Hirsutism dr Nazli Navali Associate professor of Tabriz

Hirsutism dr. Nazli Navali Associate professor of Tabriz Uivrsity of Medical Sciences Fellowship of infertility 1
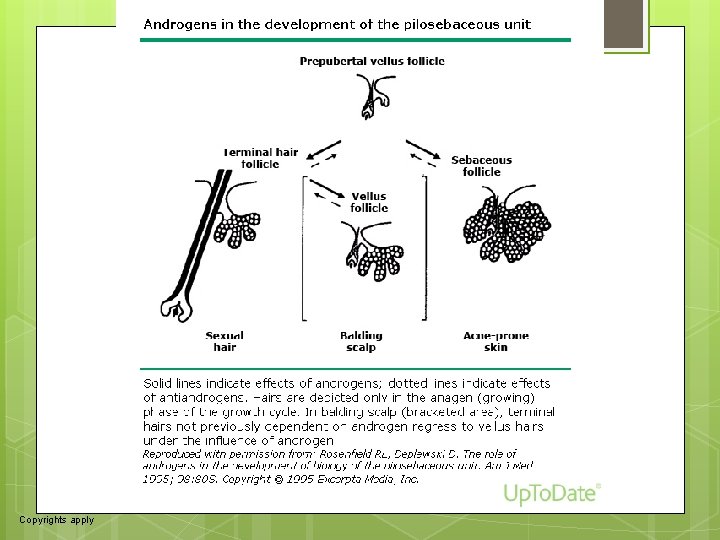
Copyrights apply
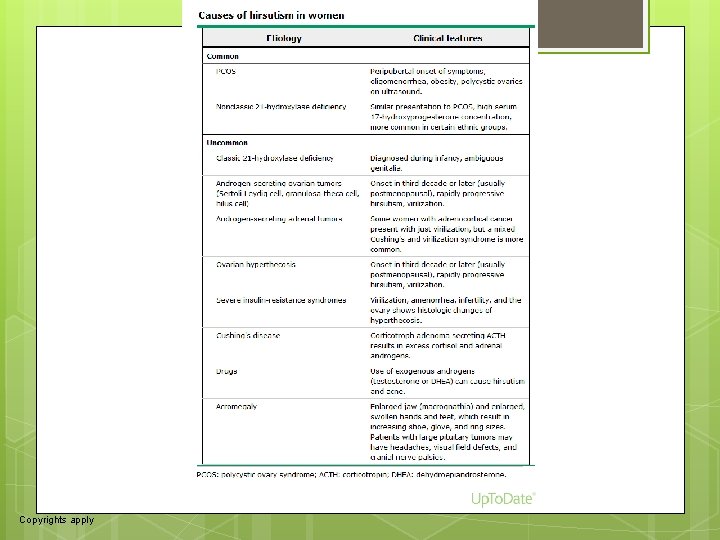
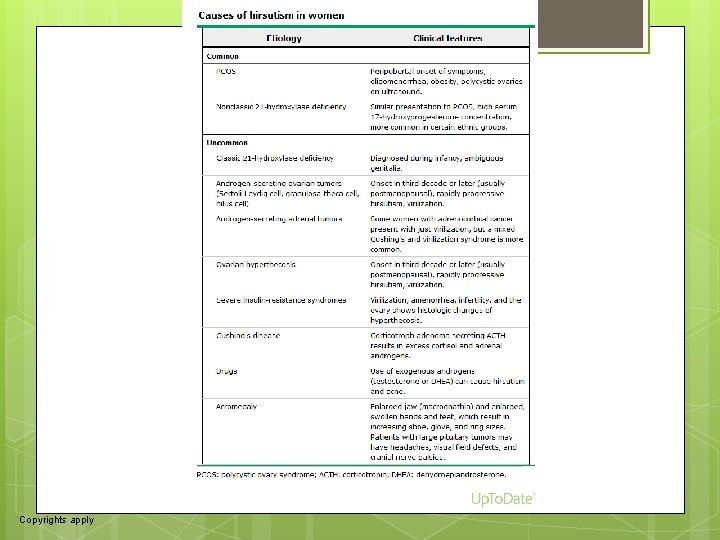
Copyrights apply

Copyrights apply

Copyrights apply

6 DEFINITION: Hirsutism is a clinical diagnosis defined by the presence of excess terminal hair growth (dark, coarse hairs) in androgendependent areas (eg, upper lip, chin, midsternum, upper and lower abdomen, upper and lower back, and buttocks)

7 Hirsutism is an important clinical problem because: ●It affects approximately 5 to 10 percent of women [3]. ●It is associated with significant emotional distress and depression [4 -6]. ●It is usually an indication of an underlying endocrine disorder (most commonly polycystic ovary syndrome [PCOS])

8 Elevated serum concentrations of androgens in women are referred to as hyperandrogenemia, which can manifest as hyperandrogenism: hirsutism, acne, androgenetic alopecia, and virilization. Hirsutism, defined as excessive growth of terminal hair in women in a male-like pattern, is the most commonly used clinical diagnostic criterion of hyperandrogenism [1, 2]. Virilization, which is rare, occurs only with severe hyperandrogenism and typically includes deepening of the voice and clitoromegaly.

9 Ferriman-Gallwey score Using this method, nine androgen-sensitive sites are graded from 0 to 4 (figure 1) [1, 10]. The criteria for identifying hirsutism using the Ferriman. Gallwey score are variable because the expression of hair growth varies among racial/ethnic groups, as follows: ●A Ferriman-Gallwey score >8 is considered abnormal for black or white women, a reasonable cutoff based upon data from two population studies in which only 5 percent of women had scores higher than 7 or 8 [1, 11]. In one study, however, many women with lower scores (>3) had evidence of an underlying androgen excess disorder [7]. Scores between 8 and 15 are usually considered to be mild hirsutism, 16 to 25 moderate, and scores >25 severe hirsutism. ●For Mediterranean, Hispanic, and Middle Eastern women, a Ferriman. Gallwey score ≥ 9 to 10 is considered abnormal and for Asian women, a score ≥ 2 [12, 13].

10 Limitations — We use the Ferriman-Gallwey score to assess the severity of hirsutism at baseline and at follow-up visits to monitor response to pharmacologic and direct hair removal therapies [ 14, 15]. However, there a number of limitations to the Ferriman-Gallwey score in clinical practice [15]: ●Many clinicians are unfamiliar with the Ferriman-Gallwey score and are therefore unlikely to use it. ●Most women have been using cosmetic measures (shaving, plucking), especially on the face, by the time they see a clinician, so their Ferriman-Gallwey score at baseline may not be an accurate reflection of the severity and distribution of their hair growth. However, details about the distribution and severity of facial hair (before cosmetic measures were used) can be obtained from the patient. In addition, nonfacial skin is less often shaved or plucked and therefore may reflect the actual hirsutism status better. ●The expression of hair growth varies among racial/ethnic groups. Most East Asian women have little body hair, white and African. American women have an intermediate amount, and most Mediterranean, South Asian, and Middle Eastern women have substantially greater quantities of body hair, even though serum androgen concentrations are similar in all groups [ 16 -18]. A much lower Ferriman. Gallwey score (≥ 2) is considered to be abnormal in Asian women than other groups as they are less likely to manifest hirsutism resulting from androgen excess [12]. (See "Pathophysiology and causes of hirsutism", section on 'Ethnicity'. ) ●There is only a modest correlation between the quantity of hair growth and serum androgen levels [7, 19]. This is thought to result from the fact that stimulation of hair growth from the follicle does not depend solely on circulating androgen concentrations but also upon local factors and variability in end-organ sensitivity to circulating androgens [8]. (See "Pathophysiology and causes of hirsutism", section on 'Role of androgens'. ) ●Intraobserver agreement appears to be relatively good when using the modified Ferriman-Gallwey score [ 20], but interobserver agreement appears to be poor [ 21]. ●Many women with low scores seek treatment because they are as distressed by their hair growth as women with high scores. In one study of black and white women, most with a score >3 considered themselves to be hirsute [ 11]. Women's self-ratings of hirsutism using the Ferriman-Gallwey score tend to be higher than clinician ratings [ 22]. Therefore, treatment decisions are often based upon the degree to which the hirsutism affects the patient's well-being and not just on the severity score

11 Other types of excess hair — There are several conditions characterized by generalized or "excess" hair growth that do not represent hirsutism and do not require biochemical evaluation with serum androgens: ●Vellus (lanugo) hair – The soft, androgen-independent unpigmented hair that covers the entire body of a fetus or newborn. It may also be seen in women with disorders that include anorexia nervosa. ●Hypertrichosis – The excessive growth of androgen-independent hair that is vellus, prominent in nonsexual areas, and most commonly familial or caused by systemic disorders (hypothyroidism, anorexia nervosa, malnutrition, porphyria, and dermatomyositis) or oral medications (phenytoin, penicillamine, diazoxide, minoxidil, or cyclosporine). Hypertrichosis is typically managed with hair removal techniques. (See "Removal of unwanted hair". ) ●"Unwanted hair" – This term refers to any hair growth (usually facial) that the patient finds bothersome. However, the hairs are typically not dark terminal hairs but rather light, unpigmented hairs. This type of hair is not a sign of androgen excess. (See "Removal of unwanted hair". )

