Honors Chemistry Organic Chemistry organic chemistry the study








- Slides: 45

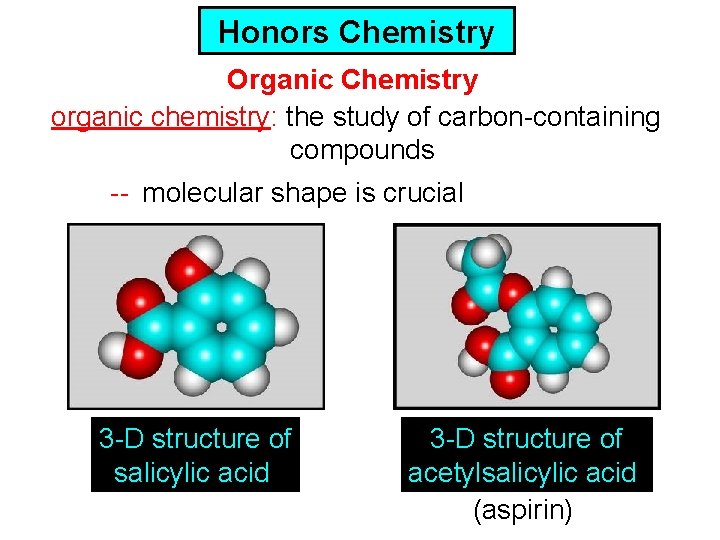
Honors Chemistry Organic Chemistry organic chemistry: the study of carbon-containing compounds -- molecular shape is crucial 3 -D structure of salicylic acid 3 -D structure of acetylsalicylic acid (aspirin)

Carbon is unique among the elements because: -- it can have up to four bonds per C atom single, double, or triple bonds –C–C– C=C –C=C– -- it can form REALLY long chains of C atoms -- its bonds are strong

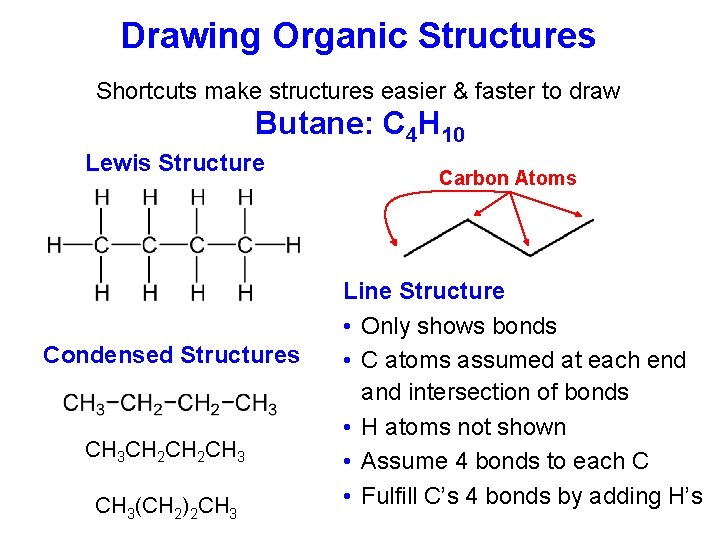
Drawing Organic Structures Shortcuts make structures easier & faster to draw Butane: C 4 H 10 Lewis Structure Condensed Structures CH 3 CH 2 CH 3(CH 2)2 CH 3 Carbon Atoms Line Structure • Only shows bonds • C atoms assumed at each end and intersection of bonds • H atoms not shown • Assume 4 bonds to each C • Fulfill C’s 4 bonds by adding H’s

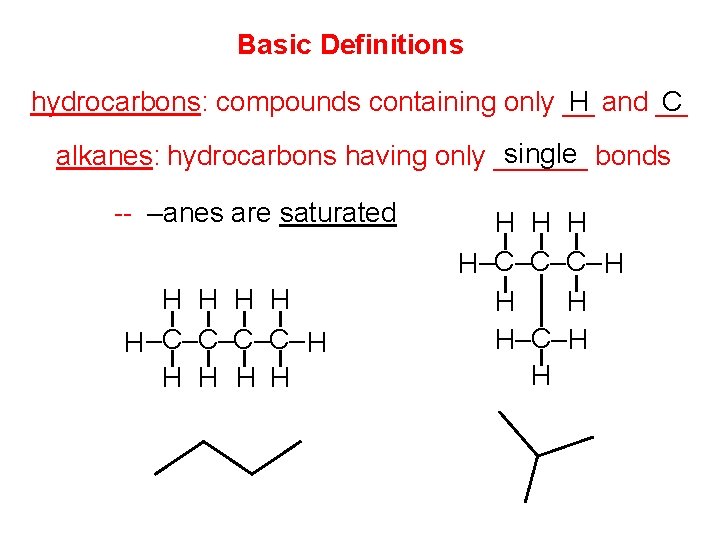
Basic Definitions hydrocarbons: compounds containing only __ H and __ C single bonds alkanes: hydrocarbons having only ______ -- –anes are saturated H H H –C–C– H H H H H–C–C–C– H H–C– H H

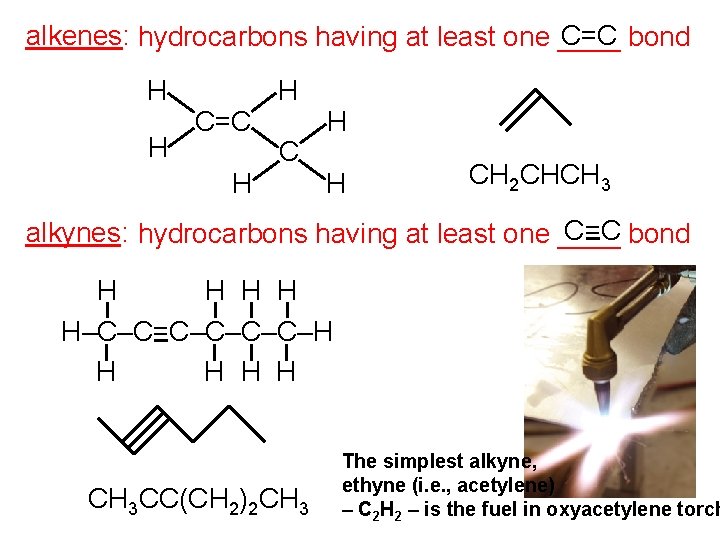
alkenes: hydrocarbons having at least one ____ C=C bond H H C=C H H H CH 2 CHCH 3 C=C bond alkynes: hydrocarbons having at least one ____ H H H–C–C=C–C–H H H CH 3 CC(CH 2)2 CH 3 The simplest alkyne, ethyne (i. e. , acetylene) – C 2 H 2 – is the fuel in oxyacetylene torch

