Homogeneous Catalysis A Personal View Matthias Beller Homogeneous

Homogeneous Catalysis – A Personal View Matthias Beller

Homogeneous Catalysis A Personal View 1. Introduction

Life Relies on Chemical Reactions What does these processes have in common ?
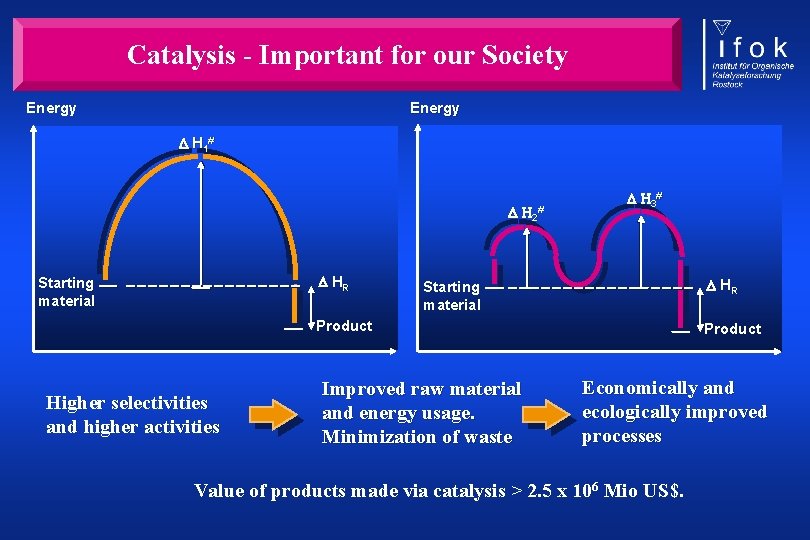
Catalysis - Important for our Society Energy H 1# H 2# HR Starting material H 3# HR Starting material Product Higher selectivities and higher activities Improved raw material and energy usage. Minimization of waste Product Economically and ecologically improved processes Value of products made via catalysis > 2. 5 x 106 Mio US$.

Worldwide Merchant Catalyst Markets Chemical 1997 Chemical 2003 2. 0 2. 2 Polymer 1. 7 2. 1 Refinery 1. 6* Environmental Billion US$ 7. 4 2. 3 Refinery Polymer 2. 1* Environmental 9. 0 * toll manufacturing fees only The Catalyst Group: The Intelligence Report: Global Shifts in the Catalyst Industry
Catalysis - Research for the 21 th Century Fine chemicals Basic chemicals New materials Health Homogeneous catalysis Biocatalysis Photocatalysis Catalysis Molecular biology Heterogeneous catalysis Electrocatalysis Food Environmental technologies Energy

Catalysis research at the If. OK Branch office in Warnemünde l l First Institute for Catalysis Research in Europe; founded 1952. 2003: ca. 90 Employees (18 permanent scientists, >20 postdocs, 13 Ph. D. students). Background and main expertise is homogeneous catalysis, based on transition metal complexes Transfer of excellent basic research to industrial applications

Homogenous versus Heterogenous Catalysis Mono phase Biphase/multiple phase Activity (of metal content) high variable selectivity high variable Conditions of reaction mild harsh Life-time of catalyst variable long Sensitivity of deactivation low high Problems due to diffusion none difficult to solve Recycling of catalyst usually difficult can easily be done Steric and electronic properties easily changed no variation possible Mechanism realistic models do exist not obvious

Homogenous Catalysis A Personal View 2. Biphasic Catalysis and Hydroformylation
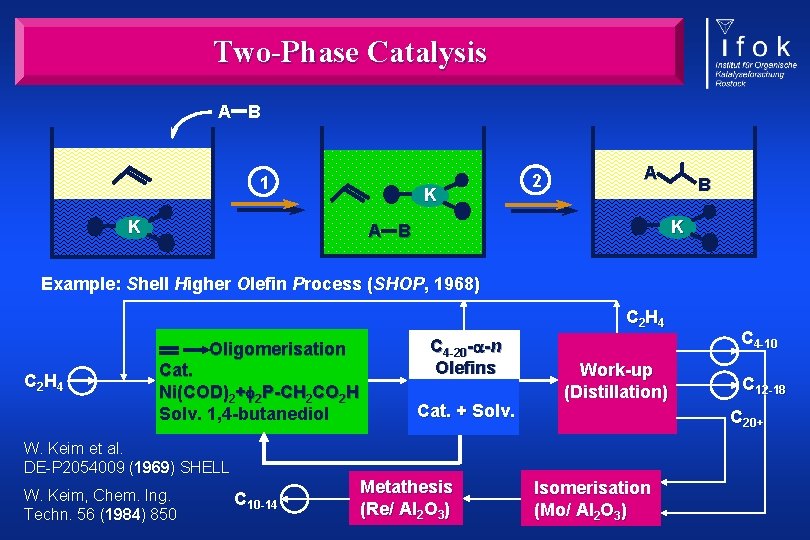
Two-Phase Catalysis A B 1 K K 2 A B K A B Example: Shell Higher Olefin Process (SHOP, 1968) C 2 H 4 Oligomerisation C 2 H 4 Cat. Ni(COD)2+ 2 P-CH 2 CO 2 H Solv. 1, 4 -butanediol C 4 -20 - -n Olefins Cat. + Solv. Work-up (Distillation) C 10 -14 Metathesis (Re/ Al 2 O 3) C 12 -18 C 20+ W. Keim et al. DE-P 2054009 (1969) SHELL W. Keim, Chem. Ing. Techn. 56 (1984) 850 C 4 -10 Isomerisation (Mo/ Al 2 O 3)
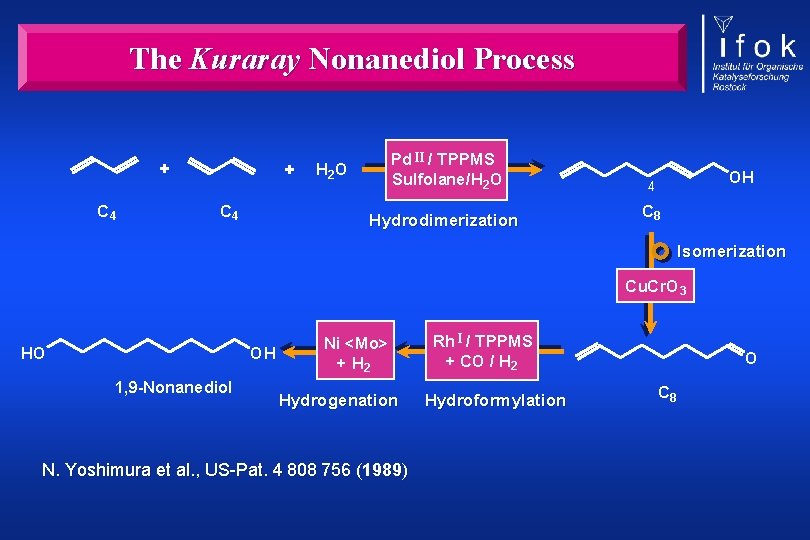
The Kuraray Nonanediol Process + C 4 Pd II / TPPMS Sulfolane/H 2 O Hydrodimerization OH 4 C 8 Isomerization Cu. Cr. O 3 HO OH 1, 9 -Nonanediol Ni <Mo> + H 2 Hydrogenation N. Yoshimura et al. , US-Pat. 4 808 756 (1989) Rh I / TPPMS + CO / H 2 Hydroformylation O C 8
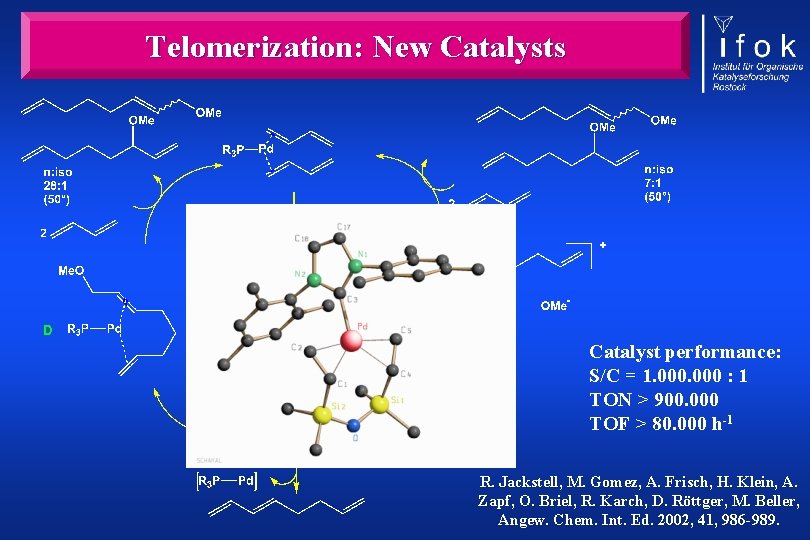
Telomerization: New Catalysts Catalyst performance: S/C = 1. 000 : 1 TON > 900. 000 TOF > 80. 000 h-1 R. Jackstell, M. Gomez, A. Frisch, H. Klein, A. Zapf, O. Briel, R. Karch, D. Röttger, M. Beller, Angew. Chem. Int. Ed. 2002, 41, 986 -989.
Hydroformylation: General Aspects Reaction discovered 1938 by Otto Roelen Catalyst metals: first Cobalt, later Rhodium In case of aliphatic olefins: linear aldehydes are preferred products One of the most important homogeneous catalytic reactions (> 7 Mio. to/a) Most important product: butyraldehyde (> 4 Mio to/a)

