Historical Atomic Theory Developments in atomic theory from















- Slides: 25

Historical Atomic Theory Developments in atomic theory from Democritus to Bohr

• How do we know what is inside of something? Open it up and look… a baseball? a golf ball? an atom? ? ? ?

• Democritus Greek philosopher – ca. 400 BC Atoms are particles in space. X Atoms are indivisible. Atoms are in motion. Things differ because of different arrangements of atoms. X X Events are predictable. Everything is made up of atoms and “the void”.

• Alchemists attempted to turn base metals into gold • Performed experiments • Recorded observations

At the age of 12!!! • John Dalton - 1808 English chemist / physicist / meteorologist / teacher Tested/experimented DALTON’S MODEL • • • All matter is made up of solid, indivisible spheres. Atoms of the same element are identical. Compounds are made up of different combinations of atoms. Atoms can not be subdivided, created, or destroyed. Chemical change is due to rearrangement of atoms. Purely Theoretical DEMOCRITUS’ IDEAS • • • Atoms are particles in space. Atoms are indivisible. Atoms are in motion. Things differ because of different arrangements of atoms. Events are predictable. Everything is made up of atoms and the void.

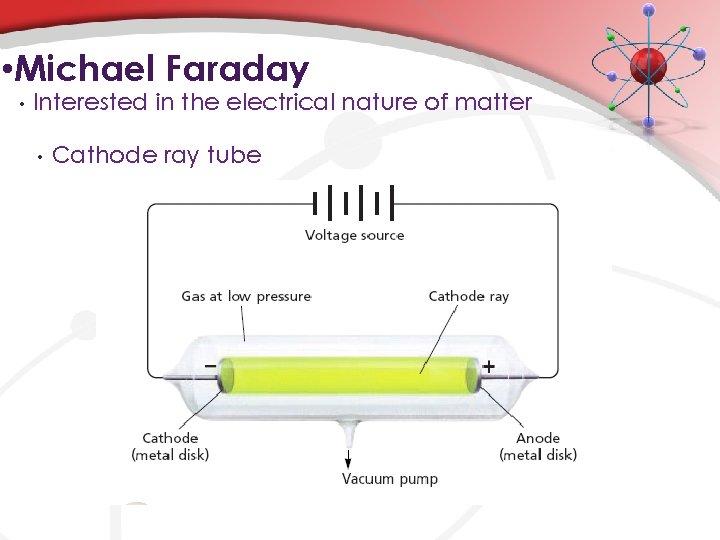
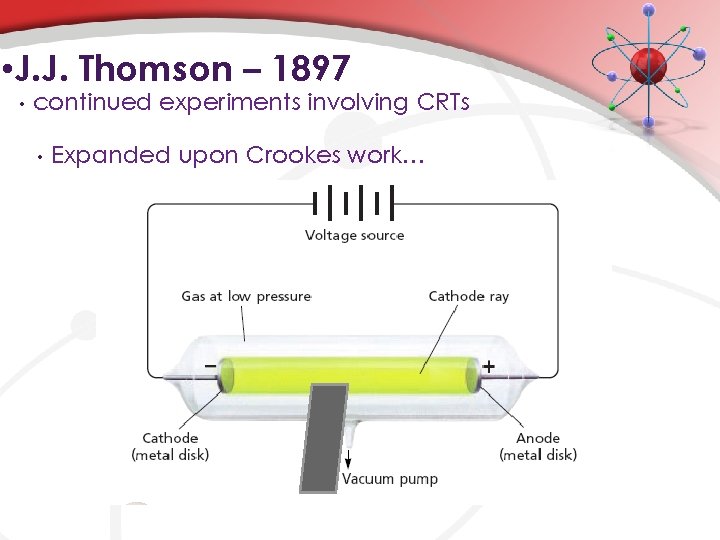
• Michael Faraday – 1820 s-1840 s+ • Interested in the electrical nature of matter • • • Used electrolysis to decompose water Thought that electricity was the “glue” that held atoms together. Popularized terms: • • electrode • cathode – negative electrode • anode – positive electrode Discovered cathode rays. • beam of light that seemed to emanate from the cathode (traveled from the cathode the anode)

• Michael Faraday • Interested in the electrical nature of matter • Cathode ray tube

