HIV PRIMARY CARE Derrick Butler MD MPH Associate

HIV PRIMARY CARE Derrick Butler, MD, MPH Associate Medical Director T. H. E. Clinic, Inc Los Angeles, CA

Quiz 1) Magic Johnson tested positive for HIV in 1992 and is now cured of the virus. a) True, he is rich and can afford the best medicine. b) False, he is still infected, but is controlled on medication. c) Don’t know, I don’t follow football.

We’ve Come a Long Way, Baby
Electron micrographic picture
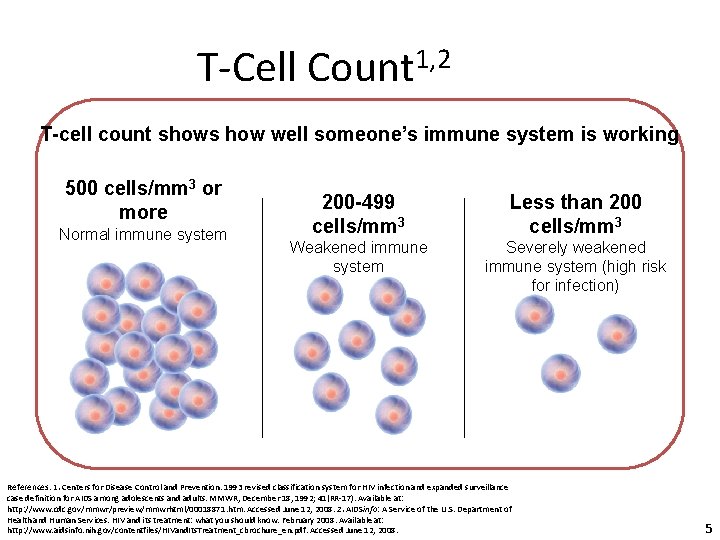
T-Cell Count 1, 2 T-cell count shows how well someone’s immune system is working 500 cells/mm 3 or more Normal immune system 200 -499 cells/mm 3 Less than 200 cells/mm 3 Weakened immune system Severely weakened immune system (high risk for infection) References: 1. Centers for Disease Control and Prevention. 1993 revised classification system for HIV infection and expanded surveillance case definition for AIDS among adolescents and adults. MMWR, December 18, 1992; 41(RR-17). Available at: http: //www. cdc. gov/mmwr/preview/mmwrhtml/00018871. htm. Accessed June 12, 2008. 2. AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. Accessed June 12, 2008. 5
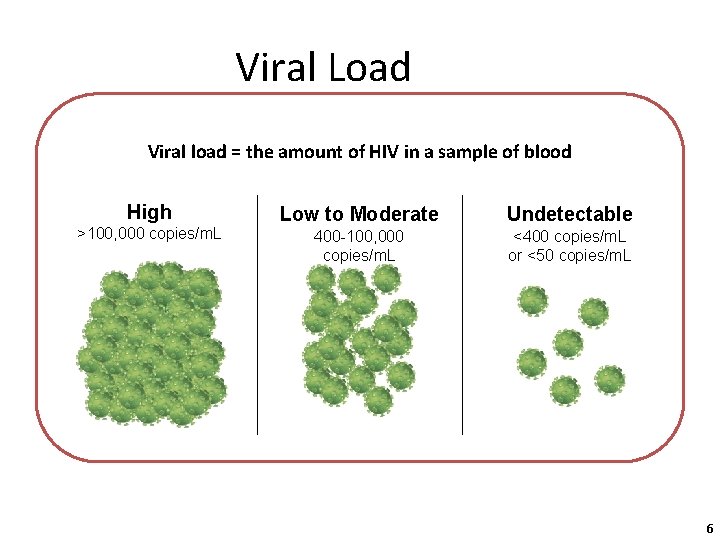
Viral Load Viral load = the amount of HIV in a sample of blood High >100, 000 copies/m. L Low to Moderate Undetectable 400 -100, 000 copies/m. L <400 copies/m. L or <50 copies/m. L 6
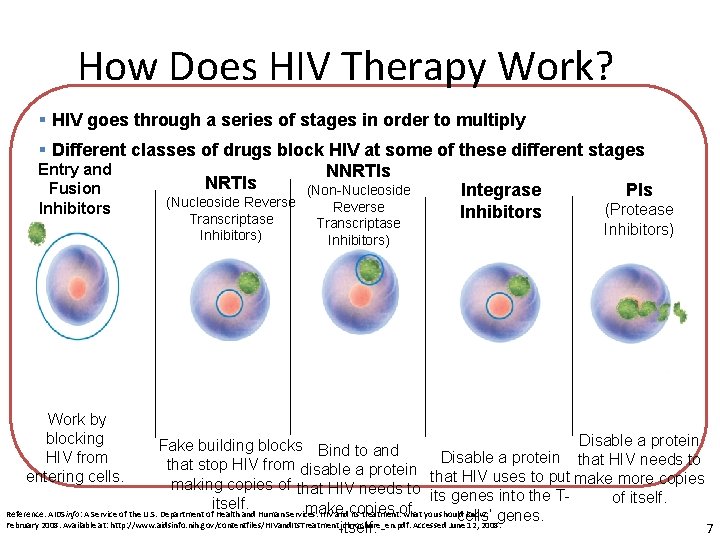
How Does HIV Therapy Work? § HIV goes through a series of stages in order to multiply § Different classes of drugs block HIV at some of these different stages Entry and NNRTIs Fusion (Non-Nucleoside PIs Integrase (Nucleoside Reverse Inhibitors (Protease Inhibitors Transcriptase Inhibitors) Work by blocking HIV from entering cells. Transcriptase Inhibitors) Disable a protein Fake building blocks Bind to and that stop HIV from disable a protein Disable a protein that HIV needs to making copies of that HIV needs to that HIV uses to put make more copies its genes into the Tof itself. make copies of Reference: AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. genes. cells’ February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. Accessed June 12, 2008. 7 itself.