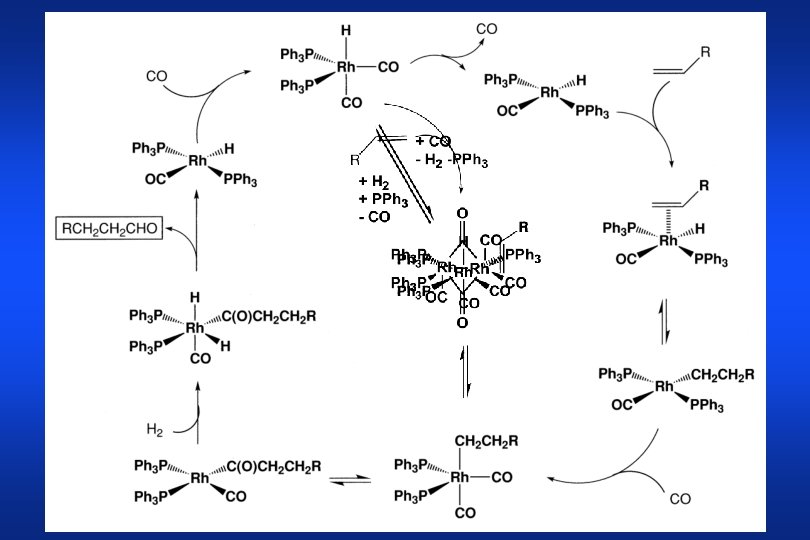
Rh-catalyzed Hydroformylation In general two classes of ligands: Phosphines (P-C-bond) Phosphites (P-O-bond) most important processes for propene hydroformylation: UCC: PPh 3 / Rh: 400 : 1, T: 85 - 90 °C, p: 15 - 18 bar, n / i: 90 % Ruhrchemie/Rhone Poulenc: TPPTS / Rh = >100 : 1, T = 120 °C, p < 20 bar actual catalyst: HRh(PR 3)3 CO studied first by Wilkinson et al. in 1968
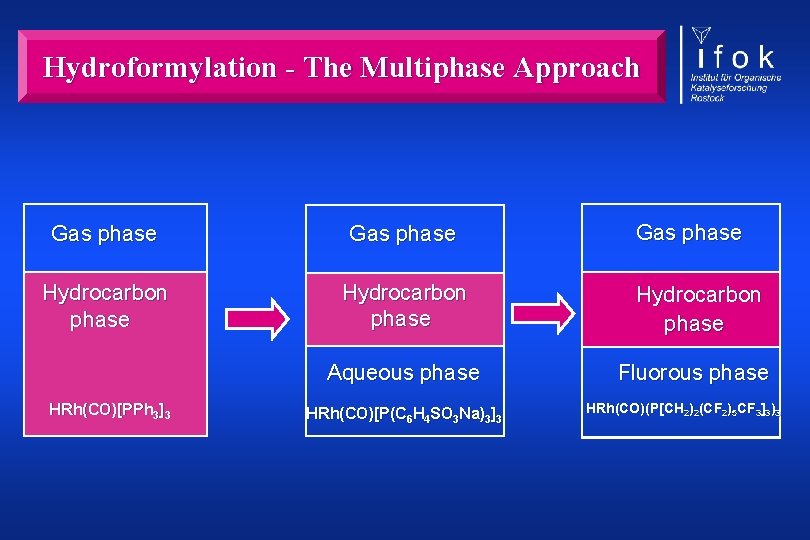
Hydroformylation - The Multiphase Approach Gas phase Hydrocarbon phase Aqueous phase Fluorous phase HRh(CO)[PPh 3]3 HRh(CO)[P(C 6 H 4 SO 3 Na)3]3 HRh(CO)(P[CH 2)2(CF 2)5 CF 3]3)3
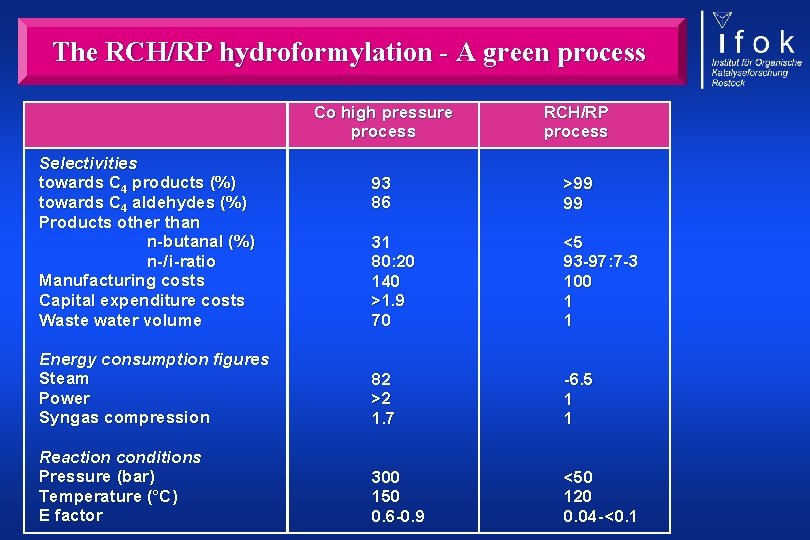
The RCH/RP hydroformylation - A green process Co high pressure process RCH/RP process 93 86 >99 99 31 80: 20 140 >1. 9 70 <5 93 -97: 7 -3 100 1 1 Energy consumption figures Steam Power Syngas compression 82 >2 1. 7 -6. 5 1 1 Reaction conditions Pressure (bar) Temperature (°C) E factor 300 150 0. 6 -0. 9 <50 120 0. 04 - <0. 1 Selectivities towards C 4 products (%) towards C 4 aldehydes (%) Products other than n-butanal (%) n-/ i-ratio Manufacturing costs Capital expenditure costs Waste water volume
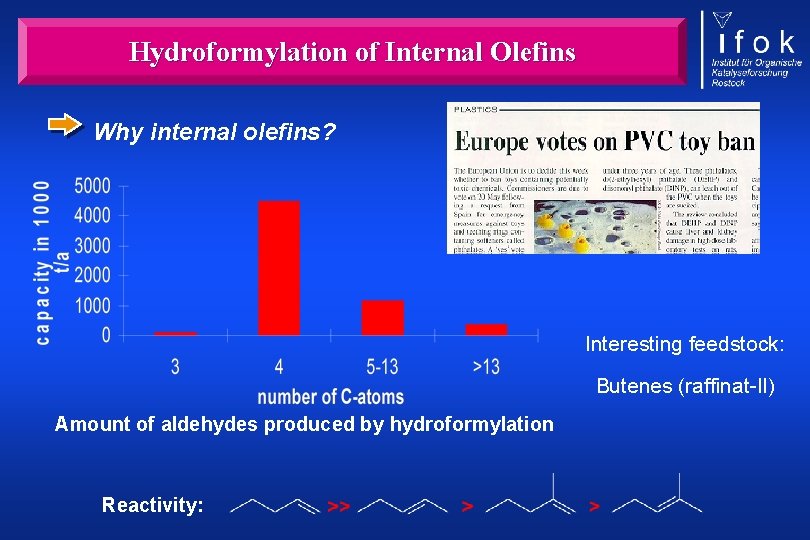
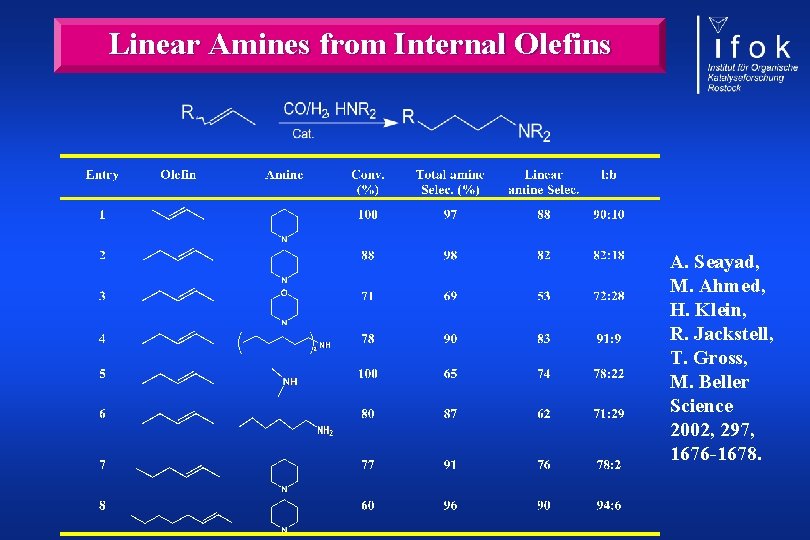
Hydroformylation of Internal Olefins Why internal olefins? Interesting feedstock: Butenes (raffinat-II) Amount of aldehydes produced by hydroformylation Reactivity:
Raising the Barr for the Perfect Reaction A. Seayad, M. Ahmed, H. Klein, R. Jackstell, T. Gross, M. Beller, Science, 2002, 297, 1676 -1678.

Composition of C 4 -Fractions steamcracker low severity 26 alkynes 2 steamcracker high severity 2 7 alkynes 2 1 47 4 7 3 32 6 20 catcracker (FCC) gasoil, zeolite 22 5 14 37 13 0, 5 15 11 12 12
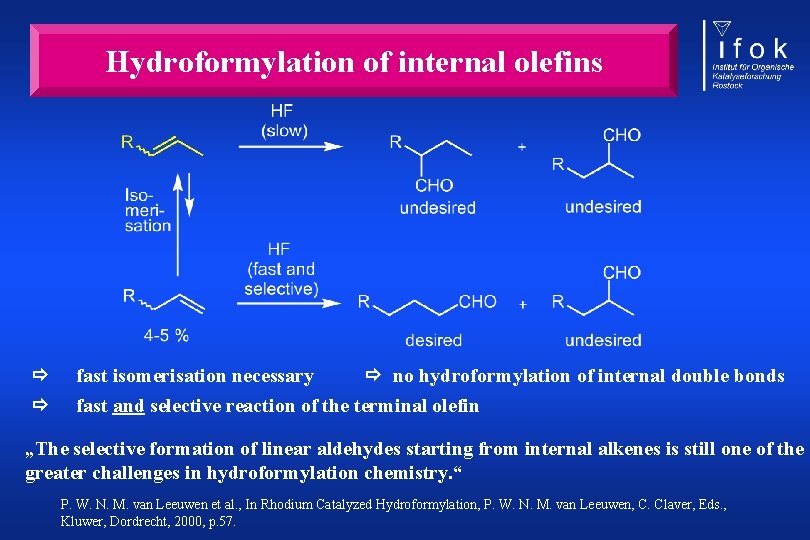
Hydroformylation of internal olefins fast isomerisation necessary no hydroformylation of internal double bonds fast and selective reaction of the terminal olefin „The selective formation of linear aldehydes starting from internal alkenes is still one of the greater challenges in hydroformylation chemistry. “ P. W. N. M. van Leeuwen et al. , In Rhodium Catalyzed Hydroformylation, P. W. N. M. van Leeuwen, C. Claver, Eds. , Kluwer, Dordrecht, 2000, p. 57.
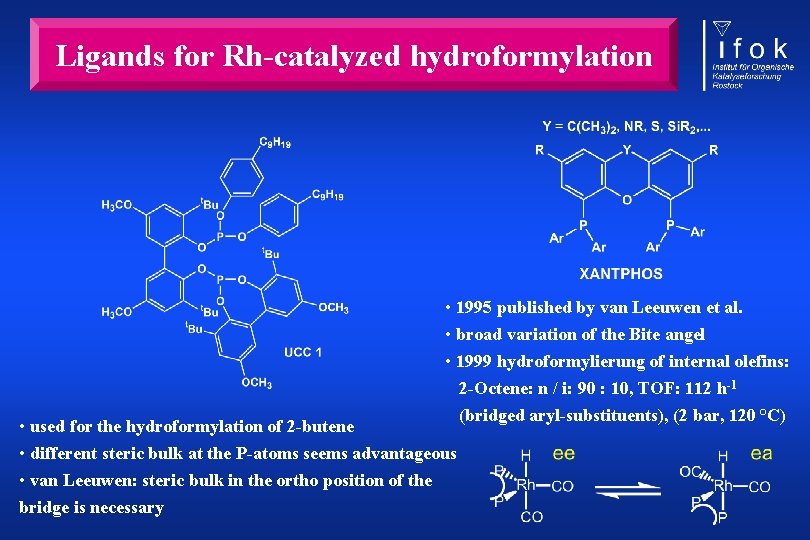
Ligands for Rh-catalyzed hydroformylation • 1995 published by van Leeuwen et al. • broad variation of the Bite angel • 1999 hydroformylierung of internal olefins: 2 -Octene: n / i: 90 : 10, TOF: 112 h-1 (bridged aryl-substituents), (2 bar, 120 °C) • used for the hydroformylation of 2 -butene • different steric bulk at the P-atoms seems advantageous • van Leeuwen: steric bulk in the ortho position of the bridge is necessary

