History Dmitri Mendeleev 1896 1951 Seaborg Developed the
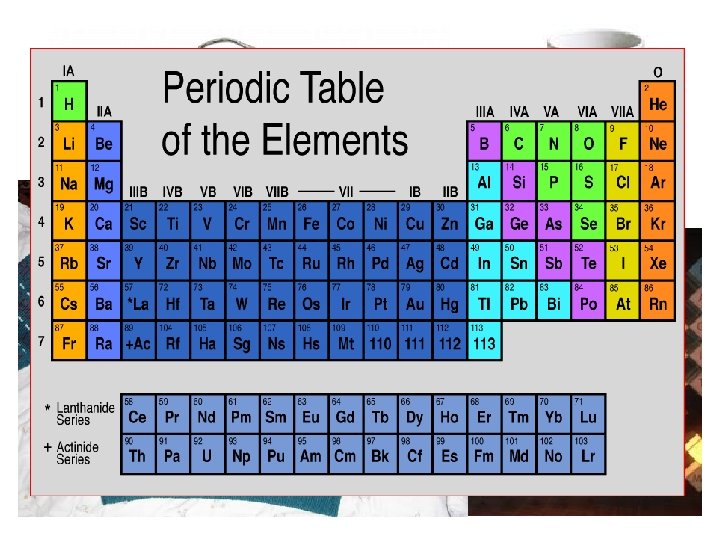

History Dmitri Mendeleev 1896
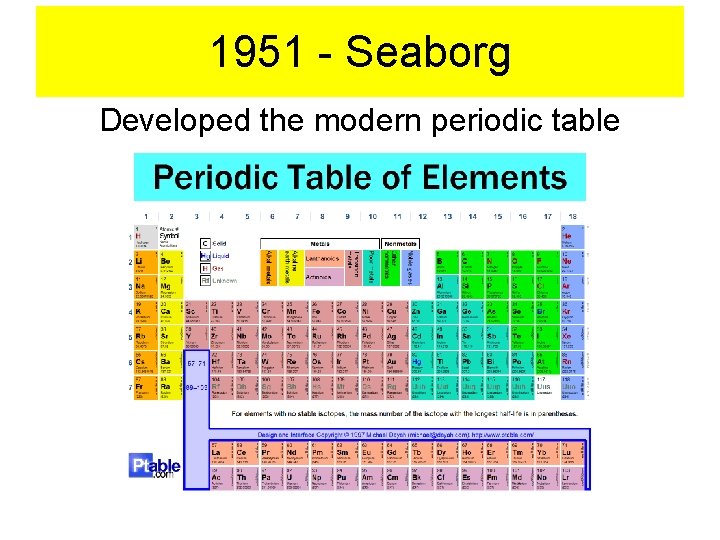
1951 - Seaborg Developed the modern periodic table
The Whole Periodic table

