Hormone Calcitonin Calcium and Phosphate Metabolism Vitamin D

Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth Dr. Noori M Luaibi

Hormone, Calcitonin, Calcium and Phosphate Metabolism, Vitamin D, Bone, and Teeth The physiology of calcium and phosphate metabolism, formation of bone and teeth, and regulation of vitamin D, parathyroid hormone (PTH), and calcitonin are all closely intertwined. Extracellular calcium ion concentration, for example, is determined by the interplay of calcium absorption from the intestine, renal excretion of calcium, and bone uptake and release of calcium, each of which is regulated by the hormones just noted. Because phosphate homeostasis and calcium homeostasis are closely associated.

Calcium and Phosphate Regulation in the Extracellular Fluid and Plasma calcium. Approximately 85 per cent of the body’s phosphate is stored in bones, 14 to 15 per cent is in the cells, and less than 1 per cent is in the extracel. Extracellular fluid calcium concentration normally is regulated very precisely, seldom rising or falling more than a few per cent from the normal value of about 9. 4 mg/dl, which is equivalent to 2. 4 mmol calcium per liter. This precise control is essential, because calcium plays a key role in many physiologic processes, including contraction of skeletal, cardiac, and smooth muscles; blood clotting; and transmission of nerve impulses, to name just a few. Excitable cells, such as neurons, are very sensitive to changes in calcium ion concentrations, and increases in calcium ion concentration above normal (hypercalcemia) cause progressive depression of the nervous system; conversely, decreases in calcium concentration (hypocalcemia) cause the nervous system to become more excited. An important feature of extracellular calcium regulation is that only about 0. 1 per cent of the total body calcium is in the extracellular fluid, about 1 per cent is in the cells, and the rest is stored in bones. Therefore, the bones can serve as large reservoirs, releasing calcium when extracellular fluid concentration decreases and storing excess lular fluid. Although extracellular fluid phosphate concentration is not nearly as well regulated as calcium concentration, phosphate serves several important functions and is controlled by many of the same factors that regulate calcium.

Calcium in the Plasma and Interstitial Fluid The calcium in the plasma is present in three forms, as shown in Figure 79– 1. (1) About 41 per cent (1 mmol/L) of the calcium is combined with the plasma proteins and in this form is nondiffusible through the capillary membrane. (2) About 9 per cent of the calcium (0. 2 mmol/L) is diffusible through the capillary membrane but is combined with anionic substances of the plasma and interstitial fluids (citrate and phosphate, for instance) in such a manner that it is not ionized. (3) The remaining 50 per cent of the calcium in the plasma is both diffusible through the capillary membrane and ionized. Thus, the plasma and interstitial fluids have a normal calcium ion concentration of about 1. 2 mmol/L (or 2. 4 m. Eq/L, because it is a divalent ion), a level only one half the total plasma calcium concentration. This ionic calcium is the form that is important for most functions of calcium in the body, including the effect of calcium on the heart, the nervous system, and bone formation.


Inorganic Phosphate in the Extracellular Fluids Inorganic phosphate in the plasma is mainly in two forms: HPO 4 - and H 2 PO 4 -. The concentration of HPO 4 - is about 1. 05 mmol/L, and the concentration of H 2 PO 4 - is about 0. 26 mmol/L. When the total quantity of phosphate in the extracellular fluid rises, so does the quantity of each of these two types of phosphate ions. Furthermore, when the p. H of the extracellular fluid becomes more acidic, there is a relative increase in H 2 PO 4 _ and a decrease in HPO 4 -, whereas the opposite occurs when the extracellular fluid becomes alkaline. Because it is difficult to determine chemically the exact quantities of HPO 4 - and H 2 PO 4 - in the blood, ordinarily the total quantity of phosphate is expressed in terms of milligrams of phosphorus per deciliter (100 ml) of blood. The average total quantity of inorganic phosphorus represented by both phosphate ions is about 4 mg/dl, varying between normal limits of 3 to 4 mg/dl in adults and 4 to 5 mg/dl in children. Non-Bone Physiologic Effects of Altered Calcium and Phosphate Concentrations in the Body Fluids Changing the level of phosphate in the extracellular fluid from far below normal to two to three times normal does not cause major immediate effects on the body. In contrast, even slight increases or decreases of calcium ion in the extracellular fluid can cause extreme immediate physiologic effects. In addition, chronic hypocalcemia or hypophosphatemia greatly decreases bone mineralization.

Hypocalcemia Causes Nervous System Excitement and Tetany. When the extracellular fluid concentration of calcium ions falls below normal, the nervous system becomes progressively more excitable, because this causes increased neuronal membrane permeability to sodium ions, allowing easy initiation of action potentials. At plasma calcium ion concentrations about 50 per cent below normal, the peripheral nerve fibers become so excitable that they begin to discharge spontaneously, initiating trains of nerve impulses that pass to the peripheral skeletal muscles to elicit tetanic muscle contraction. Consequently, hypocalcemia causes tetany. It also occasionally causes seizures because of its action of increasing excitability in the brain. Figure 79– 2 shows tetany in the hand, which usually occurs before tetany develops in most other parts of the body. This is called “carpopedal spasm. ” Tetany ordinarily occurs when the blood concentration of calcium falls from its normal level of 9. 4 mg/dl to about 6 mg/dl, which is only 35 per cent below the normal calcium concentration, and it is usually lethal at about 4 mg/dl. In laboratory animals, in which calcium can gradually be reduced beyond the usual lethal levels, very extreme hypocalcemia can cause other effects that are seldom evident in patients, such as marked dilatation of the heart, changes in cellular enzyme activities, increased membrane permeability in some cells (in addition to nerve cells), and impaired blood clotting.


Hypercalcemia Depresses Nervous System and Muscle Activity. When the level of calcium in the body fluids rises above normal, the nervous system becomes depressed and reflex activities of the central nervous system are sluggish. Also, increased calcium ion concentration decreases the QT interval of the heart and causes lack of appetite and constipation, probably because of depressed contractility of the muscle walls of the gastrointestinal tract. These depressive effects begin to appear when the blood level of calcium rises above about 12 mg/dl, and they can become marked as the calcium level rises above 15 mg/dl. When the level of calcium rises above about 17 mg/dl in the blood, calcium phosphate crystals are likely to precipitate throughout the body; this condition is discussed later in connection with parathyroid poisoning. Absorption and Excretion of Calcium and Phosphate Intestinal Absorption and Fecal Excretion of Calcium and Phosphate. The usual rates of intake are about 1000 mg/day each for calcium and phosphorus, about the amounts in 1 liter of milk. Normally, divalent cations such as calcium ions are poorly absorbed from the intestines. However, as discussed later, vitamin D promotes calcium absorption by the intestines, and about 35 per cent (350 mg/day) of the ingested calcium is usually absorbed; the calcium remaining in the intestine is excreted in the feces. An additional 250 mg/day of calcium enters the intestines via secreted gastrointestinal juices and sloughed mucosal cells. Thus, about 90 per cent (900 mg/day) of the daily intake of calcium is excreted in the feces (Figure 79– 3). Intestinal absorption of phosphate occurs very easily. Except for the portion of phosphate that is excreted in the feces in combination with nonabsorbed calcium, almost all the dietary phosphate is absorbed into the blood from the gut and later excreted in the urine.


Renal Excretion of Calcium and Phosphate Approximately 10 per cent (100 mg/day) of the ingested calcium is excreted in the urine. About 41 per cent of the plasma calcium is bound to plasma proteins and is therefore not filtered by the glomerular capillaries. The rest is combined with anions such as phosphate (9 per cent) or ionized (50 per cent) and is filtered through the glomeruli into the renal tubules. Normally, the renal tubules reabsorb 99 per cent of the filtered calcium, and about 100 mg/day is excreted in the urine. Approximately 90 per cent of the calcium in the glomerular filtrate is reabsorbed in the proximal tubules, loops of Henle, and early distal tubules. Then in the late distal tubules and early collecting ducts, reabsorption of the remaining 10 per cent is very selective, depending on the calcium ion concentration in the blood. When calcium concentration is low, this reabsorption is great, so that almost no calcium is lost in the urine. Conversely, even a minute increase in blood calcium ion concentration above normal increases calcium excretion markedly. We shall see later in the chapter that the most important factor controlling this reabsorption of calcium in the distal portions of the nephron, and therefore controlling the rate of calcium excretion, is PTH. Renal phosphate excretion is controlled by an overflow mechanism, when phosphate concentration in the plasma is below the critical value of about 1 mmol/L, all the phosphate in the glomerular filtrate is reabsorbed and no phosphate is lost in the urine. But above this critical concentration, the rate of phosphate loss is directly proportional to the additional increase. Thus, the kidneys regulate the phosphate concentration in the extracellular fluid by altering the rate of phosphate excretion in accordance with the plasma phosphate concentration and the rate of phosphate filtration by the kidneys. However, as discussed later in the chapter, PTH can greatly increase phosphate excretion by the kidneys, thereby playing an important role in the control of plasma phosphate concentration as well as calcium concentration.