Hydroformylation with NAPHOS Conditions: t: 16 h, Rh / olefin: 0, 01 mol %, P / Rh: 10 1977 published by Tamao et al. ; later on patented by Eastman Kodak and Hoechst. very good n/i-selectivity at low pressure, however activity is too low. increase of activity possible by substituted Phenyl groups?

New synthetis of substituted NAPHOS-ligands Yield (A. . . D): 44 %
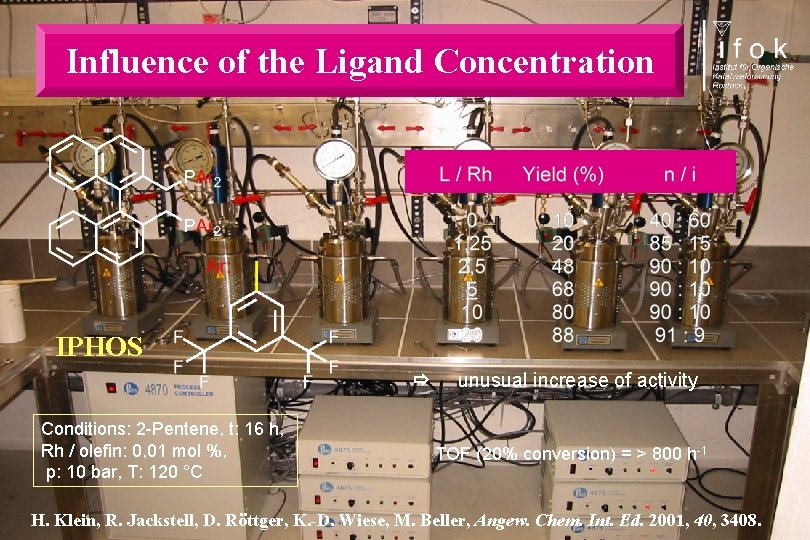
Influence of the Ligand Concentration IPHOS Conditions: 2 -Pentene, t: 16 h, Rh / olefin: 0, 01 mol %, p: 10 bar, T: 120 °C unusual increase of activity TOF (20% conversion) = > 800 h-1 H. Klein, R. Jackstell, D. Röttger, K. -D. Wiese, M. Beller, Angew. Chem. Int. Ed. 2001, 40, 3408.

Homogenous Catalysis A Personal View 3. Challenges

Ten Challenges for Industrial Catalysis Low-temperature oxidation of SO 2 to SO 3 Phenol from benzene and oxygen Direct synthesis of aromatic amines High-conversion and selective from ammonia production of olefins from alkanes Selective oxidation of methane Facile decomposition of NOX to methanol Low-temperature epoxidation of Oxidative coupling from methanol ethylene to ethylene Direct H 2 O 2 -synthesis Anti-Markovnikov addition of from H 2 and O 2 alcohols and amines to olefins Chem. Eng. News 1993, May 31, 23. 23

The Markovnikov Rule "When a hydrocarbon of unsymmetrical structure combines with a halogen hydracid the halogen adds itself to the less hydrogenated carbon atom, i. e. to the carbon which is more under the influence of other carbon atoms. " (V. Markovnikov, Comptes rendus 1875, 82, 668. ) X R + H X H R Markovnikov product Detours for anti-Markovnikov products: radical reactions hydroboration indirect paths (hydroformylation, telomerisation) H X R anti-Markovnikov product
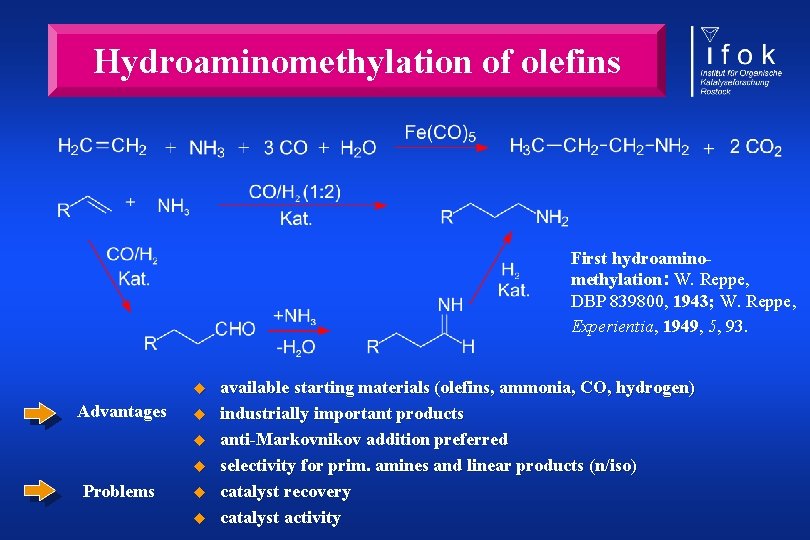
Hydroaminomethylation of olefins First hydroaminomethylation: W. Reppe, DBP 839800, 1943; W. Reppe, Experientia, 1949, 5, 93. u Advantages u u u Problems u u available starting materials (olefins, ammonia, CO, hydrogen) industrially important products anti-Markovnikov addition preferred selectivity for prim. amines and linear products (n/iso) catalyst recovery catalyst activity

A New Concept for Hydroaminomethylations Dual catalyst system: Rh / Ir / phosphine Rh: + + _ active catalyst for hydroformylation high n/iso-selektivity with excess phosphine not active for hydrogenation with excess phosphine Ir: + + active for hydrogenation with excess phosphine selective for hydrogenation of imines Use of two-phase catalysis u u u increase of selectivity via extraction of prim. amines simple catalyst recycling use of water solutions of ammonia B. Zimmermann, J. Herwig, M. Beller, Angew. Chem. Int. Ed. Engl. 1999, 38, 2372.
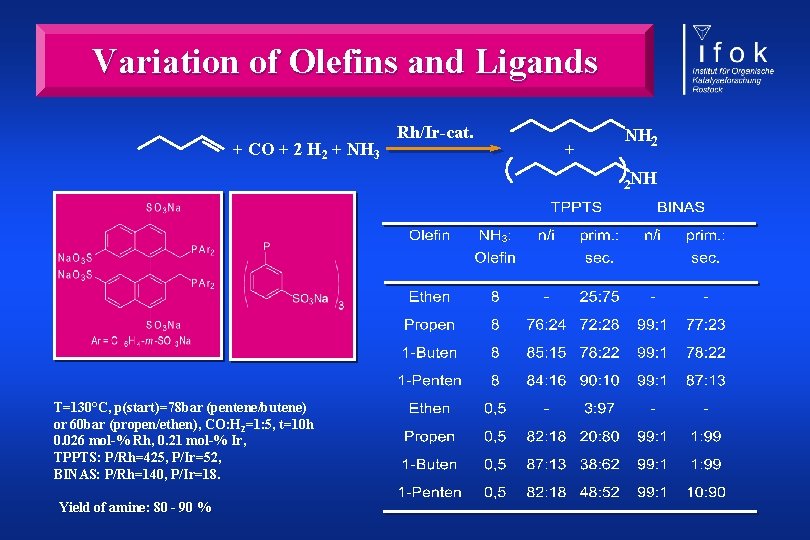
Variation of Olefins and Ligands + CO + 2 H 2 + NH 3 Rh/Ir-cat. + NH 2 2 NH T=130°C, p(start)=78 bar (pentene/butene) or 60 bar (propen/ethen), CO: H 2=1: 5, t=10 h 0. 026 mol-% Rh, 0. 21 mol-% Ir, TPPTS: P/Rh=425, P/Ir=52, BINAS: P/Rh=140, P/Ir=18. Yield of amine: 80 - 90 %
Linear Amines from Internal Olefins A. Seayad, M. Ahmed, H. Klein, R. Jackstell, T. Gross, M. Beller Science 2002, 297, 1676 -1678.
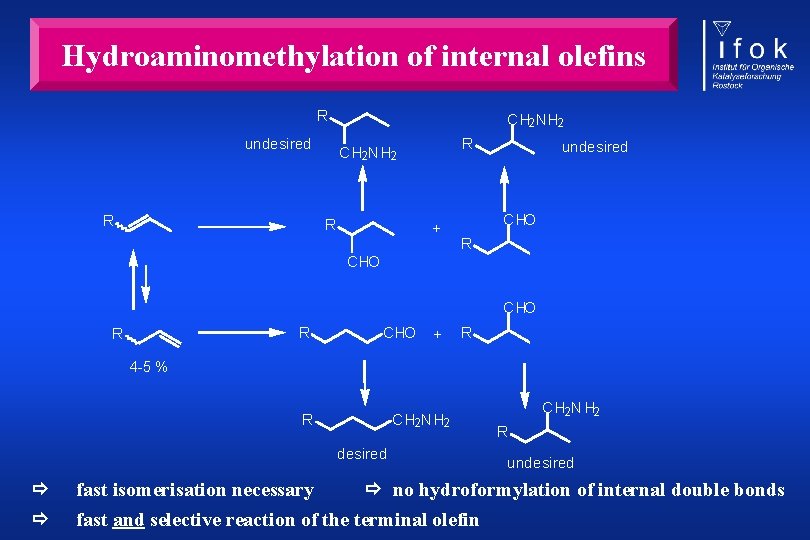
Hydroaminomethylation of internal olefins R undesired R C H 2 N H 2 R + undesired C HO R R C HO + R 4 -5 % R C H 2 N H 2 desired C H 2 N H 2 R undesired fast isomerisation necessary no hydroformylation of internal double bonds fast and selective reaction of the terminal olefin

