterone or DHT ? – Affinity difference (for receptor) – ability of a target tissue to form DHT from testosterone • Nuclear localization – Binding of the receptor-steroid complex to chromatin • Androgen response element • activates specific genes • Increased accumulation of total cellular RNA, including m. RNA, t. RNA, and r. RNA – Effects
• Enlargement of the kidney – a major target tissue – Synthesis of a number of enzymes • Stimulate the replication of cells – Benign prostatic hypertrophy • By Testosterone or DHT, in combination with E 2

Pathophysiology • Hypogonadism – Lack of testosterone synthesis – Primary hypogonadism • Testicular failure – 5α-reductase deficiency – Secondary hypogonadism • Defective secretion of the gonadotropins – No testosterone/DHT receptor – Abnormal receptor • Genetic males, phenotypic females


THE OVARIES • FEMALE SEX HORMONES • GERM CELLS • Estrogens are a family of hormones – 17β-Estradiol • Primary estrogen of ovarian origin – Estrone • Postmenopausal women – Estriol • In pregnancy • Form By – Aromatization of androgens
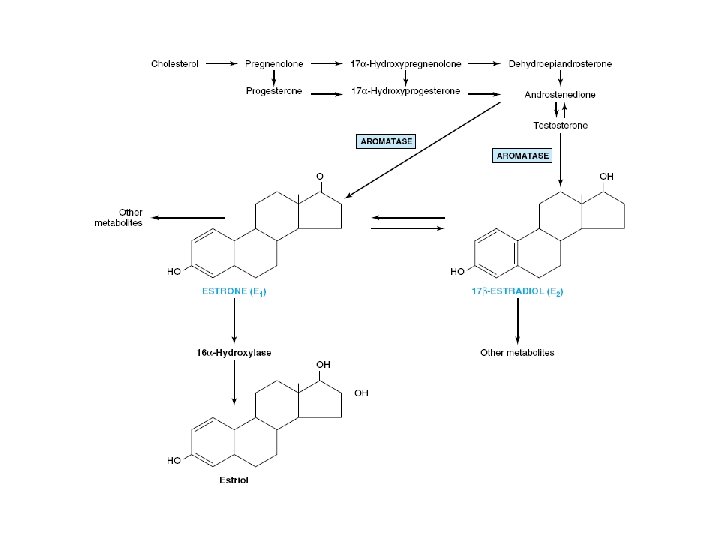

Biosynthesis of Ovarian Hormones • Theca cells – Source of androstenedione and testosterone • Granulosa cells – Estrone and estradiol • Corpus luteum – Progesterone, some estradiol • Peripheral aromatization of androgens – 80% Estradiol produced in males – 50% E 2 produced during pregnancy – In postmenopausal women

Plasma Transport • • Estrogens are bound to SHBG Progestins to CBG Provide a circulating reservoir of hormone Buffer against sudden changes

Approximate affinities of steroids for serum-binding proteins

Metabolism • By the Liver – Estradiol and estrone to estriol – Addition of glucuronide or sulfate • water-soluble • Do not bind to transport proteins • Excreted readily – In bile, feces, and urine • Progestin metabolite – Sodium pregnanediol-20 -giucuronide – actively metabolized • Ineffective when given orally

function of the ovarian hormones • Estrogens – Stimulate the development of tissues involved in reproduction • Stimulate the size and number of cells – – – Synthesis of protein, r. RNA, t. RNA, m. RNA, and DNA Uterine endometrium proliferates glands hypertrophy and elongate Myometrium develops an intrinsic, rhythmic motility breast ducts proliferate – Anabolic effects on bone and cartilage – Peripheral blood vessels • vasodilation and heat dissipation

function of the ovarian hormones • Progestins – Uterine epithelium • from proliferative to secretory – increased glycogen content – Preparing the uterine epithelium for implantation of the fertilized ovum – Breast glands • Development of the acinar portions – Decrease peripheral blood flow • Decreasing heat loss

• FSH – The main stimulus for estrogen secretion – Stimulates a follicle to ripen • The Menstrual Cycle – Results from complex interactions between the • Hypothalamus • Pituitary • Ovary – 25 and 35 days in length – Divided into • follicular phase • Luteal phase • Menstruation

Follicular Phase • Progesterone levels are very low • The LH peak heralds the end of the follicular phase

Luteal Phase • The major hormone of the luteal phase – Progesterone • Preparation and maintenance of the secretory endornetrium – Provides early nourishment for the implanted blastocyst


• Human Chorionic Gonadotropin (h. CG) – Support the corpus luteum – Peak h. CG • Reached in the middle of the first trimester – Major source of progesterone • Corpus luteum – first 6— 8 weeks of the pregnancy • Placenta • Estrogens – Gradually increase throughout pregnancy – Estriol is produced in the largest amount

Placental Hormones • • Human Chorionic Gonadotropin (h. CG) Progestins Estrogens Placental Lactogens – Biologic properties of prolactin and growth hormone

Parturition • Oxytocin – Stimulates uterine contractility – Used to facilitate delivery • Estrogen • Increase the number of Oxytocin receptors • Catecholarnines – Induction of labor • Progesterone and estriol – Decline rapidly after the placenta is delivered

Mammary Gland Development • Estrogens – Ductal growth • Progestins – Alveolar proliferation – Inhibits milk production and secretion in late pregnancy – Progesterone • Decrease abruptly after delivery

