Home Back to water As a scuba diver

Home Back to water As a scuba diver goes deeper underwater, the diver must be aware that the increased Going deeper under water, pressure affects the human body by increasing the gasses of the blood the — become more condensed Coming up too fast, and “dissolved”. A body’s temperature is like shaking a in the body B amount of dissolved gases C amount suspended solids in the body canof of coke and D concentration of minerals in the body then opening up. The body doesn’t like it. It’s called the ‘bends’. 9

Home Back to water

Home Back to water
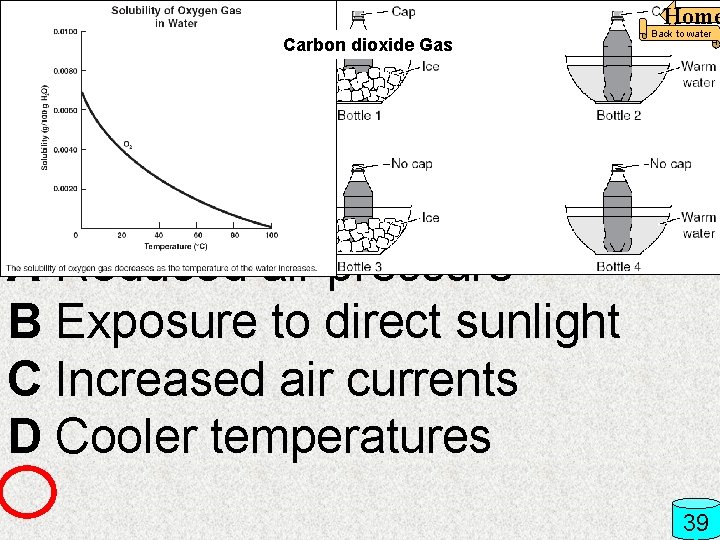
Home Over time an open soft drink will lose carbonation (dissolved CO 2). Which of these allows the CO 2 to remain in solution the longest? A Reduced air pressure B Exposure to direct sunlight C Increased air currents D Cooler temperatures Carbon dioxide Gas Back to water 39
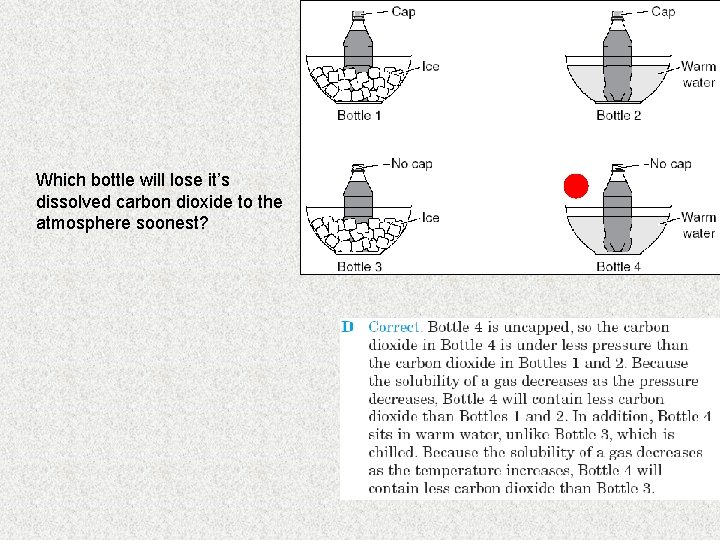
Which bottle will lose it’s dissolved carbon dioxide to the atmosphere soonest?
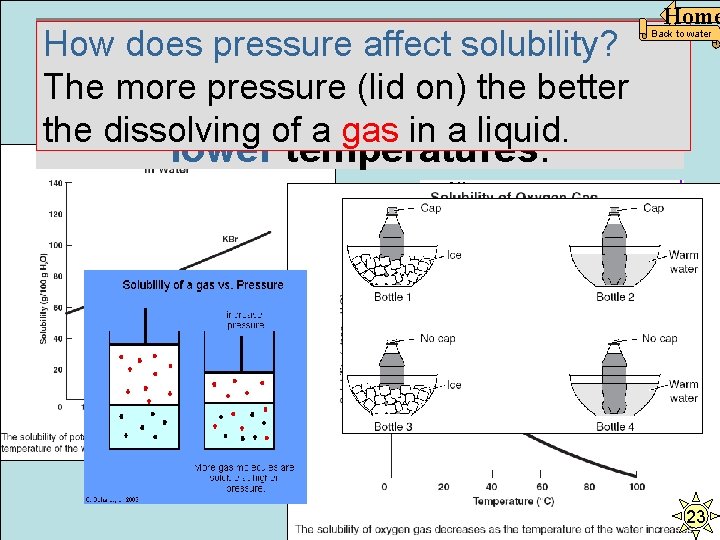
Home How does temperature affect How does pressure affect solubility? The. But more pressure (lid on) the better solubility? gases dissolve better at the dissolving of a Usually, gas inthea warmer, liquid. the better. Back to water lower temperatures. The table shows temperature and pressure in four containers holding the same amount of water. According to the table, in which container can the most sodium chloride be dissolved in the water? AQ BR Rule: With solubility of most salts, CS temperature is more important than pressure. DT 23

