HIV TESTING TECHNOLOGIES ELISA WESTERN BLOT There are

HIV TESTING TECHNOLOGIES ELISA / WESTERN BLOT

There are numbers of tests They should be used in combination (strategies) Combinations must be consistent

WHO Recommended Strategies Strategy I Test all samples with one EIA Strategy II Strategy I with all reactives retested in a more specific test with different principle and/or antigen. Strategy III Strategy II with reactives tested in a third test differing from the first two tests.

Testing Strategies AIM: To develop the logic used in establishing the use of HIV tests (testing strategies)

Objectives of Testing Strategies To achieve the correct diagnosis in the most efficient manner To maintain consistency in testing To develop baseline data for assessing changes To deliver useful results

Screening Assays § Are used to detect antibody-- specific or nonspecific § Are designed to handle large numbers of samples with rapid throughput § Must be high performance § Should include a full range of HIV antigens
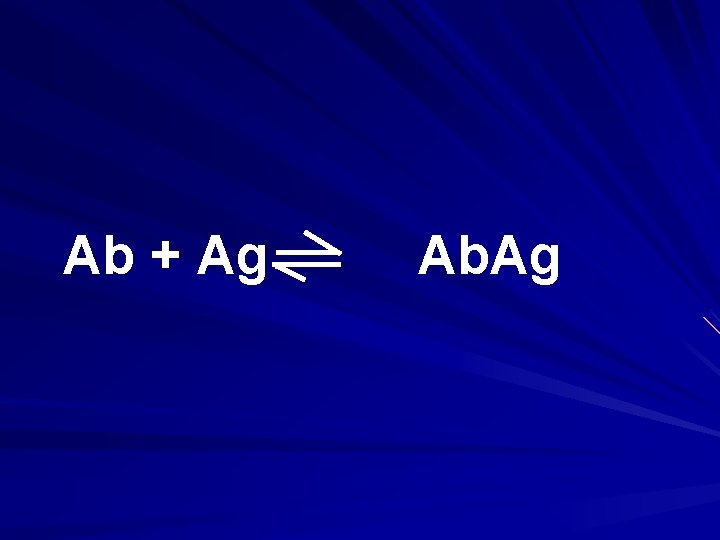
Ab + Ag Ab. Ag

Ab. Ag Ab +Ag Ab. Ag Ab +Ag

Serological Testing Strategy SCREENING TEST, highly sensitive NEG REACTIVE SUPPLEMENTAL TEST, highly sensitive & higher specificity POS ADDITIONAL TESTS NEG
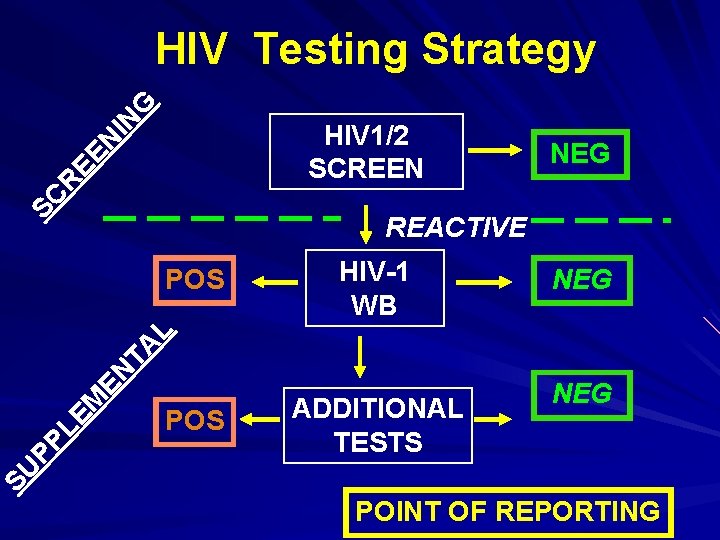
IN G HIV Testing Strategy SC R EE N HIV 1/2 SCREEN NEG REACTIVE HIV-1 WB NEG POS ADDITIONAL TESTS NEG SU PP LE M EN TA L POS POINT OF REPORTING

The Use of Screening Assays Define samples as negative for a given analyte Enable high throughput

Why Follow a Strategy?

