History of the Development of the Modern Model
























- Slides: 29

History of the Development of the Modern Model of the Atom

Dalton’s Atomic Theory (early 1800’s) 1. All elements are composed of indestructible particles called atoms 2. All atoms of an element are exactly alike 3. Atoms of one element (ex. C) are different from atoms of other elements (ex. U) 4. Compounds are formed by joining two or more different atoms in constant whole number ratios.

What did Dalton think the atom looked like?

Dalton was not quite correct. 1. atoms are NOT indestructible particles 2. All atoms of an element are NOT exactly alike

Thomson and the Cathode Ray Tube (late 1800’s) • Thomson concluded that the atom was made of smaller particles. • Discovered particles with negative charge v. Electron • No matter what metals were used they all produced a negative beam of light v. Therefore atoms of all elements contain electrons

Video • Cathode Ray Tube –Animation (53 sec) • Cathode Ray Tube –Demo (2: 48)

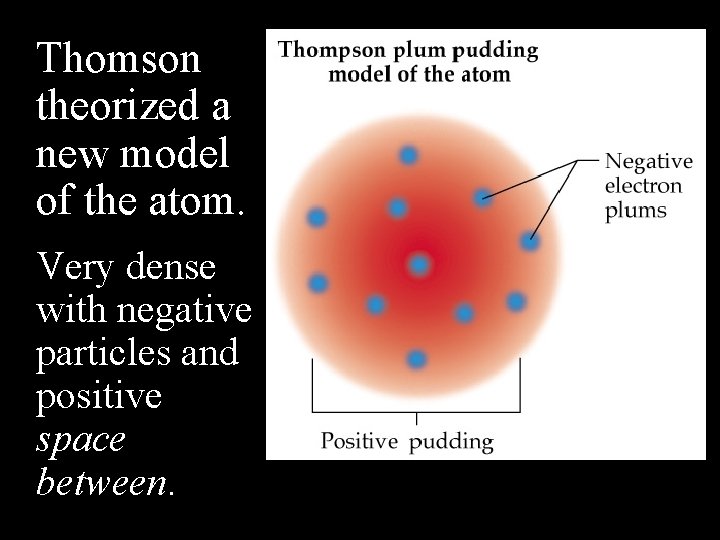
Thomson theorized a new model of the atom. Very dense with negative particles and positive space between.

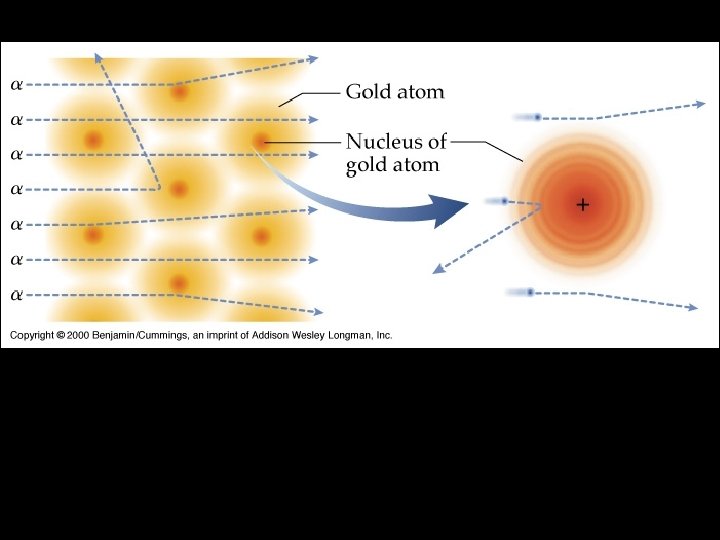
Rutherford’s Gold Foil Experiment


OBSERVATIONS • most particles went straight through CONCLUSIONS • atoms are mostly empty space • few particles were deflected or bounced back • few particles came close to or hit a positive nucleus in the atom