Mammary Gland Development • Prolactin – Increases during pregnancy – Fall rapidly postpartum • Suckling • release of oxytocin • release of prolactin

Menopause • Cease having regular menstrual cycles • loss of all follicles and ovarian estrogen production • No alternative source of progesterone • estrone, are produced by the peripheral aromatization • Increases of LH and FSH • Health problem – Osteoporosis

Synthetic Agonists and Antagonists • Estrogens – Antagonists • act by competing with estradiol • Breast cancer – Nafoxidine and tamoxifen » Form very stable complexes with the estrogen receptor • Progestins – 17α-alkyl-substituted I 9 -nortestosterone derivatives

• There is cross-talk between the sex hormone receptors – Progesterone binds to the androgen receptor • Weak androgen – Androgens bind to the estrogen receptor – Sequences of the hormone response elements are similar

• Estrogens – Increase estrogen & progesterone receptor • Progesterone – Enhance the rate of turnover of its receptor

PATHOPHYSIOLOGY • Hypogonadism – Decreased hormone production – Primary • Directly involve the ovaries – Secondary • Loss of pituitary gonadotropin function • Polycytic ovary syndrome – Overproduction of androgens

PATHOPHYSIOLOGY • Leydig cell tumors and arrhenoblastoma – Produce testosterone • Granulosa-theca cell tumors – Produce estrogens • Intraovarian adrenal rests – Produce cortisol • Hydatidiform mole, choriocarcinoma – enormous quantities of h. CG

• Estradiol is the main hormone produced during the follicular phase • Progesterone – Major hormone involved in sustaining pregnancy – Made in • Corpus luteum – Under the influence of pituitary LH – and later by h. CG • The placenta – After the second month of pregnancy


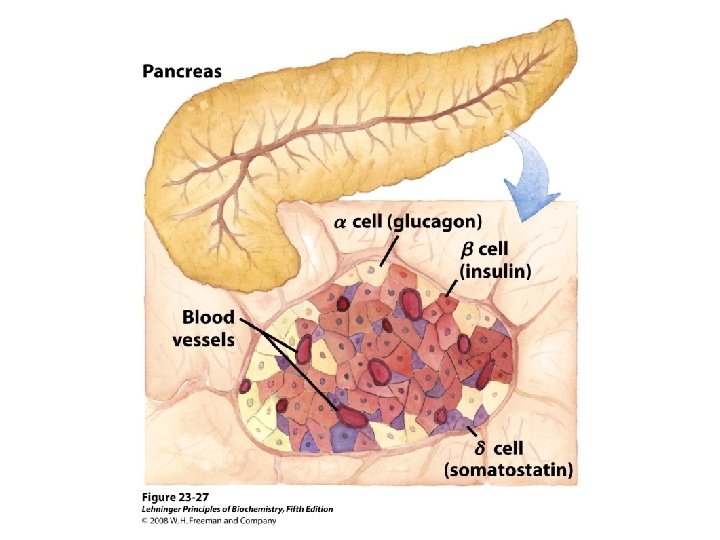
Hormones of pancrease • Hormones of Islets of Langerhans • Insulin and glucagon – Primary Target • Liver – Primary action • Regulating carbohydrate metabolism • Somatostatin – Local regulation of insulin and glucagon secretion • Pancreatic polypeptide – affects gastrointestinal secretion

Clinical significance • Diabetes mellitus – insufficient action of insulin • Its absence • resistance to its action – Glucagon • Aggravates this condition