Bone and Its Relation to Extracellular Calcium and Phosphate Bone is composed of a tough organic matrix that is greatly strengthened by deposits of calcium salts. Average compact bone contains by weight about 30 per cent matrix and 70 per cent salts. Newly formed bone may have a considerably higher percentage of matrix in relation to salts. Organic Matrix of Bone The organic matrix of bone is 90 to 95 per cent collagen fibers, and the remainder is a homogeneous gelatinous medium called ground substance. The collagen fibers extend primarily along the lines of tensional force and give bone its powerful tensile strength. The ground substance is composed of extracellular fluid plus proteoglycans, especially chondroitin sulfate and hyaluronic acid. The precise function of each of these is not known, although they do help to control the deposition of calcium salts.

Bone Salts The crystalline salts deposited in the organic matrix of bone are composed principally of calcium and phosphate. The formula for the major crystalline salt, known as hydroxyapatite, is the following: Ca 10 (PO 4)6(OH)2 Each crystal—about 400 angstroms long, 10 to 30 angstroms thick, and 100 angstroms wide—is shaped like a long, flat plate. The relative ratio of calcium to phosphorus can vary markedly under different nutritional conditions, the Ca/P ratio on a weight basis varying between 1. 3 and 2. 0. Magnesium, sodium, potassium, and carbonate ions are also present among the bone salts, although x-ray diffraction studies fail to show definite crystals formed by them. Therefore, they are believed to be conjugated to the hydroxyapatite crystals rather than organized into distinct crystals of their own. This ability of many types of ions to conjugate to bone crystals extends to many ions normally foreign to bone, such as strontium, uranium, plutonium, the other transuranic elements, lead, gold, other heavy metals, and at least 9 of 14 of the major radioactive products released by explosion of the hydrogen bomb. Deposition of radioactive substances in the bone can cause prolonged irradiation of the bone tissues, and if a sufficient amount is deposited, an osteogenic sarcoma (bone cancer) eventually develops in most cases.

Tensile and Compressional Strength of Bone Each collagen fiber of compact bone is composed of repeating periodic segments every 640 angstroms along its length; hydroxyapatite crystals lie adjacent to each segment of the fiber, bound tightly to it. This intimate bonding prevents “shear” in the bone; that is, it prevents the crystals and collagen fibers from slipping out of place, which is essential in providing strength to the bone. In addition, the segments of adjacent collagen fibers overlap one another, also causing hydroxyapatite crystals to be overlapped like bricks keyed to one another in a brick wall. The collagen fibers of bone, like those of tendons, have great tensile strength, whereas the calcium salts have great compressional strength. These combined properties plus the degree of bondage between the collagen fibers and the crystals provide a bony structure that has both extreme tensile strength and extreme compressional strength.

Precipitation and Absorption of Calcium and Phosphate in Bone—Equilibrium with the Extracellular Fluids Hydroxyapatite Does Not Precipitate in Extracellular Fluid Despite Super saturation of Calcium and Phosphate Ions The concentrations of calcium and phosphate ions in extracellular fluid are considerably greater than those required to cause precipitation of hydroxyapatite. However, inhibitors are present in almost all tissues of the body as well as in plasma to prevent such precipitation; one such inhibitor is pyrophosphate. Therefore, hydroxyapatite crystals fail to precipitate in normal tissues except in bone despite the state of super saturation of the ions.

Mechanism of Bone Calcification The initial stage in bone production is the secretion of collagen molecules (called collagen monomers) and ground substance (mainly proteoglycans) by osteoblasts. The collagen monomers polymerize rapidly to form collagen fibers; the resultant tissue becomes osteoid, a cartilage-like material differing from cartilage in that calcium salts readily precipitate in it. As the osteoid is formed, some of the osteoblasts become entrapped in the osteoid and become quiescent. At this stage they are called osteocytes. Within a few days after the osteoid is formed, calcium salts begin to precipitate on the surfaces of the collagen fibers. The precipitates first appear at intervals along each collagen fiber, forming minute nidi that rapidly multiply and grow over a period of days and weeks into the finished product, hydroxyapatite crystals. The initial calcium salts to be deposited are not hydroxyapatite crystals but amorphous compounds (noncrystalline), a mixture of salts such as Ca. HPO 4 · 2 H 2 O, Ca 3(PO 4)2 · 3 H 2 O, and others. Then by a process of substitution and addition of atoms, or reabsorption and reprecipitation, these salts are converted into the hydroxyapatite crystals over a period of weeks or months. A few per cent may remain permanently in the amorphous form. This is important because these amorphous salts can be absorbed rapidly when there is need for extra calcium in the extracellular fluid. The mechanism that causes calcium salts to be deposited in osteoid is not fully understood. One theory holds that at the time of formation, the collagen fibers are specially constituted in advance for causing precipitation of calcium salts. The osteoblasts supposedly also secrete a substance into the osteoid to neutralize an inhibitor (believed to be pyrophosphate) that normally prevents hydroxyapatite crystallization. Once the pyrophosphate has been neutralized, the natural affinity of the collagen fibers for calcium salts causes the precipitation.

Precipitation of Calcium in Nonosseous Tissues Under Abnormal Conditions Although calcium salts almost never precipitate in normal tissues besides bone, under abnormal conditions, they do precipitate. For instance, they precipitate in arterial walls in the condition called arteriosclerosis and cause the arteries to become bonelike tubes. Likewise, calcium salts frequently deposit in degenerating tissues or in old blood clots. Presumably, in these instances, the inhibitor factors that normally prevent deposition of calcium salts disappear from the tissues, thereby allowing precipitation. Calcium Exchange Between Bone and Extracellular Fluid If soluble calcium salts are injected intravenously, the calcium ion concentration may increase immediately to high levels. However, within 30 minutes to 1 hour or more, the calcium ion concentration returns to normal. Likewise, if large quantities of calcium ions are removed from the circulating body fluids, the calcium ion concentration again returns to normal within 30 minutes to about 1 hour. These effects result in great part from the fact that the bone contains a type of exchangeable calcium that is always in equilibrium with the calcium ions in the extracellular fluids. A small portion of this exchangeable calcium is also the calcium found in all tissue cells, especially in highly permeable types of cells such as those of the liver and the gastrointestinal tract. However, most of the exchangeable calcium is in the bone. It normally amounts to about 0. 4 to 1 per cent of the total bone calcium. This calcium is deposited in the bones in a form of readily mobilizable salt such as Ca. HPO 4 and other amorphous calcium salts. The importance of exchangeable calcium is that it provides a rapid buffering mechanism to keep the calcium ion concentration in the extracellular fluids from rising to excessive levels or falling to very low levels under transient conditions of excess or decreased availability of calcium.

Deposition and Absorption of Bone—Remodeling of Bone Deposition of Bone by the Osteoblasts Bone is continually being deposited by osteoblasts, and it is continually being absorbed where osteoclasts are active (Figure 79– 4). Osteoblasts are found on the outer surfaces of the bones and in the bone cavities. A small amount of osteoblastic activity occurs continually in all living bones (on about 4 per cent of all surfaces at any given time in an adult), so that at least some new bone is being formed constantly.


Absorption of Bone—Function of the Osteoclasts Bone is also being continually absorbed in the presence of osteoclasts, which are large phagocytic, multinucleated cells (as many as 50 nuclei), derivatives of monocytes or monocyte-like cells formed in the bone marrow. The osteoclasts are normally active on less than 1 per cent of the bone surfaces of an adult. Later in the chapter we see that PTH controls the bone absorptive activity of osteoclasts. Histologically, bone absorption occurs immediately adjacent to the osteoclasts. The mechanism of this absorption is believed to be the following: The osteoclasts send out villus-like projections toward the bone, forming a so-called ruffled border adjacent to the bone. The villi secrete two types of substances: (1) proteolytic enzymes, released from the lysosomes of the osteoclasts, and (2) several acids, including citric acid and lactic acid, released from the mitochondria and secretory vesicles. The enzymes digest or dissolve the organic matrix of the bone, and the acids cause solution of the bone salts. The osteoclastic cells also imbibe by phagocytosis minute particles of bone matrix and crystals, eventually also dissoluting these and releasing the products into the blood.

