HIV1 integrase inhibitors resistance among antiretroviral treatment experienced
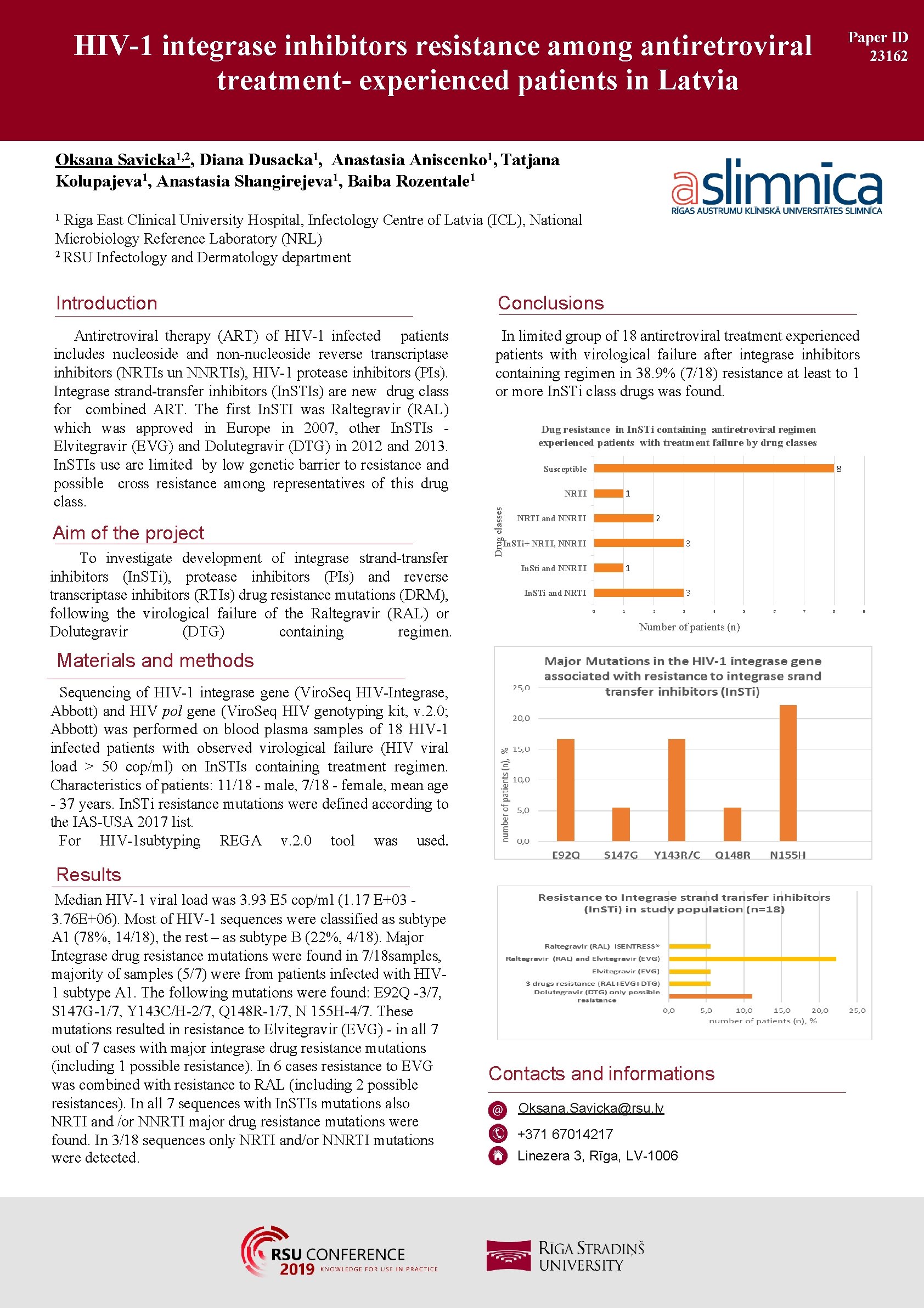
HIV-1 integrase inhibitors resistance among antiretroviral treatment- experienced patients in Latvia Paper ID 23162 Oksana Savicka 1, 2, Diana Dusacka 1, Anastasia Aniscenko 1, Tatjana Kolupajeva 1, Anastasia Shangirejeva 1, Baiba Rozentale 1 Riga East Clinical University Hospital, Infectology Centre of Latvia (ICL), National Microbiology Reference Laboratory (NRL) 2 RSU Infectology and Dermatology department 1 Introduction Conclusions Antiretroviral therapy (ART) of HIV-1 infected patients includes nucleoside and non-nucleoside reverse transcriptase inhibitors (NRTIs un NNRTIs), HIV-1 protease inhibitors (PIs). Integrase strand-transfer inhibitors (In. STIs) are new drug class for combined ART. The first In. STI was Raltegravir (RAL) which was approved in Europe in 2007, other In. STIs Elvitegravir (EVG) and Dolutegravir (DTG) in 2012 and 2013. In. STIs use are limited by low genetic barrier to resistance and possible cross resistance among representatives of this drug class. In limited group of 18 antiretroviral treatment experienced patients with virological failure after integrase inhibitors containing regimen in 38. 9% (7/18) resistance at least to 1 or more In. STi class drugs was found. To investigate development of integrase strand-transfer inhibitors (In. STi), protease inhibitors (PIs) and reverse transcriptase inhibitors (RTIs) drug resistance mutations (DRM), following the virological failure of the Raltegravir (RAL) or Dolutegravir (DTG) containing regimen. 8 Susceptible 1 NRTI Drug classes Aim of the project Dug resistance in In. STi containing antiretroviral regimen experienced patients with treatment failure by drug classes 2 NRTI and NNRTI 3 In. STi+ NRTI, NNRTI 1 In. Sti and NNRTI 3 In. STi and NRTI 0 1 2 3 4 Number of patients (n) Materials and methods Sequencing of HIV-1 integrase gene (Viro. Seq HIV-Integrase, Abbott) and HIV pol gene (Viro. Seq HIV genotyping kit, v. 2. 0; Abbott) was performed on blood plasma samples of 18 HIV-1 infected patients with observed virological failure (HIV viral load > 50 cop/ml) on In. STIs containing treatment regimen. Characteristics of patients: 11/18 - male, 7/18 - female, mean age - 37 years. In. STi resistance mutations were defined according to the IAS-USA 2017 list. For HIV-1 subtyping REGA v. 2. 0 tool was used. Results Median HIV-1 viral load was 3. 93 E 5 cop/ml (1. 17 E+03 3. 76 E+06). Most of HIV-1 sequences were classified as subtype A 1 (78%, 14/18), the rest – as subtype B (22%, 4/18). Major Integrase drug resistance mutations were found in 7/18 samples, majority of samples (5/7) were from patients infected with HIV 1 subtype A 1. The following mutations were found: E 92 Q -3/7, S 147 G-1/7, Y 143 C/H-2/7, Q 148 R-1/7, N 155 H-4/7. These mutations resulted in resistance to Elvitegravir (EVG) - in all 7 out of 7 cases with major integrase drug resistance mutations (including 1 possible resistance). In 6 cases resistance to EVG was combined with resistance to RAL (including 2 possible resistances). In all 7 sequences with In. STIs mutations also NRTI and /or NNRTI major drug resistance mutations were found. In 3/18 sequences only NRTI and/or NNRTI mutations were detected. Contacts and informations Oksana. Savicka@rsu. lv +371 67014217 Linezera 3, Rīga, LV-1006 5 6 7 8 9
- Slides: 1