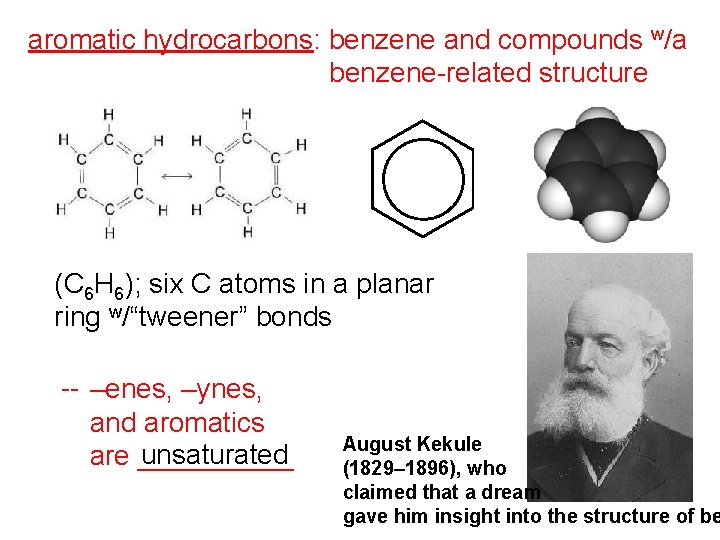
aromatic hydrocarbons: benzene and compounds w/a benzene-related structure (C 6 H 6); six C atoms in a planar ring w/“tweener” bonds -- –enes, –ynes, and aromatics unsaturated are _____ August Kekule (1829– 1896), who claimed that a dream gave him insight into the structure of be

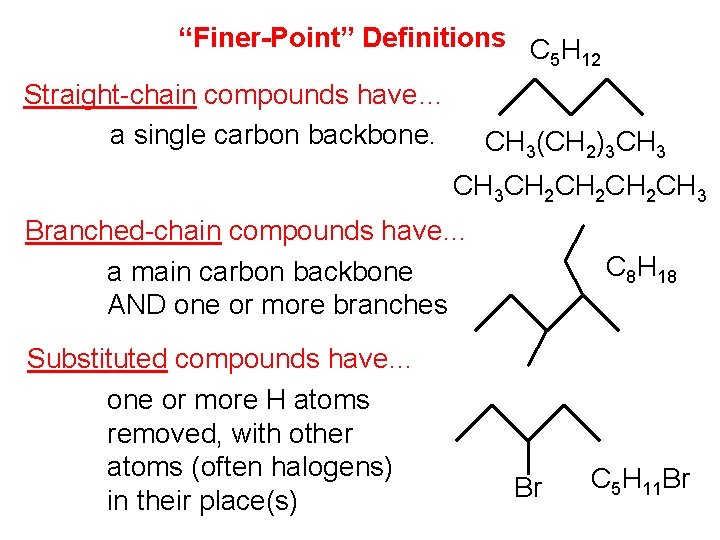
“Finer-Point” Definitions C H 5 12 Straight-chain compounds have… a single carbon backbone. CH 3(CH 2)3 CH 3 CH 2 CH 2 CH 3 Branched-chain compounds have… a main carbon backbone AND one or more branches Substituted compounds have… one or more H atoms removed, with other atoms (often halogens) in their place(s) C 8 H 18 Br C 5 H 11 Br

Branches and H-replacing atoms/groups are collectively called… substituents. Cl H –C–C–C– H H H Polyvinylchloride (PVC) is a material made of long-chain carbons that have many chloride substituents.

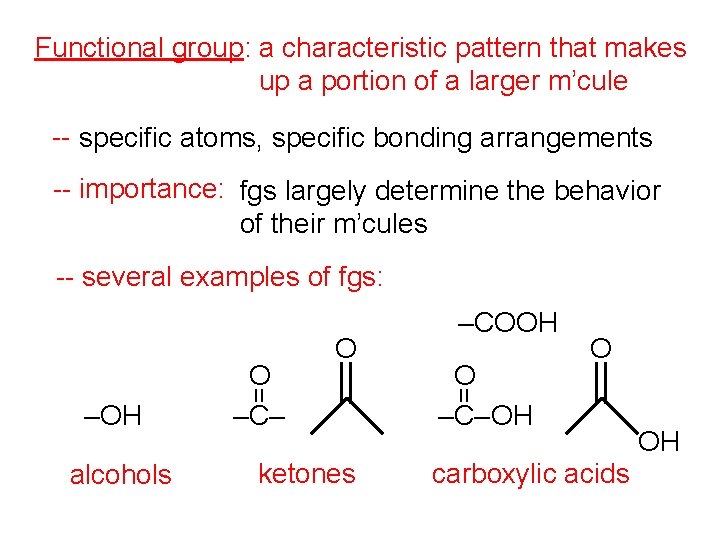
Functional group: a characteristic pattern that makes up a portion of a larger m’cule -- specific atoms, specific bonding arrangements -- importance: fgs largely determine the behavior of their m’cules -- several examples of fgs: alcohols ketones O –C– OH O = = –OH O –C– O –COOH carboxylic acids OH

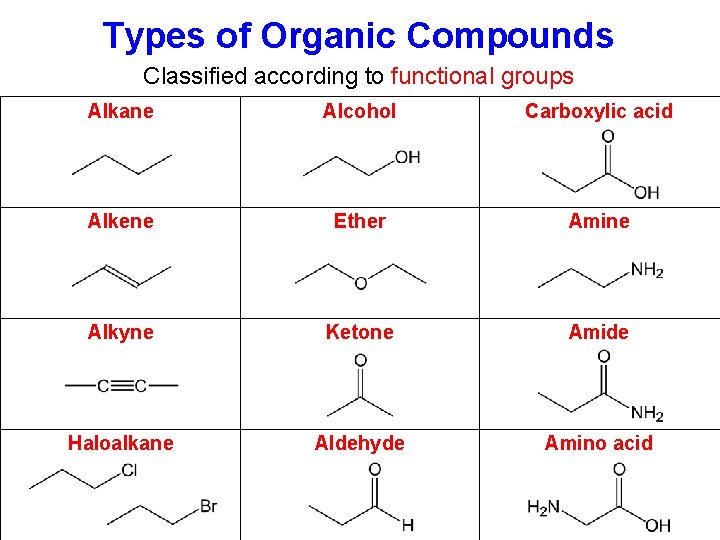
Types of Organic Compounds Classified according to functional groups Alkane Alcohol Carboxylic acid Alkene Ether Amine Alkyne Ketone Amide Haloalkane Aldehyde Amino acid

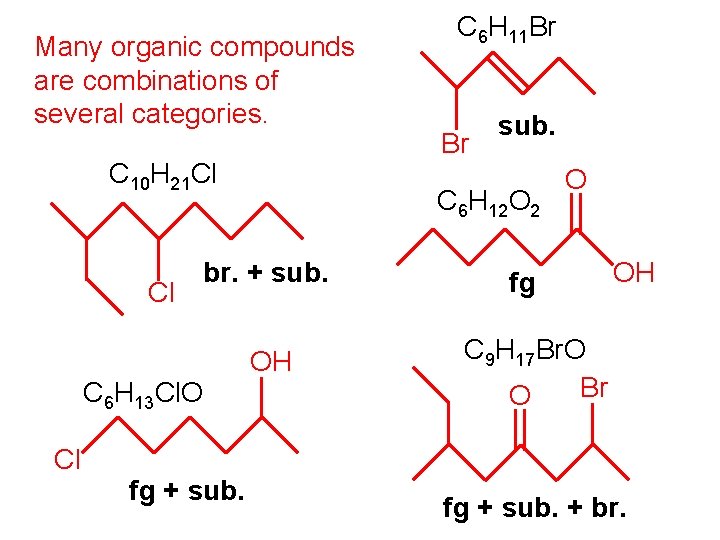
Many organic compounds are combinations of several categories. C 10 H 21 Cl Cl Cl fg + sub. Br sub. C 6 H 12 O 2 br. + sub. C 6 H 13 Cl. O C 6 H 11 Br OH O fg OH C 9 H 17 Br. O Br O fg + sub. + br.