The Importance of Maintaining a Strategy Consistency of laboratory records Consistency of results Clarity of results to doctors Maintaining data base to assess performances Avoiding common false reactivity Avoiding technical errors Reducing costs

WHO Recommended Strategies Strategy I Test all samples with one EIA Strategy II Strategy I with all reactives retested in a more specific test with different principle and/or antigen. Strategy III Strategy II with reactives tested in a third test differing from the first two tests.

Objectives for HIV testing Diagnosis Surveillance Blood transfusion safety

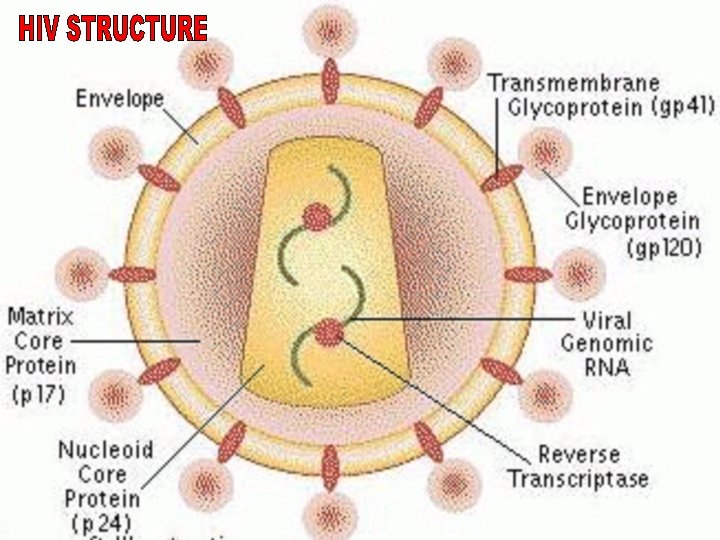
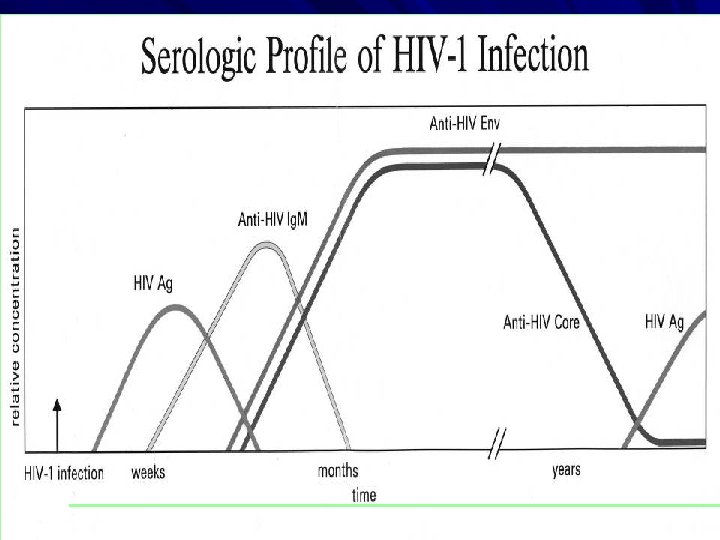
Kinetics of Antibody Response to HIV KNOWLEDGE VIRAL STRUCTURE STRUCTURAL PROTEIN OF HIV 1 AND HIV 2 HIV 1 ENV – gp 41, 120, 160 core – p 55, 18, 24 pol – p 31, 51, 65 HIV 2 ENV – gp 36, 140, core – p 56, 26, 16 pol – 68, 53, 34 Viral entry, Target cell (CD 4) Window period Ig. M. Ig. G

Different Test for HIV DIRECT INDIRECT v PRE-TEST COUNSELING, INFORMED CONSENT, CONFIDENTIALITY.

Challenges of HIV Testing Sensitivity - Early diagnostic ( window period) Specificity- Cross reactivity Easy to perform, low cost Détection of HIV-1 & HIV-2 and discrimination between the two viruses One test can not fulfill these requirements Need to perform a combination of HIV tests for screening and confirmation

Current HIV technologies Detection of antibodies Screening tests Enzyme immunosorbent assays (EIAs) Simple/rapid immuno-diagnostics assays Confirmatory or supplemental tests Western blot (WB) Alternatives to confirmatory tests Repetitive EIA or rapid assays

