Homogeneous Mixtures A mixture that appears to be

Homogeneous Mixtures A mixture that appears to be the same throughout. It is “well mixed”. The particles in the mixture are very small and not easily recognizable. Examples: sugar water salt water milk toothpaste

Solutions A solution is a type of homogeneous mixture formed when one substance dissolves in another. It is the best mixed of all mixtures. A solution always has a substance that is dissolved and a substance that does the dissolving. The substance that is dissolved is the solute and the substance that does the dissolving is the solvent.

Water: The Universal Solvent Many liquid solutions contain water as the solvent. Ocean water is basically a water solution that contains many salts. Body fluids are also water solutions.

Types of Solutions Solute Gas Solvent Gas Liquid Ex. Air Soda water Solute Oxygen Carbon dioxide Solvent Nitrogen Water Solid Liquid Salt Water Solid Ocean water Gold jewelry Copper Gold

What is Solubility? Solubility: the maximum amount of solute that Solubility: will dissolve in a certain amount of solvent at a given temperature Solute = substance dissolved Solute Solvent = does the dissolving Solvent Example: __ grams of salt (Na. Cl) dissolved in 100 g of water at __ ˚C.

What affects Solubility? Surface Area: It affects how quickly a substance dissolves. The greater the surface area, the quicker it dissolves. Agitation: This means how much energy is being put into mixing the solute once put into the solvent. The more energy, the quicker the rate of solubility. Temperature

Temperature and Solubility Temperature affects how much of the solute can Temperature be dissolved by the solvent. Note: Increasing the temperature does not always increase the solubility…think about what always happens to dissolved gas in your soda when its heated Solubility curves: Are used to show the curves solubility of a substance changes with temperature.
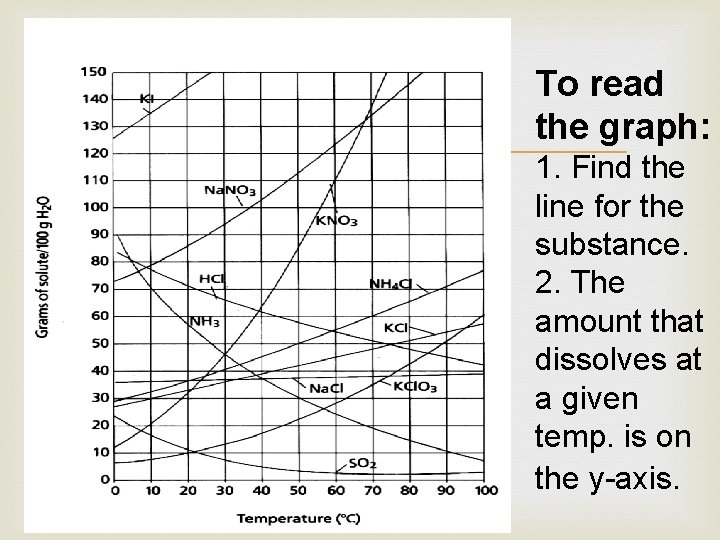
To read the graph: 1. Find the line for the substance. 2. The amount that dissolves at a given temp. is on the y-axis.