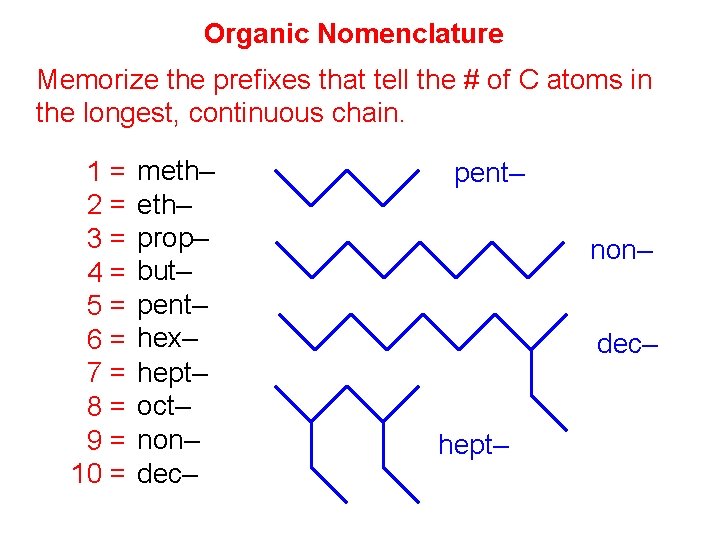
Organic Nomenclature Memorize the prefixes that tell the # of C atoms in the longest, continuous chain. 1= 2= 3= 4= 5= 6= 7= 8= 9= 10 = meth– prop– but– pent– hex– hept– oct– non– dec– pent– non– dec– hept–

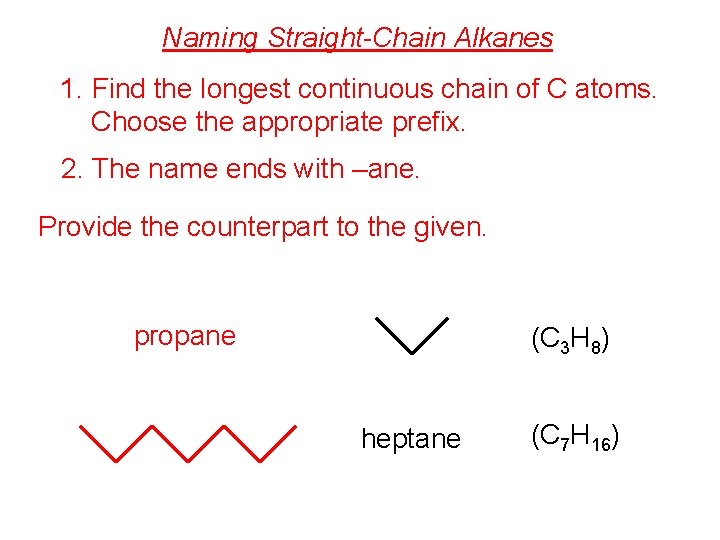
Naming Straight-Chain Alkanes 1. Find the longest continuous chain of C atoms. Choose the appropriate prefix. 2. The name ends with –ane. Provide the counterpart to the given. propane (C 3 H 8) heptane (C 7 H 16)

Alkanes: modification for substituent hydrocarbon (HC) groups (i. e. , branches) 1. Number the “longest chain” carbons. Start with the end nearest a branch. 2. Name and give the #ed location of each substituent. -- HC substituent groups use the prefixes, but end in –yl. 3. List substituents in alphabetical order.

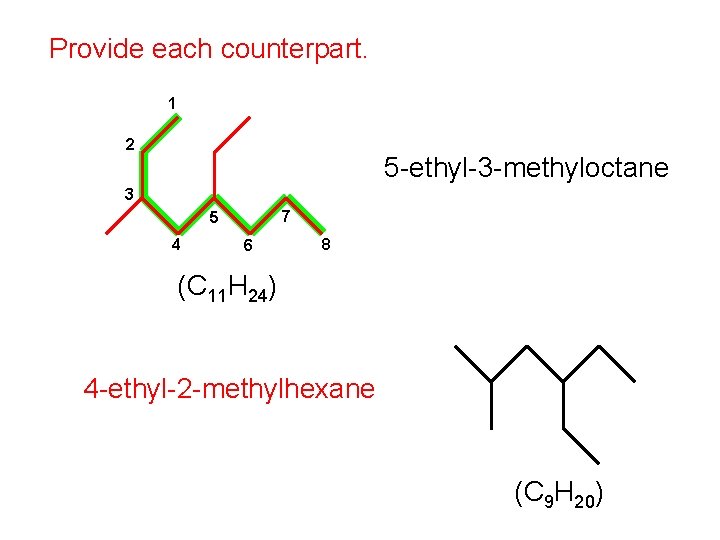
Provide each counterpart. 1 2 5 -ethyl-3 -methyloctane 3 7 5 4 6 8 (C 11 H 24) 4 -ethyl-2 -methylhexane (C 9 H 20)

Alkanes: modification for non-HC substitutions 1. The “longest chain” MUST “hold” the substituent. -- example substituents: –NO 2 nitro –NH 2 amino –F fluoro –Br bromo –I iodo 2. Number the chain carbons, starting with the end nearest a substituent. -- A non-HC substituent takes precedence over an HC branch. 3. Name and give the #ed location of each substituent. -- If necessary, choose #s so that their sum is as low as possible.

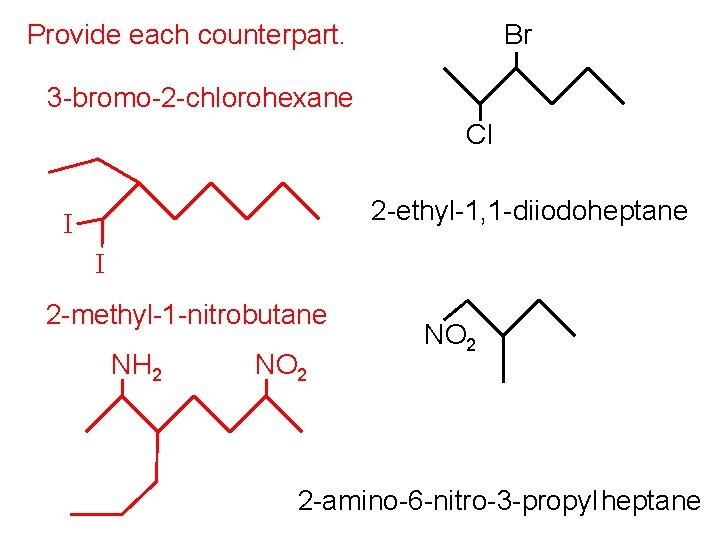
Provide each counterpart. Br 3 -bromo-2 -chlorohexane Cl 2 -ethyl-1, 1 -diiodoheptane I I 2 -methyl-1 -nitrobutane NH 2 NO 2 2 -amino-6 -nitro-3 -propyl heptane