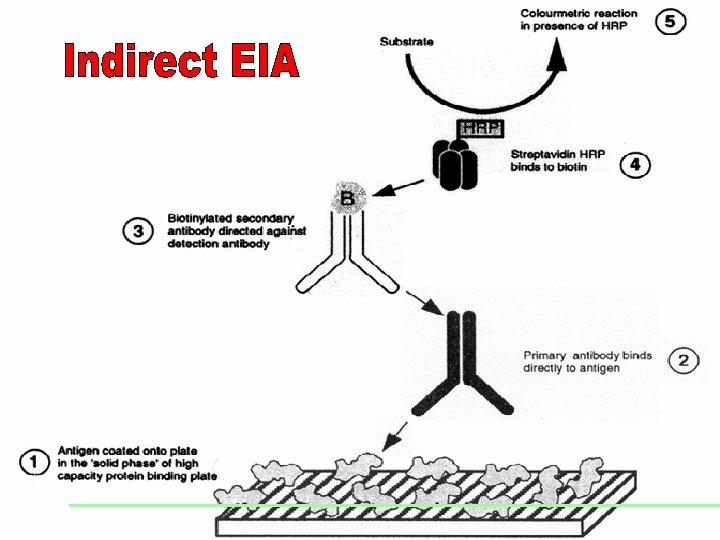
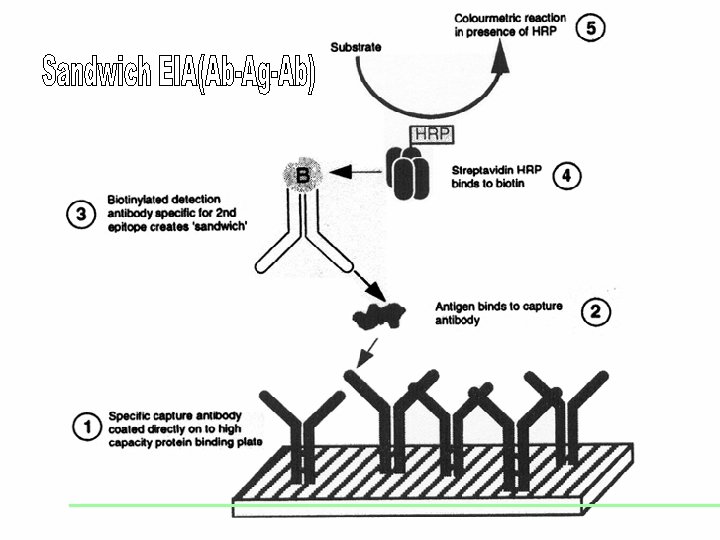
EIAs (Enzyme Immunosorbent Assays) This term describes a variety of assays that are based on the binding of antibodies with their antigens and the detection of this reaction using a component conjugated with an active enzyme. This enzyme acts on its substrate to produce a colour change. Test results are measured by measuring this colour. Four immunologic principles Indirect Competition Sandwich Immuno-capture

Competitive EIA A measured amount of known enzyme-labeled component (being measured) is added to the reaction at the same time patient sample is added. The labeled component therefore competes against the unlabeled component in the patient sample for binding sites. Results Negative Reaction has color change Positive Reaction no color change
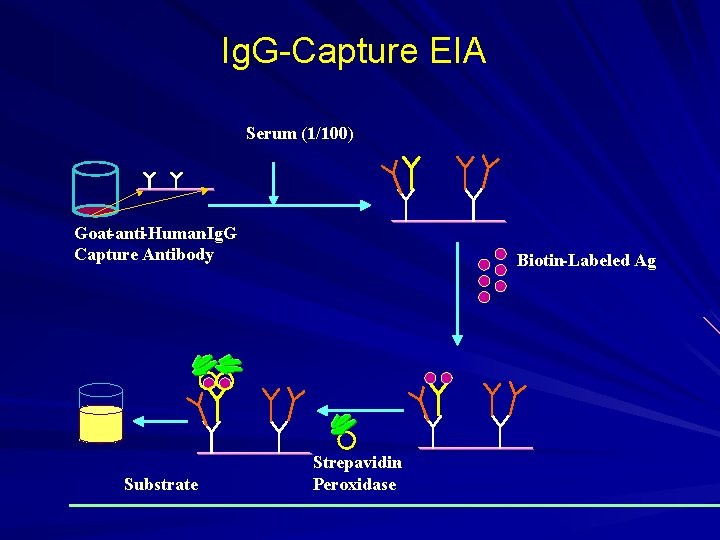
Ig. G-Capture EIA Serum (1/100) Goat-anti-Human-Ig. G Capture Antibody Substrate Biotin-Labeled Ag Strepavidin. Peroxidase