Chemical Compounds KI = Potassium iodide Na. NO 3 = Sodium nitrate KNO 3 = Potassium nitrate HCl = Hydrogen chloride NH 3 = Ammonia SO 2 = Sulfur dioxide NH 4 Cl = Ammonia chloride KCl = Potassium chloride Na. Cl = Sodium chloride (salt) KCl. O 3 = Potassium chlorate KBr = Potassium bromide Na 2 SO 3 = Sodium sulfite
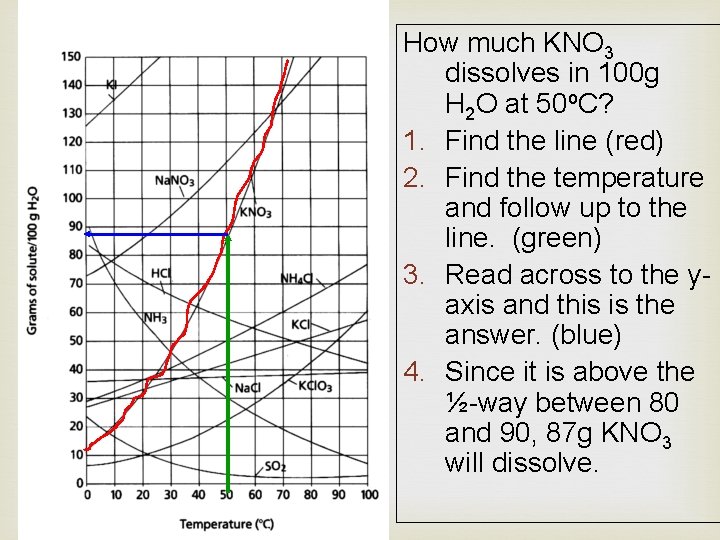
How much KNO 3 dissolves in 100 g H 2 O at 50 o. C? 1. Find the line (red) 2. Find the temperature and follow up to the line. (green) 3. Read across to the yaxis and this is the answer. (blue) 4. Since it is above the ½-way between 80 and 90, 87 g KNO 3 will dissolve.

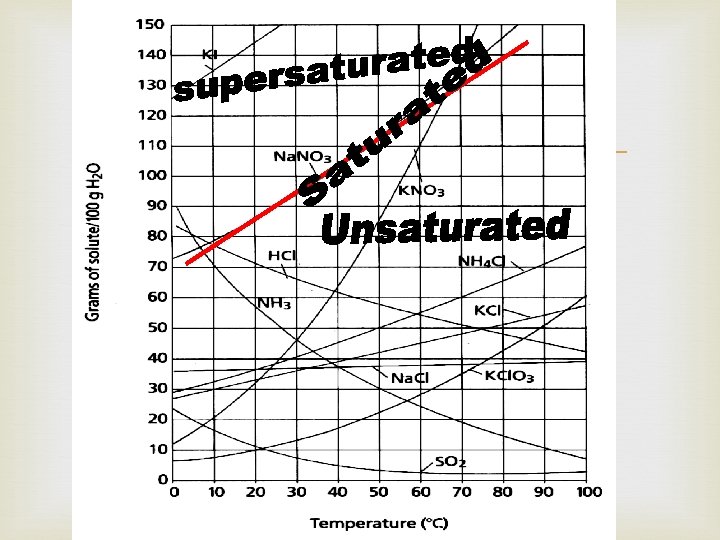
Types of Solutions: Saturated solution: point on the line Contains maximum amount of solute at given temp Contains what it should hold Supersaturated: above the line Contains more solute than a saturated solution Contains more than it should hold Unsaturated: below the line is Contains less solute than saturated solution Contains less solute than it could hold
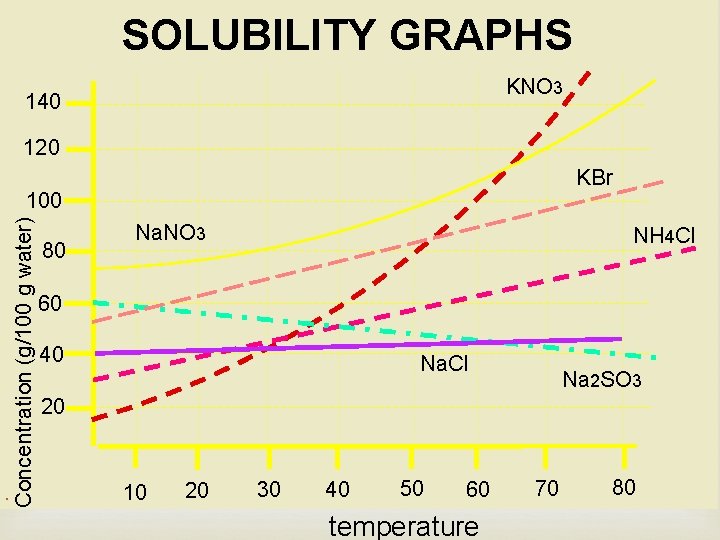
SOLUBILITY GRAPHS KNO 3 140 120 KBr . Concentration (g/100 g water) 100 80 Na. NO 3 NH 4 Cl 60 40 Na. Cl Na 2 SO 3 20 10 20 30 40 50 60 temperature 70 80
Solubility Curve Video Log onto Discovery Ed before clicking on the link below: https: //app. discoveryeducation. com/learn/player/76 fb b 860 -6 dc 5 -472 d-abf 7 -9 fff 73 b 5 ee 3 d