12 APPROACH TO THE PATIENT Although women with hirsutism typically present because of concerns regarding their excessive hair growth, most also have an underlying endocrine disorder that should be identified (table 1). Important clues to the severity of androgen excess and the presence of an underlying disorder can be obtained by the history, physical examination, and appropriate laboratory testing (algorithm 1) [23, 24]. Goals — The most important goals of the evaluation include (algorithm 1): ●Distinguish true hirsutism from other causes of bothersome hair growth. (See 'Other types of excess hair' above. ) ●Determine if the patient is taking any drugs that could cause excessive hair growth (oral minoxidil, androgenic steroids). (See "Use of androgens and other hormones by athletes". ) ●Identify the most serious causes of hirsutism, including androgen-secreting tumors (ovarian or adrenal) and ovarian hyperthecosis. Androgen-secreting tumors are rare, particularly in premenopausal women (0. 2 percent of women with hirsutism) [ 13], but 50 percent are malignant at the time of diagnosis. Ovarian hyperthecosis, a disorder characterized by severe hyperandrogenism and insulin resistance, is seen primarily in postmenopausal women but occasionally in premenopausal women. (See 'Additional evaluation for severe hyperandrogenemia' below and "Ovarian hyperthecosis". )

13 ●Identify polycystic ovary syndrome (PCOS), the underlying disorder in approximately 75 to 80 percent of women who present with hirsutism (table 2). The diagnosis of PCOS is important because, in addition to oligomenorrhea and hyperandrogenism, PCOS is associated with other important medical conditions including obesity, insulin resistance, and an increased risk for type 2 diabetes, dyslipidemia, hypertension, and obstructive sleep apnea. The clinical features and evaluation of patients with suspected PCOS are reviewed in detail separately. (See "Clinical manifestations of polycystic ovary syndrome in adults" and "Diagnosis of polycystic ovary syndrome in adults". ) ●Identify nonclassic congenital adrenal hyperplasia (NCCAH) due to 21 -hydroxylase deficiency (<5 percent of women with hirsutism, depending upon population). Although this diagnosis will not alter the approach to managing hirsutism in most cases, it is important for future genetic counseling when pursuing fertility. If her partner has NCCAH (which is asymptomatic in adult males) or is a heterozygote carrier, there is a risk that their offspring could have classic 21 -hydroxylase deficiency (the severe form of the disease). (See "Diagnosis and treatment of nonclassic (lateonset) congenital adrenal hyperplasia due to 21 -hydroxylase deficiency". )

14 ●Identify other uncommon causes of hirsutism that would require different management than PCOS, NCCAH, or idiopathic hirsutism, such as Cushing's syndrome or acromegaly (table 1). (See 'Other' below. ) ●Determine the degree of emotional distress caused by the excess hair as this has important implications for treatment. (See "Management of hirsutism in premenopausal women". )

15 Overview of our approach ●The shistory hould focus initially on the time course of symptoms, whether the patient has become virilized, and the degree of emotional distress caused by the excess hair. (See 'History' below. ) ●The physical examination should focus on determining if the patient has evidence of virilization, an indicator of severe hyperandrogenism, in addition to determining whether the patient has true hirsutism (and its severity if present) (figure 1). (See 'Physical examination' below. ) ●We agree with the 2018 Endocrine Society Clinical Guidelines and suggest measuring a serum total testosterone in all women with an abnormal hirsutism score (algorithm 1). We do not measure testosterone in women with normal menstrual cycles and "unwanted local hair growth. " Some women who present with hirsutism are already on combination oral contraceptives (COCs). Their management is described below. (See 'Women already taking pharmacologic therapy' below. )

16 The choice of additional tests is based upon the patient's presentation (time of onset, pace of progression, presence or absence of virilization, and menstrual cycle status). As noted, 75 to 80 percent of women who present with hirsutism have PCOS, but it is essential to identify those who have a more serious cause (androgen-secreting tumors and ovarian hyperthecosis). (See 'Biochemical testing' below. ) ●Further evaluation is indicated in all women when an ovarian androgensecreting tumor is suspected (serum total testosterone >150 ng/d. L [5. 2 nmol/L]) (algorithm 1). We suggest pelvic ultrasound as the next diagnostic step. If the ultrasound is negative and/or serum dehydroepiandrosterone sulfate (DHEAS) is greater than 700 mcg/d. L (18. 9 micromol/L), we suggest adrenal computed tomography (CT) to look for an androgen-secreting adrenal tumor. (See 'Additional evaluation for severe hyperandrogenemia' below. )

17 History — The history should focus initially on the time course of symptoms, whether the patient has become virilized, and the degree of emotional distress caused by the excess hair. Age of onset — Women with PCOS, the most common cause of hirsutism, typically have a peripubertal onset of hirsutism. This is not always the case, however, as adolescent girls with PCOS often take COCs for irregular periods and/or for acne, which may mask or delay the recognition of hirsutism. Women with NCCAH due to 21 -hydroxylase deficiency and idiopathic hirsutism have a similar age of symptom onset. (See "Clinical manifestations of polycystic ovary syndrome in adults", section on 'Hyperandrogenism'. ) In contrast, women with androgen-secreting tumors or ovarian hyperthecosis develop hirsutism in the third decade of life or later. Both diagnoses are most common after menopause but sometimes occur before. (See "Sex cord-stromal tumors of the ovary: Epidemiology, clinical features, and diagnosis in adults" and "Ovarian hyperthecosis". )

18 Stable versus progressive hair growth — Once women with PCOS develop hirsutism, the pattern of hair growth may be variable. Hirsutism is stable in some women and may worsen in others, but changes occur over years, not weeks or months. Women with NCCAH due to 21 -hydroxylase deficiency and idiopathic hirsutism have a similar presentation. In contrast, women with androgen-secreting tumors present with recent-onset, short-duration (typically less than one year), or rapidly progressive hirsutism (algorithm 1).

19 Virilization — Signs of virilization, including frontal balding, clitoromegaly, increased muscle mass, or deepening of the voice, are seen when the serum testosterone concentration is more severely elevated (eg, over 150 ng/d. L), which occurs in ovarian hyperthecosis androgen-secreting ovarian or adrenal tumors. Although serum testosterone levels may be somewhat elevated in women with PCOS, they are not in a range that can cause virilization. Some disorders cause virilization during pregnancy. (See "Ovarian hyperthecosis" and "Gestational hyperandrogenism" and "Sex cord-stromal tumors of the ovary: Epidemiology, clinical features, and diagnosis in adults". )

20 Emotional distress/depression — Hirsutism is associated with significant emotional distress and depression [4 -6]. Those with PCOS are more likely to have mood disorders and are at risk for eating disorders, eg, binge eating [25, 26]. Therefore, women should be asked about mood symptoms, and a dietary history should be obtained. The degree of emotional distress caused by the hirsutism can affect treatment decisions. (See "Clinical manifestations of polycystic ovary syndrome in adults", section on 'Psychosocial issues' and "Management of hirsutism in premenopausal women". )

21 Other — Other components of the history that help determine the underlying etiology include the following: ●Menstrual history – Does the patient have oligomenorrhea or amenorrhea? The presence of menstrual dysfunction suggests PCOS, but most disorders that cause hirsutism can be associated with irregular menstrual cycles, with the exception of idiopathic hirsutism, whose diagnosis requires the presence of regular menstrual cycles (algorithm 1). ●Family history – A family history of hirsutism, acne, menstrual irregularity, infertility, early cardiovascular disease, and obesity suggests the possibility of PCOS in the patient. (See "Epidemiology, phenotype, and genetics of the polycystic ovary syndrome in adults", section on 'Epidemiology'. ) ●Weight history – Obese women have increased androgen production and clearance rates [27], each of which aggravates hirsutism and increases the likelihood of menstrual irregularity, especially in those with PCOS. (See "Clinical manifestations of polycystic ovary syndrome in adults", section on 'Obesity and insulin resistance'. )