Bone Deposition and Absorption Are Normally in Equilibrium Normally, except in growing bones, the rates of bone deposition and absorption are equal to each other, so that the total mass of bone remains constant. Osteoclasts usually exist in small but concentrated masses, and once a mass of osteoclasts begins to develop, it usually eats away at the bone for about 3 weeks, creating a tunnel that ranges in diameter from 0. 2 to 1 millimeter and is several millimeters long. At the end of this time, the osteoclasts disappear and the tunnel is invaded by osteoblasts instead; then new bone begins to develop. Bone deposition then continues for several months, the new bone being laid down in successive layers of concentric circles (lamellae) on the inner surfaces of the cavity until the tunnel is filled. Deposition of new bone ceases when the bone begins to encroach on the blood vessels supplying the area. The canal through which these vessels run, called the haversian canal, is all that remains of the original cavity. Each new area of bone deposited in this way is called an osteon, as shown in Figure 79– 5.


Value of Continual Bone Remodeling The continual deposition and absorption of bone have several physiologically important functions. First, bone ordinarily adjusts its strength in proportion to the degree of bone stress. Consequently, bones thicken when subjected to heavy loads. Second, even the shape of the bone can be rearranged for proper support of mechanical forces by deposition and absorption of bone in accordance with stress patterns. Third, because old bone becomes relatively brittle and weak, new organic matrix is needed as the old organic matrix degenerates. In this manner, the normal toughness of bone is maintained. Indeed, the bones of children, in whom the rates of deposition and absorption are rapid, show little brittleness in comparison with the bones of the elderly, in whom the rates of deposition and absorption are slow.

Control of the Rate of Bone Deposition by Bone “Stress. ” Bone is deposited in proportion to the compressional load that the bone must carry. For instance, the bones of athletes become considerably heavier than those of nonathletes. Also, if a person has one leg in a cast but continues to walk on the opposite leg, the bone of the leg in the cast becomes thin and as much as 30 per cent decalcified within a few weeks, whereas the opposite bone remains thick and normally calcified. Therefore, continual physical stress stimulates osteoblastic deposition and calcification of bone. Bone stress also determines the shape of bones under certain circumstances. For instance, if a long bone of the leg breaks in its center and then heals at an angle, the compression stress on the inside of the angle causes increased deposition of bone, and increased absorption occurs on the outer side of the angle where the bone is not compressed. After many years of increased deposition on the inner side of the angulated bone and absorption on the outer side, the bone can become almost straight, especially in children because of the rapid remodeling of bone at younger ages.

Repair of a Fracture Activates Osteoblasts Fracture of a bone in some way maximally activates all the periosteal and intraosseous osteoblasts involved in the break. Also, immense numbers of new osteoblasts are formed almost immediately from osteoprogenitor cells, which are bone stem cells in the surface tissue lining bone, called the “bone membrane. ” Therefore, within a short time, a large bulge of osteoblastic tissue and new organic bone matrix, followed shortly by the deposition of calcium salts, develops between the two broken ends of the bone. This is called a callus. Many bone surgeons use the phenomenon of bone stress to accelerate the rate of fracture healing. This is done by use of special mechanical fixation apparatuses for holding the ends of the broken bone together so that the patient can continue to use the bone immediately. This causes stress on the opposed ends of the broken bones, which accelerates osteoblastic activity at the break and often shortens convalescence.

Vitamin D has a potent effect to increase calcium absorption from the intestinal tract; it also has important effects on both bone deposition and bone absorption, However, vitamin D itself is not the active substance that actually causes these effects. Instead, vitamin D must first be converted through a succession of reactions in the liver and the kidneys to the final active product, 1, 25 -dihydroxycholecalciferol, also called 1, 25(OH)2 D 3. Figure 79– 6 shows the succession of steps that lead to the formation of this substance from vitamin D. Let us discuss these steps.


Cholecalciferol (Vitamin D 3) Is Formed in the Skin Several compounds derived from sterols belong to the vitamin D family, and they all perform more or less the same functions. Vitamin D 3 (also called cholecalciferol) is the most important of these and is formed in the skin as a result of irradiation of 7 -dehydrocholesterol, a substance normally in the skin, by ultraviolet rays from the sun. Consequently, appropriate exposure to the sun prevents vitamin D deficiency. The additional vitamin D compounds that we ingest in food are identical to the cholecalciferol formed in the skin, except for the substitution of one or more atoms that do not affect their function. Cholecalciferol Is Converted to 25 -Hydroxycholecalciferol in the Liver The first step in the activation of cholecalciferol is to convert it to 25 hydroxycholecalciferol; this occurs in the liver. The process is a limited one, because the 25 -hydroxycholecalciferol has a feedback inhibitory effect on the conversion reactions. This feedback effect is extremely important for two reasons. First, the feedback mechanism precisely regulates the concentration of 25 hydroxycholecalciferol in the plasma, an effect that is shown in Figure 79– 7. Note that the intake of vitamin D 3 can increase many times, and yet the concentration of 25 -hydroxycholecalciferol remains nearly normal. This high degree of feedback control prevents excessive action of vitamin D when intake of vitamin D 3 is altered over a wide range. Second, this controlled conversion of vitamin D 3 to 25 -hydroxycholecalciferol conserves the vitamin D stored in the liver for future use. Once it is converted, it persists in the body for only a few weeks, whereas in the vitamin D form, it can be stored in the liver for many months.


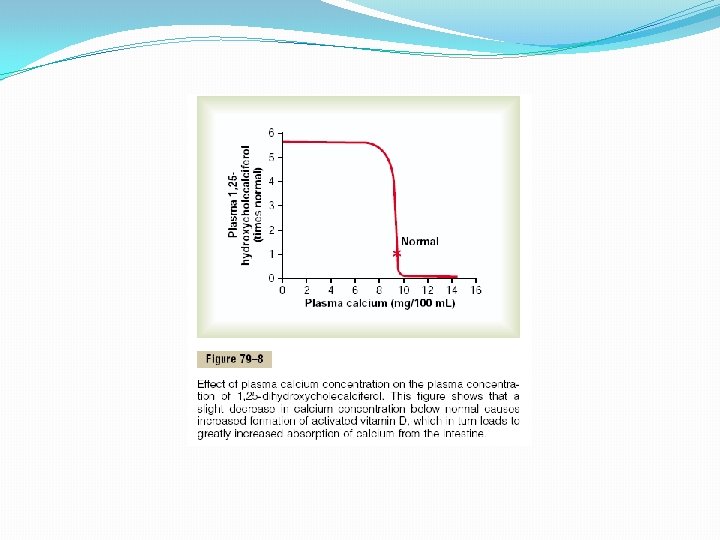
Formation of 1, 25 -Dihydroxycholecalciferol in the Kidneys and Its Control by Parathyroid Hormone. Figure 79– 6 also shows the conversion in the proximal tubules of the kidneys of 25 hydroxycholecalciferol to 1, 25 - dihydroxycholecalciferol. This latter substance is by far the most active form of vitamin D, because the previous products in the scheme of Figure 79– 6 have less than 1/1000 of the vitamin D effect. Therefore, in the absence of the kidneys, vitamin D loses almost all its effectiveness. Note also in Figure 79– 6 that the conversion of 25 - hydroxycholecalciferol to 1, 25 dihydroxycholecalciferol requires PTH. In the absence of PTH, almost none of the 1, 25 dihydroxycholecalciferol is formed. Therefore, PTH exerts a potent influence in determining the functional effects of vitamin D in the body. Calcium Ion Concentration Controls the Formation of 1, 25 - Dihydroxycholecalciferol Figure 79– 8 demonstrates that the plasma concentration of 1, 25 -dihydroxycholecalciferol is inversely affected by the concentration of calcium in the plasma. There are two reasons for this. First, the calcium ion itself has a slight effect in preventing the conversion of 25 hydroxycholecalciferol to 1, 25 -dihydroxycholecalciferol. Second, and even more important, as we shall see later in the chapter, the rate of secretion of PTH is greatly suppressed when the plasma calcium ion concentration rises above 9 to 10 mg/100 ml. Therefore, at calcium concentrations below this level, PTH promotes the conversion of 25 hydroxycholecalciferol to 1, 25 -dihydroxycholecalciferol in the kidneys. At higher calcium concentrations, when PTH is suppressed, the 25 -hydroxycholecalciferol is converted to a different compound— 24, 25 -dihydroxycholecalciferol—that has almost no vitamin D effect. When the plasma calcium concentration is already too high, the formation of 1, 25 -dihydroxycholecalciferol is greatly depressed. Lack of this in turn decreases the absorption of calcium from the intestines, the bones, and the renal tubules, thus causing the calcium ion concentration to fall back toward its normal level.