Homogenous Catalysis A Personal View 4. Fine Chemicals

The „Ideal“ Chemical Synthesis Most organic syntheses are fare away from being efficient. New methods need to be developed and known methods must be improved. Environment Produce a minimum of waste; use available feedstocks Efficiency Apply energy-efficient processes with 100% atom economy; use domino processes and mcr´s Selectivity Highly selective (chemo-, regio-, stereoselective) processes (100% yield) Economics Perform reactions in standard equipment; minimize catalyst costs (“real catalytic processes); look for high space/time yields

Environmental Acceptability: the E Factor kg waste product E= kg desired product Industry Product volume (t/a) Ratio kg by-products per kg product oil refining 106 - 108 0. 1 bulk chemicals 104 - 106 <1 -5 fine chemicals 102 - 104 5 -50 pharmaceuticals 101 - 103 25 -100+ R. A. Sheldon, Chem. Tech 1994, 24, 38.
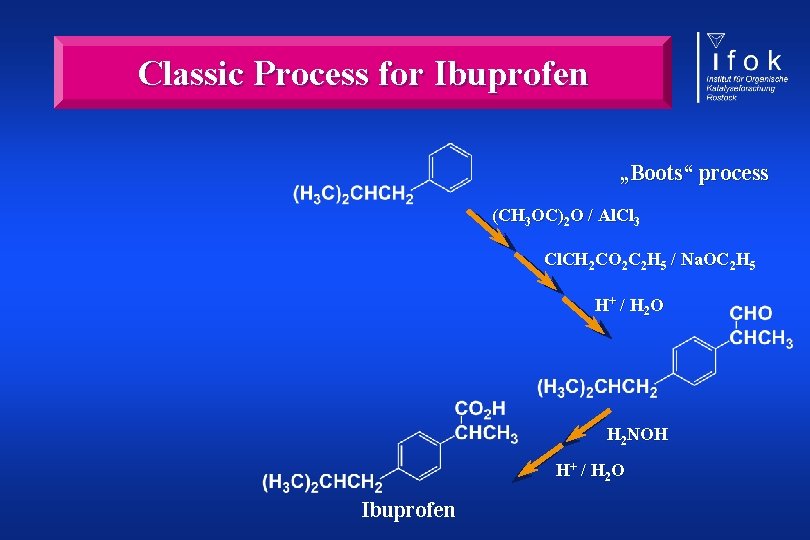
Classic Process for Ibuprofen „Boots“ process (CH 3 OC)2 O / Al. Cl 3 Cl. CH 2 CO 2 C 2 H 5 / Na. OC 2 H 5 H+ / H 2 O H 2 NOH H+ / H 2 O Ibuprofen
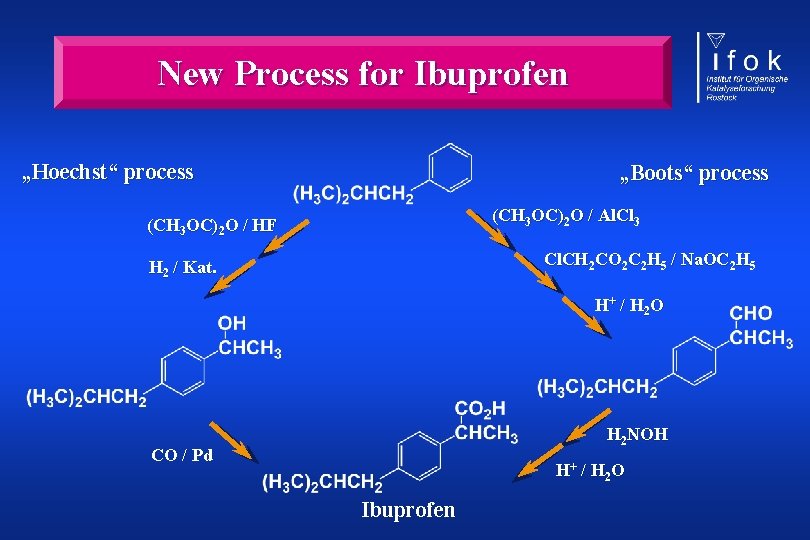
New Process for Ibuprofen „Hoechst“ process „Boots“ process (CH 3 OC)2 O / Al. Cl 3 (CH 3 OC)2 O / HF Cl. CH 2 CO 2 C 2 H 5 / Na. OC 2 H 5 H 2 / Kat. H+ / H 2 O H 2 NOH CO / Pd H+ / H 2 O Ibuprofen
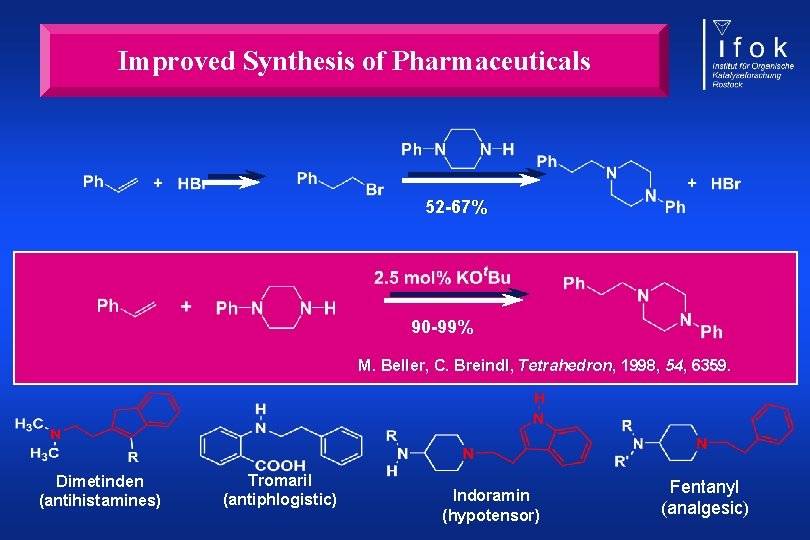
Improved Synthesis of Pharmaceuticals 52 -67% 90 -99% M. Beller, C. Breindl, Tetrahedron, 1998, 54, 6359. Dimetinden (antihistamines) Tromaril (antiphlogistic) Indoramin (hypotensor) Fentanyl (analgesic)
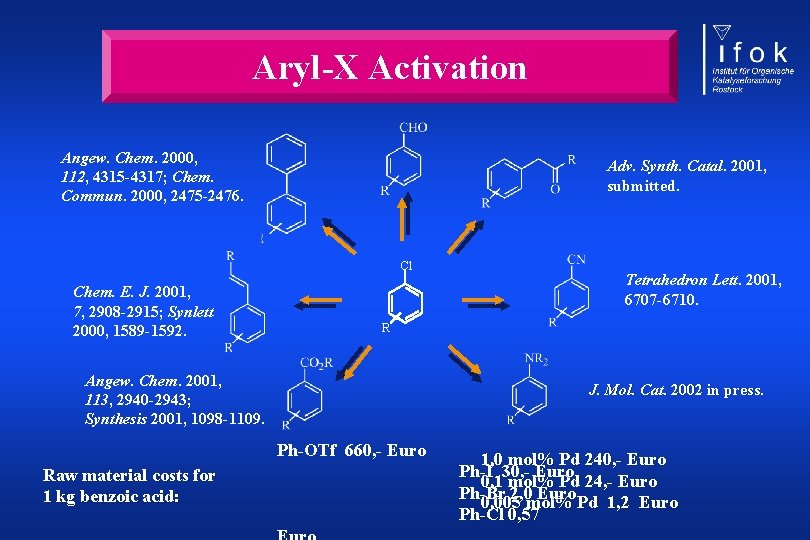
Aryl-X Activation Angew. Chem. 2000, 112, 4315 -4317; Chem. Commun. 2000, 2475 -2476. Adv. Synth. Catal. 2001, submitted. Cl Chem. E. J. 2001, 7, 2908 -2915; Synlett 2000, 1589 -1592. R Angew. Chem. 2001, 113, 2940 -2943; Synthesis 2001, 1098 -1109. J. Mol. Cat. 2002 in press. Ph-OTf 660, - Euro Raw material costs for 1 kg benzoic acid: Tetrahedron Lett. 2001, 6707 -6710. 1, 0 mol% Pd 240, - Euro Ph-I 30, - Euro 0, 1 mol% Pd 24, - Euro Ph-Br 2, 0 Euro 0, 005 mol% Pd 1, 2 Euro Ph-Cl 0, 57
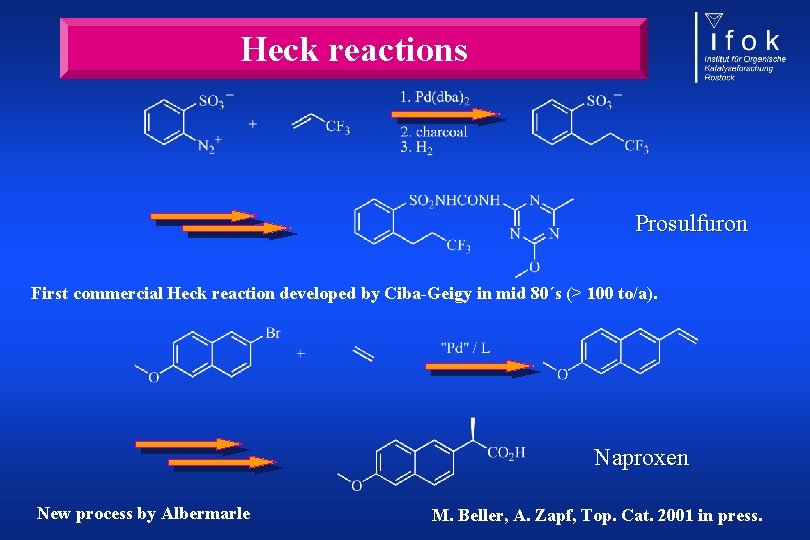
Heck reactions Prosulfuron First commercial Heck reaction developed by Ciba-Geigy in mid 80´s (> 100 to/a). Naproxen New process by Albermarle M. Beller, A. Zapf, Top. Cat. 2001 in press.
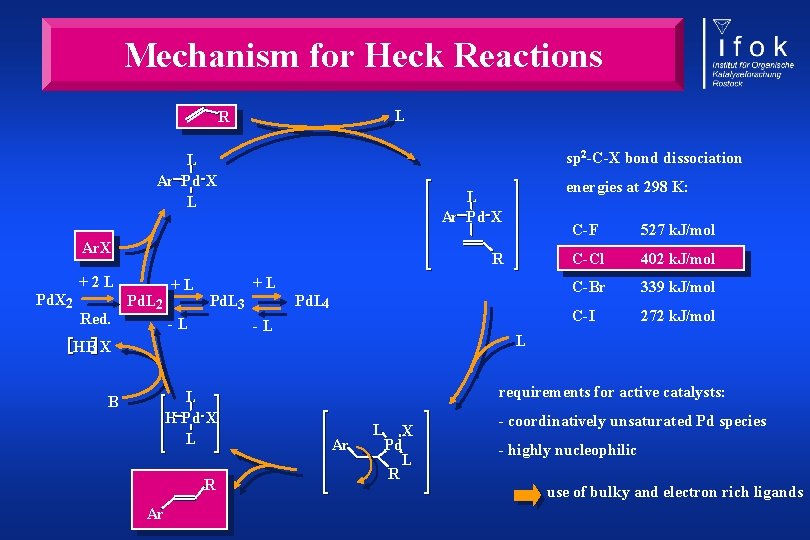
Mechanism for Heck Reactions L R sp 2 -C-X bond dissociation L Ar Pd X L Ar. X R +2 L Pd. X 2 Red. energies at 298 K: L Ar Pd X Pd. L 2 +L +L Pd. L 3 -L Pd. L 4 -L L H Pd X L R Ar 527 k. J/mol C-Cl 402 k. J/mol C-Br 339 k. J/mol C-I 272 k. J/mol L HB X B C-F requirements for active catalysts: Ar L X Pd L R - coordinatively unsaturated Pd species - highly nucleophilic use of bulky and electron rich ligands