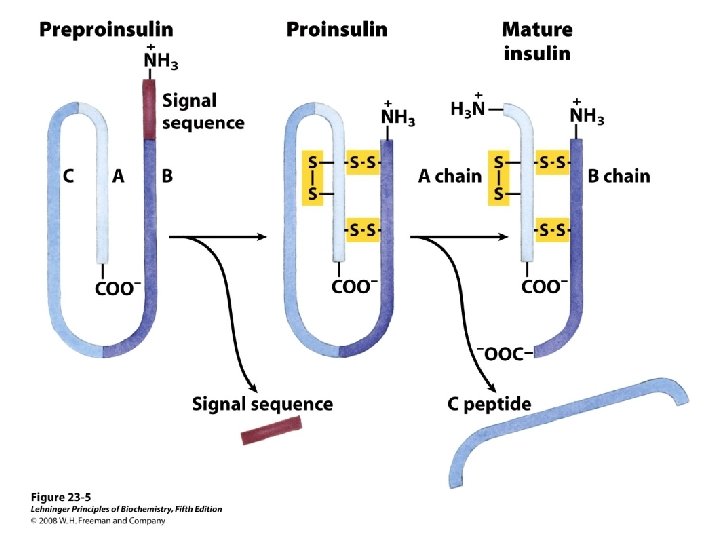
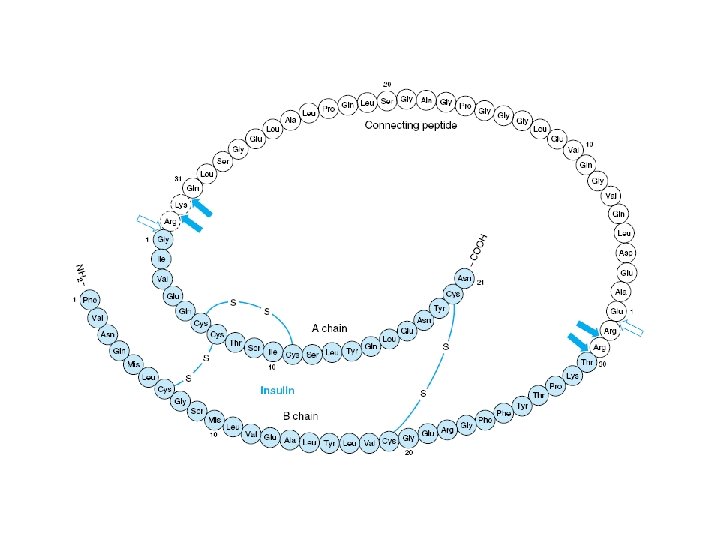
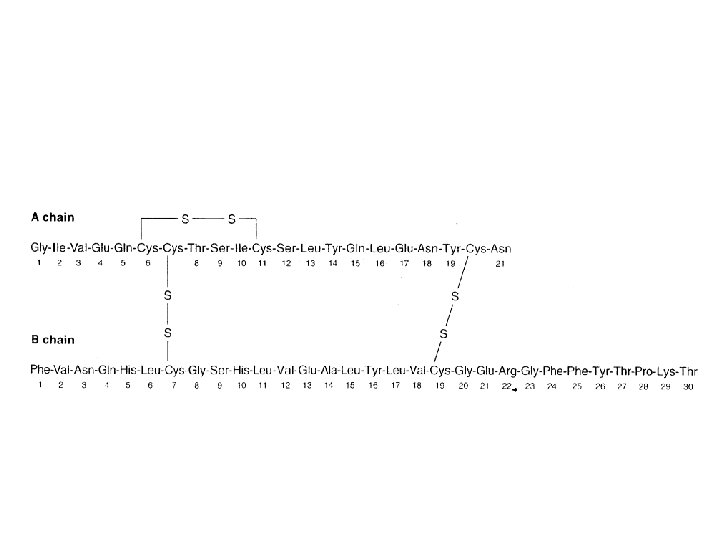
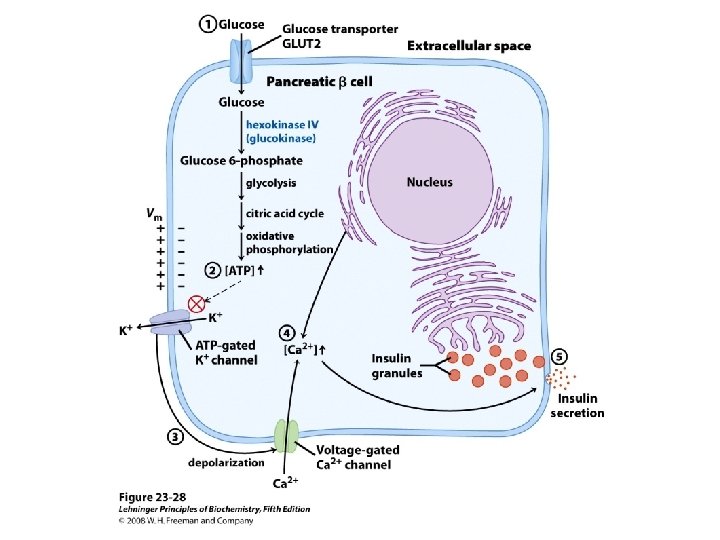