NRTI Current ARV Medications ● Abacavir (ABC) ● Didanosine (dd. I) ● Emtricitabine (FTC) ● Lamivudine (3 TC) ● Stavudine (d 4 T) ● Tenofovir (TDF) ● Zidovudine (AZT, ZDV) PI ● Atazanavir (ATV) ● Darunavir (DRV) ● Fosamprenavir (FPV) ● Indinavir (IDV) ● Lopinavir (LPV) NNRTI ● Nelfinavir (NFV) ● Delavirdine (DLV) ● Ritonavir (RTV) ● Efavirenz (EFV) ● Saquinavir (SQV) ● Etravirine (ETR) ● Tipranavir (TPV) ● Nevirapine (NVP) ●Rilpivirine Fusion Inhibitor ● Enfuvirtide (ENF, T-20) CCR 5 Antagonist ● Maraviroc (MVC) Integrase Inhibitor ● Raltegravir (RAL) ● Elvitegravir (EVG) ● Dolutegravir (DTG)*
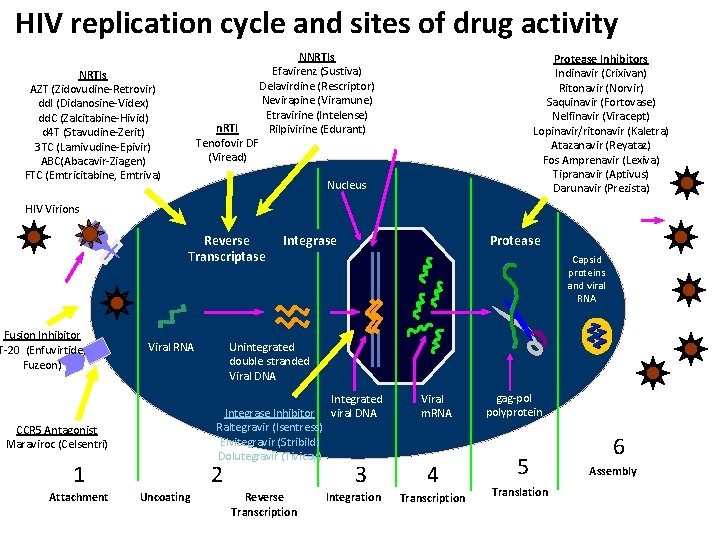
HIV replication cycle and sites of drug activity NNRTIs Efavirenz (Sustiva) Delavirdine (Rescriptor) Nevirapine (Viramune) Etravirine (Intelense) n. RTI Rilpivirine (Edurant) Cellular DNA Tenofovir DF (Viread) NRTIs AZT (Zidovudine-Retrovir) dd. I (Didanosine-Videx) dd. C (Zalcitabine-Hivid) d 4 T (Stavudine-Zerit) 3 TC (Lamivudine-Epivir) ABC(Abacavir-Ziagen) FTC (Emtricitabine, Emtriva) Protease Inhibitors Indinavir (Crixivan) Ritonavir (Norvir) Saquinavir (Fortovase) Nelfinavir (Viracept) Lopinavir/ritonavir (Kaletra) Atazanavir (Reyataz) Fos Amprenavir (Lexiva) Tipranavir (Aptivus) Darunavir (Prezista) Nucleus HIV Virions Reverse Transcriptase Protease Integrase Capsid proteins and viral RNA CD 4 Receptor Fusion Inhibitor T-20 (Enfuvirtide, Fuzeon) Viral RNA 1 Attachment Unintegrated double stranded Viral DNA Integrase Inhibitor Raltegravir (Isentress) Elvitegravir (Stribild) Dolutegravir (Tivicay) CCR 5 Antagonist Maraviroc (Celsentri) 2 Uncoating New HIV particles Reverse Transcription Integrated viral DNA 3 Integration Viral m. RNA 4 Transcription gag-pol polyprotein 5 Translation 6 Assembly and Release