Home Back to water

Home Back to water
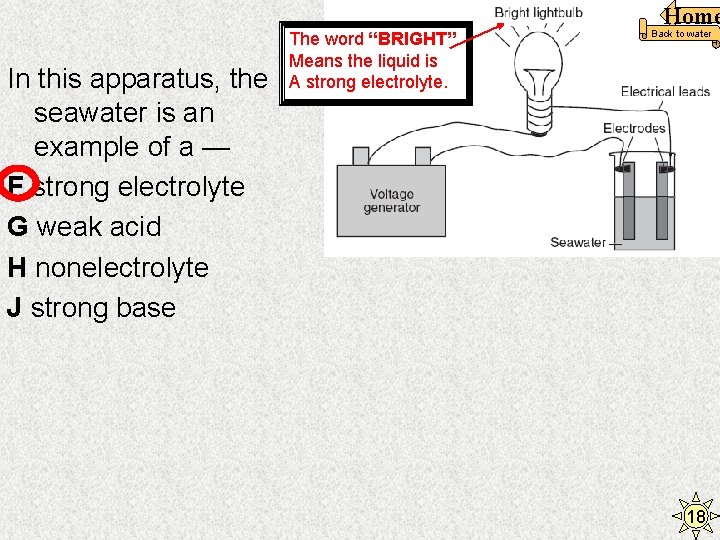
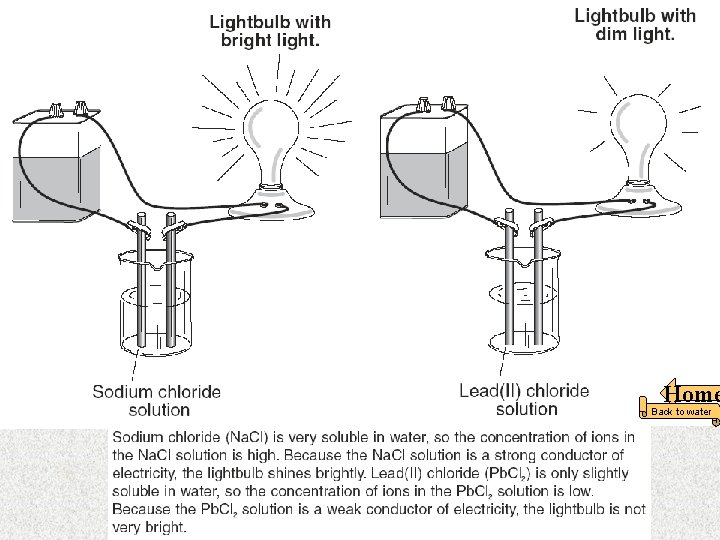
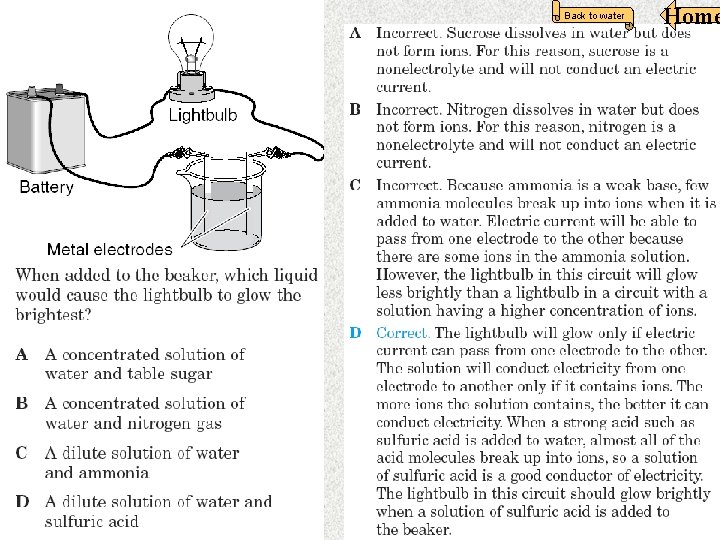
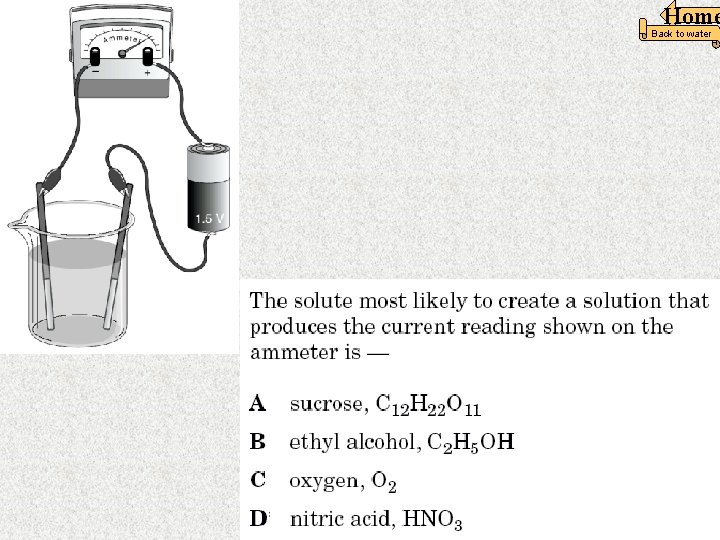
In this apparatus, the seawater is an example of a — F strong electrolyte G weak acid H nonelectrolyte J strong base The word “BRIGHT” Means the liquid is A strong electrolyte. Home Back to water 18