22 ●Ethnicity – The evaluation of body hair must consider the individual's ethnic background. Most Asian and Native American women have little body hair, while Mediterranean women on average have substantially greater quantities of body hair even though serum androgen concentrations are similar in the three groups [ 16]. Thus, an Asian woman with a few peri-areolar hairs may warrant further evaluation, whereas a Mediterranean woman with some hair growth on her upper lip might be considered normal. (See 'Ferriman-Gallwey score' above. ) ●Medication history – Drugs that cause hirsutism or have other androgenic effects include testosterone and dehydroepiandrosterone (DHEA) (sometimes used for the management of sexual dysfunction) or "androgenic steroids, " used by athletes to improve their performance. Valproate has been associated with the development of PCOS (table 1). (See "Overview of sexual dysfunction in women: Management", section on 'Androgens' and "Use of androgens and other hormones by athletes". ) ●Other endocrine disorders associated with hirsutism – These usually present with the typical manifestations of the specific endocrinopathy, such as weight gain and hypertension (Cushing's syndrome) (table 1). (See "Pathophysiology and causes of hirsutism", section on 'Causes'. )

23 Women with NCCAH due to 21 -hydroxylase deficiency have a clinical presentation that is indistinguishable from PCOS. One clue, however, is ethnicity. NCCAH is most common in Mediterranean, Hispanic, and Ashkenazi Jewish women. A family history of irregular menses and hirsutism may be present in women with NCCAH [28]. (See "Genetics and clinical presentation of nonclassic (late-onset) congenital adrenal hyperplasia due to 21 hydroxylase deficiency" and "Diagnosis and treatment of nonclassic (lateonset) congenital adrenal hyperplasia due to 21 -hydroxylase deficiency", section on 'Who should be tested? '. ) ●Some women present with hirsutism and normal ovulatory cycles. While some will meet PCOS criteria after further evaluation (if there is evidence of biochemical hyperandrogenism or polycystic ovaries on ultrasound), others will have normal androgens and a normal ultrasound. These women are considered to have "idiopathic hirsutism. " (See "Pathophysiology and causes of hirsutism", section on 'Idiopathic hirsutism'. )

24 Physical examination ●Evidence of virilization – In addition to determining whether the patient has true hirsutism (picture 1) and assessing its severity (figure 1), the most important goal of the physical examination is to determine if the patient has evidence of virilization, an indicator of severe hyperandrogenism. Findings include deepening of the voice, temporal and/or crown balding, increased muscle mass, and clitoromegaly. There is significant variability in clitoral size in normal women [29, 30]. Clitoral enlargement is typically determined on the basis of clitoral length (of the glans) or the clitoral index (length times width): length >10 mm or an index >35 mm 2 is considered above normal [31]. Women with virilization are likely to have a serious cause for their hirsutism, either an androgen-secreting tumor (ovarian or adrenal) or ovarian hyperthecosis. (See 'Additional evaluation for severe hyperandrogenemia' below and "Pathophysiology and causes of hirsutism", section on 'Causes'. )

25 ●Other important findings – Other aspects of the physical examination that are important in the evaluation of hirsutism include: • Other skin findings – Acne or seborrhea (which are additional signs of androgen excess), acanthosis nigricans, striae, thin skin, or bruising should be looked for on exam. The last three findings suggest the possible presence of Cushing's syndrome, whereas acanthosis nigricans suggests insulin resistance. • Body mass index (BMI) – Height, weight, and a calculation of BMI should be obtained. Many women with PCOS are obese (BMI ≥ 30 kg/m 2). In addition, the pattern of body fat distribution (truncal obesity, cervicodorsal fat deposition, and supraclavicular fat) may suggest the presence of Cushing's syndrome. (See "Epidemiology and clinical manifestations of Cushing's syndrome". ) • Abdominal and pelvic exam – Abdominal and pelvic examination should be performed to look for mass lesions that could indicate an androgensecreting tumor.

26 Biochemical testing — Our approach is consistent with the 2018 Endocrine Society Clinical Guidelines on hirsutism. We suggest measuring a serum total testosterone in all women with clinical evidence of hirsutism (defined by the presence of excess terminal hair growth [dark, coarse hairs] in androgen-dependent areas [ 15]. The choice of additional tests is based upon the patient's presentation (time of onset, pace of progression, presence or absence of virilization, and menstrual cycle status). As noted, 75 to 80 percent of women who present with hirsutism have PCOS, but it is essential to identify those who have a more serious cause (androgen-secreting tumors and ovarian hyperthecosis). "Unwanted hair" — Excess hair that is not true hirsutism, ie, is not dark, coarse, and in androgen-dependent areas, does not need biochemical evaluation and does not respond to the pharmacologic therapies for androgen-dependent hair. Hirsutism and normal menstrual cycles — Women with hirsutism and normal menstrual cycles are most likely to fall into the category of PCOS or idiopathic hirsutism and are unlikely to have a more serious cause for their hirsutism. We suggest measuring only a serum total testosterone. This is consistent with guidelines from expert groups (algorithm 1) [15]. (See "Pathophysiology and causes of hirsutism", section on 'Causes'. )

27 Hirsutism with oligomenorrhea/amenorrhea — Women with hirsutism and irregular menstrual cycles have an underlying endocrine disorder. Many, if not most, women will be diagnosed with PCOS. We suggest measuring serum total testosterone as well as a routine evaluation for oligo/amenorrhea (human chorionic gonadotropin [h. CG], prolactin, follicle-stimulating hormone [FSH], thyroid-stimulating hormone [TSH]) and an early morning 17 -hydroxyprogesterone (around 8 AM). Although the menstrual dysfunction will almost always be due to PCOS or, in some cases, NCCAH due to 21 -hydroxylase deficiency, the possibilities of pregnancy, hyperprolactinemia, primary ovarian insufficiency, and thyroid disease should not be overlooked (algorithm 1). (See "Evaluation and management of secondary amenorrhea", section on 'Initial laboratory testing'. )

28 Serum androgens — Suggestions for serum androgen testing include the following: ●Serum total testosterone – We measure serum total testosterone in women with any degree of hirsutism. It is best assessed by liquid chromatography-tandem mass spectroscopy (LC-MS/MS), an accurate and specific method. With LC-MS/MS, the upper limit of normal for serum testosterone in women is in the 45 to 60 ng/d. L range (1. 6 to 2. 1 nmol/L). The immunoassays that are available in most hospital laboratories are not suitable to accurately measure testosterone in women [32, 33]. Women with PCOS have serum testosterone concentrations that may be in the normal range (up to the upper limit of normal, which varies between 45 and 60 ng/d. L depending upon the laboratory [0. 69 to 2. 1 nmol/L]), or elevated but below 150 ng/d. L (5. 2 nmol/L). Although the degree of hyperandrogenemia may not correlate well with the severity of hirsutism, it does appear to correlate with metabolic risks observed in women with PCOS [34].

29 ● 17 -hydroxyprogesterone – We also suggest testing for NCCAH due to 21 -serum hydroxylase deficiency in hyperandrogenemic women by measuring an 8 AM 17 hydroxyprogesterone level. In clinical practice, it is unusual to identify NCCAH in a woman having an endocrine work-up for hirsutism unless a circulating androgen is elevated. This test is most important in high-risk women, including Mediterranean, Hispanic, and Ashkenazi Jewish women [28]. For women who have some spontaneous menstrual cycles, this should be done in the early follicular phase, while for those without cycles, it can be drawn on a random day. A morning value of 17 -hydroxyprogesterone greater than 200 ng/d. L in the early follicular phase strongly suggests the diagnosis, which may be confirmed by a high-dose (250 mcg) corticotropin (ACTH) 1 -24 (cosyntropin) stimulation test. The response to cosyntropin is exaggerated, with most patients having values exceeding 1500 ng/d. L (43 nmol/L) 60 minutes after administration of cosyntropin. (See "Diagnosis and treatment of nonclassic (late-onset) congenital adrenal hyperplasia due to 21 -hydroxylase deficiency". ) ●Free testosterone – We do not routinely measure free testosterone. Free testosterone measured by equilibrium dialysis is a sensitive test for detecting hyperandrogenic disorders, but it must be performed in a specialty laboratory. Free testosterone by analog method ("direct") is not accurate and should not be ordered.