What Is HAART? • HAART stands for Highly Active Antiretroviral Therapy • HAART combines drugs from different classes, slowing HIV replication down at different stages • HAART is also called combination therapy, a “cocktail, ” or a “regimen” Examples of HAART regimens: NNRTI + or PI Reference: AIDSinfo: A Service of the U. S. Department of Health and Human Services. HIV and its treatment: what you should know. February 2008. Available at: http: //www. aidsinfo. nih. gov/contentfiles/HIVand. Its. Treatment_cbrochure_en. pdf. Accessed June 12, 2008. 10
Therapy is Easier, More Potent, and Less Toxic in Single-Tablet Regimens
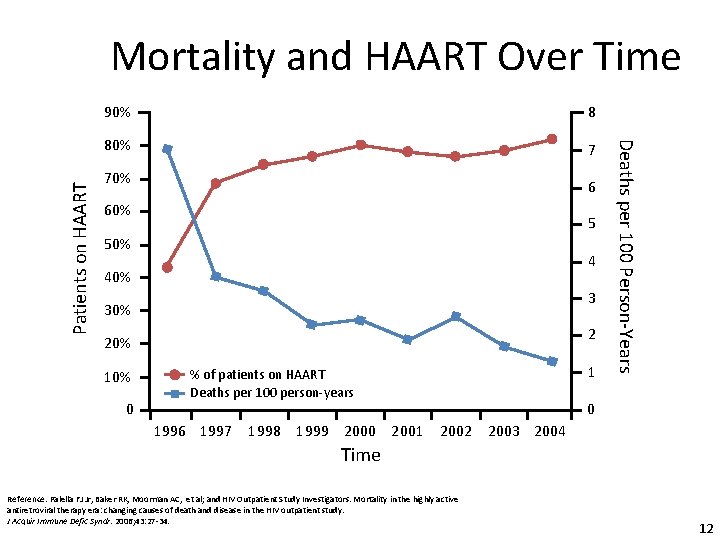
90% 8 80% 7 70% 6 60% 5 50% 4 40% 3 30% 2 20% 10% 0 1 % of patients on HAART Deaths per 100 person-years 1996 1997 1998 1999 2000 2001 Deaths per 100 Person-Years Patients on HAART Mortality and HAART Over Time 0 2002 2003 2004 Time Reference: Palella FJ Jr, Baker RK, Moorman AC, et al; and HIV Outpatient Study Investigators. Mortality in the highly active antiretroviral therapy era: changing causes of death and disease in the HIV outpatient study. J Acquir Immune Defic Syndr. 2006; 43: 27 -34. 12

HIV Patients: Baseline Evaluation • • General history HIV disease characteristics Mental health history Substance abuse history Sexual history Psychosocial assessment Review of systems

Newly Diagnosed Patient: Recommended Laboratory Tests for Initial Visit At Entry Into Care Follow-Up Before HAART CD 4 Cell count and percentage Every 3 to 6 months HIV RNA Every 3 to 6 months Drug resistance testing Recommended for all persons with an HIV RNA >1000 copies/m. L, regardless of treatment initiation Consider if HIV RNA 500 to 1000 copies/m. L Genotypic assay is preferred Genotypic test for all pregnant women DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012.

Newly Diagnosed Patient: Recommended Laboratory Tests for Initial Visit Entry Into Care Follow-Up Before HAART Basic chemistry Serum NA, K, HCO 3, Cl, BUN, creatinine, glucose (preferable fasting)* Every 6 to 12 months Liver function (ALT, AST) Bilirubin (total and direct) Every 6 to 12 months CBC with differential Every 3 to 6 months Fasting lipid profile If normal, annually Fasting glucose If normal, annually Urinalysis *Renal function determination: include estimation of creatinine clearance using Cockcroft & Gault equation of glomerular filtration rate based on MDRD equation. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012.

DHHS Treatment Guidelines When to Start

ART is Recommended for All HIV-Infected Individuals (DHHS) • To reduce the risk of disease progression – CD 4 <350 cells/mm 3 – CD 4 350 -500 cells/mm 3 – CD 4 >500 cells/mm 3 • To prevent transmission of HIV – Perinatal transmission – Heterosexual transmission – Other transmission risk groups Patients starting ART should be willing and able to commit to treatment and understand the benefits and risks of therapy and the importance of adherence (AIII). Patients may choose to postpone therapy, and providers, on a case-by-case basis, may elect to defer therapy on the basis of clinical and/or psychosocial factors DHHS. http: //aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision May 1, 2014.

DHHS Guidelines: Regimen Classification for Treatment-Naïve Patients • Recommended regimens – Regardless of baseline HIV RNA or CD 4 – Additional options for baseline HIV RNA <100 K copies/m. L • Alternative regimens • ART no longer recommended for initial therapy – – NRTI: zidovudine NNRTI: nevirapine PI: unboosted atazanavir, fosamprenavir or saquinavir + ritonavir Entry inhibitor: maraviroc DHHS. http: //aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision May 1, 2014.

