Homeostasis the Plasma Membrane Structure of the Plasma

Homeostasis & the Plasma Membrane

Structure of the Plasma Membrane v 2 layers of lipids with proteins embedded within each layer v. The lipids are called “phospholipids” v. They have a polar head and a non-polar tail v. HEAD v phosphate group which is soluble in water (hydrophilic) v. TAIL vfatty acid chain that is not soluble in water (hydrophobic)
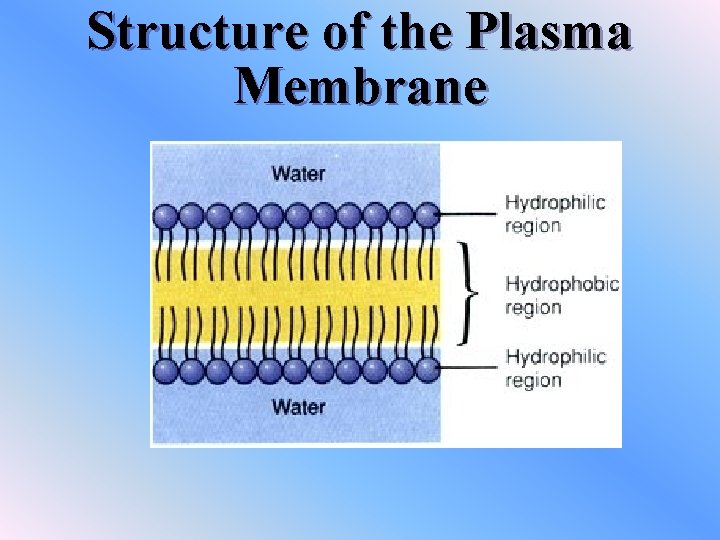
Structure of the Plasma Membrane

Structure of the Plasma Membrane v. The plasma membrane is referred to as a fluid mosaic model The plasma membrane is made up of many molecules which are free to move sideways v. Fatty acid chains can be saturated or unsaturated v. The more unsaturated fatty acids a membrane has, the more fluid it is. v. Saturated or cholesterol chains are more rigid and stable
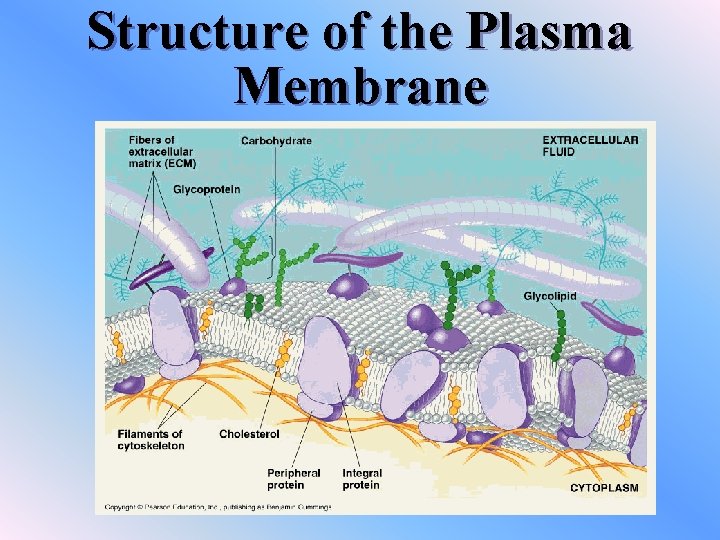
Structure of the Plasma Membrane

Maintaining a Balance v. Living cells maintain a balance by controlling what goes into and out of the cell v. The plasma membrane is a selectively permeable membrane – it has the ability to allow some materials to pass through and keep others out v. This also allows some cells to carry out different jobs than other cells

Maintaining a Balance v. Some molecules can readily pass across the membrane while others cannot. v. The size, polarity, and nature of the molecules will determine whether it passes readily or not