Actions of Vitamin D The active form of vitamin D, 1, 25 -dihydroxycholecalciferol, has several effects on the intestines, kidneys, and bones that increase absorption of calcium and phosphate into the extracellular fluid and contribute to feedback regulation of these substances. “Hormonal” Effect of Vitamin D to Promote Intestinal Calcium Absorption. 1, 25 -Dihydroxycholecalciferol itself functions as a type of “hormone” to promote intestinal absorption of calcium. It does this principally by increasing, over a period of about 2 days, formation of a calcium-binding protein in the intestinal epithelial cells. This protein functions in the brush border of these cells to transport calcium into the cell cytoplasm, and the calcium then moves through the basolateral membrane of the cell by facilitated diffusion. The rate of calcium absorption is directly proportional to the quantity of this calciumbinding protein. Furthermore, this protein remains in the cells for several weeks after the 1, 25 -dihydroxycholecalciferol has been removed from the body, thus causing a prolonged effect on calcium absorption. Other effects of 1, 25 -dihydroxycholecalciferol that might play a role in promoting calcium absorption are the formation of (1) a calcium-stimulated ATPase in the brush border of the epithelial cells and (2) an alkaline phosphatase in the epithelial cells. The precise details of all these effects are unclear.

Vitamin D Promotes Phosphate Absorption by the Intestines Although phosphate is usually absorbed easily, phosphate flux through the gastrointestinal epithelium is enhanced by vitamin D. It is believed that this results from a direct effect of 1, 25 -dihydroxycholecalciferol, but it is possible that it results secondarily from this hormone’s action on calcium absorption, the calcium in turn acting as a transport mediator for the phosphate. Vitamin D Decreases Renal Calcium and Phosphate Excretion Vitamin D also increases calcium and phosphate absorption by the epithelial cells of the renal tubules, thereby tending to decrease excretion of these substances in the urine. However, this is a weak effect and probably not of major importance in regulating the extracellular fluid concentration of these substances. Effect of Vitamin D on Bone and Its Relation to Parathyroid Hormone Activity Vitamin D plays important roles in both bone absorption and bone deposition. The administration of extreme quantities of vitamin D causes absorption of bone. In the absence of vitamin D, the effect of PTH in causing bone absorption is greatly reduced or even prevented. The mechanism of this action of vitamin D is not known, but it is believed to result from the effect of 1, 25 -dihydroxycholecalciferol to increase calcium transport through cellular membranes. Vitamin D in smaller quantities promotes bone calcification. One of the ways in which it does this is to increase calcium and phosphate absorption from the intestines. However, even in the absence of such increase, it enhances the mineralization of bone. Here again, the mechanism of the effect is unknown, but it probably also results from the ability of 1, 25 -dihydroxycholecalciferol to cause transport of calcium ions through cell membranes—but in this instance, perhaps in the opposite direction through the osteoblastic or osteocytic cell membranes.

Parathyroid Hormone Parathyroid hormone provides a powerful mechanism for controlling extracellular calcium and phosphate concentrations by regulating intestinal reabsorption, renal excretion, and exchange between the extracellular fluid and bone of these ions. Excess activity of the parathyroid gland causes rapid absorption of calcium salts from the bones, with resultant hypercalcemia in the extracellular fluid; conversely, hypofunction of the parathyroid glands causes hypocalcemia, often with resultant tetany. Physiologic Anatomy of the Parathyroid Glands Normally there are four parathyroid glands in humans; they are located immediately behind the thyroid gland—one behind each of the upper and each of the lower poles of the thyroid. Each parathyroid gland is about 6 millimeters long, 3 millimeters wide, and 2 millimeters thick and has a macroscopic appearance of dark brown fat. The parathyroid glands are difficult to locate during thyroid operations because they often look like just another lobule of the thyroid gland. For this reason, before the importance of these glands was generally recognized, total or subtotal thyroidectomy frequently resulted in removal of the parathyroid glands as well. Removal of half the parathyroid glands usually causes no major physiologic abnormalities. However, removal of three of the four normal glands causes transient hypoparathyroidism. But even a small quantity of remainiof hypertrophying satisfactorily to perform the function of all the glands. The parathyroid gland of the adult human being, shown in Figure 79– 9, contains mainly chief cells and a small to moderate number of oxyphil cells, but oxyphil cells are absent in many animals and in young humans. The chief cells are believed to secrete most, if not all, of the PTH. The function of the oxyphil cells is not certain, but they are believed to be modified or depleted chief cells that no longer secrete hormone.


Chemistry of Parathyroid Hormone PTH has been isolated in a pure form. It is first synthesized on the ribosomes in the form of a preprohormone, a polypeptide chain of 110 amino acids. This is cleaved first to a prohormone with 90 amino acids, then to the hormone itself with 84 amino acids by the endoplasmic reticulum and Golgi apparatus, and finally is packaged in secretory granules in the cytoplasm of the cells. The final hormone has a molecular weight of about 9500. Smaller compounds with as few as 34 amino acids adjacent to the N terminus of the molecule have also been isolated from the parathyroid glands that exhibit full PTH activity. In fact, because the kidneys rapidly remove the whole 84 -amino acid hormone within minutes but fail to remove many of the fragments for hours, a large share of the hormonal activity is caused by the fragments. Effect of Parathyroid Hormone on Calcium and Phosphate Concentrations in the Extracellular Fluid Figure 79– 10 shows the approximate effects on the blood calcium and phosphate concentrations caused by suddenly infusing PTH into an animal and continuing this for several hours. Note that at the onset of infusion the calcium ion concentration begins to rise and reaches a plateau in about 4 hours. The phosphate concentration, however, falls more rapidly than the calcium rises and reaches a depressed level within 1 or 2 hours. The rise in calcium concentration is caused principally by two effects: (1) an effect of PTH to increase calcium and phosphate absorption from the bone and (2) a rapid effect of PTH to decrease the excretion of calcium by the kidneys. The decline in phosphate concentration is caused by a strong effect of PTH to increase renal phosphate excretion, an effect that is usually great enough to override increased phosphate absorption from the bone.


Parathyroid Hormone Increases Phosphate Absorption from the Bone Calcium and PTH has two effects on bone in causing absorption of calcium and phosphate. One is a rapid phase that begins in minutes and increases progressively for several hours. This phase results from activation of the already existing bone cells (mainly the osteocytes) to promote calcium and phosphate absorption. The second phase is a much slower one, requiring several days or even weeks to become fully developed; it results from proliferation of the osteoclasts, followed by greatly increased osteoclastic reabsorption of the bone itself, not merely absorption of the calcium phosphate salts from the bone. �

Rapid Phase of Calcium and Phosphate Absorption—Osteolysis When large quantities of PTH are injected, the calcium ion concentration in the blood begins to rise within minutes, long before any new bone cells can be developed. Histological and physiologic studies have shown that PTH causes removal of bone salts from two areas in the bone: (1) from the bone matrix in the vicinity of the osteocytes lying within the bone itself and (2) in the vicinity of the osteoblasts along the bone surface. One does not usually think of either osteoblasts or osteocytes functioning to cause bone salt absorption, because both these types of cells are osteoblastic in nature and normally associated with bone deposition and its calcification. However, studies have shown that the osteoblasts and osteocytes form a system of interconnected cells that spreads all through the bone and over all the bone surfaces except the small surface areas adjacent to the osteoclasts. In fact, long, filmy processes extend from osteocyte to osteocyte throughout the bone structure, and these processes also connect with the surface osteocytes and osteoblasts. This extensive system is called the osteocytic membrane system, and it is believed to provide a membrane that separates the bone itself from the extracellular fluid. Between the osteocytic membrane and the bone is a small amount of bone fluid. Experiments suggest that the osteocytic membrane pumps calcium ions from the bone fluid into the extracellular fluid, creating a calcium ion concentration in the bone fluid only one third that in the extracellular fluid. When the osteocytic pump becomes excessively activated, the bone fluid calcium concentration falls even lower, and calcium phosphate salts are then absorbed from the bone. This effect is called osteolysis, and it occurs without absorption of the bone’s fibrous and gel matrix. When the pump is inactivated, the bone fluid calcium concentration rises to a higher level, and calcium phosphate salts are redeposited in the matrix. But where does PTH fit into this picture? First, the cell membranes of both the osteoblasts and the osteocytes have receptor proteins for binding PTH can activate the calcium pump strongly, thereby causing rapid removal of calcium phosphate salts from those amorphous bone crystals that lie near the cells. PTH is believed to stimulate this pump by increasing the calcium permeability of the bone fluid side of the osteocytic membrane, thus allowing calcium ions to diffuse into the membrane cells from the bone fluid. Then the calcium pump on the other side of the cell membrane transfers the calcium ions the rest of the way into the extracellular fluid.