Home Back to water
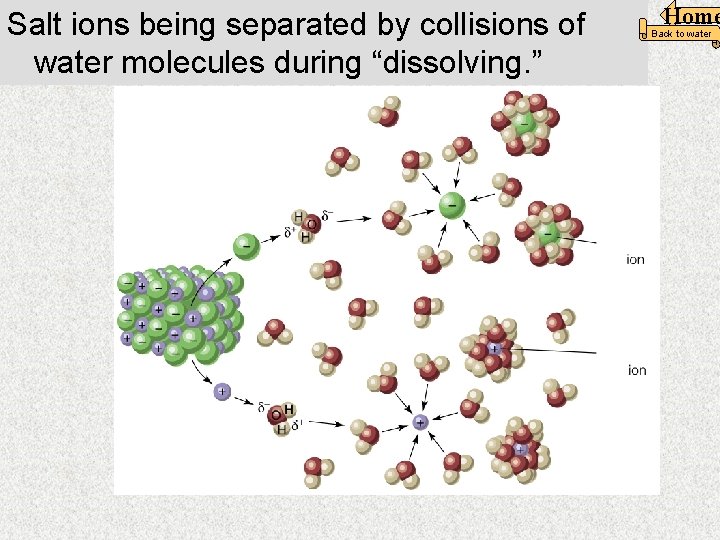
Salt ions being separated by collisions of water molecules during “dissolving. ” Home Back to water
Home Back to water
Back to water Home
Home Back to water

Home Back to water

Home Bathwater normally has electrolytic behaviors even though distilled water does not. This is because bathwater — F contains isotopes of hydrogen G has been heated H is separated into H+ and OH– Elements of water are covalently bonded that they resist becoming ions meaning Some always do, but they do not conduct electricity because they are so attracted to the J contains opposite ion. dissolved minerals 24 Back to water Isotopes are elements with different number of neutrons although they have the same number of electrons and protons. Heating water does NOT make the water an electricity carrier. Ions in the water, make water carry electricity.