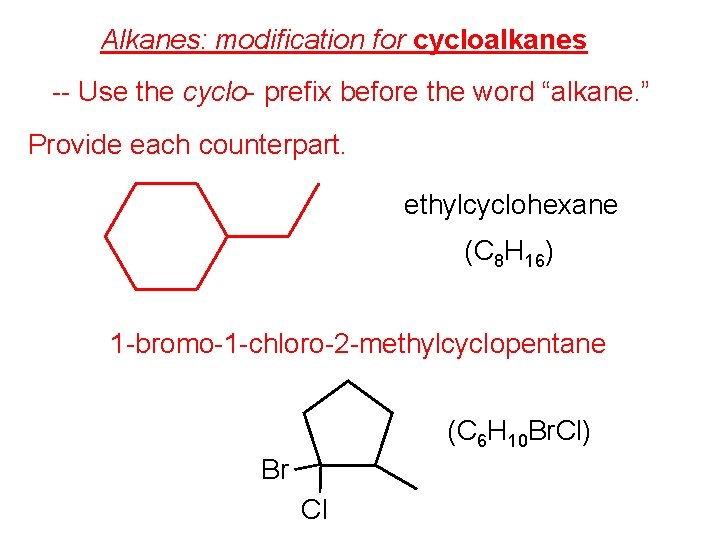
Alkanes: modification for cycloalkanes -- Use the cyclo- prefix before the word “alkane. ” Provide each counterpart. ethylcyclohexane (C 8 H 16) 1 -bromo-1 -chloro-2 -methylcyclopentane (C 6 H 10 Br. Cl) Br Cl

Naming Alkenes and Alkynes 1. The C-chain MUST include the multiple bond. Use –ene or –yne, as appropriate. 2. Number so that you get to the multiple bond ASAP. -- The multiple bond takes precedence over branching or substituents. 3. Use di- or tri- right before –ene or –yne if you have two or three multiple bonds. Polyunsaturated fats and oils contain double and/or triple bonds.

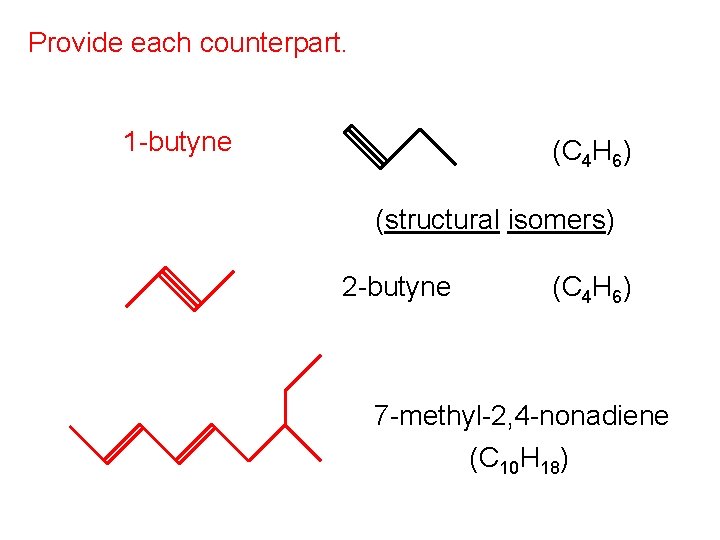
Provide each counterpart. 1 -butyne (C 4 H 6) (structural isomers) 2 -butyne (C 4 H 6) 7 -methyl-2, 4 -nonadiene (C 10 H 18)

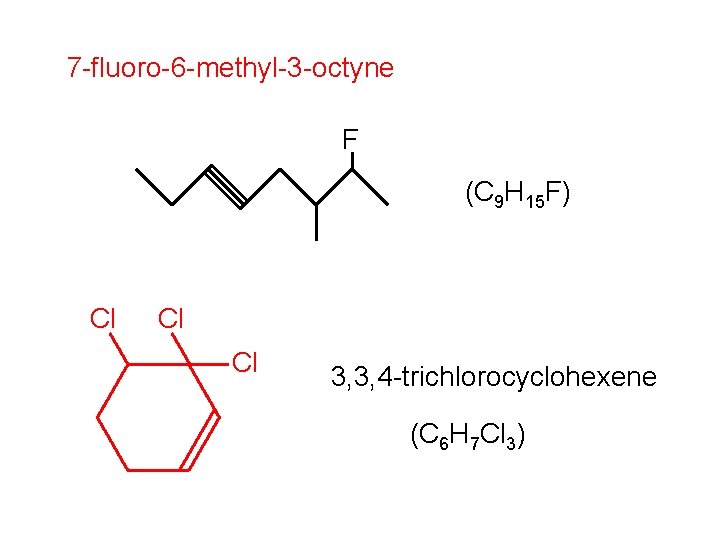
7 -fluoro-6 -methyl-3 -octyne F (C 9 H 15 F) Cl Cl Cl 3, 3, 4 -trichlorocyclohexene (C 6 H 7 Cl 3)

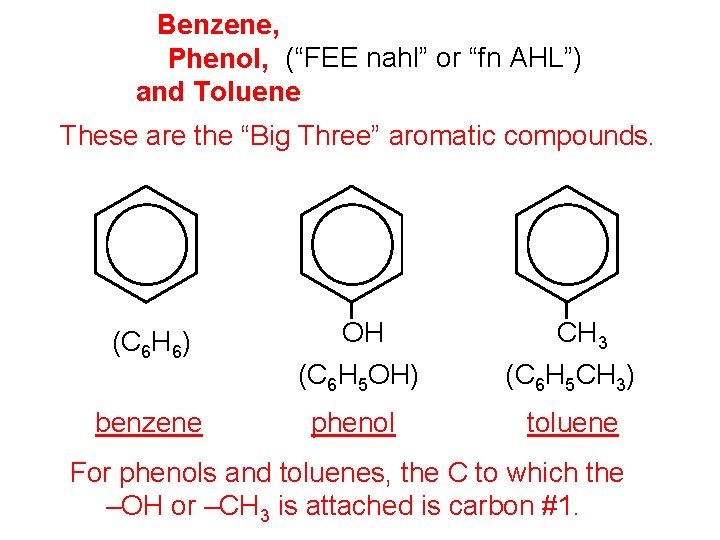
Benzene, Phenol, (“FEE nahl” or “fn AHL”) and Toluene These are the “Big Three” aromatic compounds. (C 6 H 6) OH (C 6 H 5 OH) CH 3 (C 6 H 5 CH 3) benzene phenol toluene For phenols and toluenes, the C to which the –OH or –CH 3 is attached is carbon #1.

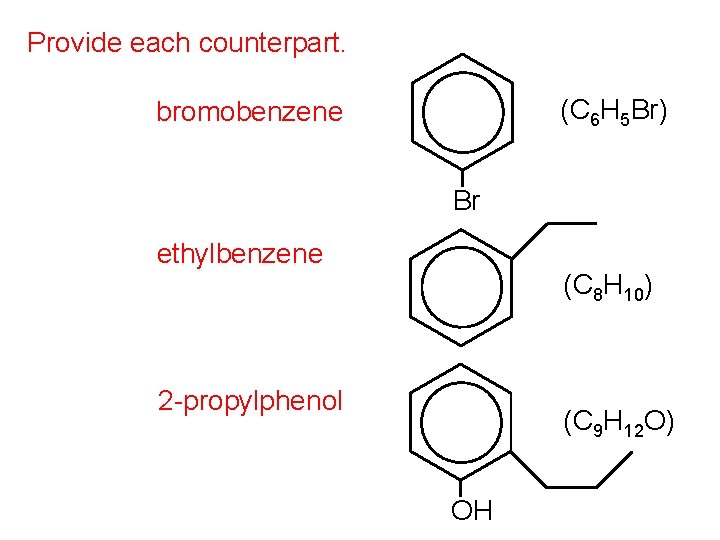
Provide each counterpart. (C 6 H 5 Br) bromobenzene Br ethylbenzene (C 8 H 10) 2 -propylphenol (C 9 H 12 O) OH