Slow Phase of Bone Absorption and Calcium Phosphate Release—Activation of the Osteoclasts A much better known effect of PTH and one for which the evidence is much clearer is its activation of the osteoclasts. Yet the osteoclasts do not themselves have membrane receptor proteins for PTH. Instead, it is believed that the activated osteoblasts and osteocytes send a secondary but unknown “signal” to the osteoclasts, causing them to set about their usual task of gobbling up the bone over a period of weeks or months. Activation of the osteoclastic system occurs in two stages: (1) immediate activation of the osteoclasts that are already formed and (2) formation of new osteoclasts. Several days of excess PTH usually cause the osteoclastic system to become well developed, but it can continue to grow for months under the influence of strong PTH stimulation. After a few months of excess PTH, osteoclastic resorption of bone can lead to weakened bones and secondary stimulation of the osteoblasts that attempt to correct the weakened state. Therefore, the late effect is actually to enhance both osteoblastic and osteoclastic activity. Still, even in the late stages, there is more bone absorption than bone deposition in the presence of continued excess PTH. Bone contains such great amounts of calcium in comparison with the total amount in all the extracellular fluids (about 1000 times as much) that even when PTH causes a great rise in calcium concentration in the fluids, it is impossible to discern any immediate effect on the bones. Prolonged administration or secretion of PTH—over a period of many months or years—finally results in very evident absorption in all the bones and even development of large cavities filled with large, multinucleated osteoclasts.

Parathyroid Hormone Decreases Calcium Excretion and Increases Phosphate Excretion by the Kidneys Administration of PTH causes rapid loss of phosphate in the urine owing to the effect of the hormone to diminish proximal tubular reabsorption of phosphate ions. PTH also increases renal tubular reabsorption of calcium at the same time that it diminishes phosphate reabsorption. Moreover, it increases the rate of reabsorption of magnesium ions and hydrogen ions while it decreases the reabsorption of sodium, potassium, and amino acid ions in much the same way that it affects phosphate. The increased calcium absorption occurs mainly in the late distal tubules, the collectingtubules, the early collecting ducts, and possibly the ascending loop of Henle to a lesser extent. Were it not for the effect of PTH on the kidneys to increase calcium reabsorption, continual loss of calcium into the urine would eventually deplete both the extracellular fluid and the bones of this mineral. Parathyroid Hormone Increases Intestinal Absorption of Calcium and Phosphate At this point, we should be reminded again that PTH greatly enhances both calcium and phosphate absorption from the intestines by increasing the formation in the kidneys of 1, 25 -dihydroxycholecalciferol from vitamin D, as discussed earlier in the chapter. Cyclic Adenosine Monophosphate Mediates the Effects of Parathyroid Hormone A large share of the effect of PTH on its target organs is mediated by the cyclic adenosine monophosphate (c. AMP) second messenger mechanism. Within a few minutes after PTH administration, the concentration of c. AMP increases in the osteocytes, osteoclasts, and other target cells. This c. AMP in turn is probably responsible for such functions as osteoclastic secretion of enzymes and acids to cause bone reabsorption and formation of 1, 25 -dihydroxycholecalciferol in the kidneys. There are probably other direct effects of PTH that function independently of the second messenger mechanism.

Control of Parathyroid Secretion by Calcium Ion Concentration Even the slightest decrease in calcium ion concentration in the extracellular fluid causes the parathyroid glands to increase their rate of secretion within minutes; if the decreased calcium concentration persists, the glands will hypertrophy, sometimes fivefold or more. For instance, the parathyroid glands become greatly enlarged in rickets, in which the level of calcium is usually depressed only a small amount; also, they become greatly enlarged in pregnancy, even though the decrease in calcium ion concentration in the mother’s extracellular fluid is hardly measurable; and they are greatly enlarged during lactation because calcium is used for milk formation. Conversely, conditions that increase the calcium ion concentration above normal cause decreased activity and reduced size of the parathyroid glands. Such conditions include (1) excess quantities of calcium in thediet, (2) increased vitamin D in the diet, and (3) bone absorption caused by factors other than PTH (for example, bone absorption caused by disuse of the bones). Figure 79– 11 shows the approximate relation between plasma calcium concentration and plasma PTH concentration. The solid red curve shows the acute effect when the calcium concentration is changed over a period of a few hours. This shows that even small decreases in calcium concentration from the normal value can double or triple the plasma PTH. The approximate chronic effect that one finds when the calcium ion concentration changes over a period of many weeks, thus allowing time for the glands to hypertrophy greatly, is shown by the dashed red line; this demonstrates that a decrease of only a fraction of a milligram per deciliter in plasma calcium concentration can double PTH secretion. This is the basis of the body’s extremely potent feedback system for longterm control of plasma calcium ion concentration.


Calcitonin, a peptide hormone secreted by the thyroid gland, tends to decrease plasma calcium concentration and, in general, has effects opposite to those of PTH. However, the quantitative role of calcitonin is far less than that of PTH in regulating calcium ion concentration. Synthesis and secretion of calcitonin occur in the parafollicular cells, or C cells, lying in the interstitial fluid between the follicles of the thyroid gland. These cells constitute only about 0. 1 per cent of the human thyroid gland are the remnants of the ultimobrachial glands of lower animals such as fish, amphibians, reptiles, and birds. Calcitonin is a 32 amino acid peptide with a molecular weight of about 3400. Increased Plasma Secretion. Calcium Concentration Stimulates Calcitonin The primary stimulus for calcitonin secretion is increased plasma calcium ion concentration. This contrasts with PTH secretion, which is stimulated by decreased calcium concentration. In young animals, but much less so in older animals and in humans, an increase in plasma calcium concentration of about 10 per cent causes an immediate twofold or more increase in the rate of secretion of calcitonin, which is shown by the blue line in Figure 79– 11. This provides a second hormonal feedback mechanism for controlling the plasma calcium ion concentration, but one that is relatively weak and works in a way opposite that of the PTH system.

Calcitonin Decreases Plasma Calcium Concentration In some young animals, calcitonin decreases blood calcium ion concentration rapidly, beginning within minutes after injection of the calcitonin, in at least two ways. 1. The immediate effect is to decrease the absorptive activities of the osteoclasts and possibly the osteolytic effect of the osteocytic membrane throughout the bone, thus shifting the balance in favor of deposition of calcium in the exchangeable bone calcium salts. This effect is especially significant in young animals because of the rapid interchange of absorbed and deposited calcium. 2. The second and more prolonged effect of calcitonin is to decrease the formation of new osteoclasts. Also, because osteoclastic resorption of bone leads secondarily to osteoblastic activity, decreased numbers of osteoclasts are followed by decreased numbers of osteoblasts. Therefore, over a long period, the net result is reduced osteoclastic and osteoblastic activity and, consequently, very little prolonged effect on plasma calcium ion concentration. That is, the effect on plasma calcium is mainly a transient one, lasting for a few hours to a few days at most. Calcitonin also has minor effects on calcium handling in the kidney tubules and the intestines. Again, the effects are opposite those of PTH, but they appear to be of such little import that they are seldom considered.