Home Back to water Sour Slippery

Home Back to water p. H indicators Litmus paper tells acid or base p. H paper tells p. H numbers

Home A certain commercial product used for cleaning ovens must be handled with rubber gloves. The product is slippery and turns litmus paper blue. It probably contains — F an acid G a base H a salt J an isotope Back to water 34

Home Two clear solutions are placed in separate beakers. The first solution has a p. H of 4, and the p. H of the second solution is unknown. If the two solutions are mixed and the resulting p. H is 5, the second solution must have — Back to water A fewer suspended solids 33 B a lower temperature C more dissolved salt (Na. Cl) particles D a higher concentration of OH– ions
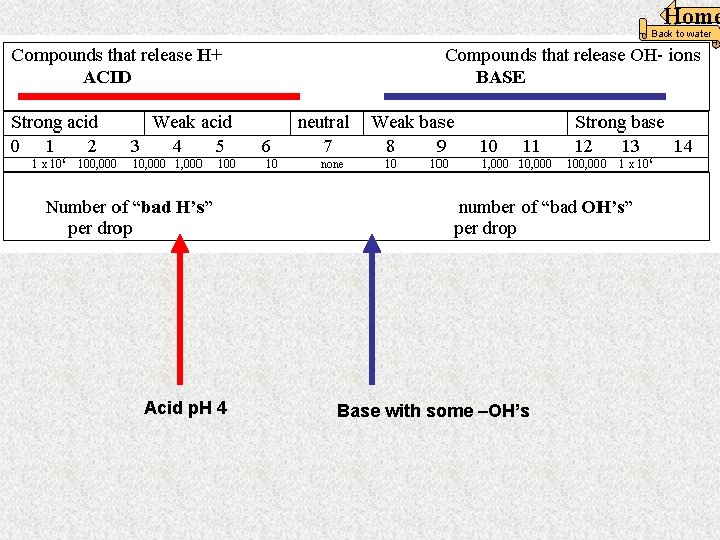
Home Back to water Acid p. H 4 Base with some –OH’s

Home Back to water Acid + Base becomes Salt + Water

Many pitcher-plant species grow well in soil with a low p. H. Soil p. H could be decreased by adding a solution of — A carbonic acid B potassium hydroxide C ammonia D sodium chloride Home Back to water

physical and chemical change Home

Which of the following processes is an example of a physical change associated with an oak tree? A Decomposition of bark by bracket fungi B Starches and sugars being broken down during energy production C Water and carbon dioxide being converted to glucose D Evaporation of water from the surfaces of leaves Home Chemical change Physical change 31

Home Chemical changes: Bonding partners are changed. Milk sugar + proteins → carbon dioxide + water + other chemicals Which of the following is an example of a chemical change? F Ice cracking G Sugar dissolving H Milk souring J Lead melting Physical changes: Getting smaller Dissolving Changing phases…solid, liquid, gas 22

Which of these changes in rocks is a physical change? A Acid rain damaging marble B Iron in rock combining with oxygen to form hematite C Carbonic acid weathering limestone D An ice wedge shattering a slab of shale Home 13

Home Which process in the rock cycle is most likely responsible for moon rocks being converted to lunar soil? A Metamorphism B Weathering C Sedimentation D Volcanism 7 Reheating of rocks. Weathering changes rock to soil on the earth, so it might apply to the moon as well.

Home Which of these describes a pollution-producing process that involves Normal only environment a physical change? Coal with + S +a Oxygen → water COcontent SOto Hot added A Coal high sulfur is. Hburned, 2 is+ 3 a+river 2 O → H 2 SO 4 or lake. producing gases that cause acid rain. Warmed Oxygen O B Chlorofluorocarbons are released, O 3 → O 2 + O 1 with the bubbles to changing ozone in the upper atmosphere. into oxygen. C Hot wastewater is. Thermal discharged into a lake, pollution causes a physical change. lowering oxygen levels in the water. D NO Nitrogen oxide emissions combine with + H O → HNO 2 2 3 water vapor, producing nitric. Decrease acid. in dissolved oxygen 2 causes fish kill. 31