Knowledge Understanding is getting familiar with facts and thoughts. 50°C, 1 bar, H 2 O, p. H 9. 5 Celanese Corp. , GB-B 1028940, 1966. 2% 70°C, 1 bar, H 2 O / dioxane, p. H 8 Exxon Corp. , US-A 4496779, 1984. 3 -5% 100°C, 28 bar, H 2 O / Me. CN J. F. Cairns, H. L. Roberts, J. Chem. Soc. C 1968, 640 -642.
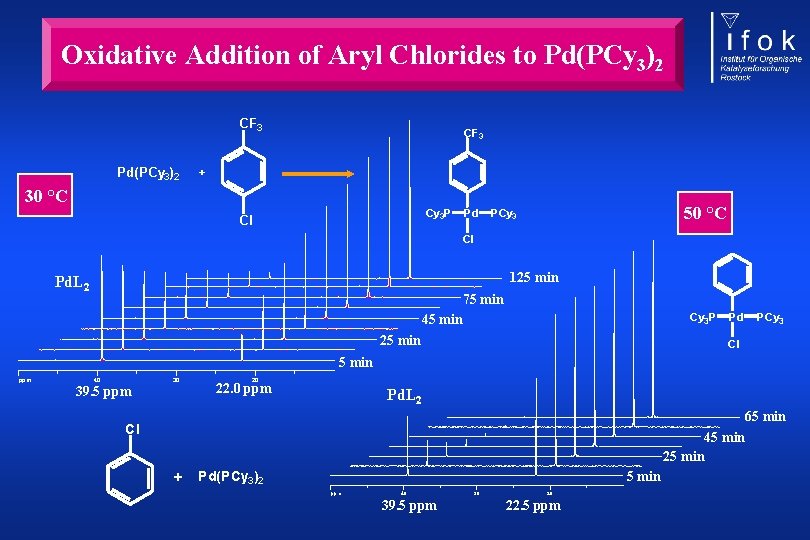
Oxidative Addition of Aryl Chlorides to Pd(PCy 3)2 CF 3 + 30 °C Cy 3 P Cl Pd 50 °C PCy 3 Cl 125 min Pd. L 2 75 min 45 min Cy 3 P 25 min Pd PCy 3 Cl 5 min ppm 40 30 39. 5 ppm 20 22. 0 ppm Pd. L 2 65 min 45 min Cl 25 min + 5 min Pd(PCy 3)2 ppm 40 39. 5 ppm 30 20 22. 5 ppm
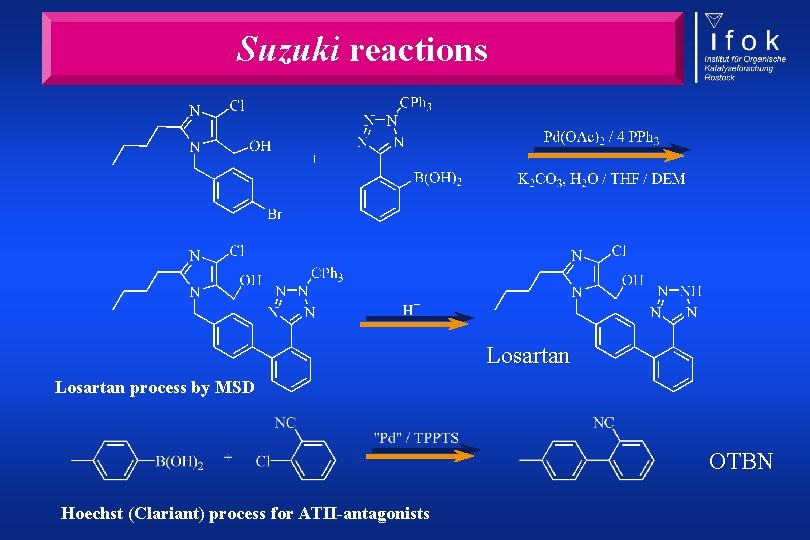
Suzuki reactions Losartan process by MSD OTBN Hoechst (Clariant) process for ATII-antagonists

Bu. PAd 2 - Variation of Substrates R Cl + B(OH)2 0. 005 mol% Pd(OAc)2 0. 01 mol% Bu. PAd 2 R toluene, K 3 PO 4 100 °C, 20 h a) 4 h A. Zapf, A. Ehrentraut, M. Beller, Angew. Chem. 2000, 112, 4315. Bu. PAd 2

Cross Coupling of Grignard Reagents 1. 5 mol% Pd 2(dba)3 4 mol % L·HCl Entry L Solvent Temp. [°C] Time [h] Yield [%] 1 IMes Et 2 O / THF 45 20 35 2 IPr Et 2 O / THF 45 20 97 3 IPr Toluene / THF 80 20 10 4 IPr THF 80 5 86 5 none Dioxane / THF 80 3 0 6 IMes Dioxane / THF 80 3 41 7 IPr Dioxane / THF 80 3 99 Lit. : Huang, J. ; Nolan, S. P. J. Am. Chem. Soc. 1999, 1999 121, 9889 -9890. L = IPr