Regulation of insulin secretion • Hormonal Factors – α-Adrenergic agonists • Principally epinephrine, inhibit insulin release – β- Adrenergic agonists • Stimulate insulin release – Chronic exposure to excessive levels of • Growth hormone, Cortisol, placental lactogen, estrogens, and progestins – Stimulate insulin release • Pharmacologic Agents – Sulfonylurea Compounds • stimulate insulin secretion
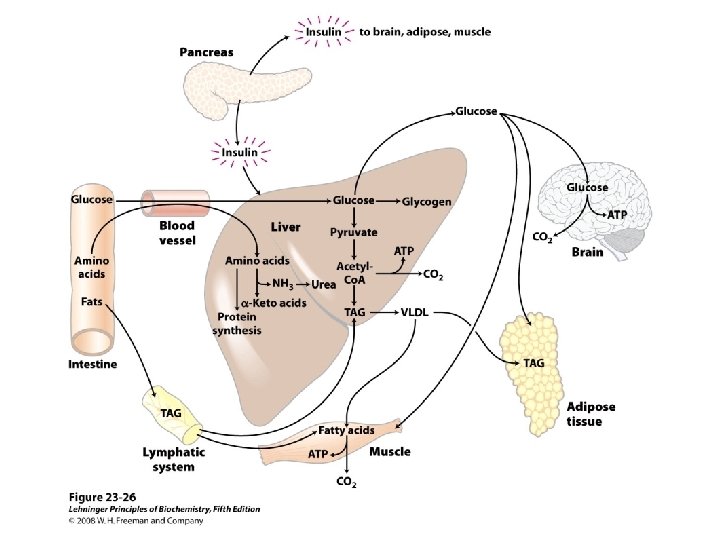
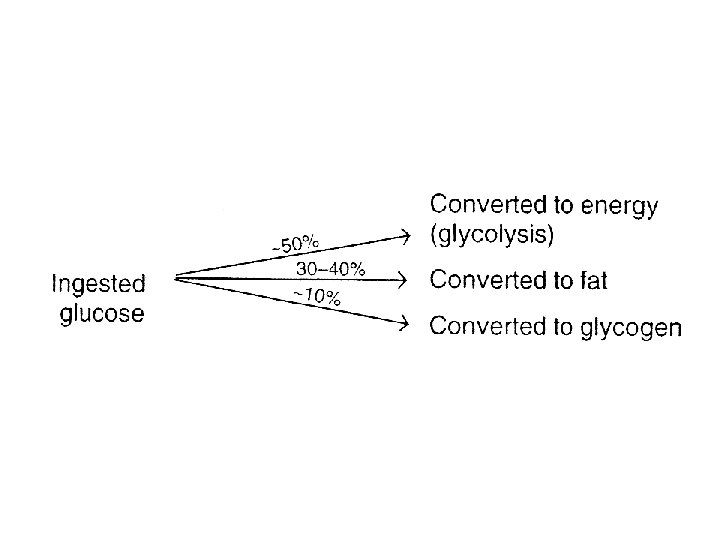
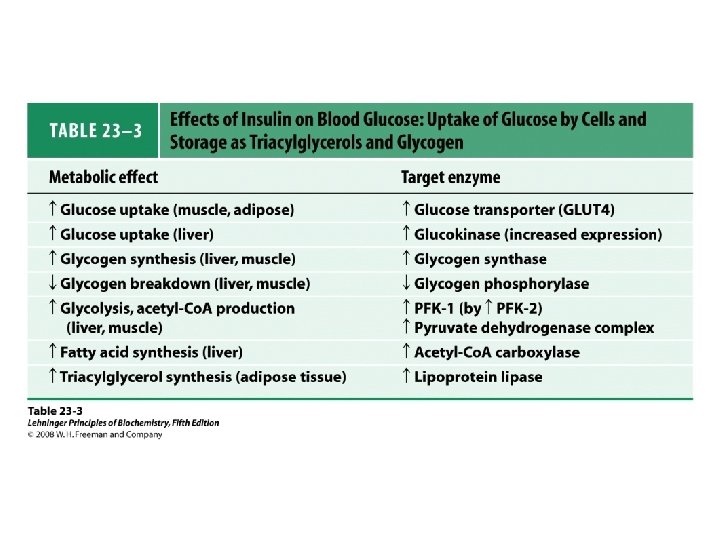