Home
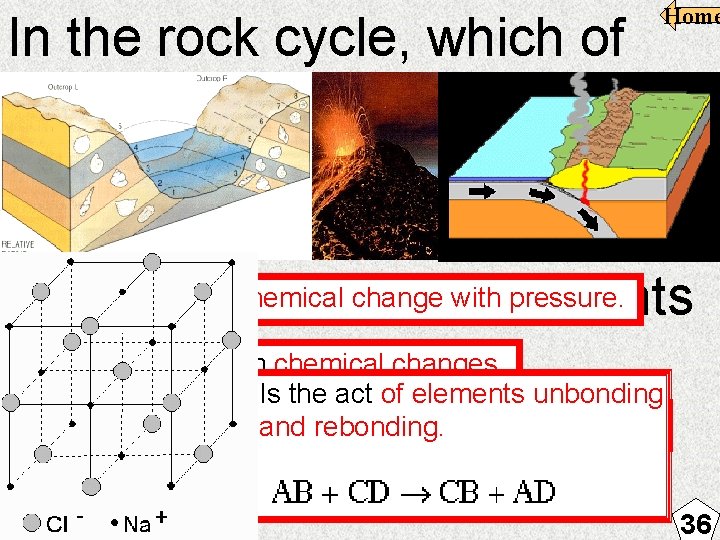
Home In the rock cycle, which of these is a chemical change involved with the formation of igneous rocks? May cause a chemical change with pressure. F Compression of sediments May resultloss from chemical changes. G Heat from lava Is the act of elements unbonding May cause a chemical change with heat/friction. and rebonding. H Subduction of plates J Formation of minerals 36

Home

Home density
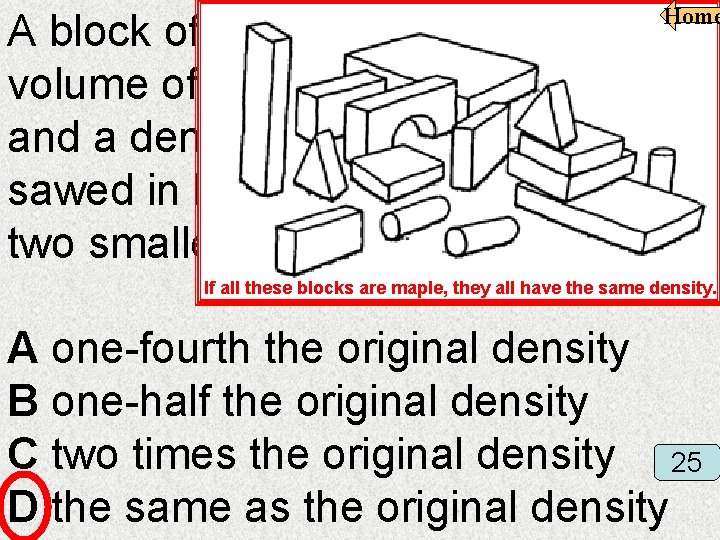
A block of maple wood with a volume of 405 cubic centimeters and a density of 0. 67 g/cm 3 is sawed in half. The density of the two smaller blocks is now — Home If all these blocks are maple, they all have the same density. A one-fourth the original density B one-half the original density C two times the original density 25 D the same as the original density

Home A sample of an element has a volume of 78. 0 m. L and a density of 1. 85 g/m. L. What is Density = mass ÷ volume 1. 85 = ? ÷ 78 the mass 1. 85 x 78 in = ? grams of the sample? Record and bubble in your answer to the nearest tenth on the answer document. 20 144. 3 grams

Home Which procedure is best to use when determining the density of a rock? F Place the rock in a water-filled beaker and find the height at which the rock floats above the water. G Use a ruler to measure the rock’s dimensions and then find its mass using an analytical balance. H Measure the mass of the rock on a balance and then find the volume of water it displaces in a graduated cylinder. J Place the rock in three liquids with different known densities and observe which liquid the rock floats in. 6

Home 161. 02 – 22. 35 = 139. 67 Density = mass / volume Density = 139. 67 g / 20. 1 m. L 60. 4 – 40. 3 = 20. 1