30 ●Serum DHEAS – We do not suggest measuring serum DHEAS, because mildly elevated levels of DHEAS are unlikely to affect management. In addition, elevated DHEAS concentrations do not discriminate as well as serum testosterone between malignant and benign causes of adrenal hyperandrogenism. (See 'Severe hyperandrogenism' below. ) ●Serum androstenedione – The role of serum androstenedione in the evaluation of PCOS and/or hirsutism is unclear [35]. However, its measurement in some populations with PCOS, for example, Icelandic women, may be important for documenting hyperandrogenism [36]. Two studies have reported isolated elevations of serum androstenedione concentrations in women with PCOS, but the percent of women with this biochemical pattern is variable (20 of 86 [23 percent] [37] and 49 of 1128 [4. 1 percent] [38]). In the smaller study, serum androstenedione was negatively associated with insulin sensitivity. (See "Clinical manifestations of polycystic ovary syndrome in adults". ) ●Sex hormone-binding globulin (SHBG) – Some experts calculate a free testosterone concentration from the total testosterone, SHBG, and albumin concentrations. However, there are many different equations for this calculation that give different results, some of which reflect the results obtained by equilibrium dialysis better than others. As noted, most women with hirsutism and irregular menses will be diagnosed with PCOS. Once the diagnosis of PCOS is made, we suggest additional evaluation including a cardiometabolic risk assessment, as well as screening for mood disorders and sleep apnea. (See "Diagnosis of polycystic ovary syndrome in adults", section on 'Further evaluation after diagnosis'. )

31 Women already taking pharmacologic therapy — Some women with hirsutism seek advice when they are already taking pharmacotherapy, usually COCs. Measuring serum androgens is generally not useful in this setting, because COCs suppress serum gonadotropins and ovarian androgens, most importantly testosterone. However, if an evaluation is done, it is best to discontinue the COC for at least 8 to 12 weeks as it takes at least this length of time for serum androgens and SHBG levels to return to basal values [39]. Biochemical evaluation is usually not necessary unless the patient is experiencing severe or rapid worsening of her hirsutism despite COC therapy. In this setting, marked elevation of testosterone in spite of COC use strongly suggests a testosterone-secreting ovarian or adrenal tumor (algorithm 1). More commonly, the patient has had an inadequate response to COCs alone so a second medication is added, usually spironolactone. (See "Management of hirsutism in premenopausal women". )

32 Women with features of other endocrine disorders — Rarely, hirsute women have symptoms and signs of cortisol excess (such as centripetal obesity, hypertension, purple striae) that suggest Cushing's syndrome, or growth hormone excess that suggests acromegaly. In addition to measuring serum total testosterone, women with suspected Cushing's syndrome should undergo testing for hypercortisolism. Options for firstline tests include a 24 -hour urinary excretion of free cortisol, late-night salivary cortisol, and the low-dose dexamethasone suppression test. At least two of these first-line tests should be abnormal to establish the diagnosis of excess cortisol production. For women with possible growth hormone excess, a serum insulin-like growth factor-1 (IGF-1) should be measured first. (See "Establishing the diagnosis of Cushing's syndrome", section on 'Available tests' and "Diagnosis of acromegaly", section on 'Serum IGF-1 concentration'. )

33 Severe hyperandrogenism — For women with features of severe hyperandrogenism (virilization and/or hirsutism of recent onset that is rapidly progressive), we suggest measuring serum total testosterone, ideally using LC-MS/MS as described above, and serum DHEAS. In addition, these patients should be referred to an endocrinologist or reproductive endocrinologist for further evaluation. These women usually have an androgen-secreting tumor (ovarian or adrenal) or ovarian hyperthecosis (although both are more common in postmenopausal than premenopausal women) (algorithm 1). (See "Evaluation and management of postmenopausal hyperandrogenism", section on 'Women with virilization or severe hyperandrogenemia'. )

34 ●Serum total testosterone – Serum total testosterone is the best test for detecting androgen-secreting tumors, in particular ovarian neoplasms. Although serum free testosterone may be the more sensitive test for the presence of hyperandrogenic disorders [40], an elevated total testosterone level alone is usually sufficient to identify those women who need further evaluation for a possible androgen-secreting tumor [41]. In addition, free testosterone measured by an analog method, which is the assay most commonly offered by hospital and commercial laboratories, does not correlate at all with the results of equilibrium dialysis, the most accurate method for measuring free testosterone [41]. (See 'Serum androgens' above. ) Total testosterone values above 150 ng/d. L (5. 2 nmol/L) should prompt additional evaluation for a testosterone-secreting ovarian or adrenal tumor [42 -45] as well as ovarian hyperthecosis, a disorder characterized by severe hyperandrogenism and insulin resistance. Both disorders are seen more commonly in postmenopausal women but occur occasionally in premenopausal women. (See 'Additional evaluation for severe hyperandrogenemia' below and "Evaluation and management of postmenopausal hyperandrogenism", section on 'Women with virilization or severe hyperandrogenemia' and "Ovarian hyperthecosis", section on 'Biochemical findings'. )

35 ●Serum DHEAS – Serum DHEAS is an adrenal androgen and is often measured to try to identify an adrenal source of excess androgens, most importantly adrenal androgen-secreting neoplasms. Mildly elevated levels of DHEAS are unlikely to affect management. Elevated DHEAS concentrations do not discriminate as well as serum testosterone between malignant and benign causes of adrenal hyperandrogenism , and it is unclear whether DHEAS is the best marker for androgen-secreting carcinomas. However, we do suggest its measurement in women who present with signs and symptoms suggestive of an androgen-secreting tumor. Serum DHEAS values above 700 mcg/d. L (18. 9 micromol/L) require further evaluation [13, 15, 42, 43, 46 -48]. (See 'Additional evaluation for severe hyperandrogenemia' below. ) Adults with hormone-secreting adrenocortical carcinomas usually present with Cushing's syndrome alone (45 percent) or a mixed Cushing's and virilization syndrome due to overproduction of both glucocorticoids androgens ( 25 percent). Fewer than 10 percent present with virilization alone, but the presence of virilization in a patient with an adrenal neoplasm suggests an adrenal carcinoma rather than an adenoma. (See "Clinical presentation and evaluation of adrenocortical tumors", section on 'Clinical presentation'. )

36 Additional evaluation for severe hyperandrogenemia — Further evaluation is indicated in all women when an ovarian androgen-secreting tumor is suspected (serum total testosterone >150 ng/d. L [5. 2 nmol/L]). We suggest pelvic ultrasound as the next diagnostic step. If the ultrasound is negative and/or serum DHEAS is greater than 700 mcg/d. L (18. 9 micromol/L), we suggest adrenal CT to look for an adrenal androgen-secreting tumor. Pelvic ultrasonography — Pelvic ultrasonography is indicated in patients with an elevated serum testosterone concentration when an ovarian androgen-secreting tumor is suspected (total testosterone >150 ng/d. L [5. 2 nmol/L]). However, failure to identify a tumor does not rule out its presence, as these tumors may be very small. High-resolution pelvic ultrasonography with a transvaginal probe can identify ovarian follicles and cysts as small as 3 to 5 mm in diameter. It is therefore a safe and effective way to look for polycystic ovary morphology and to screen for ovarian androgen-secreting tumors. Suspicious findings include large cysts, solid masses, and complex cysts that do not resolve spontaneously in two to four weeks [49].

37 Ultrasonography in women with ovarian hyperthecosis usually shows a bilateral increase in ovarian stroma. Unlike PCOS, where the ovaries characteristically have a multifollicular appearance with 12 or more antral follicles of 2 to 9 mm per ovary and/or ovarian volume >10 cm 3, few cysts are seen in severe hyperthecosis, and the ovaries appear more solid. (See "Ovarian hyperthecosis", section on 'Imaging'. ) The role of pelvic ultrasound in the evaluation of severe hyperandrogenism is reviewed in more detail separately. (See "Evaluation and management of postmenopausal hyperandrogenism", section on 'Pelvic ultrasonography' and "Ultrasound differentiation of benign versus malignant adnexal masses". )

38 Adrenal imaging — Adrenal imaging is indicated to look for an adrenal mass if the woman has a markedly elevated serum testosterone (if pelvic ultrasound is negative) or for a serum DHEAS concentration >700 mcg/d. L [18. 9 micromol/L]). Adrenal CT is the imaging test of choice and should readily identify androgen-secreting adrenal lesions [48, 50]. Radiologic testing is otherwise not indicated and may be misleading, because nonfunctioning adrenal masses (adrenal incidentalomas) are common. (See "Evaluation and management of the adrenal incidentaloma". ) Adrenal disorders that may be diagnosed during the evaluation for hirsutism and require surgery include adrenal adenomas and adrenal carcinoma (uncommon in premenopausal women). (See "Clinical presentation and evaluation of adrenocortical tumors". )

39 Ovarian and adrenal vein sampling — Combined ovarian and adrenal vein sampling (selective venous sampling) is sometimes (but not routinely) performed for further evaluation in women with high serum testosterone concentrations (testosterone >150 ng/d. L [5. 2 nmol/L]), as well as normal pelvic ultrasonography and adrenal imaging [51]. In this setting, the ovary is likely to be the source of androgen hypersecretion because adrenal tumors are almost always visualized on adrenal CT [48, 50], while ovarian tumors are often too small to be seen on imaging studies [49]. (See 'Adrenal imaging' above and 'Pelvic ultrasonography' above. ) In premenopausal women, we typically suggest this procedure to help successfully localize the ovarian tumor so that the patient can have a unilateral oophorectomy. The role of ovarian and adrenal vein sampling in the evaluation of women with severe hyperandrogenemia is reviewed in detail separately. (See "Evaluation and management of postmenopausal hyperandrogenism", section on 'Ovarian and adrenal vein sampling'. )