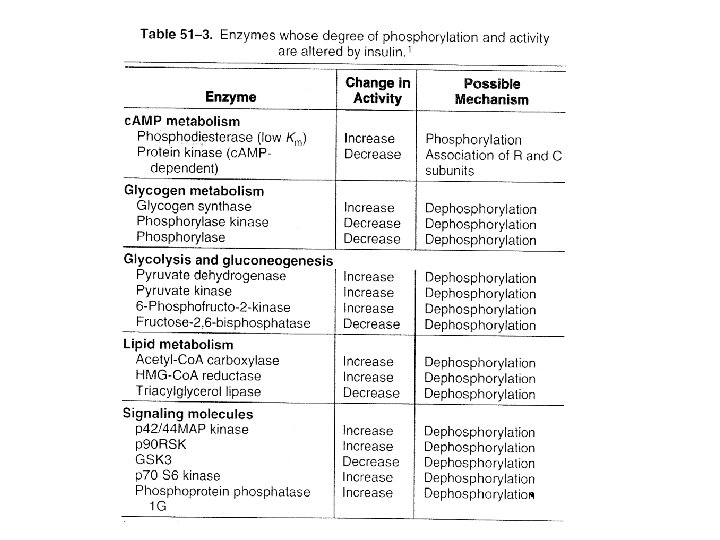
• Insulin promotes – entry of amino acids into cells – the movement of K+, Ca 2+, nucleosides, and inorganic phosphate
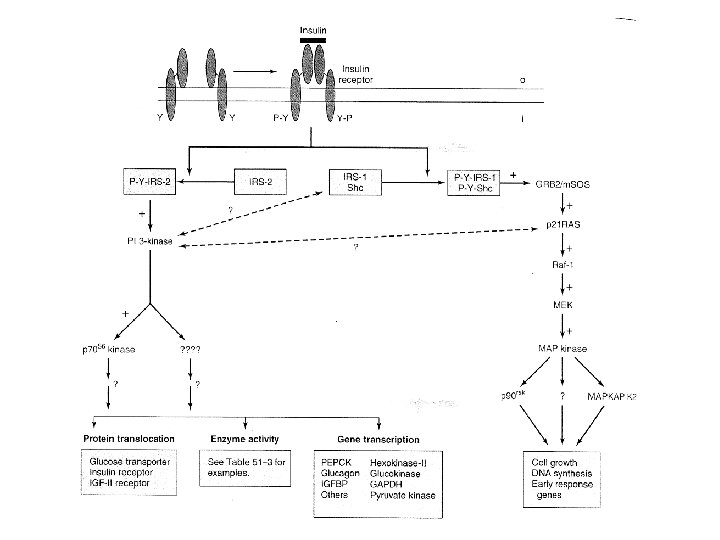
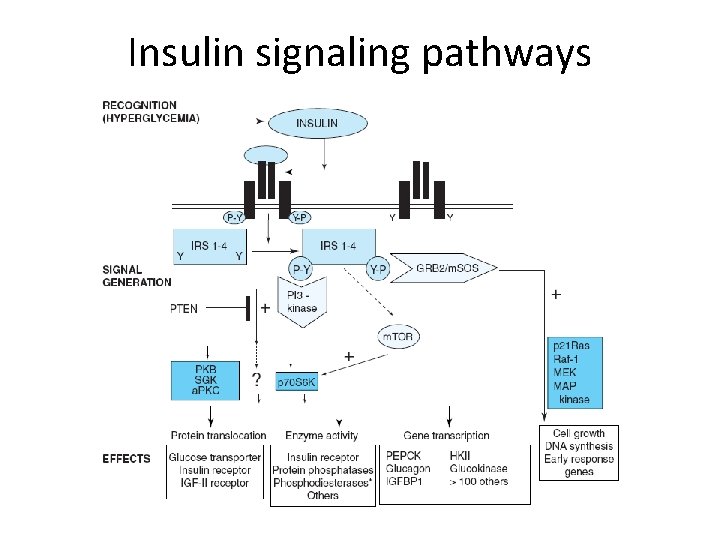
Insulin signaling pathways
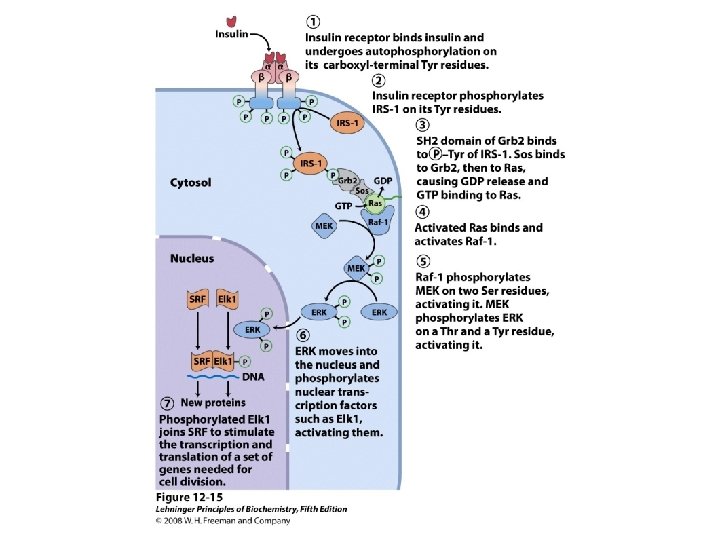
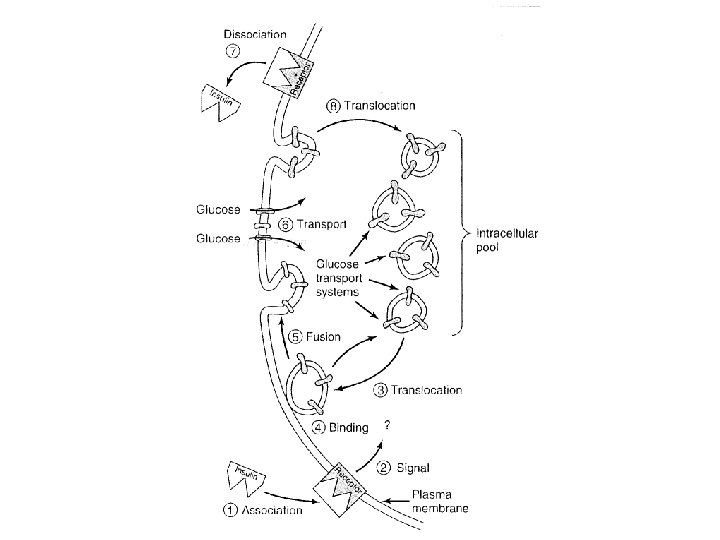

Metabolism of insulin • Plasma half-life is less than 3— 5 minutes • Liver, kidneys, and placenta • Mechanisms – Insulin-specific protease – Glutathione-insulin transhydrogenase
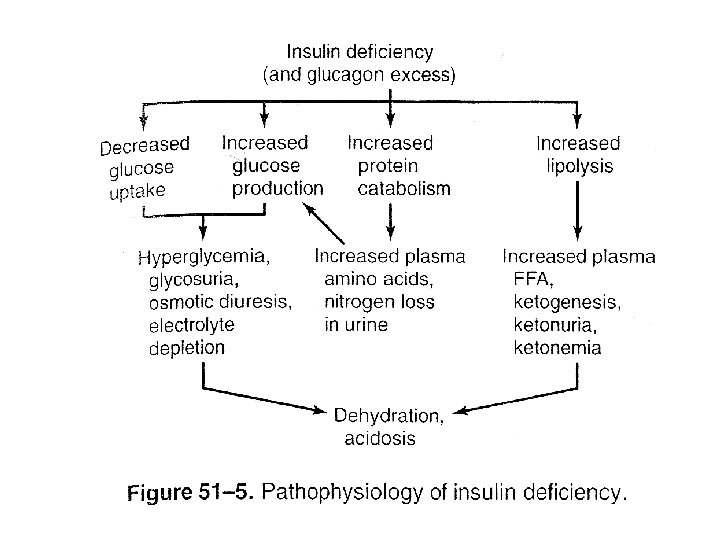