Calcitonin Has a Weak Effect on Plasma Calcium Concentration in the Adult Human The reason for the weak effect of calcitonin on plasma calcium is twofold. First, any initial reduction of the calcium ion concentration caused by calcitonin leads within hours to a powerful stimulation of PTH secretion, which almost overrides the calcitonin effect. When the thyroid gland is removed and calcitonin is no longer secreted, the long term blood calcium ion concentration is not measurably altered, which again demonstrates the overriding effect of the PTH system of control. Second, in the adult, the daily rates of absorption and deposition of calcium are small, and even after the rate of absorption is slowed by calcitonin, this still has only a small effect on plasma calcium ion concentration. The effect of calcitonin in children is much greater because bone remodeling occurs rapidly in children, with absorption and deposition of calcium as great as 5 grams or more per day—equal to 5 to 10 times the total calcium in all the extracellular fluid. Also, in certain bone diseases, such as Paget’s disease, in which osteoclastic activity is greatly accelerated, calcitonin has a much more potent effect of reducing the calcium absorption.

Summary of Control of Calcium Ion Concentration At times, the amount of calcium absorbed into or lost from the body fluids is as much as 0. 3 gram in 1 hour. For instance, in cases of diarrhea, several grams of calcium can be secreted in the intestinal juices, passed into the intestinal tract, and lost into the feces each day. Conversely, after ingestion of large quantities of calcium, particularly when there is also an excess of vitamin D activity, a person may absorb as much as 0. 3 gram in 1 hour. This figure compares with a total quantity of calcium in all the extracellular fluid of about 1 gram. The addition or subtraction of 0. 3 gram to or from such a small amount of calcium in the extracellular fluid would cause serious hypercalcemia or hypocalcemia. However, there is a first line of defense to prevent this from occurring even before the parathyroid and calcitonin hormonal feedback systems have a chance to act. Buffer Function of the Exchangeable Calcium in Bones—the First Line of Defense The exchangeable calcium salts in the bones, discussed earlier in this chapter, are amorphous calcium phosphate compounds, probably mainly Ca. HPO 4 or some similar compound loosely bound in the bone and in reversible equilibrium with the calcium and phosphate ions in the extracellular fluid. The quantity of these salts that is available for exchange is about 0. 5 to 1 per cent of the total calcium salts of the bone, a total of 5 to 10 grams of calcium. Because of the ease of deposition of these exchangeable salts and their ease of resolubility, an increase in the concentrations of extracellular fluid calcium and phosphate ions above normal causes immediate deposition of exchangeable salt. Conversely, a decrease in these concentrations causes immediate absorption of exchangeable salt. This reaction is rapid because the amorphous bone crystals are extremely small and their total surface area exposed to the fluids of the bone is perhaps 1 acre or more. Also, about 5 per cent of all the blood flows through the bones each minute—that is, about 1 per cent of all the extracellular fluid each minute. Therefore, about one half of any excess calcium that appears in the extracellular fluid is removed by this buffer function of the bones in about 70 minutes. In addition to the buffer function of the bones, the mitochondria of many of the tissues of the body, especially of the liver and intestine, contain a reasonable amount of exchangeable calcium (a total of about 10 grams in the whole body) that provides an additional buffer system for helping to maintain constancy of the extracellular fluid calcium ion concentration.

Hormonal Control of Calcium Ion Concentration—the Second Line of Defense At the same time that the exchangeable calcium mechanism in the bones is “buffering” the calcium in the extracellular fluid, both the parathyroid and the calcitonin hormonal systems are beginning to act. Within 3 to 5 minutes after an acute increase in the calcium ion concentration, the rate of PTH secretion decreases. As already explained, this sets into play multiple mechanisms for reducing the calcium ion concentration back toward normal. At the same time that PTH decreases, calcitonin increases. In young animals and possibly in young children (but probably to a smaller extent in adults), the calcitonin causes rapid deposition of calcium in the bones, and perhaps in some cells of other tissues. Therefore, in very young animals, excess calcitonin cause a high calcium ion concentration to return to normal perhaps considerably more rapidly than can be achieved by the exchangeable calcium-buffering mechanism alone. In prolonged calcium excess or prolonged calcium deficiency, only the PTH mechanism seems to be really important in maintaining a normal plasma calcium ion concentration. When a person has a continuing deficiency of calcium in the diet, PTH often can stimulate enough calcium absorption from the bones to maintain a normal plasma calcium ion concentration for 1 year or more, but eventually, even the bones will run out of calcium. Thus, in effect, the bones are a large buffer-reservoir of calcium that can be manipulated by PTH. Yet, when the bone reservoir either runs out of calcium or, oppositely, becomes saturated with calcium, the long-term control of extracellular calcium ion concentration resides almost entirely in the roles of PTH and vitamin D in controlling calcium absorption from the gut and calcium excretion in the urine.

Pathophysiology of Parathyroid Hormone, Vitamin D, and Bone Disease Hypoparathyroidism When the parathyroid glands do not secrete sufficient PTH, the osteocytic reabsorption of exchangeable calcium decreases and the osteoclasts become almost totally inactive. As a result, calcium reabsorption from the bones is so depressed that the level of calcium in the body fluids decreases. Yet, because calcium and phosphates are not being absorbed from the bone, the bone usually remains strong. When the parathyroid glands are suddenly removed, the calcium level in the blood falls from the normal of 9. 4 mg/dl to 6 to 7 mg/dl within 2 to 3 days, and the blood phosphate concentration may double. When this low calcium level is reached, the usual signs of tetany develop. Among the muscles of the body especially sensitive to tetanic spasm are the laryngeal muscles. Spasm of these muscles obstructs respiration, which is the usual cause of death in tetany unless appropriate treatment is applied. Treatment of Hypoparathyroidism with PTH and Vitamin D PTH is occasionally used for treating hypoparathyroidism. However, because of the expense of this hormone, because its effect lasts for a few hours at most, and because the tendency of the body to develop antibodies against it makes it progressively less and less effective, hypoparathyroidism is usually not treated with PTH administration. In most patients with hypothyroidism, the administration of extremely large quantities of vitamin D, to as high as 100, 000 units per day, along with intake of 1 to 2 grams of calcium, keeps the calcium ion concentration in a normal range. At times, it might be necessary to administer 1, 25 -dihydroxycholecalciferol instead of the nonactivated form of vitamin D because of its much more potent and much more rapid action. This can also cause unwanted effects, because it is sometimes difficult to prevent overactivity by this activated form of vitamin D.

Primary Hyperparathyroidism In primary hyperparathyroidism, an abnormality of the parathyroid glands causes inappropriate, excess PTH secretion. The cause of primary hyperparathyroidism ordinarily is a tumor of one of the parathyroid glands; such tumors occur much more frequently in women than in men or children, mainly because pregnancy and lactation stimulate the parathyroid glands and therefore predispose to the development of such a tumor. Hyperparathyroidism causes extreme osteoclastic activity in the bones. This elevates the calcium ion concentration in the extracellular fluid while usually depressing the concentration of phosphate ions because of increased renal excretion of phosphate. Bone Disease in Hyperparathyroidism. Although in mild hyperparathyroidism new bone can be deposited rapidly enough to compensate for the increased osteoclastic reabsorption of bone, in severe hyperparathyroidism the osteoclastic absorption soon far outstrips osteoblastic deposition, and the bone may be eaten away almost entirely. Indeed, the reason a hyperparathyroid person seeks medical attention is often a broken bone. Radiographs of the bone show extensive decalcification and, occasionally, large punched-out cystic areas of the bone that are filled with osteoclasts in the form of socalled giant cell osteoclast “tumors. ” Multiple fractures of the weakened bones can result from only slight trauma, especially where cysts develop. The cystic bone disease of hyperparathyroidism is called osteitis fibrosa cystica. Osteoblastic activity in the bones also increases greatly in a vain attempt to form enough new bone to make up for the old bone absorbed by the osteoclastic activity. When the osteoblasts become active, they secrete large quantities of alkaline phosphatase. Therefore, one of the important diagnostic findings in hyperparathyroidism is a high level of plasma alkaline phosphatase.

Effects of Hypercalcemia in Hyperparathyroidism can at times cause the plasma calcium level to rise to 12 to 15 mg/dl and, rarely, even higher. The effects of such elevated calcium levels, as detailed earlier in the chapter, are depression of the central and peripheral nervous systems, muscle weakness, constipation, abdominal pain, peptic ulcer, lack of appetite, and depressed relaxation of the heart during diastole. Parathyroid Poisoning and Metastatic Calcification When, on rare occasions, extreme quantities of PTH are secreted, the level of calcium in the body fluids rises rapidly to high values. Even the extracellular fluid phosphate concentration often rises markedly instead of falling, as is usually the case, probably because the kidneys cannot excrete rapidly enough all the phosphate being absorbed from the bone. Therefore, the calcium and phosphate in the body fluids become greatly supersaturated, so that calcium phosphate (Ca. HPO 4) crystals begin to deposit in the alveoli of the lungs, the tubules of the kidneys, the thyroid gland, the acid-producing area of the stomach mucosa, and the walls of the arteries throughout the body. This extensive metastatic deposition of calcium phosphate can develop within a few days. Ordinarily, the level of calcium in the blood must rise above 17 mg/dl before there is danger of parathyroid poisoning, but once such elevation develops along with concurrent elevation of phosphate, death can occur in only a few days.