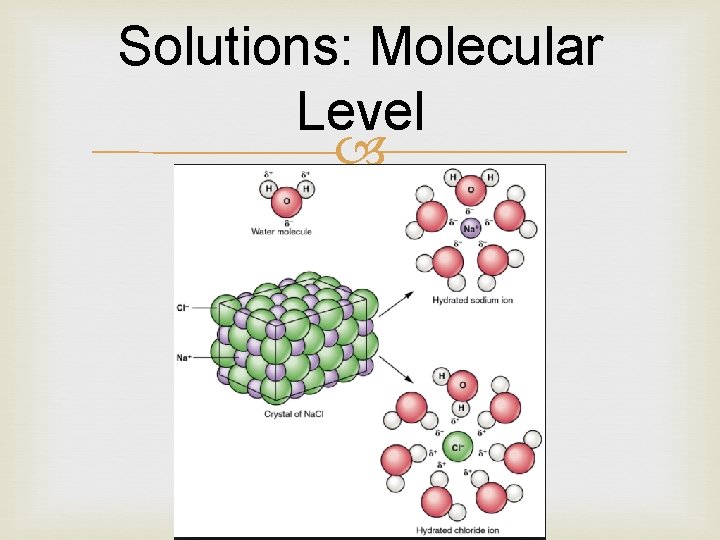
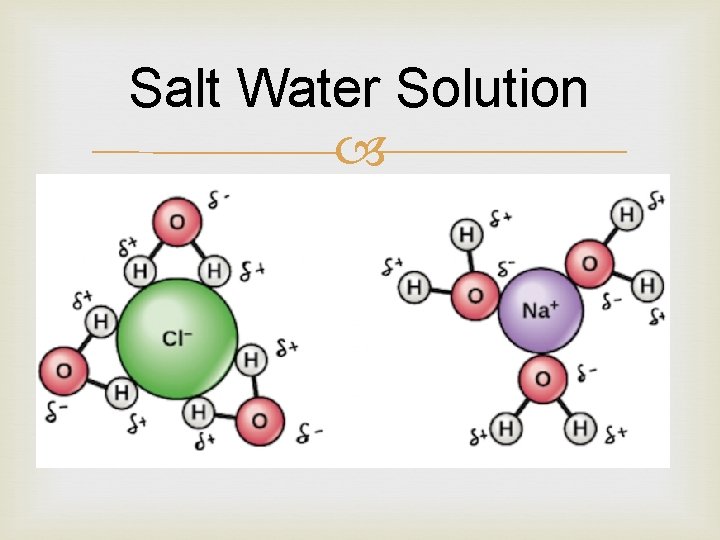
What happens in solutions? If you stir table salt into water, the crystal lattice of Na. Cl will begin to dissociate into Na+ and Cl− Dissociation is just a name for the process in which compound or molecule breaks apart to form ions. Water molecules form hydration shells around the ions: positively charged Na+ ions are surrounded by partial negative charges from the oxygen ends of the water molecules, while negatively charged Cl− ions are surrounded by partial positive charges from the hydrogen ends. As the process continues, all of the ions in the table salt crystals are surrounded by hydration shells and dispersed in solution.
Solutions: Molecular Level
Salt Water Solution

Discovery Ed Video Log onto Discovery Ed before clicking on the link: https: //app. discoveryeducation. com/learn/player/624 a 8392 -bf 24 -45 cb-aa 2 f-7233 b 628 d 031
- Slides: 18