History of the Master Chemical Mechanism MCM and


















- Slides: 25

History of the Master Chemical Mechanism (MCM) and its development protocols Mike Jenkin EPSR Group Department of Environmental Science and Technology m. jenkin@imperial. ac. uk

1993 – the birth of the MCM l University of Leeds Sam Saunders, Mike Pilling l AEA Technology Mike Jenkin, Colin Johnson l UK Meteorological Office Dick Derwent l Work commissioned by the Department of the Environment, Do. E (Air Quality Division), to improve the treatment of organic chemistry in ozone policy models

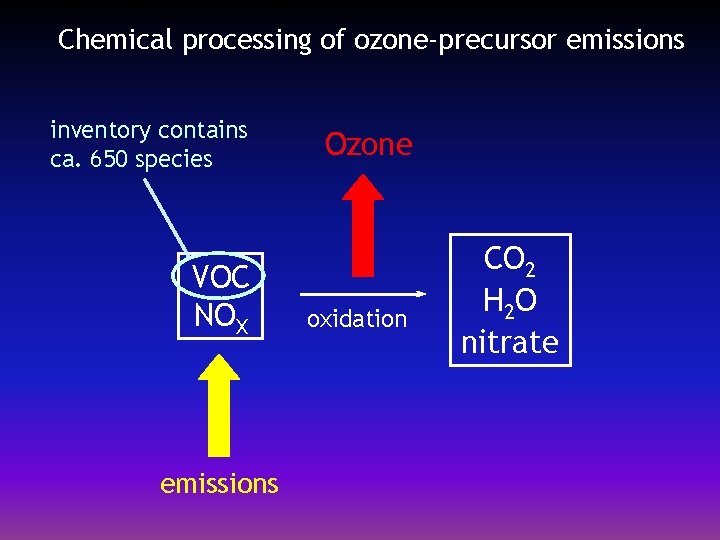
Chemical processing of ozone-precursor emissions inventory contains ca. 650 species VOC NOX emissions Ozone oxidation CO 2 H 2 O nitrate

Chemistry in Do. E ozone models in 1993 Photochemical Trajectory Model l l chemistry of 95 VOC represented although reasonably detailed, the chemistry did not reflect the current status of kinetic and mechanistic data, e. g. - no formation of organic nitrates from RO 2 + NO - RO 2 + HO 2 reactions not included (except for CH 3 O 2) - incomplete degradation of some VOC - - many VOC degraded via products known to be wrong (i. e. incorrect RO reactions applied) very limited representation of photolysis of organics

1993 -1996: Master Chemical Mechanism (MCM v 1) Philosophy l l l to use information on the kinetics and products of elementary reactions relevant to VOC oxidation to build up an explicit representation of the degradation mechanisms. - the resultant formation of ozone and other gas-phase secondary pollutants apply measured and evaluated parameters (e. g. rate coefficients; branching ratios) from the literature where possible. use analogy and ‘structure-reactivity correlations’ to define the other reactions and parameters. ‘Mechanism Development Protocol’ - Atmospheric Environment, 31, 81 -104, 1997

1996: Master Chemical Mechanism (MCM v 1) l l l Degradation of CH 4 and 119 non-methane VOC ca. 2, 500 chemical species ca. 7, 000 chemical reactions l l l 22 alkanes (C 1 -C 12) 16 alkenes (C 2 -C 6) 2 dienes (C 4 -C 5) 1 alkyne (C 2) 18 aromatics (C 6 -C 11) 6 aldehydes (C 1 -C 5) l l l 10 ketones (C 3 -C 6) 17 alcohols (C 1 -C 6) 10 ethers (C 2 -C 7) 8 esters (C 2 -C 6) 3 carboxylic acids (C 1 -C 3) 8 halocarbons (C 1 -C 3) MCM website launched in March 1997

MCM timeline 1996 MCM v 1 - 120 VOC; 7000 reactions; 2500 species 1999 MCM v 2 - 123 VOC; 10500 reactions; 3500 species 101 non-aromatic anthropogenic species 18 aromatics (provisional chemistry) 1 biogenic species (isoprene) 103 non-aromatic anthropogenic species 18 aromatics (extended provisional chemistry) 2 biogenic species (isoprene: a-pinene) 2002 MCM v 3 - 125 VOC; 12700 reactions; 4400 species 104 non-aromatic anthropogenic species 18 aromatics (first rigorous representation) 3 biogenic species (isoprene: a-pinene: b-pinene) Contributions to MCM v 2 and v 3 activities at Leeds: Nic Carslaw, Stephen Pascoe, Volker Wagner