Groups vs. Periods Need to Know: Halogens Noble Gas Alkali Metals Alkaline Earth Transition Metals Lanthanides Actanides Groups: Up and Down Periods: Left to Right
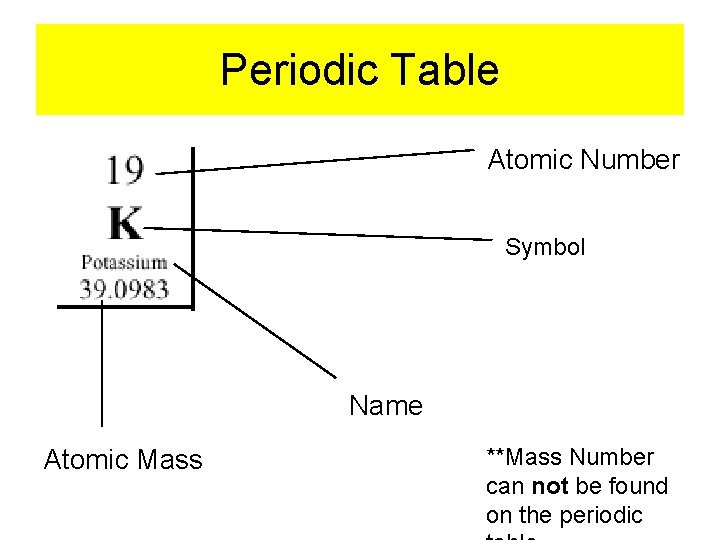
Periodic Table Atomic Number Symbol Name Atomic Mass **Mass Number can not be found on the periodic
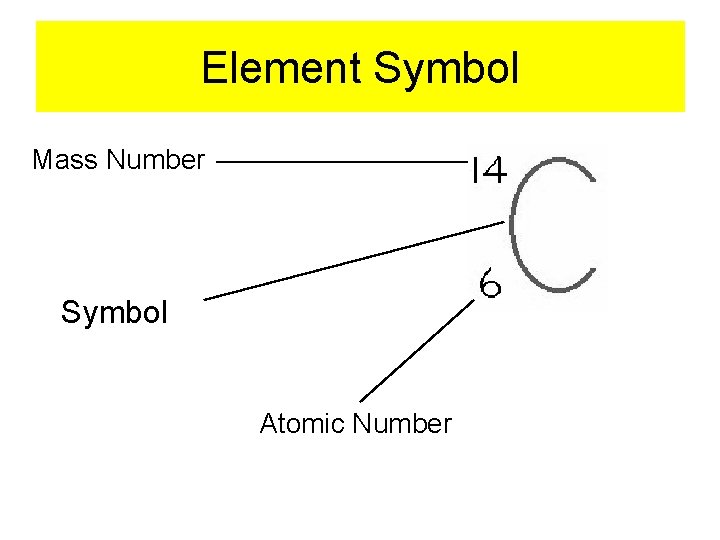
Element Symbol Mass Number Symbol Atomic Number
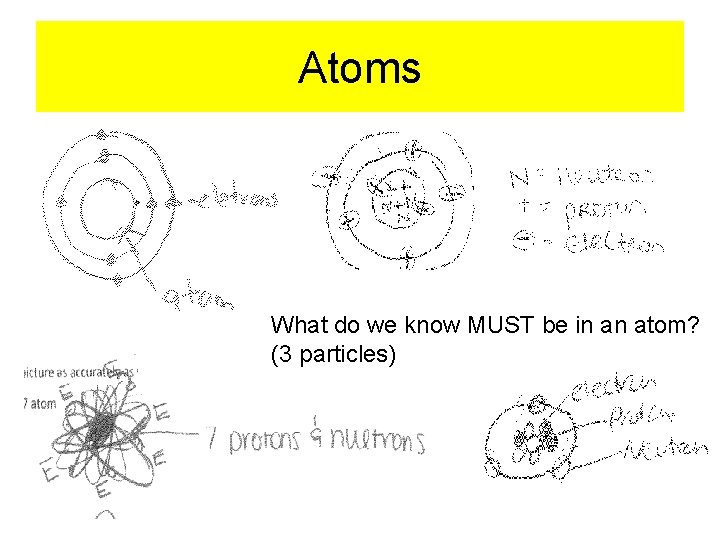
Atoms What do we know MUST be in an atom? (3 particles)

Ions How is an ion formed?

Ions If an ion has a charge of -2, what has happened to the atom? The atom has gained two electrons If an ion has a charge of +2, what has happened to the atom? The atom has lost two electrons
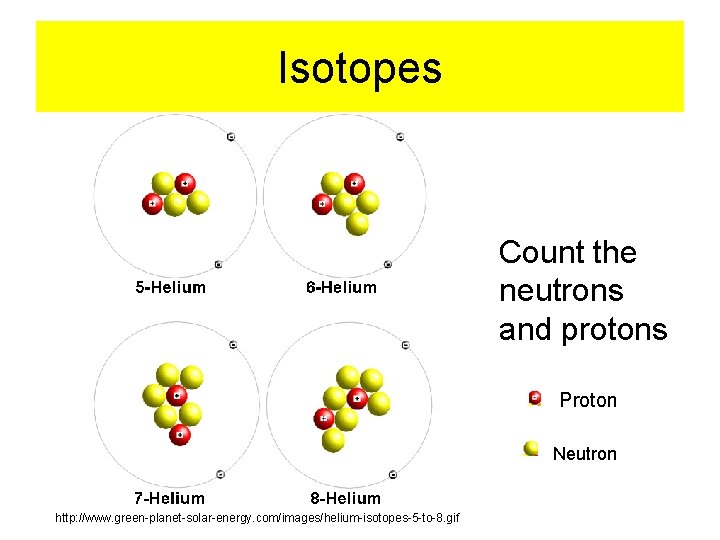
Isotopes Count the neutrons and protons Proton Neutron http: //www. green-planet-solar-energy. com/images/helium-isotopes-5 -to-8. gif
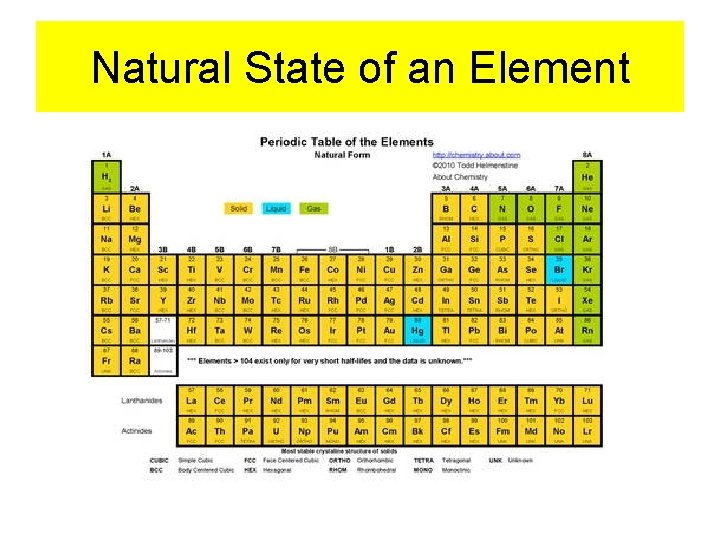
Natural State of an Element