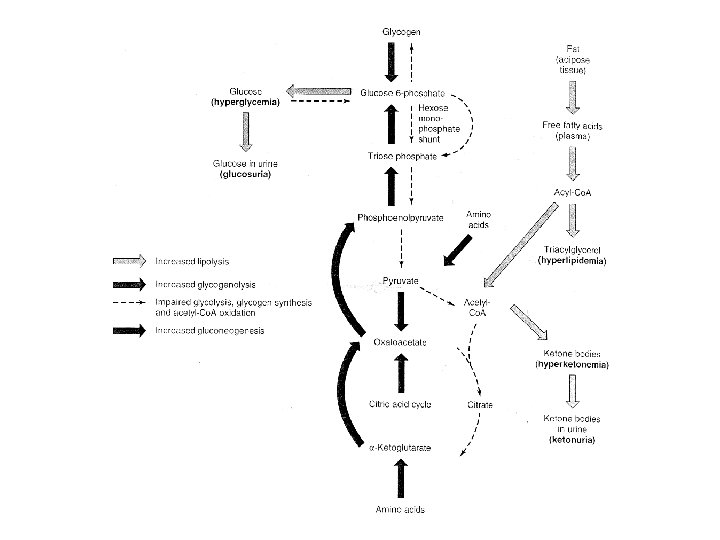
• Insulin deficiency – Accelerated lipolysis • Hyperlipidemia • Ketonuria – Glycolysis is inhibited – Accelerated glycogenolysis • Glucose release – Accelerated gluconeogenesis – Increased availability of amino acids – Increased amount of PEPCK – Glucose production


Pathophysiology • Diabetes Mellitus – Insulin deficiency – Resistance to the action of insulin • Non-insu. Iindependent(type 2) diabetes mellitus (NIDDM) – 90% of diabetes – Usually obese – Elevated plasma insulin – Down-regulated insulin receptors

Pathophysiology • Insulin-dependent (type 1) diabetes mellitus (IDDM) – 10% of diabetes • Antibodies against insulin receptors • Insulin resistance • Hyperinsulinism – Tumors of B cell – Severe hypoglycemia

Pathophysiology • Leprechaunism – Insulin resistance – Elevated biologically active insulin – Lack insulin receptors – Or defective receptors


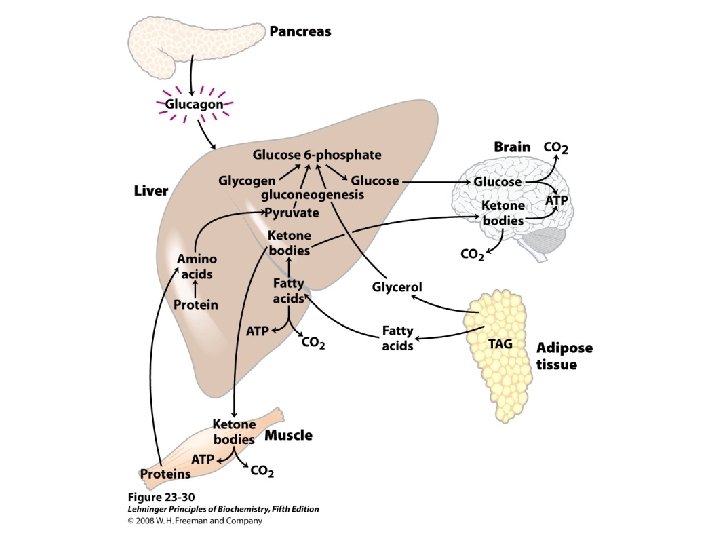
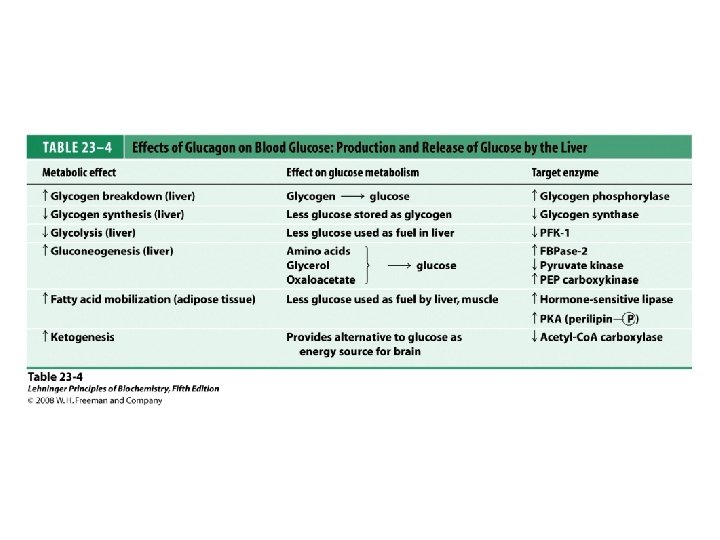
Glucagon is an Insulin Antagonist • Synthesis – as a Precursor Molecule • Proglucagon precursor – 29 amino acids • Half-life – about 5 minutes • Glucagon is inactivated by the liver – In portal vein is much higher