2002: Master Chemical Mechanism (MCM v 3) l l l l 22 alkanes (C 1 -C 12) 16 alkenes (C 2 -C 6) 2 dienes (C 4 -C 5) 2 monoterpenes (C 10) 1 alkyne (C 2) 18 aromatics (C 6 -C 11) 6 aldehydes (C 1 -C 5) l l l l 10 ketones (C 3 -C 6) 17 alcohols (C 1 -C 6) 10 ethers (C 2 -C 7) 8 esters (C 2 -C 6) 3 carboxylic acids (C 1 -C 3) 2 other oxygenates (C 3) 8 halocarbons (C 1 -C 3) Supplementary protocols: • Atmospheric Chemistry and Physics, 3, 161 -180, 2003 (non-aromatic VOC) • Atmospheric Chemistry and Physics, 3, 181 -193, 2003 (aromatic VOC)

MCM construction methodology

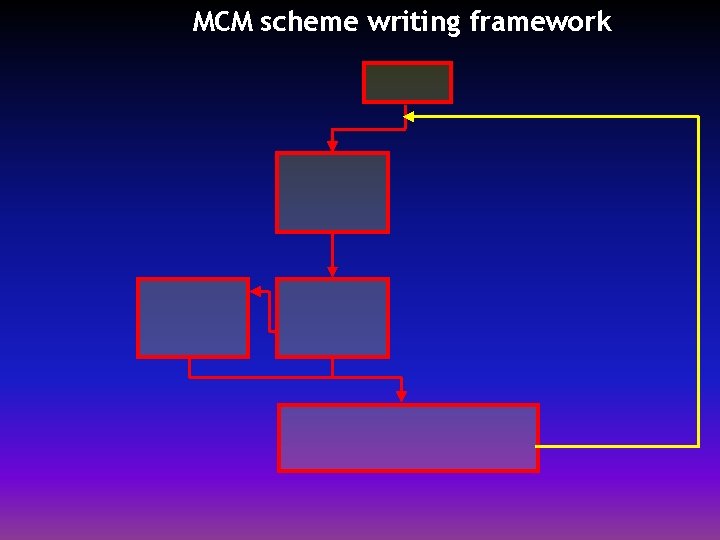
MCM scheme writing framework

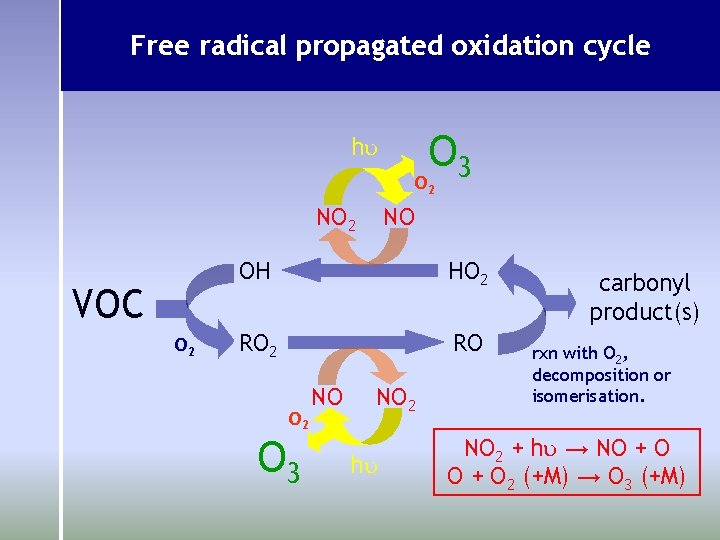
Free radical propagated oxidation cycle O 3 hu O 2 NO 2 VOC O 2 NO OH HO 2 RO O 2 O 3 NO NO 2 hu carbonyl product(s) rxn with O 2, decomposition or isomerisation. NO 2 + hu → NO + O 2 (+M) → O 3 (+M)

