History of the periodic table Learning Objectives B

History of the periodic table Learning Objectives: B 1. 3 Name the person who invented the modern periodic table. B 4. 2 Compare our version of the periodic table with Mendeleev’s. B 2. 4 Explain how Mendeleev developed the periodic table and why he left gaps in his version. B 3. 3 Describe and predict the chemical properties of elements Starter: Thinking ideas (TW) Why is it important to understand the structure of the atom? Why are atoms ‘electronically neutral’? Why is the periodic table useful?
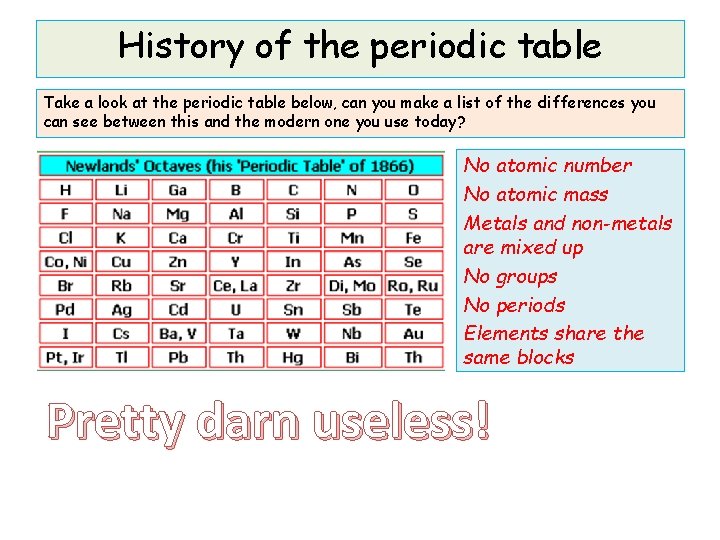
History of the periodic table Take a look at the periodic table below, can you make a list of the differences you can see between this and the modern one you use today? No atomic number No atomic mass Metals and non-metals are mixed up No groups No periods Elements share the same blocks Pretty darn useless!

History of the periodic table Take a look at the periodic table below, can you make a list of the differences you can see between this and the modern one you use today? The table you just saw was made by John Newlands Born 1837, died 1898. Won the Davy Medal in 1887. In the early 1800 s, scientists had no idea of atomic structure or of protons or electrons, so there was no such thing as atomic number to them. Back then, the only thing they could measure was relative atomic mass. Newlands attempted to arrange the elements into a table for everybody to use in 1864. He arranged them in order of relative atomic mass. He noticed a pattern in the properties of the elements; every eighth element had similar properties. These sets of eight elements were called Newlands’ Octaves. (Named after musical notes)

I am Dmitri Mendeleev! I made the PERIODIC TABLE ! Born: 8 February 1834, Died: 2 February 1907 (aged 72) Nationality: Russian Fields: Chemistry, physics and adjacent fields Notable awards: Davy Medal (1882)

History of the periodic table Watch the video: https: //www. youtube. com/watch? v=nsb. Xp 64 YPRQ 1. What did Mendeleev arrange the elements in order of? Atomic number (protons) 2. How did Newlands arrange the elements into a table? Atomic mass and in groups of 8 3. Why did Mendeleev leave gaps in his periodic table? To allow the pattern to fit, some had not been discovered 4. How are elements arranged in the modern periodic table? Increasing atomic number 5. Give two reasons why Newlands’ version of the periodic table was heavily criticised. Metals and non-metals mixed Easily proven wrong 6. Give one piece of evidence that strongly suggested Mendeleev’s system of classifying elements was correct. When electrons were discovered it matched the table 7. Suggest why many scientists didn’t initially take the periodic table seriously. Didn’t understand it or see any use for it 8. What happened to make scientists believe that the periodic table is a useful summary of atomic structure? Electrons were discovered

Using the periodic table So now we know where it came from, how can we use it? What is the reactivity trend of group 1 with water? What is the trend of group 7? (halogens) What are noble gases? Explain their reactivity.

Now you are almost as smart as I am! But not as handsome! Man, I look GOOD! I am working this beard!
- Slides: 7