Reason for EIA This test supplied as kit Easy to Perform Use to screen large number of sample Sensitive Specific Cost effective

Reason for EIA This test supplied as kit Easy to Perform Use to screen large number of sample Sensitive Specific Cost effective

Components of Commercially Available EIA Kits Solid–Phase Support Antigens bound to polystyrene microtiter plates (passive absorption) Blocking is necessary to reduce nonspecific binding (test accuracy) 96 microwell format Antigens The use of cloned antigens has reduced nonspecific binding Antibodies Monoclonals of high titer, affinity and avidity

Components of Commercially Available EIA Kits Conjugates Ab conjugated with enzyme (without effecting binding site) Enzyme HRP (horseradish peroxidase) Substrate (chromogenic) Colorless chromogen reacts with enzyme ( color) • Stop Solution • Typically an acid, stabilizes the color for a limited time

Sources of Error for HIV EIA Tests Documented Sources of False Negative Results Operator Error FAIL TO ADD SERUM OR REAGENT TO THE CORRECT WELL REAGENT DILUTED IN WRONG DILUENT IN WRONG DILUTION

Equipment : 1 - Pipettes Single and Multi channel Pipettes should be calibrated on a monthly basis. This can be done using a balance. Inaccurate pipetting

Equipment : 2 - Microplate Washers Daily Prime the washer with wash solution before running sample plates Set the washer to wash the recommended number of times (with correct volume) Check for accurate dispensing and complete aspiration in each plate well, if not clean the washer head Listen for changes in the sound the washer makes, this can indicate a vacuum leak At the end of the day prime the washer with DI water

Equipment : Micro-plate Washers Weekly If a washer is not used during the week rinse it out with DI water to reduce microbial growth. Monthly Run a 10% solution of ethanol through the washer to disinfect. This can also be done if the washer exhibits signs of contamination (high background). Thoroughly rinse the washer after alcohol is used.

Equipment : Micro-plate Reader Daily – Each time a reader is turned on it runs a self test, it will then report any errors. Weekly – Run a control plate weekly. Variations in positive or negative specimens could be a sign of a bad diode or a spill on a diode.

Source of False Positive Results MULTIPLE PREGNANCY MULTIPLE TRANSFUSION AUTO IMMUNE DISORDER CHRONIC HEPATITIS, CHRONIC ALCOHOLIC HBV VACCINATION ANTIBODY TO POLYSTERENE

Cross contamination Can be caused by: Reusing pipette tips (contaminated with + plasma) Splashes from one well to another During removal of plate covers

Sample Quality Properly collected (no haemolysis) Transport conditions Storage conditions Number of freeze/thaw cycles Age of sample

Validation and Interpretation of Results Product inserts provide guidelines Positive and Negative controls must fall within a certain range. Controls are used to calculate a cut-off. Samples below cut-off are negative, those above are positive

Western Blot (Immunoblotting) Solid-phase EIA with immobilized viral antigens to detect antibodies to specific HIV proteins.

Principle AIDS is caused by at least 2 etiological agents HIV-1 & HIV-2 Inactivated and denatured protein of HIV-1 are fractioned by polyacrylamide gel electrophoresis Protein bands are transferred into nitrocellulose strips HIV-1 sample diluted with buffer are then incubated with the strip

Conjugate peroxidase labeled anti human Ig. G is added It will bind to the antibodies already bound to the strip Chromogen is then added forming color reaction Reaction is then stopped by aspiration and reaction