Home Which of the following objects will float on water? 55/50 = greater than 1 45/40 = greater than 1 Density greater than “ 1” sinks. Density less than “ 1” floats. 50/45 = greater than 1 60/65 = less than 1 27
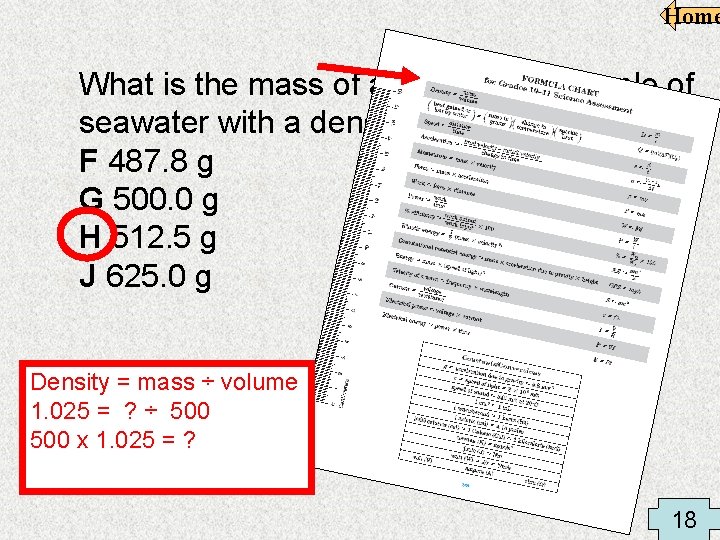
Home What is the mass of a 500. 00 m. L sample of seawater with a density of 1. 025 g/m. L? F 487. 8 g G 500. 0 g H 512. 5 g J 625. 0 g Density = mass ÷ volume 1. 025 = ? ÷ 500 x 1. 025 = ? 18

Home Gases have a greater volume than liquids. Compared to 250 g of gaseous nitrogen, 250 g of liquid nitrogen has greater — A volume B temperature Density is mass / volume C mass D density Gas N 2 Liquid N 2 250/big number 250/small number 35
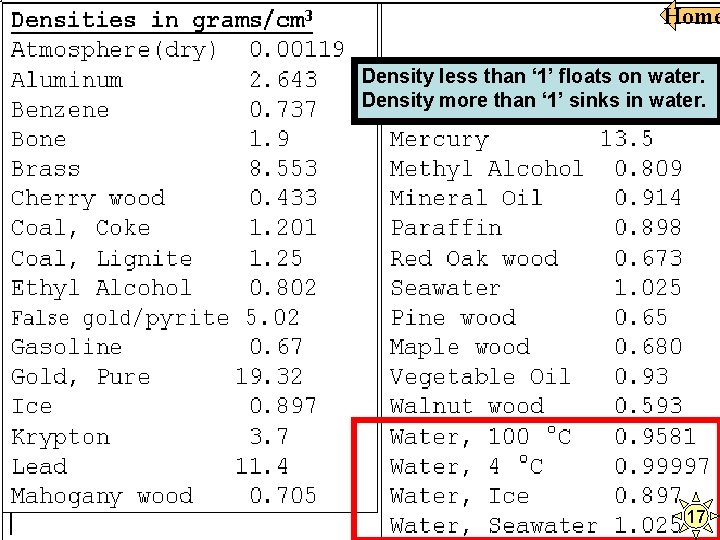
Home Density less than ‘ 1’ floats on water. Density more than ‘ 1’ sinks in water. 17 Swimmers find that they can float more easily in the ocean than in a freshwater pond. The most likely reason for this phenomenon is that the — A viscosity of pond water is greater than that of ocean water B density of ocean water is higher than that of pond water C temperature of pond water is lower than that of ocean water D mass of ocean water is greater than that of pond water. 17

Home From 1942 to 1945, U. S. nickels were made of an alloy that contained 35% silver, 9. 0% manganese, and the rest copper, by mass. If one of these nickels has a mass of 5. 0 grams, what is the mass of the copper? copper = 100% A 0. 5 g Silver + manganese + Copper = 100 - 35 - 9 B 1. 8 g Copper = 56% or 56/100 or. 56 C 2. 2 g ÷ D 2. 8 g 56 / 100 x 5 grams = 2. 8 grams or. 56 x 5 = 2. 8 23

What is the density at 20°C of 12. 0 milliliters of a liquid Density = mass ÷ volume that has a mass of 4. 05 ? = 4. 05 g ÷ 12 m. L grams? A 0. 338 g/m. L B 2. 96 g/m. L C 16. 1 g/m. L D 48. 6 g/m. L Home Don’t need temp. 31

Home This pipette is filled with a 20% Na. OH solution. The solution is at 20°C and has a density of 1. 23 g/m. L. According to this information, what is the mass of this Na. OH solution? A 3. 88 g B 15. 7 g C 23. 9 g D=m÷V D x V =gm D 24. 6 1. 23 x amount in graduated cylinder = mass 27

Home

Home

Home
- Slides: 47