Amidocarbonylation Reactions Wakamatsu reaction (Co-catalyzed) Pd-catalyzed (Jägers, Knoll 1989, Beller et al. since 1995) CO CO / H 2 (10 - 60 bar) (50 - 100 bar) [Pd] HCo(CO)4 (0. 01 - 0. 5 mol%) (1 - 5 mol%) N-Acyl Amino Acid H + (1 mol%) X - (5 - 35 mol%) One-pot Synthesis of N-Acyl Amino Acids with 100 % Atom Utilization High Selectivity and Chemical Yield Tolerance of various functional groups M. Beller, M. Eckert, Angew. Chem. 2000, 112, 1027; Angew. Chem. Int. Ed. 2000, 39, 1010.
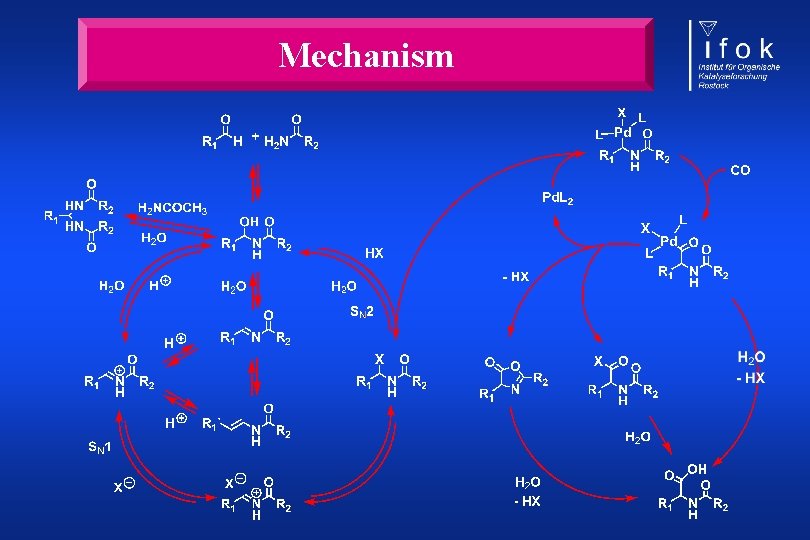
Mechanism
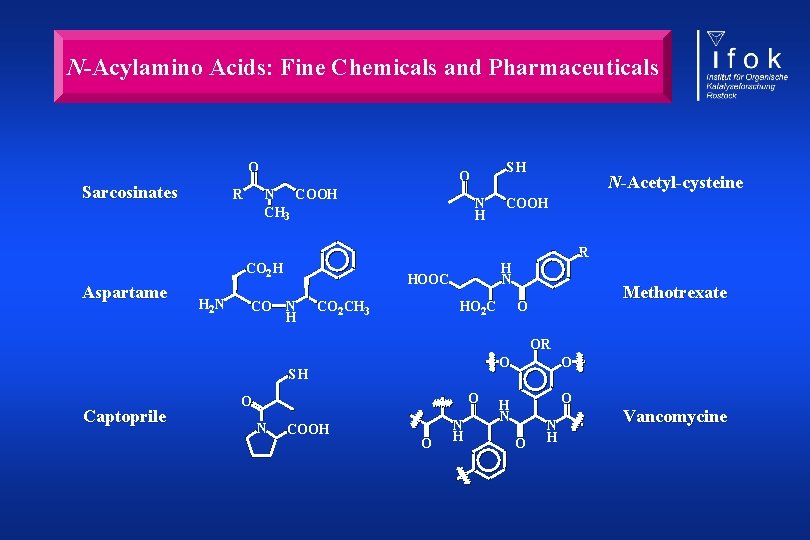
N-Acylamino Acids: Fine Chemicals and Pharmaceuticals O Sarcosinates R N COOH CH 3 H 2 N CO N H N-Acetyl-cysteine COOH R CO 2 H Aspartame SH O H N HOOC N H CO 2 CH 3 HO 2 C Methotrexate O OR SH Captoprile O O N COOH O N H O O H N O O N H Vancomycine
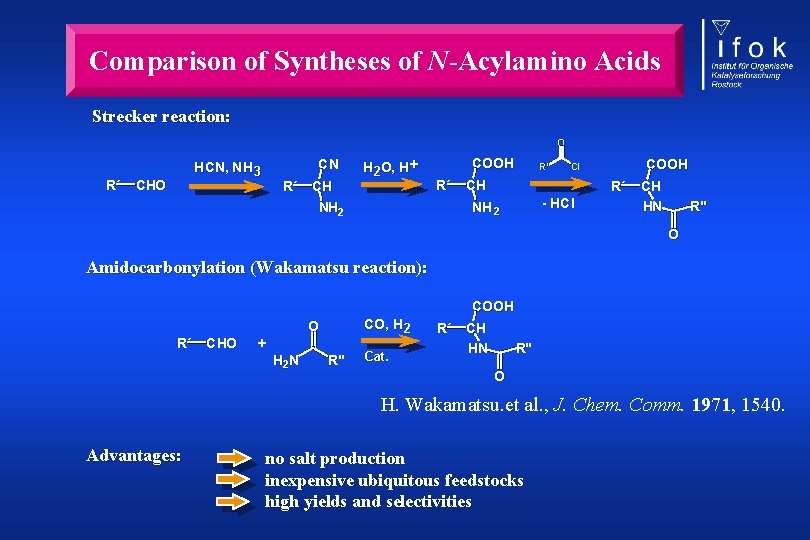
Comparison of Syntheses of N-Acylamino Acids Strecker reaction: O R´ CN HCN, NH 3 CHO R´ CH H 2 O, H + COOH R´ R" CH R´ - HCl NH 2 COOH Cl CH HN R" O Amidocarbonylation (Wakamatsu reaction): COOH CO, H 2 O R´ CHO + H 2 N R" Cat. R´ CH HN R" O H. Wakamatsu. et al. , J. Chem. Comm. 1971, 1540. Advantages: no salt production inexpensive ubiquitous feedstocks high yields and selectivities
![Amidocarbonylation Reactions [Co] [Pd] Angew. Chem. 1999, 111, 1562. J. Org. Chem. 1998, 63, Amidocarbonylation Reactions [Co] [Pd] Angew. Chem. 1999, 111, 1562. J. Org. Chem. 1998, 63,](http://slidetodoc.com/presentation_image_h/7c4ac088cf1485ec4b34abfb1932452f/image-52.jpg)
Amidocarbonylation Reactions [Co] [Pd] Angew. Chem. 1999, 111, 1562. J. Org. Chem. 1998, 63, 5658. [Pd] Tetrahedron Lett. 1999, 40, 4523. [Pd] [Co] Chem. Eur. J. 1998, 4, 935. Angew. Chem. 1997, 109, 1534.