DHHS Guidelines: Recommended Regimens Regardless of Baseline HIV RNA Level or CD 4 Count NNRTI PI INSTI Efavirenz/emtricitabine/tenofovir DF* Atazanavir + ritonavir + emtricitabine/tenofovir DF Darunavir + ritonavir (qd) + emtricitabine/tenofovir DF Raltegravir + emtricitabine/tenofovir DF Elvitegravir/cobicistat/emtricitabine/tenofovir DF* Dolutegravir + abacavir/lamivudine Dolutegravir + emtricitabine/tenofovir DF *Available as a once-daily, single-tablet regimen. Notes: Efavirenz: avoid use in women trying to conceive or are sexually active and not using contraception. Lamivudine may substitute for emtricitabine or visa versa. Tenofovir DF: use with caution in patients with renal insufficiency. Atazanavir + RTV: absorption depends on food and low gastric p. H. Elvitegravir/cobicistat/emtricitabine/tenofovir DF: only for patients with pre-ART creatinine clearance >70 m. L/min. Abacavir: only for patients who are HLA-B*5701 negative. DHHS. http: //aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision May 1, 2014.

DHHS Guidelines: Recommended Regimens Additional Options When Baseline HIV RNA <100 K Copies/m. L NNRTI PI Efavirenz + abacavir/lamivudine Rilpivirine/emtricitabine/tenofovir DF* Atazanavir + ritonavir + abacavir/lamivudine *Available as a once-daily, single-tablet regimen. Notes: Efavirenz: avoid use in women trying to conceive or are sexually active and not using contraception. Abacavir: only for patients who are HLA-B*5701 negative. Lamivudine may substitute for emtricitabine or visa versa. Tenofovir DF: use with caution in patients with renal insufficiency. Rilpivirine/emtricitabine/tenofovir DF: only for patients with pre-ART CD 4 >200 cells/mm 3. Atazanavir + RTV: absorption depends on food and low gastric p. H. DHHS. http: //aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision May 1, 2014.

DHHS Guidelines: Alternative Regimens May Be the Preferred Regimen for Some Patients PI Darunavir + ritonavir + abacavir/lamivudine Lopinavir/r (qd or bid) + abacavir/lamivudine or emtricitabine/tenofovir DF INSTI Raltegravir + abacavir/lamivudine Notes: Abacavir: only for patients who are HLA-B*5701 negative. Lamivudine may substitute for emtricitabine or visa versa. DHHS. http: //aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision May 1, 2014.

Recommended Laboratory Tests at HAART Initiation and Follow-Up Repeat Every HAART Initiation 2 -8 Weeks Post HAART 3 -6 Months 12 Months * * Treatment Failure Clinically Indicated CD 4 HIV RNA Resistance Basic chemistry LFT, bilirubin CBC+differential (ZDV) Fasting lipids Fasting glucose Urinalysis HLA-B*5701 (ABC) Pregnancy (EFV) Tropism 1 1 2 2 (HIVAN) (TDF) (CCR 5 antagonist) *Every 6 to 12 months: on a suppressive regimen and CD 4 counts well above threshold for OI risk. 1 Borderline or abnormal at last measurement; 2 normal at last measurement. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012.

Opportunistic Infections (O. I. ) Candidiasis of bronchi, trachea, or lungs Candidiasis esophageal Cervical cancer (invasive) Coccidioidomycosis, disseminated or extrapulmonary Cryptococcosis, outside of the lungs Cryptosporidiosis, chronic intestinal for longer than 1 month Cytomegalovirus disease (other than liver, spleen or lymph nodes) Encephalopathy (HIV-related) Herpes simplex: chronic ulcer(s) (for more than 1 month); or bronchitis, pneumonitis, or esophagitis Histoplasmosis, disseminated or outside the lung Isosporiasis, chronic intestinal (for more than 1 month) Kaposi's sarcoma (skin cancer –internal and external Lymphoma (Burkitt's), immunoblastic or if primary location is the brain Mycobacterium avium complex (MAC) Mycobacterium, other species, disseminated or if found outside the lungs Pneumocystis jiroveci pneumonia (formerly Pneumocystis carinii) Pneumonia (recurring, persistent infections) Progressive multifocal leukoencephalopathy (PML) Salmonella septicemia (recurrent) Toxoplasmosis of the brain Tuberculosis, disseminated (widespread or outside the lung) Wasting syndrome due to HIV

O. I. Screening • At Baseline: -TB Screening (PPD, CXR, Quantiferon) - Toxoplasmosis - Cryptococcus • Yearly: - TB Screening