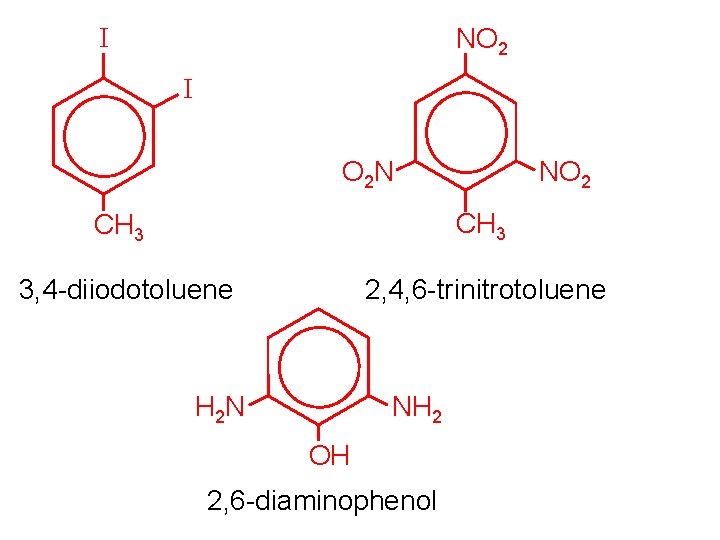
I NO 2 I O 2 N NO 2 CH 3 3, 4 -diiodotoluene 2, 4, 6 -trinitrotoluene H 2 N NH 2 OH 2, 6 -diaminophenol

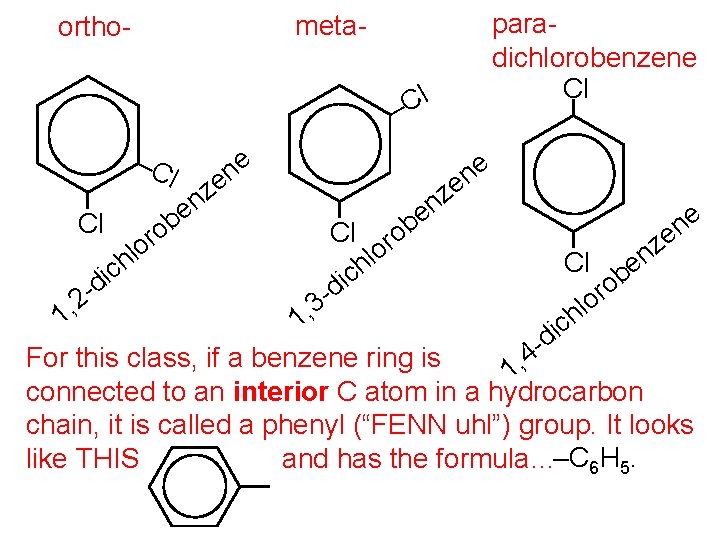
paradichlorobenzene Cl meta- ortho- l C – –C l Cl b o r o hl c di , 2 z n e e n e Cl d 3 ic r o hl ob z n e Cl b o r o l ich e n e z n e 1 1, d 4 For this class, if a benzene ring is 1, connected to an interior C atom in a hydrocarbon chain, it is called a phenyl (“FENN uhl”) group. It looks like THIS and has the formula…–C 6 H 5.

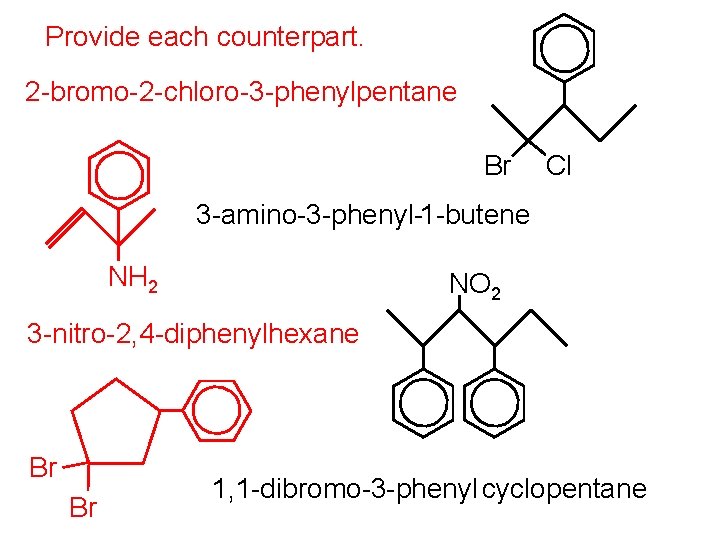
Provide each counterpart. 2 -bromo-2 -chloro-3 -phenylpentane Br Cl 3 -amino-3 -phenyl-1 -butene NH 2 NO 2 3 -nitro-2, 4 -diphenylhexane Br Br 1, 1 -dibromo-3 -phenyl cyclopentane

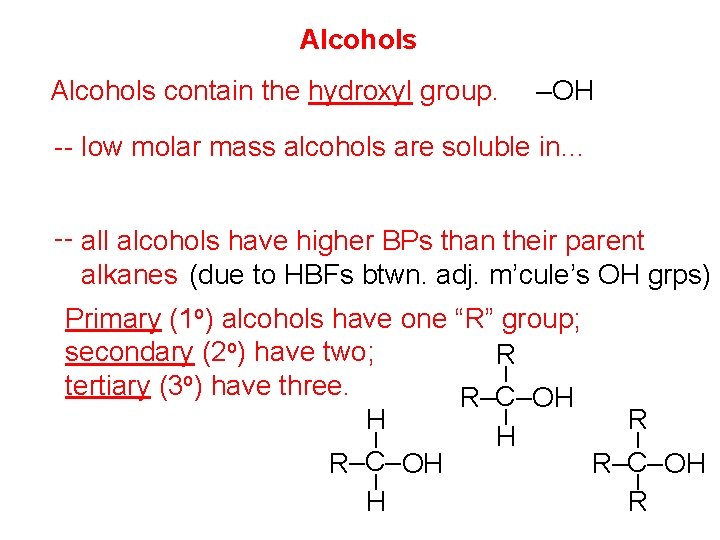
Alcohols contain the hydroxyl group. –OH -- low molar mass alcohols are soluble in… polar solvents, due to the polar nature of –OH -- all alcohols have higher BPs than their parent alkanes (due to HBFs btwn. adj. m’cule’s OH grps) – – – Primary (1 o) alcohols have one “R” group; secondary (2 o) have two; R tertiary (3 o) have three. R–C– OH H R–C– OH H R