Glucagon • Secretion of Glucagon – Glucose • Inhibition – Other substances • • Amino acids Fatty acids and ketones Gastrointestinal tract hormones Neurotransmitters

Glucagon Actions • Oppose Those of Insulin – Rapid mobilization of potential energy sources • Glycogenolysis • Lipolysis



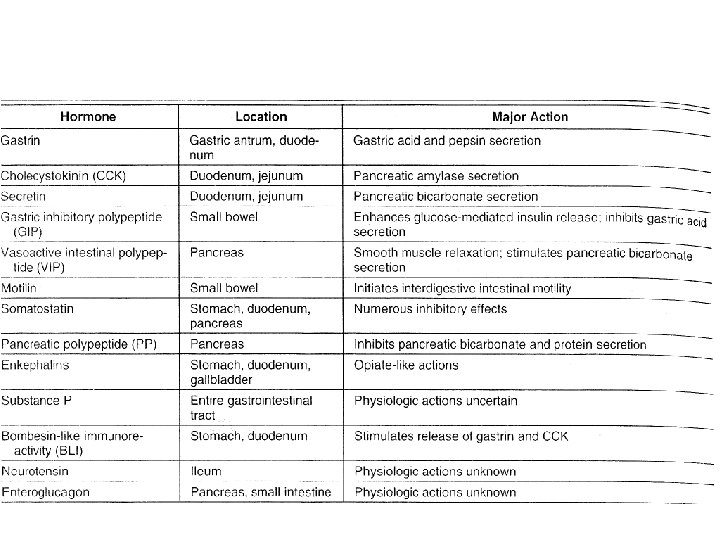
Gastrointestinal Hormones • Families – Gastrin family • Gastrin and cholecystokinin (CCK) – Secretin family • Secretin, glucagon, gastric inhibitory polypeptide (GIP), vasoactive intestinal polypeptide (VIP), and glicentin • The neurocrine peptides • Neurotensin, bombesin-like peptides, substance P, and somatostatin • No structural similarity to any other gastrointestinal peptide • Have very short plasma half-lives

Gastrointestinal Hormones • Mechanism of Action – Little Is Known


Catecholamine hormones • Adaptation to acute and chronic stress • Dopamine • Norepinephrine • Epinephrine • Synthesis – The adrenal medulla – Chromaffin cells – From Tyrosine
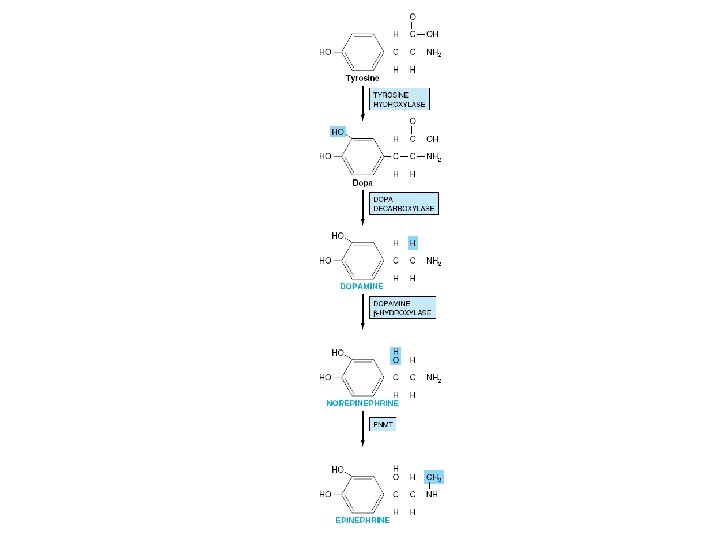
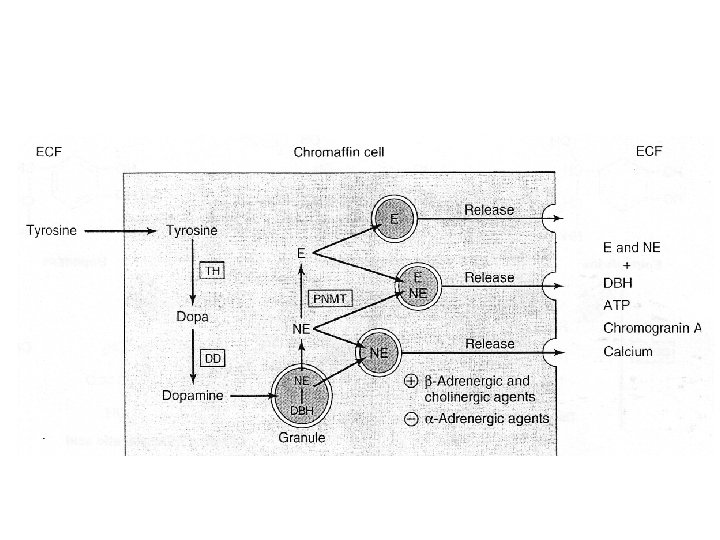

Regulation of catecholamine synthesis • Physiologic stress – Hypothalamus and brain stem • Stimulation of the splanchnic nerve – Adrenal medulla » increased synthesis & release of catecholamines • Tyrosme hydroxylase • Phosphorylation (c. AMP-dependent) • Induction (increased amount) • DBH (Induction) • PNMT(induction by glucocorticoids)