Formation of Kidney Stones in Hyperparathyroidism Most patients with mild hyperparathyroidism show few signs of bone disease and few general abnormalities as a result of elevated calcium, but they do have an extreme tendency to form kidney stones. The reason is that the excess calcium and phosphate absorbed from the intestines or mobilized from the bones in hyperparathyroidism must eventually be excreted by the kidneys, causing a proportionate increase in the concentrations of these substances in the urine. As a result, crystals of calcium phosphate tend to precipitate in the kidney, forming calcium phosphate stones. Also, calcium oxalate stones develop because even normal levels of oxalate cause calcium precipitation at high calcium levels. Because the solubility of most renal stones is slight in alkaline media, the tendency formation of renal calculi is considerably greater in alkaline urine than in acid urine. For this reason, acidotic diets and acidic drugs are frequently used for treating renal calculi. Secondary Hyperparathyroidism In secondary hyperparathyroidism, high levels of PTH occur as a compensation for hypocalcemia rather than as a primary abnormality of the parathyroid glands. This contrasts with primary hyperparathyroidism, which is associated with hypercalcemia. Secondary hyperparathyroidism can be caused by vitamin D deficiency or chronic renal disease in which the damaged kidneys are unable to produce sufficient amounts of the active form of vitamin D, 1, 25 dihydroxycholecalciferol. As discussed in more detail in the next section, the vitamin D deficiency leads to osteomalacia (inadequate mineralization of the bones), and high levels of PTH cause absorption of the bones.

Rickets—Vitamin D Deficiency Rickets occurs mainly in children. It results from calcium or phosphate deficiency in the extracellular fluid, usually caused by lack of vitamin D. If the child is adequately exposed to sunlight, the 7 -dehydrocholesterol in the skin becomes activated by the ultraviolet rays and forms vitamin D 3, which prevents rickets by promoting calcium and phosphate absorption from the intestines. Children who remain indoors through the winter in general do not receive adequate quantities of vitamin D without some supplementation in the diet. Rickets tends to occur especially in the spring months because vitamin D formed during the preceding summer is stored in the liver and available for use during the early winter months. Also, calcium and phosphate absorption from the bones can prevent clinical signs of rickets for the first few months of vitamin D deficiency. Plasma Concentrations of Calcium and Phosphate Decrease in Rickets The plasma calcium concentration in rickets is only slightly depressed, but the level of phosphate is greatly depressed. This is because the parathyroid glands prevent the calcium level from falling by promoting bone absorption every time the calcium level begins to fall. However, there is no good regulatory system for preventing a falling level of phosphate, and the increased parathyroid activity actually increases the excretion of phosphates in the urine.

Rickets Weakens the Bones During prolonged rickets, the marked compensatory increase in PTH secretion causes extreme osteoclastic absorption of the bone; this in turn causes the bone to become progressively weaker and imposes marked physical stress on the bone, resulting in rapid osteoblastic activity as well. The osteoblasts lay down large quantities of osteoid, which does not become calcified because of insufficient calcium and phosphate ions. Consequently, the newly formed, uncalcified, and weak osteoid gradually takes the place of the older bone that is being reabsorbed. Tetany in Rickets In the early stages of rickets, tetany almost never occurs because the parathyroid glands continually stimulate osteoclastic absorption of bone and, therefore, maintain an almost normal level of calcium in the extracellular fluid. However, when the bones finally become exhausted of calcium, the level of calcium may fall rapidly. As the blood level of calcium falls below 7 mg/dl, the usual signs of tetany develop, and the child may die of tetanic respiratory spasm unless intravenous calcium is administered, which relieves the tetany immediately. Treatment of Rickets. The treatment of rickets depends on supplying adequate calcium and phosphate in the diet and, equally important, on administering large amounts of vitamin D. If vitamin D is not administered, little calcium and phosphate are absorbed from the gut.

Osteomalacia—“Adult Rickets”. Adults seldom have a serious dietary deficiency of vitamin D or calcium because large quantities of calcium are not needed for bone growth as in children. However, serious deficiencies of both vitamin D and calcium occasionally occur as a result of steatorrhea (failure to absorb fat) because vitamin D is fat-soluble and calcium tends to form insoluble soaps with fat; consequently, in steatorrhea, both vitamin D and calcium tend to pass into the feces. Under these conditions, an adult occasionally has such poor calcium and phosphate absorption that adult rickets can occur, although this almost never proceeds to the stage of tetany but often is a cause of severe bone disability. Osteomalacia and Rickets Caused by Renal Disease. “Renal rickets” is a type of osteomalacia that results from prolonged kidney damage. The cause of this condition is mainly failure of the damaged kidneys to form 1, 25 -dihydroxycholecalciferol, the active form of vitamin D. In patients whose kidneys have been removed or destroyed and who are being treated by hemodialysis, the problem of renal rickets is often a severe one. Another type of renal disease that leads to rickets and osteomalacia is congenital hypophosphatemia, resulting from congenitally reduced reabsorption of phosphates by the renal tubules. This type of rickets must be treated with phosphate compounds instead of calcium and vitamin D, and it is called vitamin D–resistant rickets. Osteoporosis—Decreased Bone Matrix Osteoporosis is the most common of all bone diseases in adults, especially in old age. It is different from osteomalacia and rickets because it results from diminished organic bone matrix rather than from poor bone calcification. In osteoporosis the osteoblastic activity in the bone usually is less than normal, and consequently the rate of bone osteoid deposition is depressed. But occasionally, as in hyperparathyroidism, the cause of the diminished bone is excess osteoclastic activity.

The many common causes of osteoporosis are (1) lack of physical stress on the bones because of inactivity; (2) malnutrition to the extent that sufficient protein matrix cannot be formed; (3) lack of vitamin C, which is necessary for the secretion of intercellular substances by all cells, including formation of osteoid by the osteoblasts; (4) postmenopausal lack of estrogen secretion because estrogens decrease the number and activity of osteoclasts; (5) old age, in which growth hormone and other growth factors diminish greatly, plus the fact that many of the protein anabolic functions also deteriorate with age, so that bone matrix cannot be deposited satisfactorily; (6) Cushing’s syndrome, because massive quantities of glucocorticoids secreted in this disease cause decreased deposition of protein throughout the body and increased catabolism of protein and have the specific effect of depressing osteoblastic activity. Thus, many diseases of deficiency of protein metabolism can cause osteoporosis.

Physiology of the Teeth The teeth cut, grind, and mix the food eaten. To perform these functions, the jaws have powerful muscles capable of providing an occlusive force between the front teeth of 50 to 100 pounds and for the jaw teeth, 150 to 200 pounds. Also, the upper and lower teeth are provided with projections and facets that interdigitate, so that the upper set of teeth fits with the lower. This fitting is called occlusion, and it allows even small particles of food to be caught and ground between the tooth surfaces. Function of the Different Parts of the Teeth Figure 79– 12 shows a sagittal section of a tooth, demonstrating its major functional parts: the enamel, dentin, cementum, and pulp. The tooth can also be divided into the crown, which is the portion that protrudes out from the gum into the mouth, and the root, which is the portion within the bony socket of the jaw. The collar between the crown and the root where the tooth is surrounded by the gum is called the neck.