40 SOCIETY GUIDELINE LINKSLinks to society and government-sponsored guidelines from selected countries and regions around the world are provided separately. (See "Society guideline links: Polycystic ovary syndrome" and "Society guideline links: Hirsutism" and "Society guideline links: Classic and nonclassic congenital adrenal hyperplasia due to 21 -hydroxylase deficiency". ) INFORMATION FOR PATIENTSUp. To. Date offers two types of patient education materials, "The Basics" and "Beyond the Basics. " The Basics patient education pieces are written in plain language, at the 5 th to 6 th grade reading level, and they answer the four or five key questions a patient might have about a given condition. These articles are best for patients who want a general overview and who prefer short, easy-to-read materials. Beyond the Basics patient education pieces are longer, more sophisticated, and more detailed. These articles are written at the 10 th to 12 th grade reading level and are best for patients who want in-depth information and are comfortable with some medical jargon. Here are the patient education articles that are relevant to this topic. We encourage you to print or email these topics to your patients. (You can also locate patient education articles on a variety of subjects by searching on "patient info" and the keyword(s) of interest. ) ●Basics topics (see "Patient education: Hirsutism (excess hair growth in women) (The Basics)") ●Beyond the Basics topics (see "Patient education: Hirsutism (excess hair growth in women) (Beyond the Basics)")
Copyrights apply

Copyrights apply

Copyrights apply
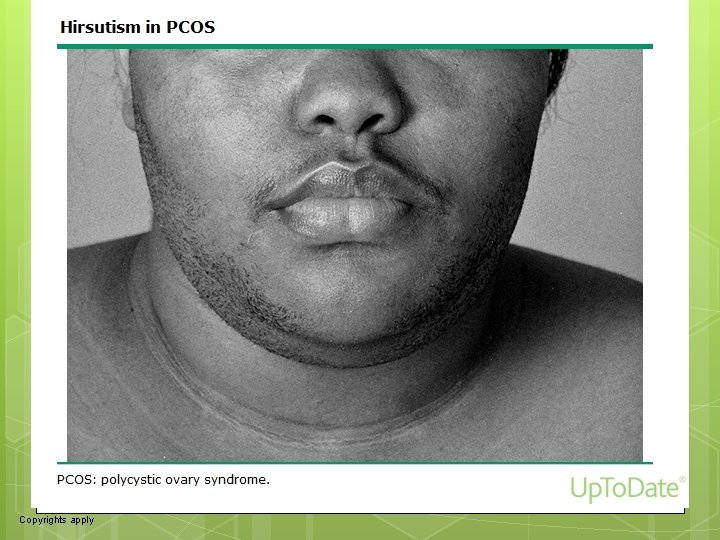
Copyrights apply

Copyrights apply

46

47

48

49 SUMMARY AND RECOMMENDATIONS ●Hirsutism affects approximately 5 to 10 percent of premenopausal women and is usually an indication of an underlying endocrine disorder (most commonly polycystic ovary syndrome [PCOS]) (table 1). (See 'Introduction' above. ) ●Hirsutism is associated with significant emotional distress and depression. (See 'Emotional distress/depression' above. ) ●Hirsutism is a clinical diagnosis defined by the presence of excess terminal hair growth (dark, coarse hairs) in androgen-sensitive areas (eg, upper lip, chin, midsternum, upper abdomen, back, and buttocks). (See 'Definition' above. ) ●A simple and commonly used method to grade hair growth makes use of the modified scale of Ferriman and Gallwey, which grades nine androgen-sensitive sites on a scale from 0 to 4 (figure 1). (See 'Ferriman-Gallwey score' above. ) ●Although women with hirsutism typically present because of cosmetic concerns, the majority also have an underlying endocrine disorder (most commonly PCOS) that should be identified. (See 'Goals' above. )

50 ●The most important goal of the evaluation is to identify the most serious causes of hirsutism, including androgen-secreting ovarian and adrenal tumors. Features that suggest a possible androgen -secreting tumor include (see 'Goals' above): • Recent-onset, short-duration (typically less than one year), progressive worsening of hirsutism • Signs of virilization, including frontal balding, acne, clitoromegaly, increased muscle mass, and deepening of the voice ●We suggest measuring a serum total testosterone in all women with hirsutism. (See 'Biochemical testing' above. ) Additional testing is based upon the patient's presentation (time of onset, pace of progression, presence or absence of virilization, and menstrual cycle status) (algorithm 1). As noted, 75 to 80 percent of women who present with hirsutism have PCOS, but it is essential to identify those who have a more serious cause (androgen-secreting tumors and ovarian hyperthecosis). (See 'Biochemical testing' above. ) ●Some women with hirsutism seek advice when they are already receiving pharmacotherapy, usually combination oral contraceptives (COCs). Measuring serum androgens is not useful in this setting, because COCs suppress serum gonadotropins and ovarian androgen, most importantly, testosterone. Biochemical evaluation is usually not necessary unless the patient is experiencing severe or rapid worsening of her hirsutism in spite of COCs (algorithm 1). More commonly, the patient has had an inadequate response to COCs alone so a second medication is added, usually spironolactone. (See 'Women already taking pharmacologic therapy' above. )

51 ●For women with severe hyperandrogenemia (see 'Additional evaluation for severe hyperandrogenemia' above): • We suggest a transvaginal ultrasound if the serum total testosterone concentration is ≥ 150 ng/d. L (5. 2 nmol/L) (see 'Pelvic ultrasonography' above). If the ultrasound is negative, an adrenal computed tomography (CT) scan should be performed. • We also suggest an adrenal CT scan to look for an adrenal androgen-secreting tumor if the serum dehydroepiandrosterone sulfate (DHEAS) concentration is ≥ 700 mcg/d. L (18. 9 micromol/L). (See 'Adrenal imaging' above. ) • If the necessary expertise is available, combined ovarian and adrenal vein sampling (selective venous sampling) is sometimes performed for further evaluation in women with high serum testosterone concentrations (testosterone >150 ng/d. L [5. 2 nmol/L]), as well as normal pelvic ultrasonography and adrenal imaging. In this setting, the ovary is likely to be the source of androgen hypersecretion because adrenal tumors are almost always visualized on adrenal CT, while ovarian tumors are often too small to be seen on imaging studies. (See 'Ovarian and adrenal vein sampling' above. ) Use of Up. To. Date is subject to the Subscription and License Agreement.

52
Copyrights apply
Copyrights apply

55 Management of hirsutism in premenopausal women INITIAL VISIT What is the patient's underlying diagnosis? — The first step before starting therapy in hirsute women is to establish or confirm the underlying diagnosis (see "Evaluation of premenopausal women with hirsutism"). Most women (80 to 85 percent) have polycystic ovary syndrome (PCOS), defined by the presence of two of the following three criteria (oligomenorrhea, hyperandrogenism, and polycystic ovaries on pelvic ultrasound) (see "Diagnosis of polycystic ovary syndrome in adults", section on 'Diagnosis'). The two main pharmacologic options for hirsutism (combined estrogen-progestin oral contraceptives [COCs] and antiandrogens) can only be used in women not seeking fertility. A much lower percentage of women with hirsutism have nonclassic congenital adrenal hyperplasia due to 21 -hydroxylase deficiency (NCCAH). Their clinical presentation is very similar and often indistinguishable from PCOS. Although NCCAH is an adrenal disorder, the management of hirsutism is largely the same as for women with PCOS. (See "Genetics and clinical presentation of classic congenital adrenal hyperplasia due to 21 -hydroxylase deficiency" and "Diagnosis and treatment of nonclassic (late-onset) congenital adrenal hyperplasia due to 21 -hydroxylase deficiency". ) Baseline Ferriman-Gallwey score — We use the modified scale of Ferriman and Gallwey (F-G) to grade the severity of hair growth objectively. We perform an assessment at baseline and at follow-up visits. We do not use these scores to identify candidates for pharmacologic treatment, but we do use them to monitor response to therapy.
![56 Nine androgen-sensitive sites are graded from 0 to 4 (figure 1) [9]. The 56 Nine androgen-sensitive sites are graded from 0 to 4 (figure 1) [9]. The](http://slidetodoc.com/presentation_image_h2/32194bb162c169105456c3ca02d28c7a/image-56.jpg)
56 Nine androgen-sensitive sites are graded from 0 to 4 (figure 1) [9]. The criteria for identifying hirsutism using the F-G score are variable because the expression of hair growth varies considerably among racial/ethnic groups. Based on population studies, an F-G score >8 is considered abnormal for black or white women. For Mediterranean, Hispanic, and Middle Eastern women, an F-G score ≥ 9 to 10 is considered abnormal, and for Asian women, a score of ≥ 2 qualifies. There a number of limitations of the F-G score including lack of clinician familiarity with its use, patient use of cosmetic hair removal methods (shaving, plucking, waxing) before the initial evaluation and while receiving therapy, and the variable scores based on ethnicity. This issue is reviewed in more detail separately. (See "Evaluation of premenopausal women with hirsutism", section on 'Ferriman-Gallwey score'. ) Assess degree of emotional distress — Regardless of the baseline F-G score, the impact of the hirsutism on the patient's quality of life should be assessed [1 -3]. Women with scores below those that are considered to be abnormal can experience significant distress from the presence of excess terminal hair, particularly on the face and neck (figure 1). We therefore treat any woman with "patientimportant" hirsutism, defined in the Endocrine Society guidelines as "unwanted sexual hair growth of any degree that causes sufficient distress for women to seek additional treatment" [10]. If not done previously, screening for depression should be performed. (See 'Management' below and "Evaluation of premenopausal women with hirsutism", section on 'Emotional distress/depression' and "Diagnosis of polycystic ovary syndrome in adults", section on 'Depression and anxiety disorders'. )