Questions?
Quiz Time Acceptable Answers Correct Answer

Good Luck You have 7 minutes to complete your quiz. When you are done, hold on to it and wait patiently.

Average. Relative Atomic Size Mass Calcs 1 atomic mass unit (amu) = 1. 66054 x 10 -27 kg, Mass of an electron = 9. 10939 x 10 -31 kg Mass of a proton = 1. 67262 x 10 -27 kg Mass of a neutron = 1. 67493 x 10 -27 kg **You will NOT need to memorize these numbers. Always given to you.
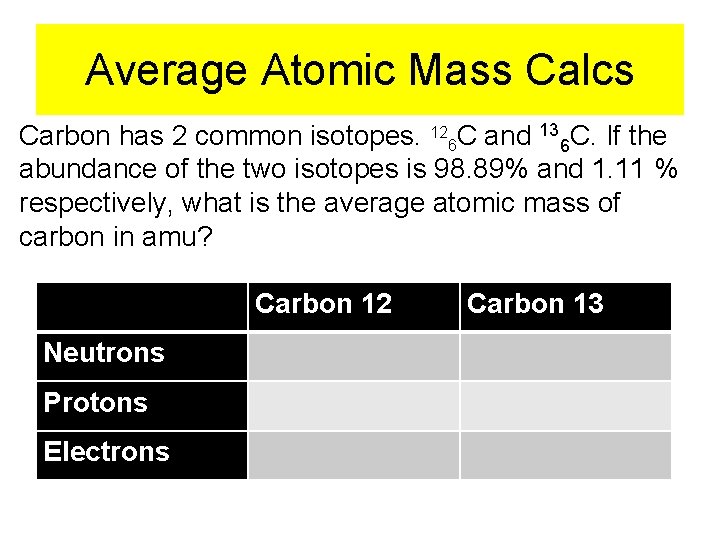
Average Atomic Mass Calcs Carbon has 2 common isotopes. 126 C and 136 C. If the abundance of the two isotopes is 98. 89% and 1. 11 % respectively, what is the average atomic mass of carbon in amu? Carbon 12 Neutrons Protons Electrons Carbon 13

12 6 C 6 protons 1. 67262 x 10 -27 kg 1 proton 6 electrons 9. 10939 x 10 -31 kg 1 electron 6 neutrons 1. 67493 x 10 -27 kg 1 neutron 1 amu = 6. 04364 amu 1. 66054 x 10 -27 kg 1 amu = 0. 0032915 amu 1. 66054 x 10 -27 kg 1 amu = 6. 05200 amu 1. 66054 x 10 -27 kg 6. 04364 amu + 6. 05200 amu + 0. 0032915 amu =12. 09893 amu

13 6 C 6 protons 1. 67262 x 10 -27 kg 1 proton 7 neutrons 1. 67493 x 10 -27 kg 1 neutron 6 electrons 9. 10939 x 10 -31 kg 1 electron 1 amu = 6. 04364 amu 1. 66054 x 10 -27 kg 1 amu = 7. 06066 amu 1. 66054 x 10 -27 kg 1 amu = 0. 0032915 amu 1. 66054 x 10 -27 kg 6. 04364 amu + 7. 06066 amu + 0. 0032915 amu = 13. 10755 amu

Putting It All Together What we know: Carbon 12: 12. 09893 amu (98. 89% abundance) Carbon 13: 13. 10755 amu (1. 11% abundance) Therefore: (12. 09893 amu x 0. 9889) + (13. 10755 amu x 0. 0111) 12. 01101 amu

Homework *Attempt problem #1 on Average Atomic Mass Calculations WS Read and notes 3. 5, 3. 7, 3. 8
- Slides: 21