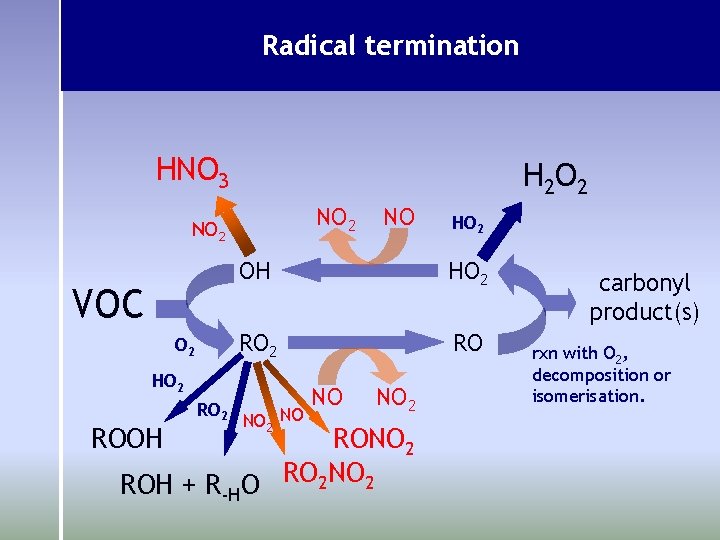
Radical termination HNO 3 H 2 O 2 NO 2 VOC O 2 ROOH HO 2 RO HO 2 RO 2 NO NO NO 2 ROH + R-HO RO 2 NO 2 carbonyl product(s) rxn with O 2, decomposition or isomerisation.

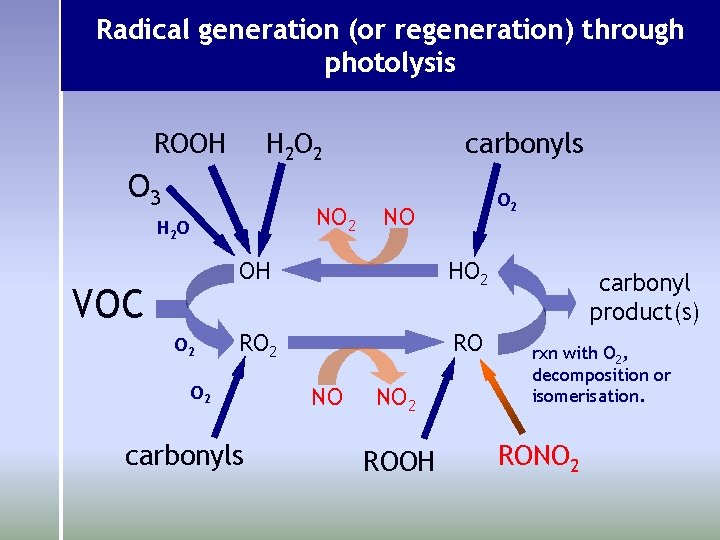
Radical generation (or regeneration) through photolysis ROOH H 2 O 2 O 3 NO 2 H 2 O VOC O 2 carbonyls O 2 NO OH HO 2 RO O 2 carbonyls NO NO 2 ROOH carbonyl product(s) rxn with O 2, decomposition or isomerisation. RONO 2


OH-initiated degradation of methane (CH 4)

OH-initiated degradation of ethane (C 2 H 6)

OH-initiated degradation of 1, 3 -butadiene

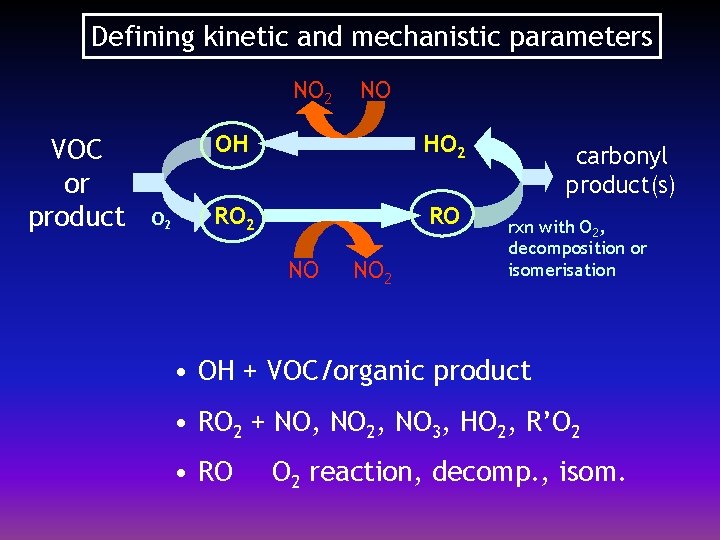
Defining kinetic and mechanistic parameters NO 2 VOC or product O 2 NO OH HO 2 RO NO NO 2 carbonyl product(s) rxn with O 2, decomposition or isomerisation • OH + VOC/organic product • RO 2 + NO, NO 2, NO 3, HO 2, R’O 2 • RO O 2 reaction, decomp. , isom.