O. I. Prophylaxis • CD 4 count <200: PCP Prophylaxis • CD 4 count <50: MAC Prophylaxis Toxo Prophylaxis CMV Screening

Immunizations • • • Tetanus/Diptheria ( Td, TDa. P) Pneumonia (Prevnar 13, Pneumovax) Influenza Hepatitis A Hepatitis B
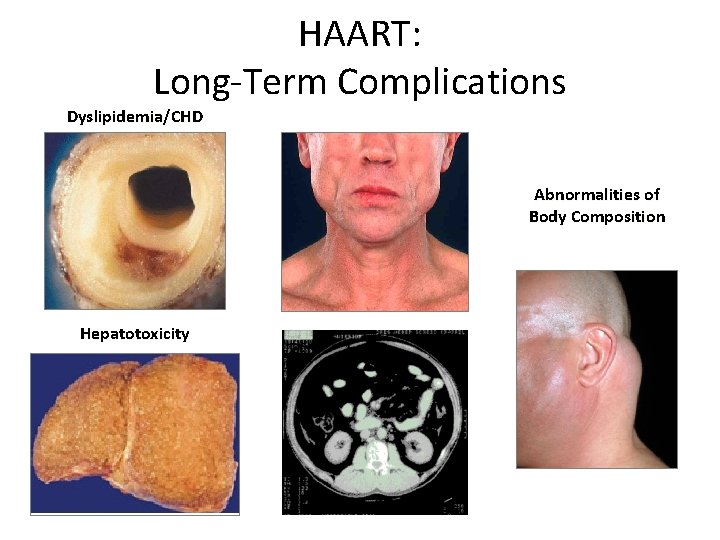
HAART: Long-Term Complications Dyslipidemia/CHD Abnormalities of Body Composition Hepatotoxicity

Screening for Coronary Heart Disease • Complete clinical history • Physical examination • Risk assessment with the Framingham Risk Equation (or similar) – Every patient without ischemic heart disease • Before HAART initiation • Annually thereafter • 12 -lead ECG – Men (>40 years) and women (>50 years of age) • Annually Hsue PY, et al. Circulation. 2008; 118: e 41 -e 47. Lundgren JD, et al. HIV Med. 2008; 9: 72 -81.
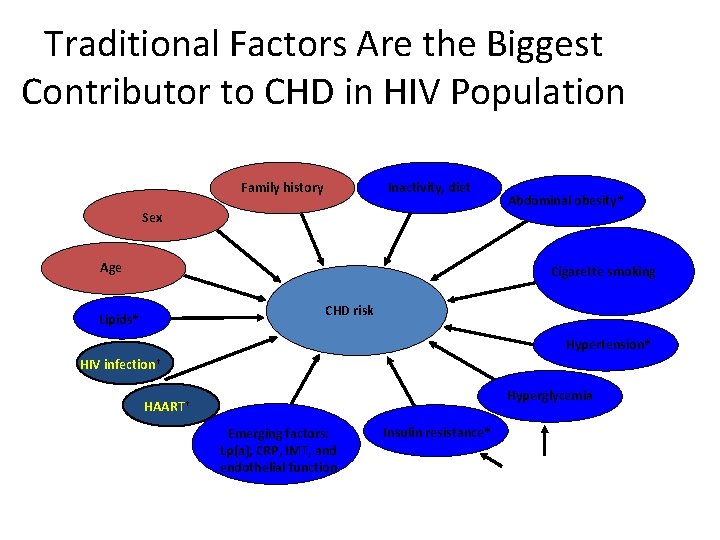
Traditional Factors Are the Biggest Contributor to CHD in HIV Population Family history Inactivity, diet Sex Age Abdominal obesity* Cigarette smoking CHD risk Lipids* Hypertension* HIV infection† Hyperglycemia HAART† Emerging factors: Lp(a), CRP, IMT, and endothelial function *Component of metabolic syndrome. †Precise contribution unclear. Insulin resistance* Diabetes
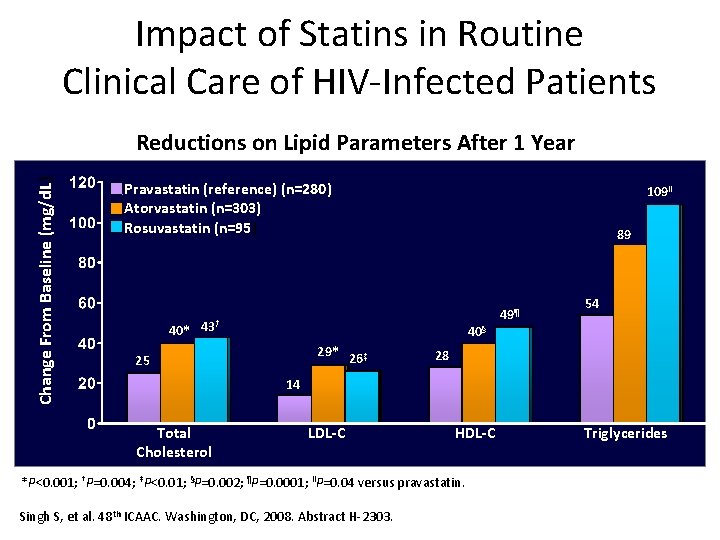
Impact of Statins in Routine Clinical Care of HIV-Infected Patients Change From Baseline (mg/d. L) Reductions on Lipid Parameters After 1 Year Pravastatin (reference) (n=280) Atorvastatin (n=303) Rosuvastatin (n=95) 109 ll 89 49¶ † 40* 43 54 40§ 29* 25 26‡ 28 14 Total Cholesterol LDL-C HDL-C *P<0. 001; †P=0. 004; ‡P<0. 01; §P=0. 002; ¶P=0. 0001; ll. P=0. 04 versus pravastatin. Singh S, et al. 48 th ICAAC. Washington, DC, 2008. Abstract H-2303. Triglycerides