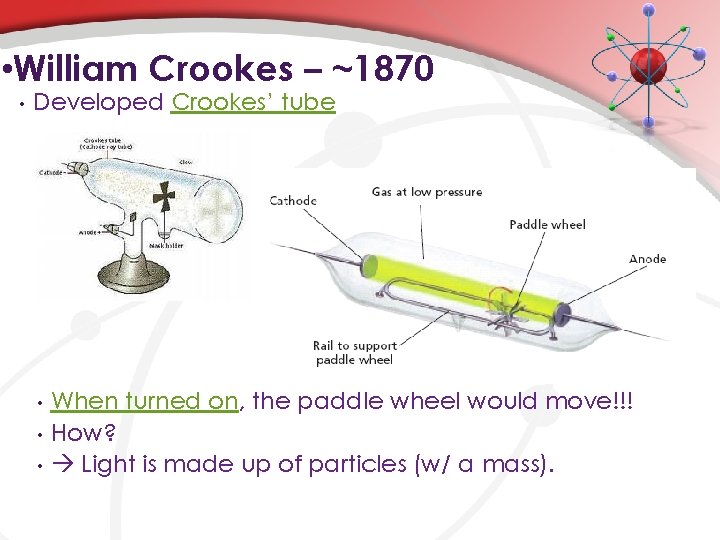
• William Crookes – ~1870 • Developed Crookes’ tube • • • When turned on, the paddle wheel would move!!! How? Light is made up of particles (w/ a mass).

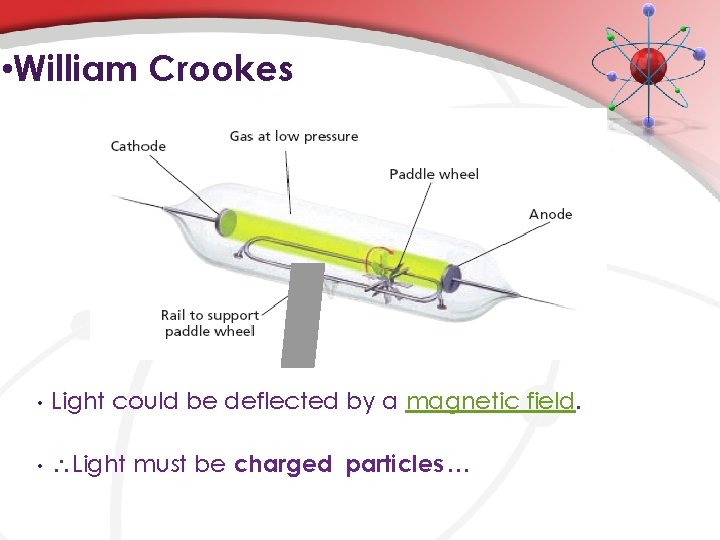
• William Crookes • Light could be deflected by a magnetic field. • Light must be charged particles …

• J. J. Thomson – 1897 • continued experiments involving CRTs • Expanded upon Crookes work…

• J. J. Thomson • • • Light could be repelled by negative charge. Light could be attracted by positive charge. Light must be negatively charged particles … called electrons back to Thomson in a few minutes…

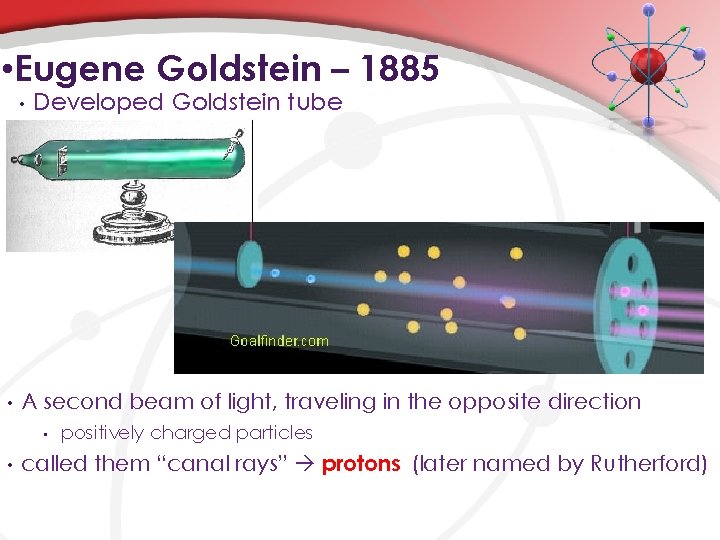
• Eugene Goldstein – 1885 • • Developed Goldstein tube A second beam of light, traveling in the opposite direction • • positively charged particles called them “canal rays” protons (later named by Rutherford)