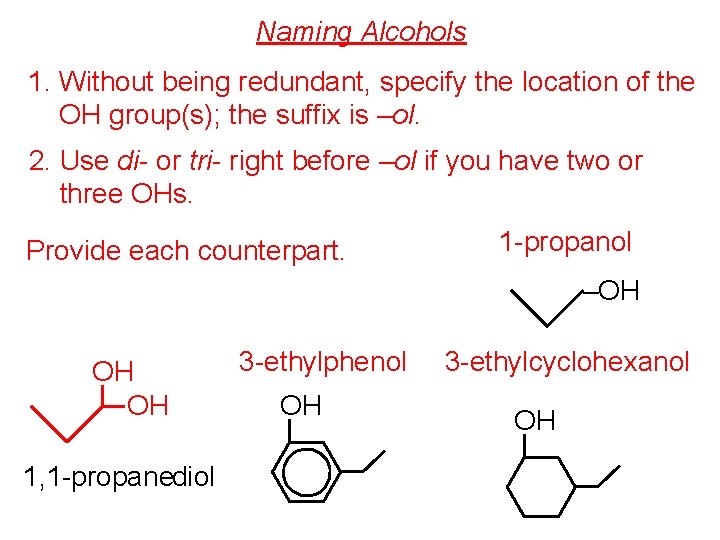
Naming Alcohols 1. Without being redundant, specify the location of the OH group(s); the suffix is –ol. 2. Use di- or tri- right before –ol if you have two or three OHs. Provide each counterpart. 1 -propanol –OH OH OH 1, 1 -propanediol 3 -ethylphenol OH 3 -ethylcyclohexanol OH

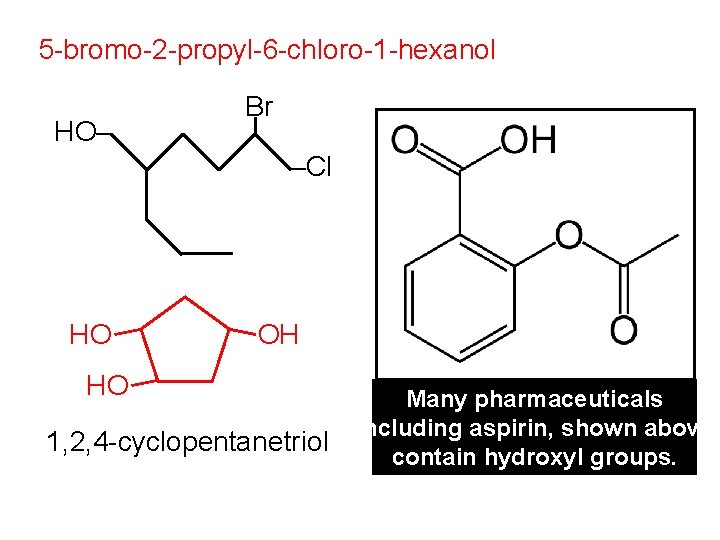
5 -bromo-2 -propyl-6 -chloro-1 -hexanol HO– Br –Cl HO HO OH Many pharmaceuticals (including aspirin, shown above) 1, 2, 4 -cyclopentanetriol contain hydroxyl groups.

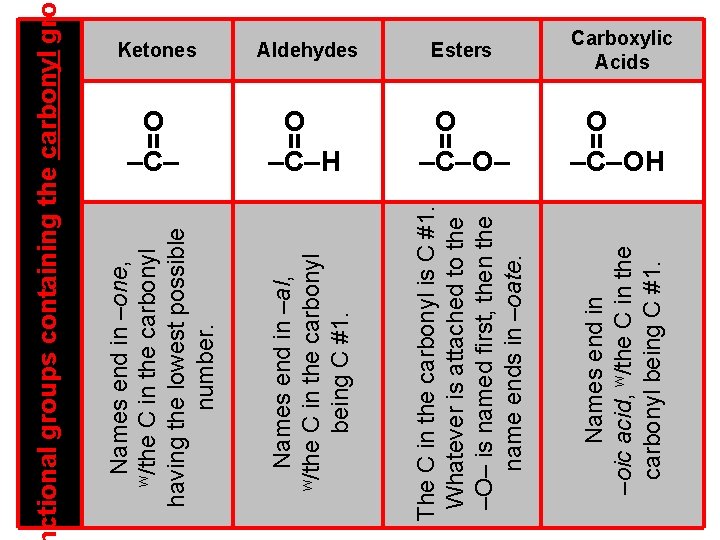
Carboxylic Acids O –C– H O –C– O– O –C– OH Names end in –oic acid, w/the C in the carbonyl being C #1. = = = Esters The C in the carbonyl is C #1. Whatever is attached to the –O– is named first, then the name ends in –oate. = Aldehydes Names end in –al, w/the C in the carbonyl being C #1. Names end in –one, w/the C in the carbonyl having the lowest possible number. ctional groups containing the carbonyl gro Ketones

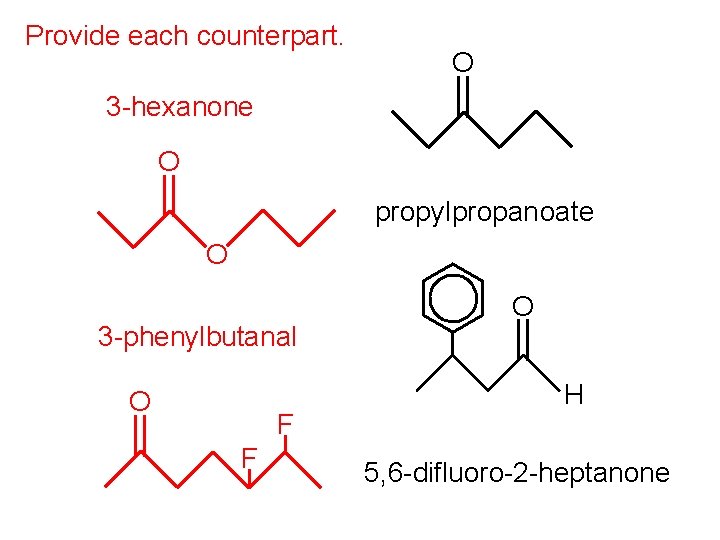
Provide each counterpart. O 3 -hexanone O propylpropanoate O 3 -phenylbutanal O F F O H 5, 6 -difluoro-2 -heptanone

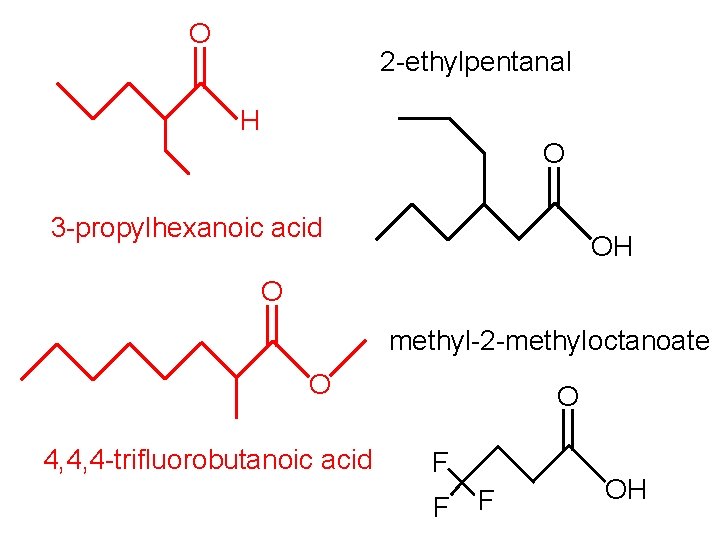
O 2 -ethylpentanal H O 3 -propylhexanoic acid OH O methyl-2 -methyloctanoate O 4, 4, 4 -trifluorobutanoic acid O F F F OH

Other Functional Groups to Recognize Ethers Amines Amides (“EETH erz”) (“uh MEENZ”) (“uh MIDZ” or “AM idz”) –N “coca-ine” “Tatoo-ine” “caffe-ine” = –O– O –C–N