Cardiovascular Risk

Diabetes: Recommended Laboratory Tests • Fasting serum glucose measurement – Before starting treatment • If normal, annually thereafter – 3 to 6 months after starting HAART if borderline or abnormal before starting treatment • If normal, repeat every 6 months • Oral glucose tolerance test – In patients with family history of diabetes, obesity or metabolic syndrome, on HAART • At the first visit • Repeat when there is a clinical suspicion of impaired glucose tolerance DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012. Florescu D, et al. Antiviral Ther. 2007; 12: 149 -162.

HIV: Pathogenic Mechanisms of Insulin Resistance • Similar in HIV and non-HIV patients – – – Genetic influences Elevated circulating free fatty acids Increased muscle and organ fat Hormones Comorbid diseases Chronic inflammatory changes (cytokines) • Specific for HIV-infected patients – Lipodystrophy – HAART components, particularly PIs and some NRTIs (didanosine, stavudine) Florescu D, et al. Antiviral Ther. 2007; 12: 149 -162.

Malignancies in HIV: Changes in Incidence Over the Past 10 Years • AIDS-related malignancies – Decreased • Kaposi sarcoma and CNS lymphoma – Increased • Non-Hodgkin lymphoma • Non-AIDS defining malignancies – Overall incidence increased by >3 -fold – Greatest increases seen in liver, larynx, anal, and lung cancers – No increase in prostate and breast cancers Mitsuyasu RT. Top HIV Med. 2008; 16: 117 -121. Engels EA, et al. Int J Cancer. 2008; 123: 187 -194. Patel P, et al. Ann Intern Med. 2008; 148: 728 -736.

CANCER: Early Detection and Prevention • Yearly intervals – – Cervical and anal Papanicolaou tests Gynecologic examinations and high-resolution anoscopy Breast examinations Prostate examinations (including prostate-specific antigen) • Periodically – Liver function tests and alpha-fetoprotein in HBV and/or HCV coinfection • Sunscreen and avoidance of overexposure to sunlight – Endothelial and epithelial cells in HIV-infected patient may be more susceptible to carcinogenesis Mitsuyasu RT. Top HIV Med. 2008; 16: 117 -121.

Cancer: Prevention • Smoking cessation • Use of hepatitis and HPV vaccines in seronegative individuals – Immunogenicity studies for HPV underway in HIVinfected persons • Maintain a high suspicion for cancer in HIVinfected persons Mitsuyasu RT. Top HIV Med. 2008; 16: 117 -121.

Osteoporosis • Higher rates of osteoporosis in HIV populations • Long term effect of ART (Tenofovir) • Higher rates of Vitamin D deficiency in HIV populations • Higher rates of smoking, alcohol. • Bone Mineral Density Screening with DEXA Scan (Men >50, Postmenopausal women, any h/o fracture) • RX- Calcium/Vit D, Biphosphpnates

Screening Strategies to Detect Asymptomatic STDs: First Visit • All patients – Serologic test for syphilis • RPR or VDRL, confirm positive test with FTA-Abs or TP-PA – Consider testing for gonorrhea, Chlamydia species, and herpes simplex based on patient sexual history – Serologic tests for hepatitis A, B, C at baseline • Female – Culture or DNA amplification test for gonorrhea – Urine sample examination for Trichomonas infection – Immunofluorescence or DNA amplification for chlamydia if: • Sexually active (<25 years of age) • At increased risk for particular situation (eg, commercial sex worker) Aberg JA, et al. Clin Infect Dis. 2004; 39: 609 -629. New York State Department of Health. Available at: http: //www. hivguidelines. org/Guide. Line. aspx? page. ID=257&guide. Line. ID=13.

Screening Strategies to Detect Asymptomatic STDs: Subsequent Visits • All sexually active patients – Screening tests for STDs should be repeated at least annually • More frequent periodic screening (at least 3 to 6 month intervals) for asymptomatic persons at higher risk – Multiple or anonymous sex partners – Past history of any STD – Behaviors associated with transmission of HIV or other STDs – Sexual or needle-sharing partners with any of the above risks – Changes in lifestyle/circumstances that are associated with increased risk behavior – High prevalence of STDs in the area or in the patient population Aberg JA, et al. Clin Infect Dis. 2004; 39: 609 -629. New York State Department of Health. Available at: http: //www. hivguidelines. org/Guide. Line. aspx? page. ID=257&guide. Line. ID=13.