• Wilhelm Röntgen – 1895 • • also working with CRTs / modified Crookes tubes conducting experiments with faint luminescence paper covered with barium platinocyanide • • some “unknown” rays causing the paper to glow called these unknown rays – x-rays

• Henri Becquerel – 1896 • • following up on the work of Röntgen with investigations of radiation had inherited a supply of uranium salts (compounds) that phosphoresce on exposure to light he set up an experiment where he wrapped some of these salts with photographic plates found that the plates were already ‘fogged’… radiation must be coming from the uranium atom FIRST CLEAR INDICATION that the atom is not featureless, providing strong evidence to the existence of electrons. radioactivity – named by Marie Curie (1898) Becquerel, Marie & Pierre Curie shared a Nobel Prize (1903)

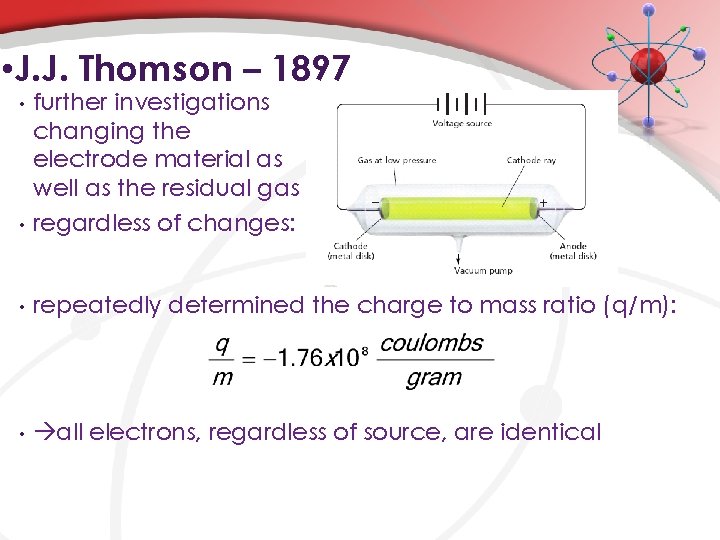
• J. J. Thomson – 1897 • further investigations changing the electrode material as well as the residual gas regardless of changes: • repeatedly determined the charge to mass ratio (q/m): • all electrons, regardless of source, are identical •

• J. J. Thomson – 1897 • • • Dalton’s model must be incorrect atom is composed of positive material with negative particles (electrons) embedded in it AKA Plum Pudding Model (1904)

• Robert Millikan – 1909 • • • American physicist Millikan oil drop experiment was able to determine the charge on the droplet with q/m ratio (Thomson) and the charge… calculated the mass of an electron = 9. 11 x 10 -28 grams

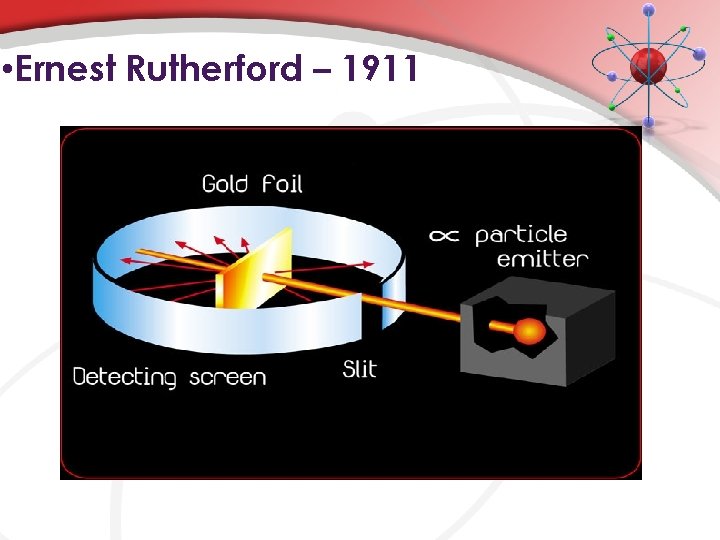
• Ernest Rutherford – 1911 • • (1908) designed an experiment to test Thomson’s model of the atom Gold foil experiment • used alpha (a) particles [product of radioactive decay] • small • dense • fast-moving • positive charge (Actually 2 protons & 2 neutrons. Same as helium nucleus – but Rutherford did not know this…)