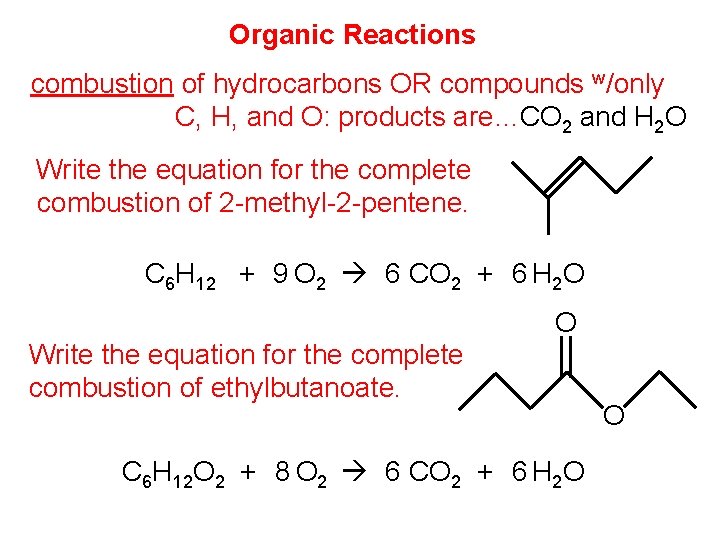
Organic Reactions combustion of hydrocarbons OR compounds w/only C, H, and O: products are…CO 2 and H 2 O Write the equation for the complete combustion of 2 -methyl-2 -pentene. C 6 H 12 + 9 O 2 6 CO 2 + 6 H 2 O Write the equation for the complete combustion of ethylbutanoate. O C 6 H 12 O 2 + 8 O 2 6 CO 2 + 6 H 2 O O

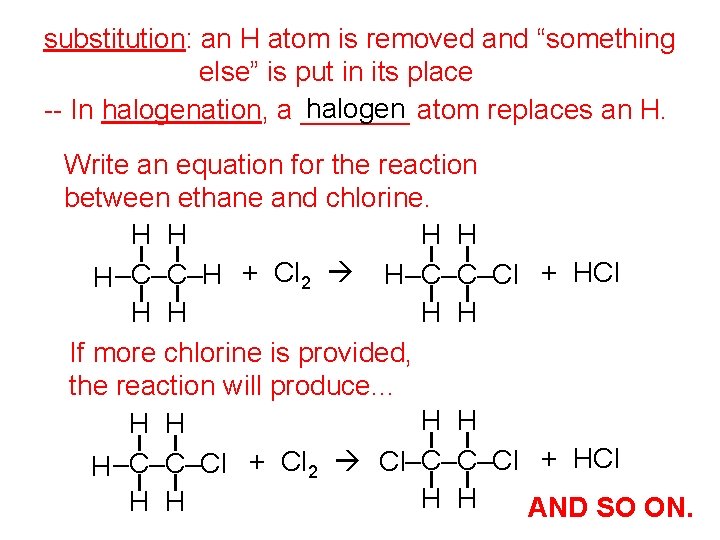
substitution: an H atom is removed and “something else” is put in its place halogen atom replaces an H. -- In halogenation, a _______ Write an equation for the reaction between ethane and chlorine. H H H –C–C–H + Cl 2 H–C–C–Cl + HCl H H If more chlorine is provided, the reaction will produce. . . H H H –C–C–Cl + Cl 2 Cl–C–C–Cl + HCl H H AND SO ON.

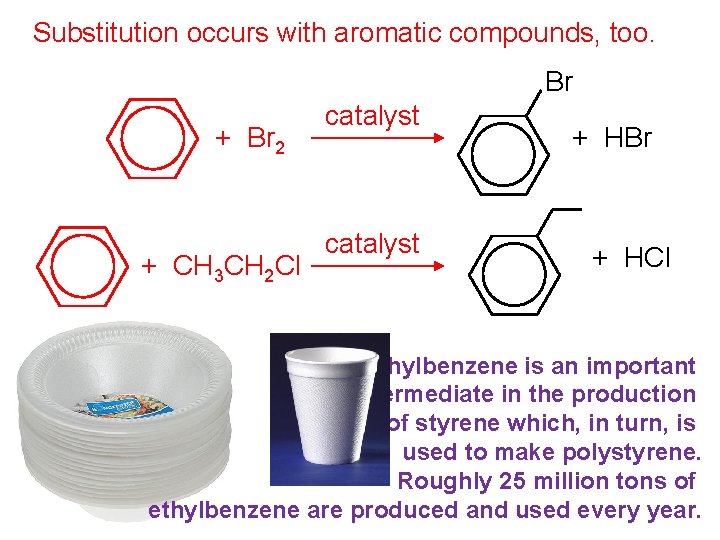
Substitution occurs with aromatic compounds, too. Br + Br 2 + CH 3 CH 2 Cl catalyst + HBr + HCl Ethylbenzene is an important intermediate in the production of styrene which, in turn, is used to make polystyrene. Roughly 25 million tons of ethylbenzene are produced and used every year.

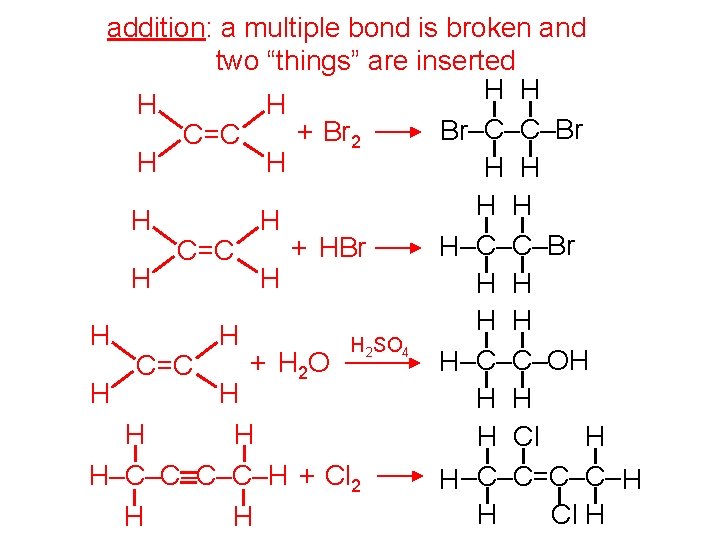
addition: a multiple bond is broken and two “things” are inserted H H Br–C–C–Br + Br 2 C=C H H H H H–C–C–Br + HBr C=C H H H H H 2 SO 4 H–C–C–OH + H 2 O C=C H H H H Cl H H–C–C–H + Cl 2 H –C–C=C–C– H H Cl H H H