Regulation of catecholamine synthesis • Tyrosine hydroxylase – Feedback inhibition by the catecholamines

Mechanism of action • The catecholamines act through two major classes of receptors – α-adrenergic • α 1 , α 2 – β-adrenergic • β 1 and β 2 • Epinephrine – Binds to and activates both α and β receptors • Norepinephrine – Primarily binds to α receptors
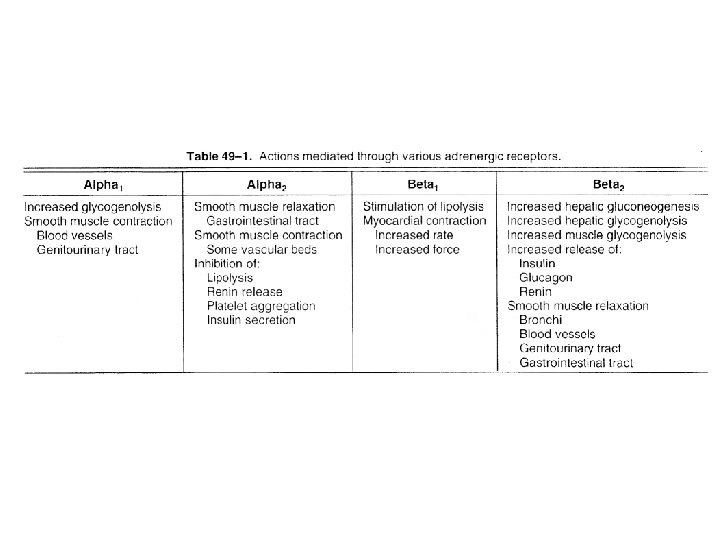

Mechanism of action • Receptors Coupled to the Adenylyl Cyclase System • β 1 and β 2 – Activate Adenylyl Cyclase • α 2 – Inhibit Adenylyl Cyclase • α 1 – alter Intracellular calcium concentrations – or modify phosphatidylinositide metabolism

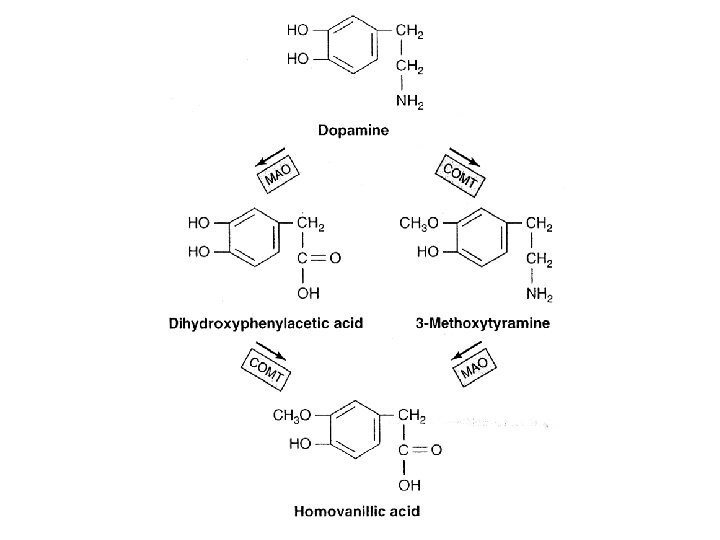
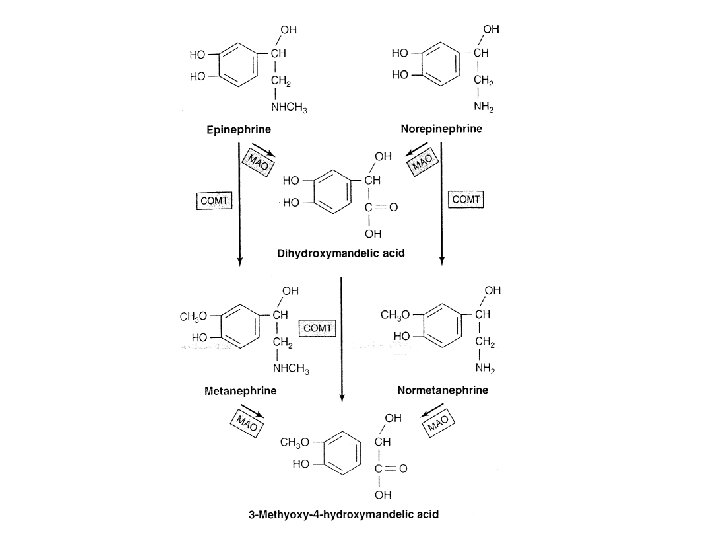
Metabolism of catecholamines • Catecholamines – Loose association with albumin – Extremely short biologic half-life (10— 30 seconds). • Catechol-O-methyltransferase – O-Methoxylated derivatives • Homovanillic acid • Normetanephrine • Metanephrine – Conjugation with glucuronic or sulfuric acid • Monoamine oxidase – MAO inhibitors • Treat hypertension and depression

Metabolism of catecholamines • Diagnostic significance (pheochromocytoma) • Metanephrines – Methoxy derivatives of epinephrine and norepinephrine • Vanillylmandelic acid (VMA) • o-methylated deaminated

Pheochromocytoma • Tumors of the adrenal medulla – Hypertension syndrome – Norepinephrine / epinephrine is often increased
- Slides: 134