• Ernest Rutherford – 1911

• Ernest Rutherford – 1911 • • (1908) designed an experiment to test Thomson’s model of the atom Gold foil experiment (click…) A closer look (click…) More explanation? ? ? Atom contains a small, positively charged center (nucleus), surrounded by empty space with electrons

• Light • At the time of Rutherford: • electrons: particles • light: wave • But in the early 1900 s, experiments began to produce evidence of: • electrons having wave-like properties • light having particle-like properties • Photoelectric effect • explained • by Einstein Hot body radiation • explained by Planck

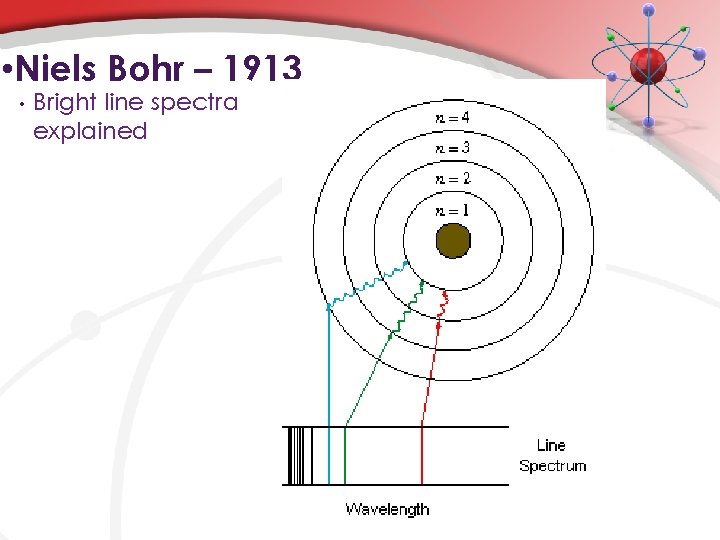
• Niels Bohr – 1913 • • • Electrons can only exist in a certain path (orbit) of a certain energy. An electron does not emit (give off) energy when in a ground state orbit. Energy is emitted when an e- goes from an excited state (higher energy level) to a lower energy level. Atoms emit (or absorb) energy only in specific amounts (quantas). The energy emitted (or absorbed) is equal to the difference in the energy between energy levels. (Angular momentum of electron is quantized)

• Niels Bohr – 1913 • Bright line spectra explained

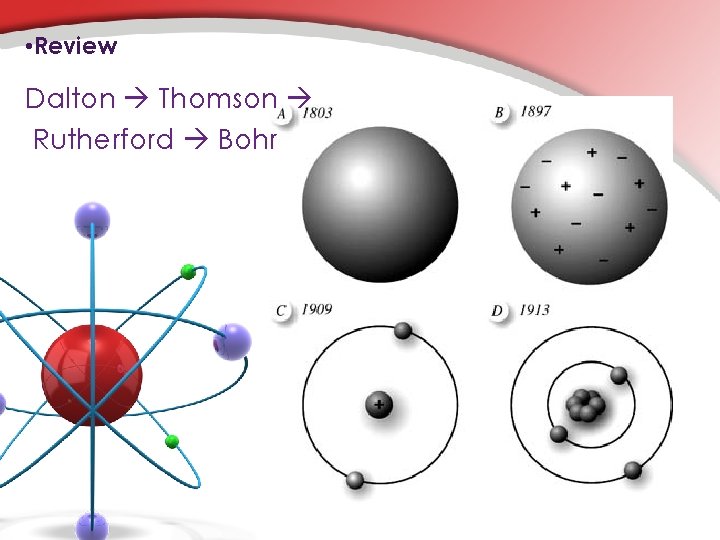
• Review Dalton Thomson Rutherford Bohr