OH radical reactions Kinetics of OH + VOC/organic products l l Rate coefficients have been measured for several hundred organics Rate coefficients for ca. 2, 000 species need to be estimated (e. g. SAR method of Atkinson, 1994; Kwok and Atkinson, 1995) Product radical distribution of OH + VOC/organic product l Mainly inferred from SAR partial rate coefficients l Scheme simplification measures applied in some cases - minor channels (<5%) ignored - single representative channel for ≥ C 7 alkanes - so called ‘minor’ products (e. g. RONO 2; ROOH) degraded to regenerate existing species

RO 2 radical reactions Kinetics of RO 2 reactions l l l Reactions with NO, NO 2, NO 3, HO 2 and other peroxy radicals (R’O 2) are included in MCM There about 1200 RO 2 radicals in MCM v 3 Kinetic data are available for only ca. 20 RO 2 – parameters assigned to majority of reactions by analogy and structure reactivity correlations Product branching ratios l Multiple channels for reactions with NO, HO 2 and R’O 2 l Scheme simplification measures applied in some cases - RO 2 from ‘minor’ products react via single channel - RO 2 + R’O 2 reaction are necessarily parameterised (explicit chemistry for 1200 radicals would require 0. 7 million reactions!)

RO radical reactions reaction with O 2 decomposition isomerisation l l There about 1200 RO radicals in MCM v 3 Relative importance of these modes of reaction largely defined by SAR methods of Carter and Atkinson (1989) and Atkinson (1997)

Simplification measure oxygenated RO radicals – exclusive decomposition assumed

VOC/product initiation reactions l Reaction with OH – all VOC and oxygenated products l Reaction with O 3 – alkenes/dienes and unsaturated products l Reaction with NO 3 – alkenes/dienes, aldehydes and cresols l Photolysis – carbonyls, RONO 2, ROOH

Organic photolysis processes • 26 photolysis processes defined • 14 parameters also used to define photolysis rates for several thousand other species

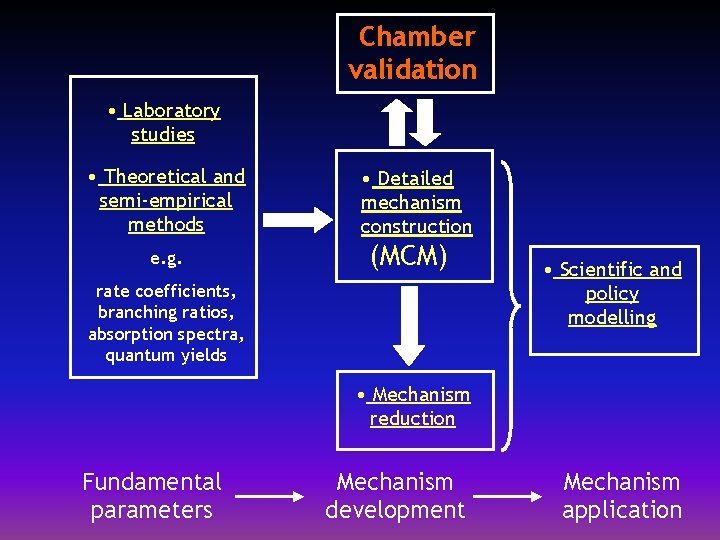
Chamber validation • Laboratory studies • Theoretical and semi-empirical methods e. g. • Detailed mechanism construction (MCM) rate coefficients, branching ratios, absorption spectra, quantum yields • Scientific and policy modelling • Mechanism reduction Fundamental parameters Mechanism development Mechanism application