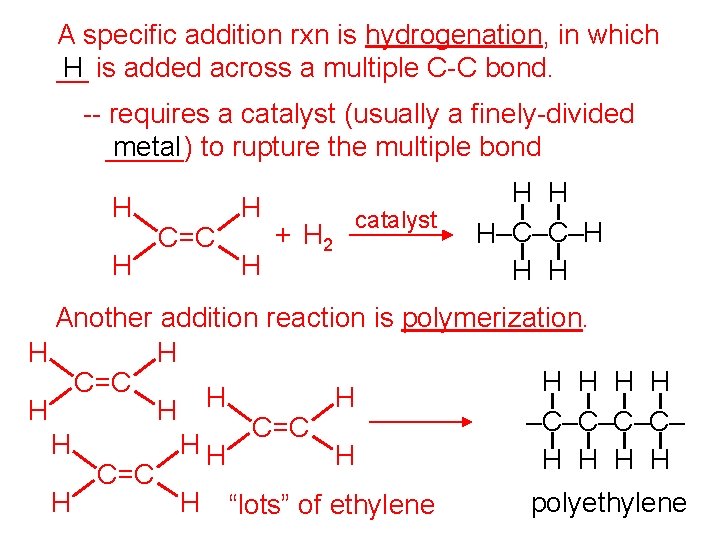
A specific addition rxn is hydrogenation, in which __ H is added across a multiple C-C bond. -- requires a catalyst (usually a finely-divided _____) metal to rupture the multiple bond H H C=C H H + H 2 catalyst H H H–C–C–H H H Another addition reaction is polymerization. H H H C=C H H –C–C– C=C H HH H H C=C H H “lots” of ethylene polyethylene

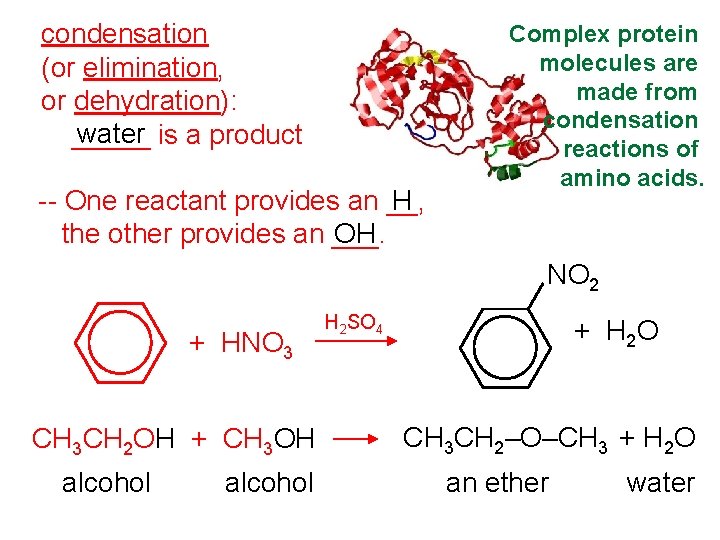
condensation (or elimination, or dehydration): water is a product _____ -- One reactant provides an __, H the other provides an ___. OH Complex protein molecules are made from condensation reactions of amino acids. NO 2 + HNO 3 CH 3 CH 2 OH + CH 3 OH alcohol H 2 SO 4 + H 2 O CH 3 CH 2–O–CH 3 + H 2 O an ether water


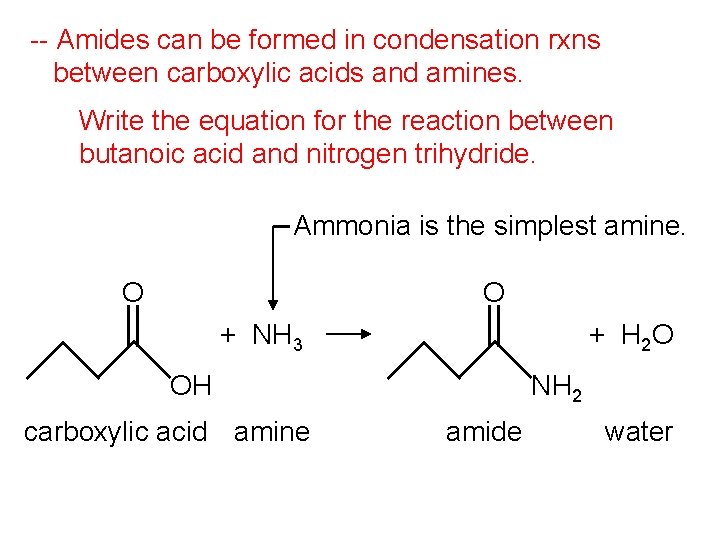
-- Amides can be formed in condensation rxns between carboxylic acids and amines. Write the equation for the reaction between butanoic acid and nitrogen trihydride. Ammonia is the simplest amine. O O + NH 3 + H 2 O OH carboxylic acid amine NH 2 amide water


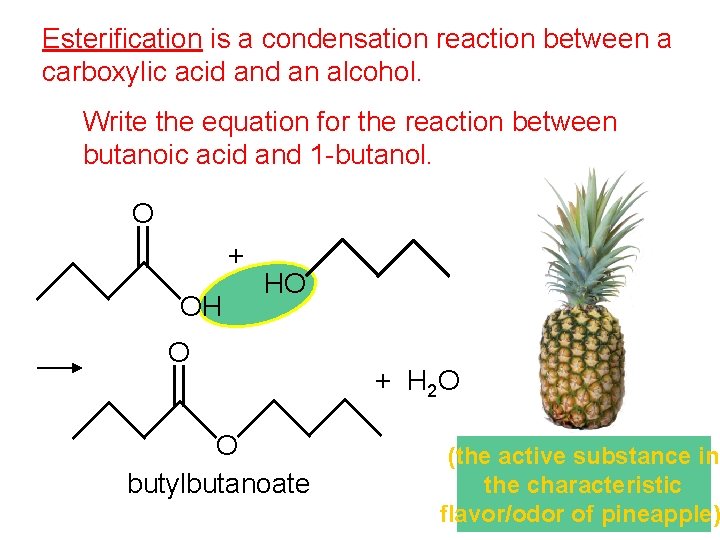
Esterification is a condensation reaction between a carboxylic acid an alcohol. Write the equation for the reaction between butanoic acid and 1 -butanol. O + OH HO O O butylbutanoate + H 2 O (the active substance in the characteristic flavor/odor of pineapple)

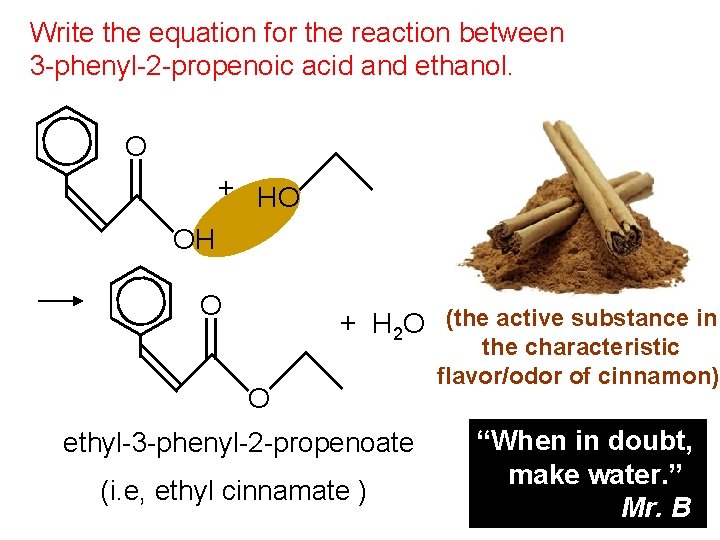
Write the equation for the reaction between 3 -phenyl-2 -propenoic acid and ethanol. O + HO OH O + H 2 O (the active substance in O ethyl-3 -phenyl-2 -propenoate (i. e, ethyl cinnamate ) the characteristic flavor/odor of cinnamon) “When in doubt, make water. ” Mr. B