Enamel. The outer surface of the tooth is covered by a layer of enamel that is formed before eruption of the tooth by special epithelial cells called ameloblasts. Once the tooth has erupted, no more enamel is formed. Enamel is composed of very large and very dense crystals of hydroxyapatite with adsorbed carbonate, magnesium, sodium, potassium, and other ions imbedded in a fine meshwork of strong and almost insoluble protein fibers that are similar in physical characteristics (but not chemically identical) to the keratin of hair. The crystalline structure of the salts makes the enamel extremely hard-much harder than the dentin. Also, the special protein fiber meshwork, although constituting only about 1 per cent of the enamel mass, makes enamel resistant to acids, enzymes, and other corrosive agents because this protein is one of the most insoluble and resistant proteins known. Dentin. The main body of the tooth is composed of dentin, which has a strong, bony structure. Dentin is made up principally of hydroxyapatite crystals similar to those in bone but much more dense. These crystals are imbedded in a strong meshwork of collagen fibers. In other words, the principal constituents of dentin are very much the same as those of bone. The major difference is its histological organization, because dentin does not contain any osteoblasts, osteocytes, osteoclasts, or spaces for blood vessels or nerves. Instead, it is deposited and nourished by a layer of cells called odontoblasts, which line its inner surface along the wall of the pulp cavity. The calcium salts in dentin make it extremely resistant to compressional forces, and the collagen fibers make it tough and resistant to tensional forces that might result when the teeth are struck by solid objects.

Cementum is a bony substance secreted by cells of the periodontal membrane, which lines the tooth socket. Many collagen fibers pass directly from the bone of the jaw, through the periodontal membrane, and then into the cementum. These collagen fibers and the cementum hold the tooth in place. When the teeth are exposed to excessive strain, the layer of cementum becomes thicker and stronger. Also, it increases in thickness and strength with age, causing the teeth to become more firmly seated in the jaws by adulthood and later. Pulp. The pulp cavity of each tooth is filled with pulp, which is composed of connective tissue with an abundant supply of nerve fibers, blood vessels, and lymphatics. The cells lining the surface of the pulp cavity are the odontoblasts, which, during the formative years of the tooth, lay down the dentin but at the same time encroach more and more on the pulp cavity, making it smaller. In later life, the dentin stops growing and the pulp cavity remains essentially constant in size. However, the odontoblasts are still viable and send projections into small dentinal tubules that penetrate all the way through the dentin; they are of importance for exchange of calcium, phosphate, and other minerals with the dentin.

Dentition. Humans and most other mammals develop two sets of teeth during a lifetime. The first teeth are called the deciduous teeth, or milk teeth, and they number 20 in humans. They erupt between the 7 th month and the 2 nd year of life, and they last until the 6 th to the 13 th year. After each deciduous tooth is lost, a permanent tooth replaces it, and an additional 8 to 12 molars appear posteriorly in the jaws, making the total number of permanent teeth 28 to 32, depending on whether the four wisdom teeth finally appear, which does not occur in everyone. Formation of the Teeth. Figure 79– 13 shows the formation and eruption of teeth. Figure 79– 13 A shows invagination of the oral epithelium into the dental lamina; this is followed by the development of a tooth producing organ. The epithelial cells above form ameloblasts, which form the enamel on the outside of the tooth. The epithelial cells below invaginate upward into the middle of the tooth to form the pulp cavity and the odontoblasts that secrete dentin. Thus, enamel is formed on the outside of the tooth, and dentin is formed on the inside, giving rise to an early tooth, as shown in Figure 79– 13 B.


Eruption of Teeth. During early childhood, the teeth begin to protrude outward from the bone through the oral epithelium into the mouth. The cause of “eruption” is unknown, although several theories have been offered in an attempt to explain this phenomenon. The most likely theory is that growth of the tooth root as well as of the bone underneath the tooth progressively shoves the tooth forward. Development of the Permanent Teeth. During embryonic life, a tooth-forming organ also develops in the deeper dental lamina for each permanent tooth that will be needed after the deciduous teeth are gone. These tooth-producing organs slowly form the permanent teeth throughout the first 6 to 20 years of life. When each permanent tooth becomes fully formed, it, like the deciduous tooth, pushes outward through the bone. In so doing, it erodes the root of the deciduous tooth and eventually causes it to loosen and fall out. Soon thereafter, the permanent tooth erupts to take the place of the original one. Metabolic Factors Influence Development of the Teeth. The rate of development and the speed of eruption of teeth can be accelerated by both thyroid and growth hormones. Also, the deposition of salts in the early forming teeth is affected considerably by various factors of metabolism, such as the availability of calcium and phosphate in the diet, the amount of vitamin D present, and the rate of PTH secretion. When all these factors are normal, the dentin and enamel will be correspondingly healthy, but when they are deficient, the calcification of the teeth also may be defective, so that the teeth will be abnormal throughout life.

Mineral Exchange in Teeth The salts of teeth, like those of bone, are composed of hydroxyapatite with adsorbed carbonates and various cations bound together in a hard crystalline substance. Also, new salts are constantly being deposited while old salts are being reabsorbed from the teeth, as occurs in bone. Deposition and reabsorption occur mainly in the dentin and cementum and to a very limited extent in the enamel. In the enamel, these processes occur mostly by diffusional exchange of minerals with the saliva instead of with the fluids of the pulp cavity. The rate of absorption and deposition of minerals in the cementum is about equal to that in the surrounding bone of the jaw, whereas the rate of deposition and absorption of minerals in the dentin is only one third that of bone. The cementum has characteristics almost identical to those of usual bone, including the presence of osteoblasts and osteoclasts, whereas dentin does not have these characteristics, as explained earlier. This difference undoubtedly explains the different rates of mineral exchange. In summary, continual mineral exchange occurs in the dentin and cementum of teeth, although the mechanism of this exchange in dentin is unclear. However, enamel exhibits extremely slow mineral exchange, so that it maintains most of its original mineral complement throughout life. Dental Abnormalities The two most common dental abnormalities are caries and malocclusion. Caries refers to erosion of the teeth, whereas malocclusion is failure of the projections of the upper and lower teeth to interdigitate properly.

Caries and the Role of Bacteria and Ingested Carbohydrates. It is generally agreed that caries result from the action of bacteria on the teeth, the most common of which is Streptococcus mutans. The first event in the development of caries is the deposit of plaque, a film of precipitated products of saliva and food, on the teeth. Large numbers of bacteria inhabit this plaque and are readily available to cause caries. These bacteria depend to a great extent on carbohydrates for their food. When carbohydrates are available, their metabolic systems are strongly activated and they multiply. In addition, they form acids (particularly lactic acid) and proteolytic enzymes. The acids are the major culprit in causing caries because the calcium salts of teeth are slowly dissolved in a highly acidic medium. And once the salts have become absorbed, the remaining organic matrix is rapidly digested by the proteolytic enzymes. The enamel of the tooth is the primary barrier to the development of caries. Enamel is far more resistant to demineralization by acids than is dentin, primarily because the crystals of enamel are dense, but also because each enamel crystal is about 200 times as large in volume as each dentin crystal. Once the carious process has penetrated through the enamel to the dentin, it proceeds many times as rapidly because of the high degree of solubility of the dentin salts. Because of the dependence of the caries bacteria on carbohydrates for their nutrition, it has frequently been taught that eating a diet high in carbohydrate content will lead to excessive development of caries. However, it is not the quantity of carbohydrate ingested but the frequency with which it is eaten that is important. If carbohydrates are eaten in many small parcels throughout the day, such as in the form of candy, the bacteria are supplied with their preferential metabolic substrate for many hours of the day, and the development of caries is greatly increased.

Role of Fluorine in Preventing Caries. Teeth formed in children who drink water that contains small amounts of fluorine develop enamel that is more resistant to caries than the enamel in children who drink water that does not contain fluorine. Fluorine does not make the enamel harder than usual, but fluorine ions replace many of the hydroxyl ions in the hydroxyapatite crystals, which in turn makes the enamel several times less soluble. Fluorine may also be toxic to the bacteria. Finally, when small pits do develop in the enamel, fluorine is believed to promote deposition of calcium phosphate to “heal” the enamel surface. Regardless of the precise means by which fluorine protects the teeth, it is known that small amounts of fluorine deposited in enamel make teeth about three times as resistant to caries as teeth without fluorine. Malocclusion is usually caused by a hereditary abnormality that causes the teeth of one jaw to grow to abnormal positions. In malocclusion, the teeth do not interdigitate properly and therefore cannot perform their normal grinding or cutting action adequately. Malocclusion occasionally also results in abnormal displacement of the lower jaw in relation to the upper jaw, causing such undesirable effects as pain in the mandibular joint and deterioration of the teeth. The orthodontist can usually correct malocclusion by applying prolonged gentle pressure against the teeth with appropriate braces. The gentle pressure causes absorption of alveolar jaw bone on the compressed side of the tooth and deposition of new bone on the tensional side of the tooth. In this way, the tooth gradually moves to a new position as directed by the applied pressure.
- Slides: 66