Homogeneous Catalysis A Personal View 6. Oxidation Chemistry

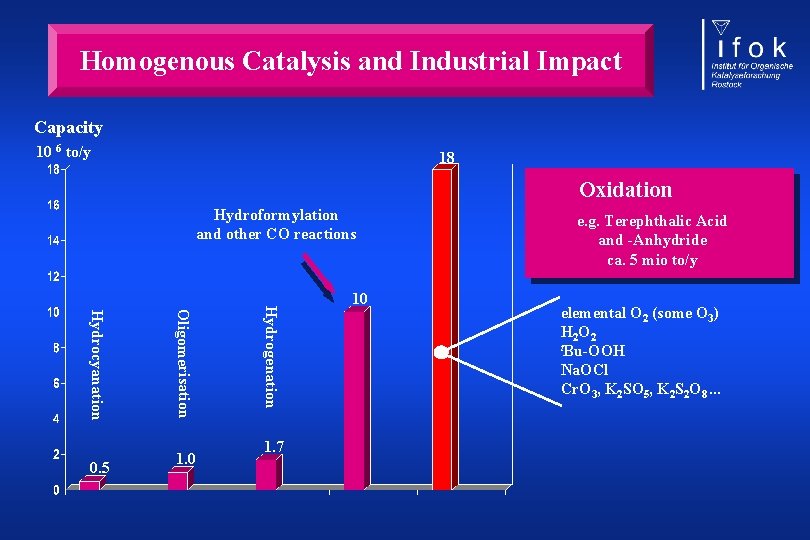
Homogenous Catalysis and Industrial Impact Capacity 10 6 to/y 18 Oxidation Hydroformylation and other CO reactions 1. 0 Hydrogenation Oligomerisation Hydrocyanation 0. 5 1. 7 10 e. g. Terephthalic Acid and -Anhydride ca. 5 mio to/y elemental O 2 (some O 3) H 2 O 2 t. Bu-OOH Na. OCl Cr. O 3, K 2 SO 5, K 2 S 2 O 8. . .
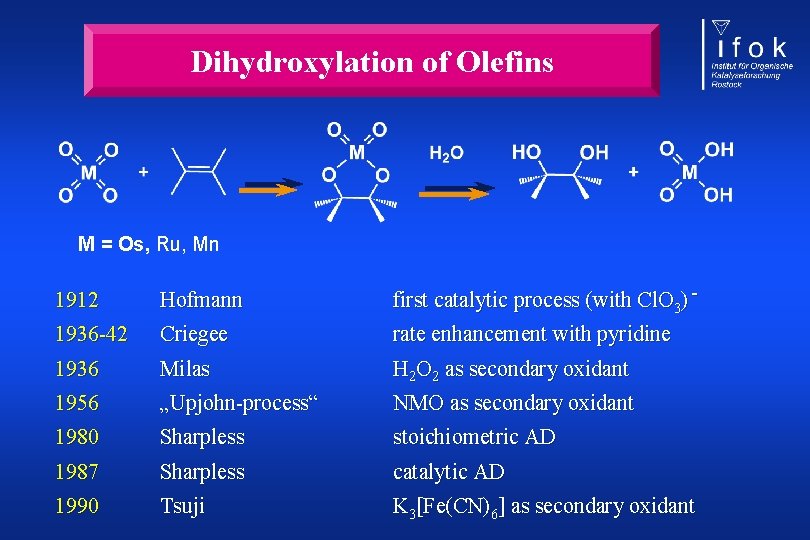
Dihydroxylation of Olefins M = Os, Ru, Mn 1912 Hofmann first catalytic process (with Cl. O 3) - 1936 -42 Criegee rate enhancement with pyridine 1936 Milas H 2 O 2 as secondary oxidant 1956 „Upjohn-process“ NMO as secondary oxidant 1980 Sharpless stoichiometric AD 1987 Sharpless catalytic AD 1990 Tsuji K 3[Fe(CN)6] as secondary oxidant
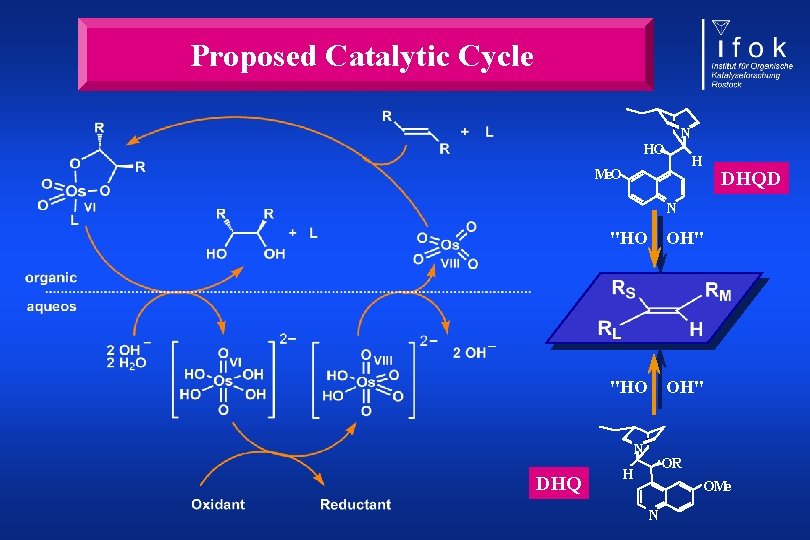
Proposed Catalytic Cycle N HO H Me. O DHQD N "HO OH" N DHQ OR H OMe N
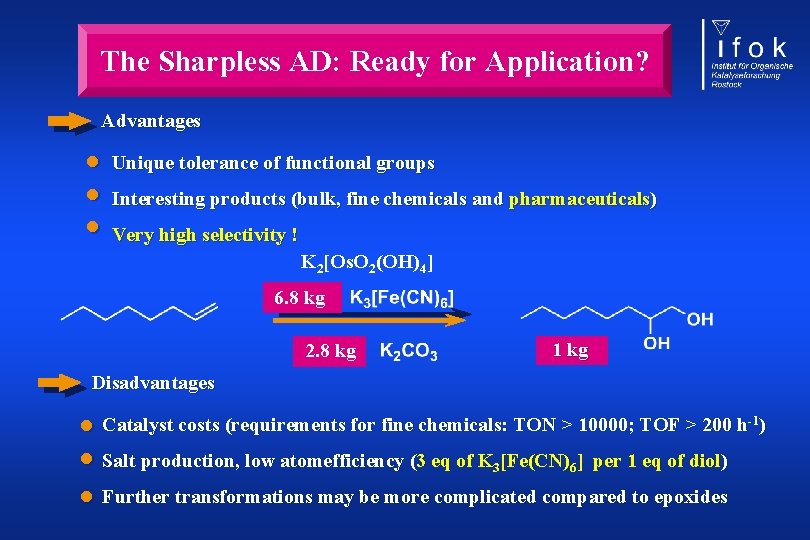
The Sharpless AD: Ready for Application? Advantages • Unique tolerance of functional groups • Interesting products (bulk, fine chemicals and pharmaceuticals) • Very high selectivity ! K 2[Os. O 2(OH)4] 6. 8 kg 1 kg 2. 8 kg Disadvantages • Catalyst costs (requirements for fine chemicals: TON > 10000; TOF > 200 h ) • Salt production, low atomefficiency (3 eq of K [Fe(CN) ] per 1 eq of diol) • Further transformations may be more complicated compared to epoxides -1 3 6
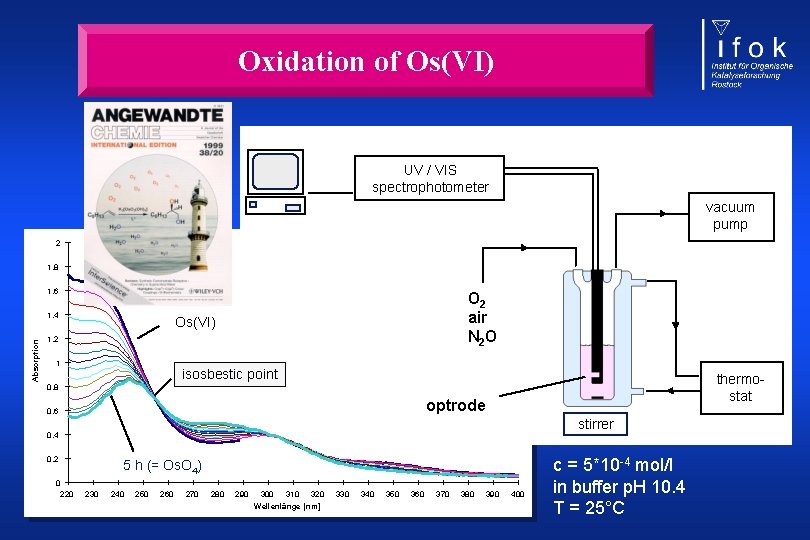
Oxidation of Os(VI) UV / VIS spectrophotometer vacuum pump 2 1, 8 1, 6 Absorption 1, 4 O 2 air N 2 O Os(VI) 1, 2 1 isosbestic point thermostat 0, 8 optrode 0, 6 stirrer 0, 4 0, 2 5 h (= Os. O 4) 0 220 230 240 250 260 270 280 290 300 310 320 Wellenlänge [nm] 330 340 350 360 370 380 390 400 c = 5*10 -4 mol/l in buffer p. H 10. 4 T = 25°C
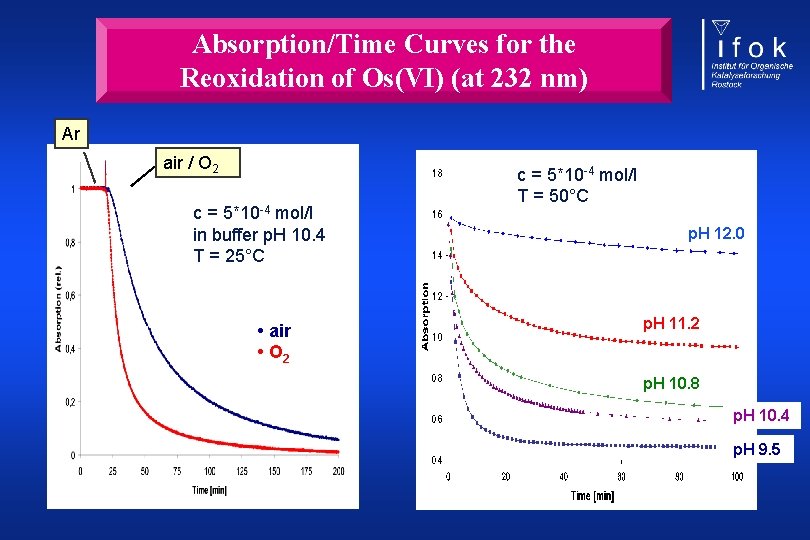
Absorption/Time Curves for the Reoxidation of Os(VI) (at 232 nm) Ar air / O 2 c = 5*10 -4 mol/l in buffer p. H 10. 4 T = 25°C • air • O 2 c = 5*10 -4 mol/l T = 50°C p. H 12. 0 p. H 11. 2 p. H 10. 8 p. H 10. 4 p. H 9. 5
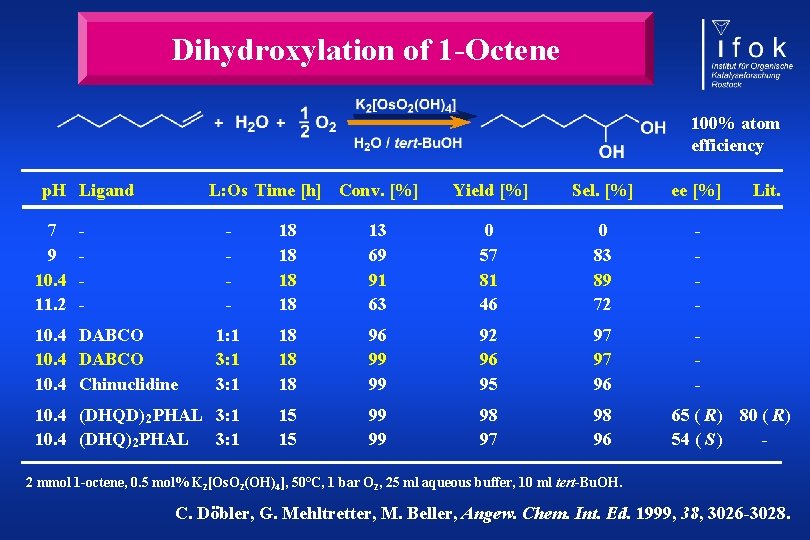
Dihydroxylation of 1 -Octene 100% atom efficiency p. H Ligand 7 9 10. 4 11. 2 L: Os Time [h] Conv. [%] - Yield [%] Sel. [%] ee [%] - 18 18 13 69 91 63 0 57 81 46 0 83 89 72 - 1: 1 3: 1 18 18 18 96 99 99 92 96 95 97 97 96 - 10. 4 (DHQD) 2 PHAL 3: 1 10. 4 (DHQ) 2 PHAL 3: 1 15 15 99 99 98 97 98 96 10. 4 DABCO 10. 4 Chinuclidine Lit. 65 ( R) 80 ( R) 54 ( S ) - 2 mmol 1 -octene, 0. 5 mol% K 2[Os. O 2(OH)4], 50°C, 1 bar O 2, 25 ml aqueous buffer, 10 ml tert-Bu. OH. C. Döbler, G. Mehltretter, M. Beller, Angew. Chem. Int. Ed. 1999, 38, 3026 -3028.

Homogeneous Catalysis A Personal View 7. Asymmetric Catalysis

Syntheses of Enantiomerically Pure Compounds * Stoichiometric Asymmetric Synthesis / * Catalysis * Biocatalyst * Chemical catalyst * Synthesis ("chiral pool") * Fermentation * Enantiomer separation Chiral chromatography Diastereomer separation Kinetic resolution

Dynamic Kinetic Resolution Classical resolution A B k. A P Dynamic kinetic resolution A P k. A krac k. B Q B k. B Q A, B : Substrate isomers P : Desired product k. X : Rate constants for the part reaction X Q : By-product Comments about dynamic kinetic resolution : 1. The yield of product is not limited to only 50% as in a classical racemisation 2. The desired product must not racemise under the reaction conditions 3. The rate of substrate racemisation should be faster than the rate of the resolution step
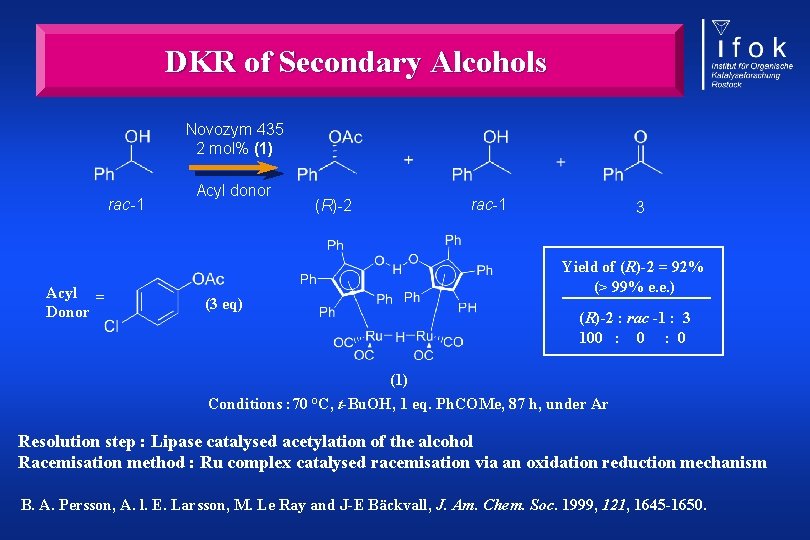
DKR of Secondary Alcohols Novozym 435 2 mol% (1) rac-1 Acyl = Donor Acyl donor rac-1 (R)-2 3 Yield of (R)-2 = 92% (> 99% e. e. ) (3 eq) (R)-2 : rac -1 : 3 100 : 0 (1) Conditions : 70 °C, t-Bu. OH, 1 eq. Ph. COMe, 87 h, under Ar Resolution step : Lipase catalysed acetylation of the alcohol Racemisation method : Ru complex catalysed racemisation via an oxidation reduction mechanism B. A. Persson, A. l. E. Larsson, M. Le Ray and J-E Bäckvall, J. Am. Chem. Soc. 1999, 121, 1645 -1650.

Criteria for an Industrial Catalyst (Hydrogenation) Enantiomeric excess, ee >99% (pharma); >80% (agro or if further enrichment easy) Catalyst productivity, ton catalyst cost >1000 (small scale, high value); >50‘ 000 (large scale, low price) Catalyst activity, tof production capacity >500/h (small scale); >10‘ 000/h (large scale)

Milestones for Industrial Enantioselective Catalysis • 1970’s L-Dopa, enamide hydrogenation (Monsanto) • 1980’s L-Menthol, C=C isomerization (Takasago) • 1990’s Glycidol, Sharpless epoxidation (Arco) • • 2000’s S-Metolachlor (Ciba-Geigy/Novartis) ? ? ?