57 MANAGEMENT Review strategies and expectations — We review the available pharmacologic options and methods of direct hair removal (photoepilation [laser and intense pulsed light] and electrolysis). ●For all pharmacologic therapies for hirsutism, we suggest a trial of at least six months before making any changes in dose, adding a medication, or switching to a new medication [10]. (See 'Six-month assessment' below. ) ●Our approach is similar to that outlined in the 2018 Endocrine Society clinical guidelines [10]. We suggest that most women with hirsutism start with pharmacologic therapy, and we consider combined estrogen-progestin oral contraceptives (COCs) to be the first-line drug. Exceptions would include women with contraindications to COC use or those who choose not to take COCs. (See 'Combined estrogen-progestin oral contraceptives' below. ) ●An antiandrogen (spironolactone) is then added if the clinical response is suboptimal after six months of therapy [10]. We try to avoid antiandrogen monotherapy unless the patient has adequate contraception (such as an intrauterine device [IUD]), because of the potential risk that a developing male fetus could be undervirilized. (See 'Antiandrogens' below. ) ●We do not typically start with combination therapy (both a COC and an antiandrogen) as initial treatment. However, in occasional patients with severe hirsutism causing significant distress or in women who have not had a good response to COC monotherapy in the past, it is reasonable to initiate combination therapy with a COC and antiandrogen [10]. (See 'Combined with COCs' below. )

58 ●Reasonable expectations should be discussed; women should be counseled that drug therapy is unlikely to completely eliminate already existing hair growth, but that hair may become less coarse, grow more slowly, and/or require less frequent use of cosmetic methods (shaving, plucking, waxing). ●For overweight and obese women, we also recommend lifestyle interventions with a goal of weight loss, in addition to the specific therapy they choose for hirsutism. Weight loss results in improved insulin sensitivity, a decrease in serum androgens, and, in some cases, a return of ovulatory cycles (see 'Obesity' below). The impact on hirsutism is less clear. The majority of women with hirsutism have polycystic ovary syndrome (PCOS). Additional metabolic evaluation for them is reviewed separately. (See "Diagnosis of polycystic ovary syndrome in adults", section on 'Cardiometabolic risk assessment'. ) ●Insulin-lowering agents are not an effective therapy for hirsutism, and we do not suggest their use [9, 11]. (See 'Treatments not routinely recommended' below. )

59 ●Direct methods of hair removal such as photoepilation (laser and intense pulsed light), are also referred to as "permanent" hair reduction techniques. Women may choose to start these options at any point; some choose this as their initial therapy. However, hyperandrogenic women are likely to experience hair regrowth because of the continued stimulation of hair follicles by endogenous androgens. Women who decide to pursue laser/intense pulsed light should also be on pharmacologic therapy to suppress androgens and prevent new hair growth. (See 'Role of direct hair removal methods' below. )

60 Combined estrogen-progestin oral contraceptives — For most women, COCs are our first-line drug; an antiandrogen can then be added if the clinical response is suboptimal after six months of therapy. Women should be aware that COCs and antiandrogens do not have US Food and Drug Administration (FDA) approval for the indication of hirsutism. Therefore, these therapies represent "off-label" use. These preparations also provide additional non-hirsutism benefits such as contraception and cycle management. It is assumed that transdermal and vaginal estrogen-progestin contraceptive preparations are also effective for hirsutism, but data are limited. Clinical trial data on the impact of COCs on hirsutism have been somewhat limited. However, in a network meta-analysis that included trials of COC therapy, the poolweighted mean difference in Ferriman-Gallwey (F-G) score versus placebo was -7. 20 (95% CI -11. 96 to -2. 52) [11]. Although there is no established minimally important difference established for the F-G score, virtually all women would consider these reductions to be clinically important [10]. COCs are also effective for the treatment of acne that often accompanies hirsutism, especially in women with PCOS. These data are reviewed separately. (See "Acne vulgaris: Management of moderate to severe acne", section on 'Oral contraceptives'. )

61 For women with PCOS, COCs provide the additional benefit of preventing the development of endometrial hyperplasia. The treatment of PCOS, including the use of COCs for hirsutism, is reviewed in greater detail separately. (See "Treatment of polycystic ovary syndrome in adults". ) Mechanisms of action in hyperandrogenism/hirsutism — The contraceptive mechanisms of COC action are well known and are discussed separately. COC therapy reduces hyperandrogenism and hirsutism by the following mechanisms (see "Combined estrogen-progestin oral contraceptives: Patient selection, counseling, and use", section on 'Noncontraceptive uses'): ●Inhibition of gonadotropin secretion – Inhibition of ovarian androgen production through suppression of luteinizing hormone (LH) secretion. COC therapy decreases LH and, to a lesser extent, follicle-stimulating hormone (FSH) secretion in hyperandrogenic women [12 -15]. In studies of gonadotropin secretion in women with PCOS, serum FSH concentrations fell more rapidly than did serum LH concentrations, but the fall in serum LH was greater (70 versus 50 percent) [ 12, 13]. The result was decreased ovarian secretion of testosterone and other androgens by the ovaries.

62 ●Increased SHBG – Stimulation of the hepatic production of sex hormone-binding globulin (SHBG), thereby increasing androgen binding in serum and reducing serum free androgen concentrations. In the circulation, testosterone (and estradiol) are bound with high affinity to SHBG and, with lesser affinity but greater extent, to albumin. In women, only approximately 1. 0 to 1. 5 percent of the testosterone in serum is in the physiologically active free form [16, 17]. Androgens and insulin both decrease the hepatic production of SHBG. Since both are often increased in hyperandrogenic women, these women have lower serum SHBG concentrations than normal [18, 19]. Estrogen (and COCs) causes a dose-dependent increase in serum SHBG concentrations over three to four weeks in hyperandrogenic women [15, 20, 21]. As serum SHBG concentrations increase, serum free testosterone concentration decreases. This increase is smaller in women treated with a COC containing a progestin that has significant androgenic properties (eg, levonorgestrel) [22]. When hormonally evaluating women who have been on a COC, it is best to discontinue the medications for at least 8, if not 12, weeks as it takes at least this length of time for measured androgens and SHBG levels to return to basal values [23].

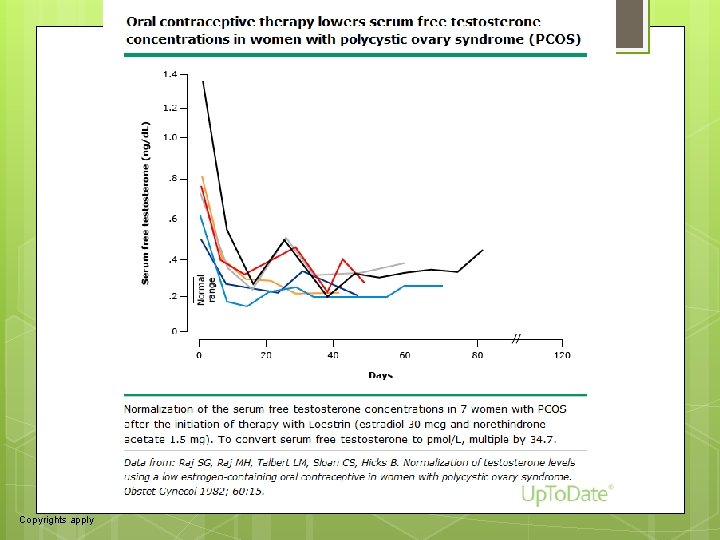
63 ●Reduction in serum total and free testosterone concentrations – As a result of both inhibition of LH secretion and increase in SHBG production, serum free testosterone concentrations decrease by approximately 50 percent in hyperandrogenic women treated with a COC (figure 2) [12 -14, 25]. Serum total testosterone concentrations also fall, but not as much as free testosterone, because of the increase in serum SHBG concentrations. These changes may occur over several weeks [12 -14], but can be slower, especially in women with marked insulin resistance in whom both ovarian androgen and hepatic SHBG production are presumably more dependent on insulin than LH or estrogen. The changes are independent of ovarian histology [25]. ●Secondary mechanisms – COCs may also ameliorate hyperandrogenism by decreasing adrenal androgen secretion [26 -29], inhibiting the peripheral conversion of testosterone to dihydrotestosterone (DHT), and inhibiting the binding of DHT to androgen receptors. However, the evidence that COCs have any of these actions is not strong, and it is unlikely that these mechanisms contribute importantly to their benefit in hyperandrogenic women.