HIV/HBV-Coinfection: Management Begins With Detection • All HIV-infected patients should be screened for HBV (anti-HBs and HBs. Ag) • Vaccination should be offered to anti-HBV-negative patients • Response to vaccine is influenced by CD 4 count and HIV RNA level – Initiate HAART first if CD 4 count is <200 cells/mm 3 and there is ongoing HIV RNA replication HBs. Ag Anti-HBs Meaning Action Negative Susceptible Vaccinate Negative Positive Immune, previous infection or vaccination None Positive Negative Chronic (or acute) hepatitis B infection HBe. Ag HBV DNA, Repeat in 6 months if suspect acute HBV USPHS Guideline. Available at: http: //www. cdc. gov/ncidod/diseases/hepatitis/b/Bserology. htm. Lok AS, et al. Hepatology. 2009; 50: 661 -662. Available at: http: //www. aasld. org/Pages/Default. aspx. EASL. J Hepatol. 2009: 50: 227 -242.

HIV/HBV-Coinfected Patients • Advise to abstain from alcohol • Should receive hepatitis A vaccine if found not to be immune at baseline – Absence of hepatitis A total or Ig. G antibody • Advise on methods to prevent HBV transmission (similar to those used to prevent HIV transmission) • Evaluate for the severity of HBV infection DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012.

HBV Treatment Options in HIV/HBV-Coinfected Patients Other Options Preferred Comments HIV and HBV need treatment HBV treatment-naïve Tenofovir DF/emtricitabine Tenofovir DF + lamivudine Lamivudine resistant Tenofovir DF/emtricitabine Tenofovir DF + entecavir HBV only needs treatment Peginterferon 2 a Adefovir Entecavir + HAART Avoid single-agent HBV therapy with 3 TC, FTC, or TDF due to the increased risk of HBV resistance Other options with limited data in HIV/HBV-coinfection: peginterferon alfa 2 a alone; adefovir + (3 TC, FTC, or telbivudine) + HAART Emtricitabine not active against lamivudine-resistant HBV Adefovir + telbivudine (monitor HBV DNA at week 24) Initiate HAART Monitor HBV DNA Monitor liver function Lok AS, et al. Hepatology. 2009; 50: 661 -662. Available at: http: //www. aasld. org/Pages/Default. aspx. DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012.

DHHS and AASLD Recommendations Prior to ART: HIV/HCV-Coinfected Patients • HCV antibody (EIA) – Performed in all HIV-infected persons • HCV RNA testing – Performed to confirm HCV infection in HIV-infected persons who are positive for anti-HCV – Performed in those who are negative and have evidence of unexplained liver disease • Patients with HIV/HCV coinfection – Advise to avoid alcohol – Use appropriate precautions to avoid transmission of both viruses – Receive HAV and HBV vaccines if susceptible DHHS. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Revision March 27, 2012. Ghany MG, et al. Hepatology. 2009; 49: 1335 -1374.

Recommended Pre-Treatment Assessments in HIV/HCV-Coinfected Patients • Liver disease status – HCV RNA, HCV genotype, AFP* and US for HCC, HBV status (HBs. Ag, anti. HBc), anti-HAV Ig. G, MELD calculation • HIV disease status – Presence or history of OIs, HIV-associated malignancy, CD 4 cell count, HIV RNA, details of HAART • Factors precluding HCV therapy or requiring control prior to initiating HCV therapy – TSH; screen for depression or other psychiatric disease; CBC; blood sugar; history of significant cardiac, renal, or pulmonary disease; fundus examination; beta HCG (women of childbearing potential); social support; treatment adherence *Most hepatologists recommend; not recommended by AASLD. Singal AK, et al. World J Gastroenterol. 2009; 15: 3713 -3724.

HIV/HCV Coinfection: Who to Treat? • Degree of Liver fibrosis or cirrhosis • Consider comorbid conditions that limit life expectancy or increase the risks associated with HCV therapy • HIV disease should be stable with or without HAART – CD 4 cell count <200 cells/mm 3 • Treat HIV and defer HCV • Interferon can exacerbate pre-existing mental illness – Evaluate patients with underlying psychiatric disease before initiating HCV treatment with interferon • Substance abuse – Active substance abuse is not a contraindication – Associated with high rates of treatment nonadherence and may compromise treatment outcomes Sulkowski MS, et al. J Viral Hepatitis. 2007; 14: 371 -386.
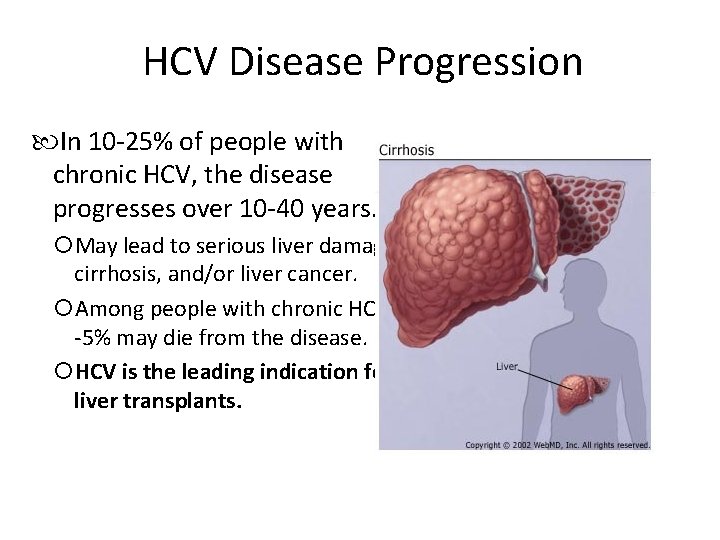
HCV Disease Progression In 10 -25% of people with chronic HCV, the disease progresses over 10 -40 years. May lead to serious liver damage, cirrhosis, and/or liver cancer. Among people with chronic HCV, 1 -5% may die from the disease. HCV is the leading indication for liver transplants.