Diffusion v. Diffusion is the net movement of particles from high concentration to low concentration v. Diffusion cannot occur unless a substance is in a higher concentration in one region than in another. This difference is called a concentration gradient. v. Dynamic equilibrium – there is continuous movement, but no overall change v. Dynamic = movement v. Equilibrium = balance
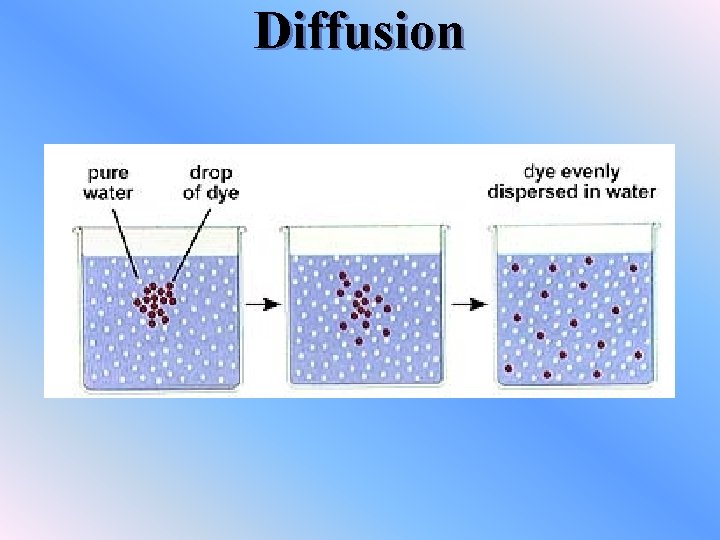
Diffusion

Osmosis v. Osmosis is the diffusion of water molecules through a selectively permeable membrane from an area of high concentration to an area of low concentration v. A cell will lose water if it is placed in an environment in which the water concentration is lower than that of the cell contents

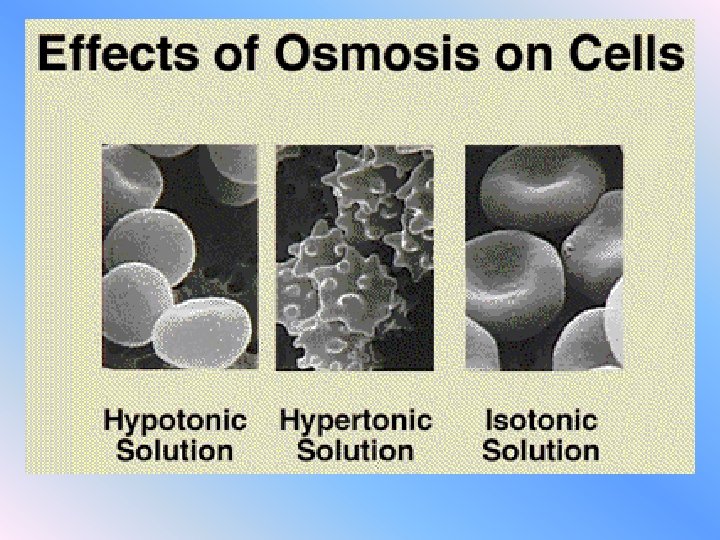
ISOTONIC SOLUTION v. The concentration of a dissolved substance is the same as the concentration inside the cell. v. Water molecules will still move, but there is no net movement of water. v. No osmosis occurs b/c the cell is in dynamic equillibrium

HYPOTONIC SOLUTION v. The concentration of dissolved substances is lower than the concentration within the cell v. Water will rush into the cell causing an increase in pressure v. This pressure is called “turgor pressure”