Rutherford’s Model • Electrons randomly orbit a large, positive nucleus

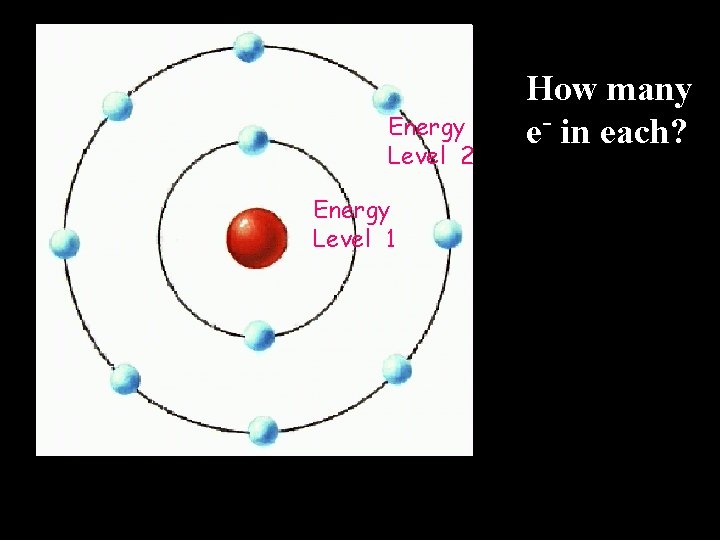
Bohr Model of the Atom (early 1900’s) • Electrons have definite, fixed orbits • Electrons have a definite energy – orbits farther from the nucleus have greater energy

Energy Level 2 Energy Level 1 How many e- in each?

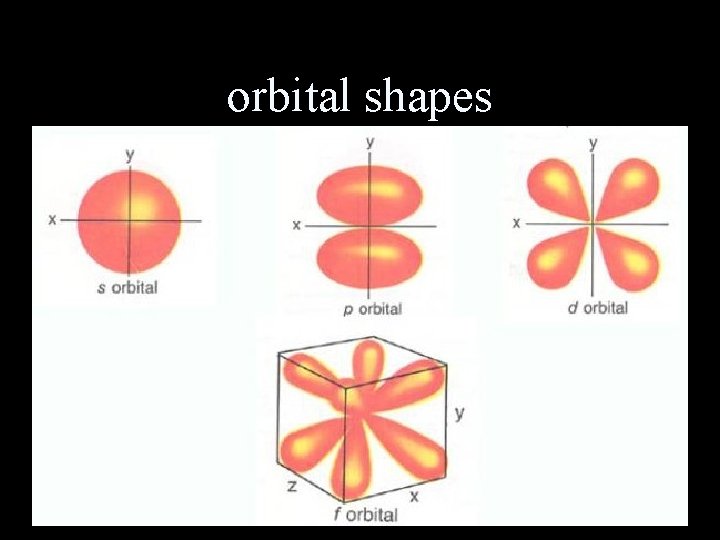
Electron-cloud model Modern Model electrons are most likely found in regions outside the nucleus called orbitals and do NOT travel in fixed paths

orbital shapes


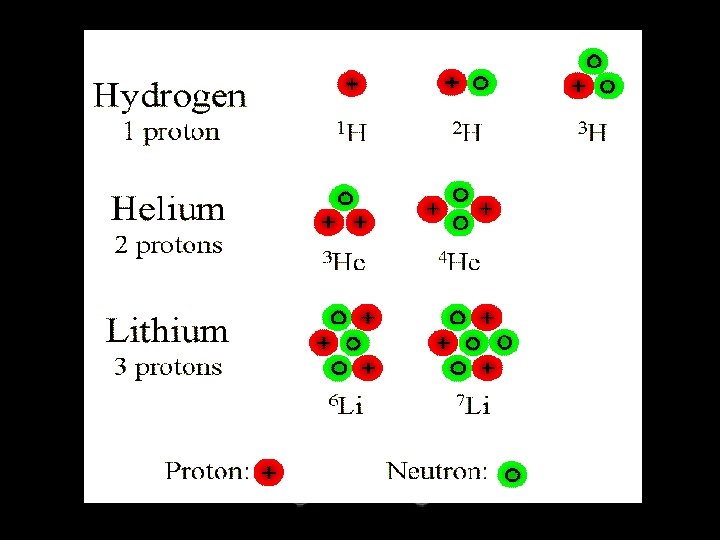
Atoms & Isotopes

Atoms: Review

Mystery Atoms

• You will need your Periodic Table • The bags represent Atomic Nuclei Protons Neutrons • Fill in data table to determine the Mystery Atom’s identity. • We’ll do A together

A

We’ll talk more about this column after Protons blue Neutrons green Mystery Atom atomic # # neutrons # electrons EXAMPLE A 7 8 7 Atomic Mass Isotopic Symbol 15 N B C

• contain the same number of protons but different numbers of neutrons. • There may be several isotopes of one element.


Isotopic Notation Mass number 14 6 Atomic number a. k. a. C C-14

• The most abundant isotope of an element is the whole number mass on the PT. • A weighted average of the element (all isotopes) is on the PT.

Weighted Averages & isotopes of Pe

Steps to Calculate a Weighted Average • You’ll calculate % (or be given) • Multiply the % by the mass (for as many isotopes as you have) • Add them together.

The End