Sample requirement: Serum sample Maximum 8 days Stored 2 o C – 8 o. C or frozen at – 25 o. C Lipemic sample must be centrifuged well Avoid heating
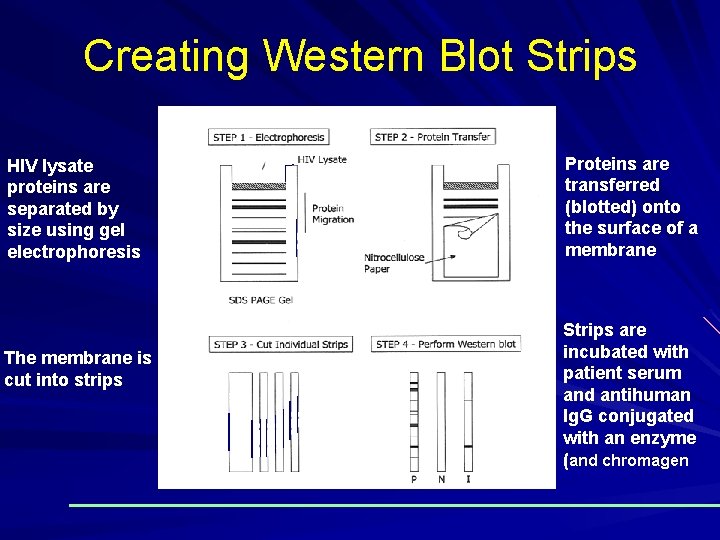
Creating Western Blot Strips HIV lysate proteins are separated by size using gel electrophoresis The membrane is cut into strips Proteins are transferred (blotted) onto the surface of a membrane Strips are incubated with patient serum and antihuman Ig. G conjugated with an enzyme (and chromagen)
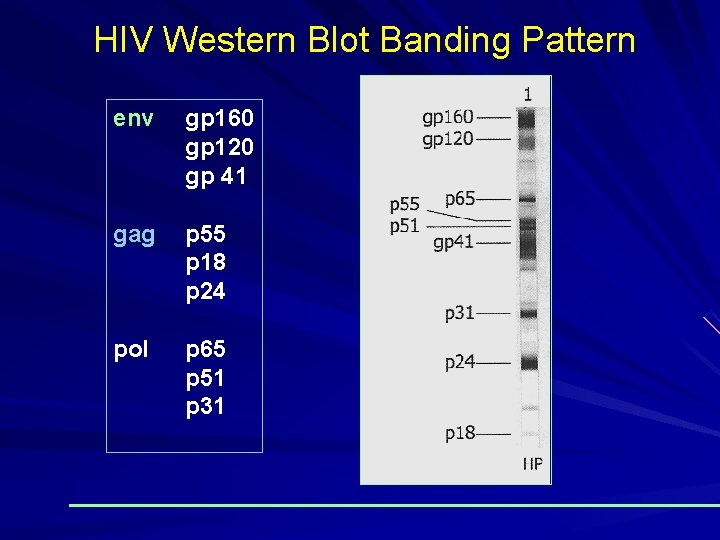
HIV Western Blot Banding Pattern env gp 160 gp 120 gp 41 gag p 55 p 18 p 24 pol p 65 p 51 p 31

Interpretation of Results (General Consensus) Negative: No bands present Positive: 2 ENV band present (WHO Guidelines) Indeterminate Any bands present but do not meet criteria for positive
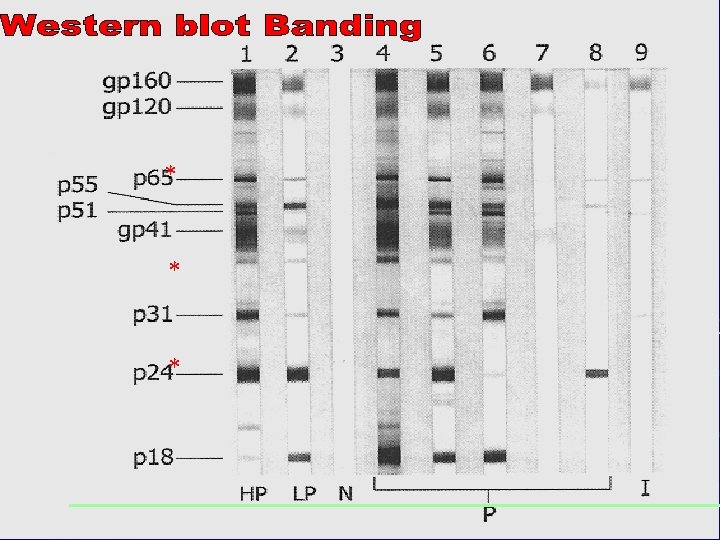

When should WB be used? Western Blot assay should not be used as a screening test. WB should be viewed as a supplemental test which can be used to confirm positive results obtained from EIA. HOWEVER: Specificity is less than that of EIA – A significant number of indeterminate blots are seen in low risk populations –

Advantages Specific interaction of antibody and antigen can be directly visualized. Disadvantages Technically demanding Expensive Subject to interpretation Presence or absence of bands Intensity of those bands
- Slides: 49