HYPERTONIC SOLUTION v. The concentration of dissolved substances is higher than the concentration inside the cell v. Water will rush out of the cell causing it to shrink v. This loss of water from the cell is called plasmolysis
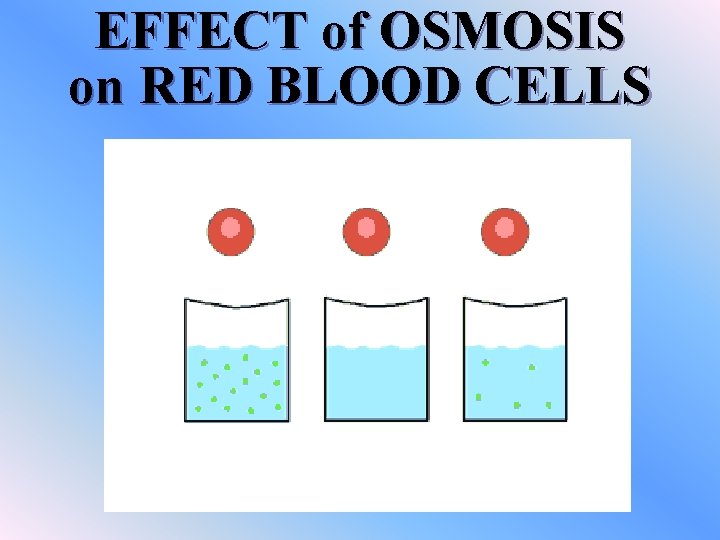
EFFECT of OSMOSIS on RED BLOOD CELLS
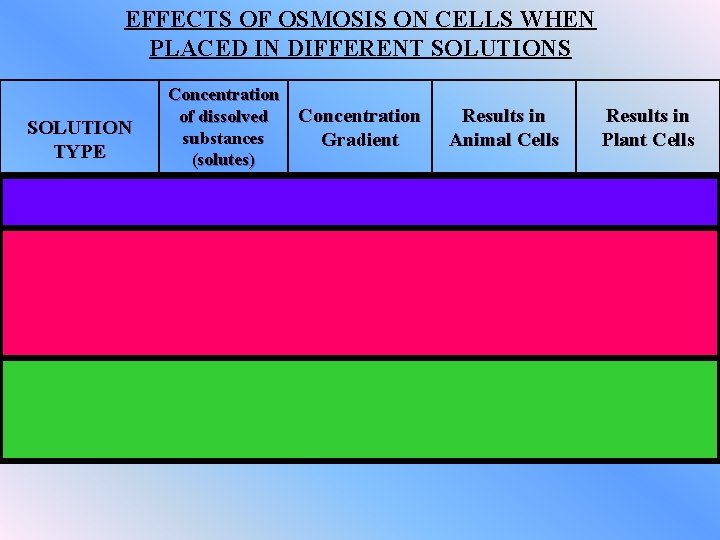
EFFECTS OF OSMOSIS ON CELLS WHEN PLACED IN DIFFERENT SOLUTIONS SOLUTION TYPE ISOTONIC HYPERTONIC Concentration of dissolved substances (solutes) Concentration Gradient Results in Animal Cells Results in Plant Cells Same as the living cell Zero No change No Change Lower than the living cell Net water movement into the cell ** Pressure created by Cell swells and excess water bursts stored in central vacuole Higher than the living cell Net water movement out of the cell Central vacuole collapses and plasmolysis occurs Cell shrinks * plasmolysis
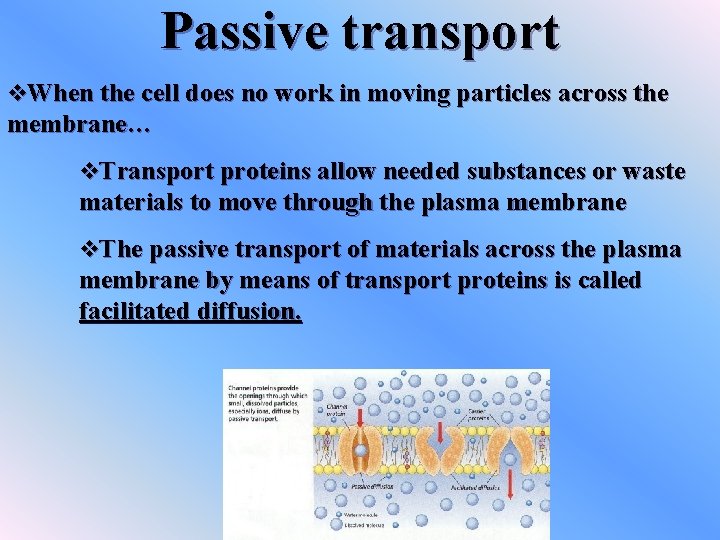
Passive transport v. When the cell does no work in moving particles across the membrane… v. Transport proteins allow needed substances or waste materials to move through the plasma membrane v. The passive transport of materials across the plasma membrane by means of transport proteins is called facilitated diffusion.

Active transport v. The transport of materials against a concentration gradient which requires energy. v. BOTH endo- and exocytosis requires energy to move materials through the membrane v. Endocytosis vthe cell surrounds and takes in materials from its environment v. Exocytosis vused to expel wastes
- Slides: 18