64 Choice of pill — Although data suggest that COCs have similar efficacy for hirsutism [10, 11], we typically choose one that contains a progestin with low or neutral androgenicity, such as norethindrone or norgestimate (table 2). Some clinicians prefer to start with a COC containing an antiandrogenic progestin, drospirenone, or cyproterone acetate (CPA). Drospirenone is structurally related to spironolactone, but it is a very weak antiandrogen at the dose employed in many contraceptives (3 mg). [30]. Data comparing COCs containing drospirenone or CPA with COCs containing other progestins are limited but do not suggest a clinically important advantage for hirsutism [10, 11, 31 -33]. In addition, there have been concerns about a possible excess risk of venous thromboembolism (VTE) with both of these newer progestins compared with other progestins such as norethindrone and levonorgestrel. (See "Combined estrogen-progestin contraception: Side effects and health concerns", section on 'Cardiovascular effects'. ) We typically avoid COC preparations containing the most androgenic progestin, levonorgestrel. Although limited clinical trial data suggest that levonorgestrel-containing COCs have similar benefits for hirsutism as other less androgenic COCs [10, 11, 34], they may have adverse metabolic effects. In one study, a COC containing desogestrel, a third-generation progestin with low androgenicity, had more favorable effects on lipid profiles when compared with a COC containing levonorgestrel [10, 35], a potential concern in women with PCOS who have metabolic concerns at baseline. (See "Treatment of polycystic ovary syndrome in adults", section on 'Metabolic effects of COCs in PCOS'. )

65 We start most patients on a COC formulation that contains 20 mcg of ethinyl estradiol, particularly women with obesity or over age 39 (risk factors for VTE). However, some clinicians may start with 30 to 35 mcg if the hirsutism is more significant and the patient has no VTE risk factors. (See "Combined estrogenprogestin contraception: Side effects and health concerns", section on 'Cardiovascular effects'. ) There had been concerns that the COCs with lower doses of ethinyl estradiol (20 mcg) would be less effective for ovarian androgen suppression. However, in a metaanalysis of 42 studies, suppression of serum total and free testosterone concentrations was similar with 20 mcg versus 30 to 35 mcg dose pills [36]. The transdermal contraceptive patch and vaginal ring suppress serum androgens to a similar degree; limited data suggest that they are beneficial for hirsutism, but their impact on hirsutism has not been studied [37]. (See "Contraception: Transdermal contraceptive patches" and "Contraception: Hormonal contraceptive vaginal rings". ) Six-month assessment — We suggest a trial of at least six months before making any changes in dose, adding a medication, or switching to a new medication. This is because the growth phase of a hair follicle is approximately six months; a significant reduction in hair growth may not occur before then.

66 In addition, we do try to obtain an F-G score at baseline and at each follow-up visit (figure 1). Although we make therapeutic changes primarily based upon the patient's assessment of her response to therapy, the F-G score may help to confirm a good response or suboptimal response. (See "Evaluation of premenopausal women with hirsutism", section on 'Ferriman-Gallwey score'. ) We do not suggest routine monitoring of serum androgens to assess the response to drug therapy. However, if there is progression of hirsutism during therapy, repeat biochemical evaluation is warranted. (See "Evaluation of premenopausal women with hirsutism". ) Good cosmetic response — For women who are satisfied with the degree of improvement in their hirsutism after six months of COC therapy, we suggest continuing the COC based upon the patient's priorities (stop if she decides to pursue fertility). (See 'Duration of drug therapy' below. ) Suboptimal response: Add antiandrogen — If the patient is not satisfied with the degree of improvement in her hirsutism after six months of monotherapy with a COC, we suggest adding an antiandrogen [10, 38].

67 Antiandrogens Combined with COCs — As noted above, we most commonly add an antiandrogen to a COC when the initial response to six months of COC monotherapy has been inadequate. Monitoring of women on combination COC-antiandrogen therapy is the same as that described for COC monotherapy. (See 'Six-month assessment' above. ) We suggest against combination therapy as the initial therapy for most women. However, in occasional patients with severe hirsutism causing significant distress or in women who have not had a good response to COC monotherapy in the past, it is reasonable to initiate combination therapy with a COC and antiandrogen [10]. Monotherapy — Although antiandrogens appear to be as effective as COCs for hirsutism [39], we suggest not using them as monotherapy, because of the potential adverse effects on a developing male fetus in utero. However, in women who cannot conceive or who are using a reliable contraceptive method, either a COC or an antiandrogen can be used as initial therapy as they appear to have similar efficacy [10]. For women with contraindications to COC use, antiandrogens may be given as initial therapy, as long as the individual is using some form of reliable contraception. For example, in a woman with a thrombophilia who cannot take an estrogen-containing contraceptive, an approach to management of hirsutism could include a long-acting reversible contraceptive such as a levonorgestrel-releasing IUD and an antiandrogen such as spironolactone. (See "Intrauterine contraception: Background and device types", section on 'Levonorgestrel IUD'

68 Available antiandrogens: Efficacy — In a network metaanalysis of pharmacologic therapies for hirsutism [11], analyses of seven trials of individual antiandrogens versus placebo (two spironolactone trials 100 mg/day; three finasteride 2. 5 to 5 mg/day; two flutamide 500 mg/day), each antiandrogen showed a significant reduction in hirsutism scores. The effects of the three drugs were similar. Of note, we suggest not using flutamide because of its potential hepatotoxicity. When results of the seven trials of the three antiandrogens were pooled, antiandrogens were significantly more effective than placebo at reducing F-G scores, with a pooled weighted mean difference of -7. 02 (95% CI -11. 51 to -2. 52).

69 Available antiandrogens that are commonly used for hirsutism include the following: ●Spironolactone, our first choice of antiandrogen, is an aldosterone androgen receptor antagonist that is structurally similar to progestin. It competes with DHT for binding to the androgen receptor and inhibits enzymes involved in androgen biosynthesis. (See 'Choice of antiandrogen' below. ) ●Finasteride inhibits 5 -alpha-reductase type 2, the enzyme that converts testosterone to DHT. Only a partial inhibitory effect occurs when used for excess hair growth because the enhanced 5 -alphareductase activity in hirsutism involves both the type 1 and type 2 enzymes. It is used by some clinicians at doses of 1 to 5 mg/day, but there are particular concerns about its inadvertent use in early pregnancy, given the essential role of DHT in the development of male external genitalia. Dutasteride, an inhibitor of both 5 -alpha-reductase types 1 and 2, would theoretically be a more effective therapy than finasteride for hirsutism. It is used for the treatment of benign prostatic hyperplasia, but we suggest against its use for women with hirsutism as there is a substantial risk of preventing the development of normal male external genitalia during early pregnancy [10]. In addition, there are no clinical trial data demonstrating its efficacy for hirsutism. ●Flutamide is a nonsteroidal androgen receptor antagonist. It is used primarily in the management of prostate cancer, but it has been used off-label for managing hirsutism. The efficacy of flutamide (250 to 750 mg/day) is similar to that of spironolactone (100 to 200 mg/day) and finasteride (5 mg/day) [4044]. However, we suggest against the use of flutamide for hirsutism since it has been associated with hepatotoxicity [45 -47], even at doses as low as 62. 5 mg (a dose that has not been shown to be effective for hirsutism in randomized clinical trials) [10, 48]. While some studies have reported that low -dose flutamide (≤ 250 mg) is not hepatotoxic [49 -52], others have identified adverse hepatic effects:

70 • In a 10 -year surveillance study of 203 women receiving flutamide (62. 5 to 125 mg/day), 22 (11 percent) experienced elevated serum concentrations of alanine aminotransferase (ALT) and/or aspartate aminotransferase (AST) [48]. • In a retrospective study of 414 women taking flutamide (125 to 250 mg/day) alone or with COCs, 6 percent stopped therapy in the first year due to elevated transaminases [53]. • In a series of seven women with acne or hirsutism who developed hepatoxicity while taking flutamide (150 to 250 mg/day), five required urgent liver transplantation (four of the five survived) [47]. ●Two weak antiandrogens, CPA (a 17 -hydroxyprogesterone derivative) and drospirenone, are the progestin component in some COCs: • Drospirenone, a progestin used in some COCs, is a very weak antiandrogen. The dose used with ethinyl estradiol in COCs (3 mg) is equivalent to approximately 25 mg of spironolactone [10]. Drospirenone-containing COCs have been associated with a higher risk of VTE than COCs containing second-generation progestins. A progestin-only contraceptive pill containing drospirenone is available, but no data are available for its possible efficacy for treating hirsutism. (See 'Choice of pill' above and "Progestin-only pills (POPs) for contraception", section on 'Formulations'. ) • CPA competes with DHT for binding to the androgen receptor and reduces serum LH and ovarian androgen concentrations. It is used in a low dose (2 mg) as the progestin component of COCs, or as a higher dose (12. 5 to 100 mg) as monotherapy or with estrogen. It is available in almost all countries but the United States. Drug regulatory agencies in Europe have recommended limiting its use to "second-line" therapy because of a perceived increase in risk of hepatotoxicity compared with other available progestins [54, 55].