How Many Industrial Processes are There? transformation production pilot bench scale Hydrogenation of enamides, C=C-COOR C=C-CH-OH and other C=C 5 18 12 Hydrogenation of ketones ( mainly a and b functionalized) 4 10 8 Hydrogenation of C=N 1 1 - Dihydroxylation of C=C - 1 4 Epoxidation of C=C, oxidation of sulfide 3 2 2 Isomerization, epoxide opening, addition 2 3 1 15 35 27 Total H. U. Blaser, F. Spindler, M. Studer, to appear in Applied Catalysis: A General – Special Issue: Industrial Catalytic Processes (W. Hölderich, ed. )
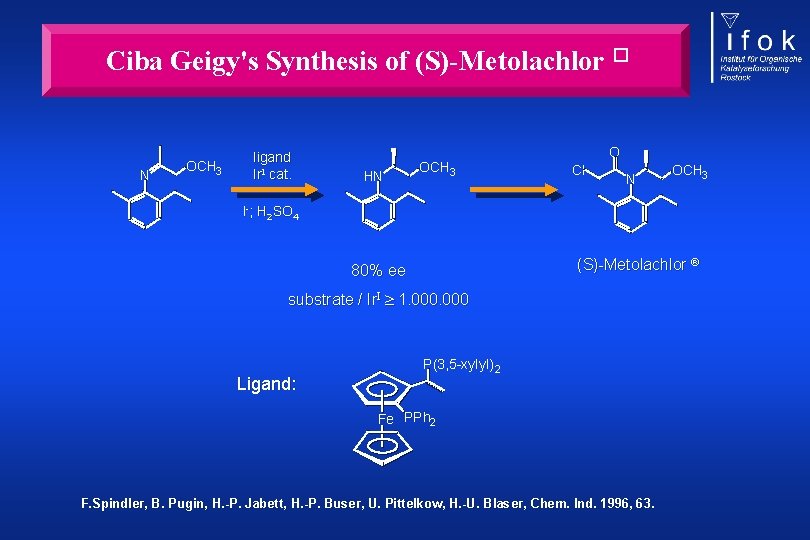
Ciba Geigy's Synthesis of (S)-Metolachlor � N OCH 3 ligand Ir. I cat. HN OCH 3 O Cl N OCH 3 I-; H 2 SO 4 (S)-Metolachlor 80% ee substrate / Ir. I 1. 000 Ligand: P(3, 5 -xylyl) 2 Fe PPh 2 F. Spindler, B. Pugin, H. -P. Jabett, H. -P. Buser, U. Pittelkow, H. -U. Blaser, Chem. Ind. 1996, 63.
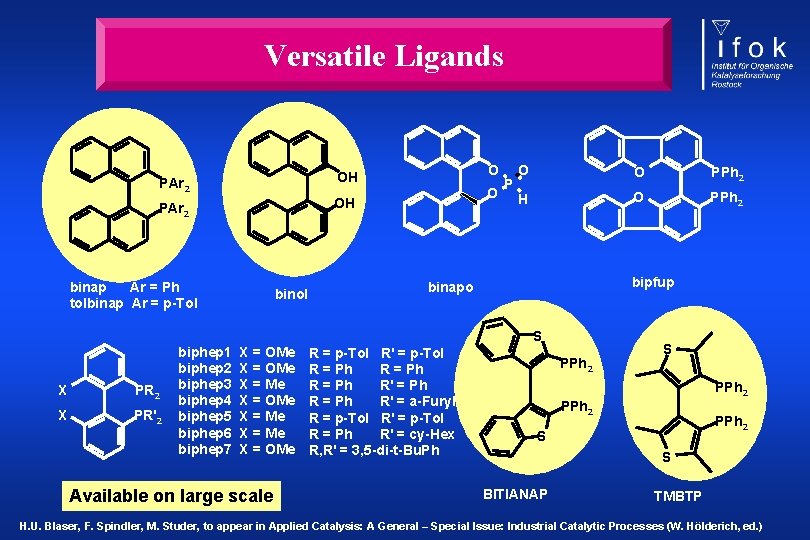
Versatile Ligands PAr 2 OH binap Ar = Ph tolbinap Ar = p-Tol binol O O PPh 2 H O PPh 2 bipfup binapo S X PR 2 X PR' 2 biphep 1 biphep 2 biphep 3 biphep 4 biphep 5 biphep 6 biphep 7 X = OMe X = Me X = OMe Available on large scale R = p-Tol R' = p-Tol R = Ph R' = Ph R' = a-Furyl R = p-Tol R' = p-Tol R = Ph R' = cy-Hex R, R' = 3, 5 -di-t-Bu. Ph PPh 2 S PPh 2 S S BITIANAP TMBTP H. U. Blaser, F. Spindler, M. Studer, to appear in Applied Catalysis: A General – Special Issue: Industrial Catalytic Processes (W. Hölderich, ed. )
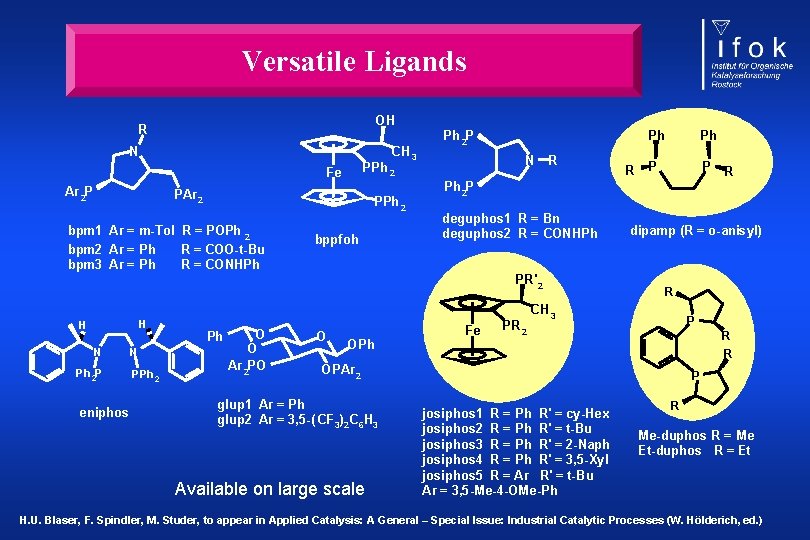
Versatile Ligands OH R N CH 3 PAr 2 PPh 2 bpm 1 Ar = m-Tol R = POPh 2 bpm 2 Ar = Ph R = COO-t-Bu bpm 3 Ar = Ph R = CONHPh N PPh 2 Fe Ar 2 P Ph 2 P bppfoh R H N Ph 2 P eniphos N PPh 2 O Ph O Ar 2 PO O OPh Ph P P deguphos 1 R = Bn deguphos 2 R = CONHPh Fe PR 2 dipamp (R = o-anisyl) R CH 3 P R R OPAr 2 glup 1 Ar = Ph glup 2 Ar = 3, 5 -( CF 3)2 C 6 H 3 Available on large scale R Ph 2 P PR' 2 H R Ph P josiphos 1 R = Ph R' = cy-Hex josiphos 2 R = Ph R' = t-Bu josiphos 3 R = Ph R' = 2 -Naph josiphos 4 R = Ph R' = 3, 5 -Xyl josiphos 5 R = Ar R' = t-Bu Ar = 3, 5 -Me-4 -OMe-Ph R Me-duphos R = Me Et-duphos R = Et H. U. Blaser, F. Spindler, M. Studer, to appear in Applied Catalysis: A General – Special Issue: Industrial Catalytic Processes (W. Hölderich, ed. )
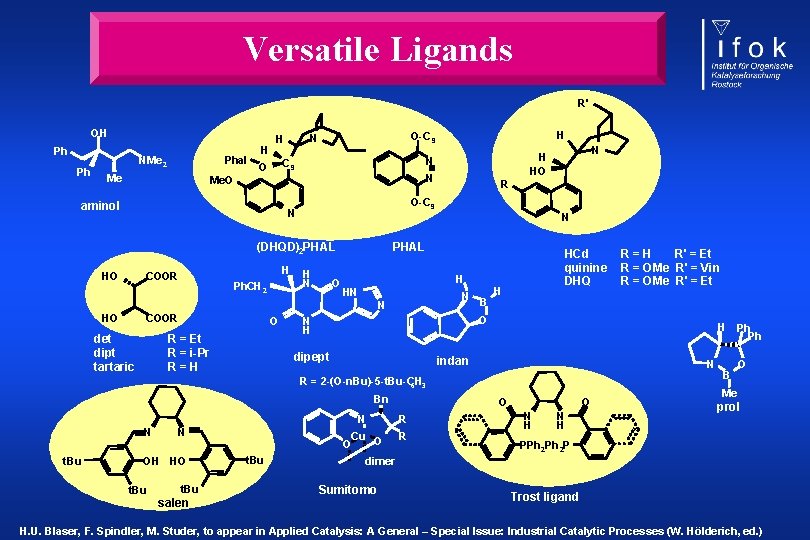
Versatile Ligands R' OH Ph Ph Phal NMe 2 Me H N Me. O aminol N (DHQD)2 PHAL H COOR Ph. CH 2 H N O PHAL COOR det dipt tartaric O R = Et R = i-Pr R=H HCd quinine DHQ H HN N N HO R O-C 9 N HO N H HO N C 9 O H O-C 9 N H B H O N H dipept H indan N R = 2 -(O-n. Bu)-5 -t. Bu-C 6 H 3 Bn N t. Bu N OH HO t. Bu salen t. Bu R=H R' = Et R = OMe R' = Vin R = OMe R' = Et O O N R Cu O O R N H Ph Ph B O Me prol PPh 2 P dimer Sumitomo Trost ligand H. U. Blaser, F. Spindler, M. Studer, to appear in Applied Catalysis: A General – Special Issue: Industrial Catalytic Processes (W. Hölderich, ed. )

Homogeneous Catalysis A Personal View 8. Outlook

„Green“ catalysis - challenges 2010 Selective functionalization of aromatics CH-activation Refinement of olefins

Thanks !!!
- Slides: 75