History of the Periodic Table 1808 Daltons Table
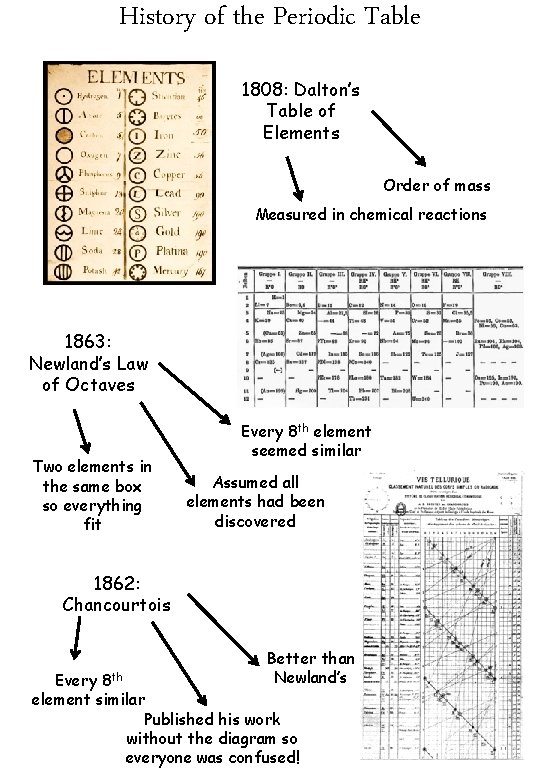
History of the Periodic Table 1808: Dalton’s Table of Elements Order of mass Measured in chemical reactions 1863: Newland’s Law of Octaves Two elements in the same box so everything fit Every 8 th element seemed similar Assumed all elements had been discovered 1862: Chancourtois Better than Newland’s Every 8 th element similar Published his work without the diagram so everyone was confused!
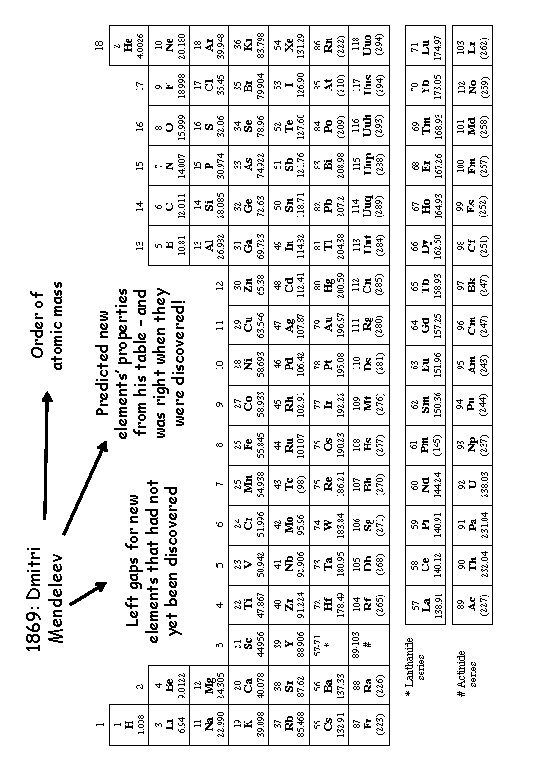
Left gaps for new elements that had not yet been discovered 1869: Dmitri Mendeleev Predicted new elements’ properties from his table – and was right when they were discovered! Order of atomic mass
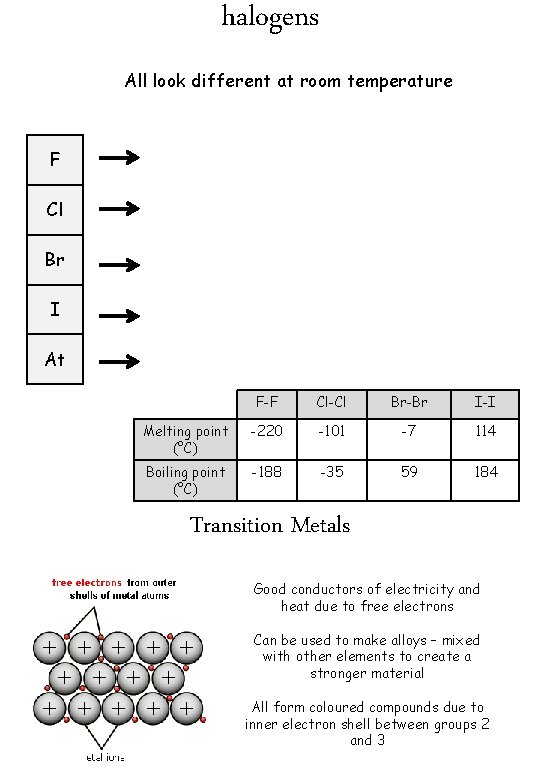
halogens All look different at room temperature F Cl Br I At F-F Cl-Cl Br-Br I-I Melting point (°C) -220 -101 -7 114 Boiling point (°C) -188 -35 59 184 Transition Metals Good conductors of electricity and heat due to free electrons Can be used to make alloys – mixed with other elements to create a stronger material All form coloured compounds due to inner electron shell between groups 2 and 3

Transition metals cont. List 5 properties of metals below: 1. 2. 3. 4. 5. Complete the following sentences using the words below: The transition metals are a block of elements that lie between Groups …… and …… in the periodic table. These elements nearly all have high …………, high …………… points and are good ……………. They are …………… reactive than the alkali metals, and often form ………… compounds. Match the metal with the correct coloured compound Iron III Copper Potassium Chromium Green Blue Reddish-brown Purple Orange We use these coloured compounds as pottery glazes, or just to give colour to buildings (e. g. copper roofs or statues!)
- Slides: 4