Hepatitis C Treatment Just Got Much Better • HIGHER CURE RATES! • EASIER! Interferon free, fewer side effects • SHORTER! 3 -6 months instead of a year
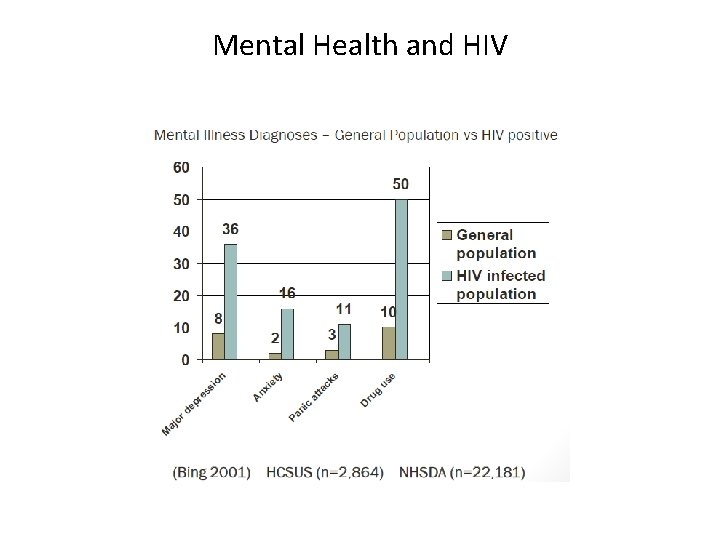
Mental Health and HIV
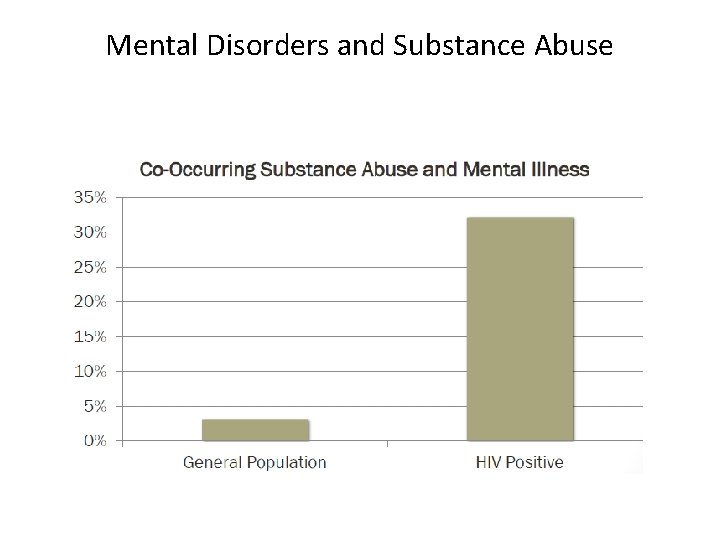
Mental Disorders and Substance Abuse

Depression and HIV

Depression and HIV

Management Issues in Older HIV-Infected Patients • Choice of HAART – Early initiation of HAART to avoid immune decline and help maintain immune function as patients age – Avoidance of metabolic and other toxicities a key issue • Need for regular screening and health maintenance – Fasting lipids and glucose, renal function, bone disease – Cancer screening as would be performed in general population • Awareness of drug-drug interactions • Management of dyslipidemia – Increased likelihood of need for lipid-lowering therapy – Recognition that HIV-infected patients may not respond as well to lipid-lowering therapy

Effect of Adherence on HIV • It is important for people to take their meds • Taking meds as prescribed helps to fight the virus – Viral load may go down – When viral load is low, T-cell count can go up Reference: Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in HIV-1 -Infected Adults and Adolescents. Department of Health and Human Services. January 29, 2008. Available at: http: //www. aidsinfo. nih. gov/Content. Files/Adultand. Adolescent. GL. pdf. Accessed June 20, 2008. 53

STIGMA Church sign in Birmingham, Alabama
- Slides: 54