71 Choice of antiandrogen — There are few comparative studies of different antiandrogens in women with hirsutism. Since they all appear to be effective for hirsutism, the choice of drug depends upon its availability, cost, side effects, and potential toxicity. ●We suggest spironolactone, as clinical trials have shown consistent benefit, and it is considered to be safe [10, 56, 57]. We start with 50 mg twice daily and increase to 100 mg twice daily as needed. The side effects of spironolactone include hyperkalemia (a rare problem in women with normal renal function and aldosterone secretion). However, we measure a serum potassium level in all patients after one month of spironolactone administration. Women with renal insufficiency should not be prescribed spironolactone, because of the high risk of hyperkalemia. Other side effects include gastrointestinal discomfort and irregular menstrual bleeding. In women taking spironolactone (200 mg daily) who develop irregular uterine bleeding, decreasing the spironolactone dose to 100 mg daily may reduce or resolve the bleeding. As noted above, we suggest against spironolactone monotherapy because of the potential risk that a developing male fetus could be undervirilized. An exception is that women with an IUD or subdermal progestin implant can use an antiandrogen monotherapy because they are at a low risk of unintended pregnancy.

72 Duration of drug therapy — Pharmacologic therapy is usually continued during the reproductive years as the underlying condition typically persists during this window, and hirsutism recurs when treatment is discontinued [58, 59]. When pregnancy is desired, all pharmacologic treatments for hirsutism must be discontinued. Antiandrogens, in particular, are contraindicated in women trying to conceive because of potential adverse effects on male sexual development. Role of direct hair removal methods — Direct or mechanical methods of hair removal, including electrolysis and photoepilation (laser and intense pulsed light), are also referred to as "permanent" hair reduction techniques. However, women with underlying hyperandrogenemia are likely to experience hair regrowth because of the continued stimulation of hair follicles by endogenous androgens. This can be prevented by suppressing endogenous androgens with pharmacologic therapy. Melanin pigment is necessary for photoepilation; therefore, hair that is naturally white or blonde is not amenable to treatment. We therefore suggest electrolysis for these patients. Patients with tanned or darkly pigmented skin are at higher risk for unintended thermal injury to the epidermis during photoepilation. Other adverse effects that occur more commonly in women with dark skin include inflammation, blistering, hyperpigmentation, hypopigmentation, and/or rarely, scarring [60]. In women with fair skin, the risk of side effects other than temporary perifollicular inflammation is low. Direct methods of hair removal, including laser and intense pulsed light, are reviewed in more detail separately. (See "Removal of unwanted hair", section on 'Laser and intense pulsed light'. ) Topical therapy — Women who desire a more rapid response to laser therapy can add eflornithine cream. Vaniqa (eflornithine hydrochloride cream 13. 9%) is a topical drug that is available for the treatment of unwanted facial hair in women. It is an inhibitor of hair growth, not a depilatory, and it must be used indefinitely to prevent regrowth [61, 62]. In two clinical trials of combined eflornithine and laser therapy, a more rapid response was observed when compared with laser treatments alone [63, 64].

73 TREATMENTS NOT ROUTINELY RECOMMENDED Metformin — Insulin-lowering drugs, which have been used for a number of indications, do not appear to be effective for hirsutism. Metformin is used primarily for blood glucose control in patients with type 2 diabetes but has also been used for some indications in women with polycystic ovary syndrome (PCOS). However, it is no longer suggested for treating hirsutism [10]. We agree with the 2018 Endocrine Society guidelines and suggest against its routine use for hirsutism as metformin has minimal or no benefit [9 -11, 65]. Gn. RH agonist or antagonist therapy — We suggest against the routine use of gonadotropin-releasing hormone (Gn. RH) analog therapy (agonist or antagonist) for hirsutism. However, Gn. RH agonists are sometimes considered for women with severe hyperandrogenemia, menstrual disturbances, and signs of virilization in addition to hirsutism (for example, ovarian hyperthecosis). It should only rarely be used in women with PCOS [66 -70]. (See "Ovarian hyperthecosis", section on 'Treatment' and "Evaluation and management of postmenopausal hyperandrogenism", section on 'Ovarian hyperthecosis'. ) The role of "add-back" estrogen and progestin therapy to prevent bone loss and hot flashes is discussed separately. (See "Endometriosis: Long-term treatment with gonadotropin-releasing hormone agonists", section on 'Gn. RH with add-back therapy'. )

74 Other ●Multidrug therapy – European investigators have studied the following combinations for the treatment of hirsutism: • A combined estrogen-progestin oral contraceptive (COC) plus metformin and low-dose flutamide [49 -51] • Metformin plus flutamide [52, 71] • A COC plus metformin in adolescents and women with hirsutism and oligomenorrhea [72] The combinations in these trials were effective in the treatment of hirsutism. In addition, flutamide reduced the accumulation of visceral fat, and metformin improved insulin sensitivity. In the absence of additional clinical trial data for these regimens, we suggest against their routine use. In addition, as noted above, we do not recommend treating hirsutism with flutamide, because of potential hepatotoxicity and because there are other effective and safer antiandrogens. (See 'Choice of antiandrogen' above. ) ●Topical antiandrogens – Limited data suggest that topical canrenone (the active metabolite of spironolactone) and finasteride provide little or no benefit for hirsutism [73, 74]. Thus, we suggest against their use. One topical antiandrogen, clascoterone, has been approved for the treatment of acne, but it has not yet been studied for hirsutism [75].

75 SPECIAL POPULATIONS Women with NCCAH — Given their relative lack of benefit for hirsutism and their potential side effects, we suggest against glucocorticoid therapy for the routine treatment of hirsutism. Like other women with hirsutism, we suggest combined estrogen-progestin oral contraceptives (COCs) as first-line therapy. An exception is in women with nonclassic congenital adrenal hyperplasia due to 21 -hydroxylase deficiency (NCCAH) who do not respond to or cannot tolerate COC and antiandrogen therapies [10]. Glucocorticoids are also indicated for ovulation induction in women with NCCAH. (See "Diagnosis and treatment of nonclassic (late-onset) congenital adrenal hyperplasia due to 21 -hydroxylase deficiency", section on 'Women'. ) Exogenous glucocorticoids are used long term to manage hirsutism and maintain ovulatory cycles in women with classic CYP 21 A 2 deficiency but have also been used to treat hirsutism in women with the nonclassic form of CYP 21 A 2 deficiency (NCCAH). In two trials of glucocorticoid therapy in women with NCCAH (one randomized, one nonrandomized), glucocorticoids were more effective than COCs or antiandrogens for suppressing serum adrenal androgen concentrations (dehydroepiandrosterone [DHEA] and dehydroepiandrosterone sulfate [DHEAS]) but less effective for decreasing hirsutism scores [76, 77]. (See "Diagnosis and treatment of nonclassic (late-onset) congenital adrenal hyperplasia due to 21 -hydroxylase deficiency". )

76 Obesity — For hirsute women with obesity, including those with polycystic ovary syndrome (PCOS), we recommend lifestyle changes in addition to starting pharmacologic therapy. We avoid COCs in women who are obese and over age 40 years because of an excess risk of venous thromboembolism (VTE) and other cardiovascular complications. A list of relative and absolute contraindications to COCs is found separately. (See "Combined estrogen-progestin contraception: Side effects and health concerns", section on 'Eligibility criteria (WHO and CDC)'. ) Coexisting depression — Depression and anxiety symptoms are common in women with hirsutism, but the optimal management of the mood disorder is unclear. It is important to monitor the patient's mood symptoms during treatment. If they do not improve (or worsen), the patient should be referred for further evaluation and treatment. Some studies suggest that treatment of the hirsutism can improve quality of life and reduce depression and anxiety symptoms [1, 78]. (See "Treatment of polycystic ovary syndrome in adults", section on 'Depression/anxiety'. ) Postmenopausal women — In postmenopausal women with new hirsutism that is severe or rapidly progressive, the possibility of an androgen-secreting tumor or ovarian hyperthecosis must be excluded before initiating treatment. If there is no evidence of a tumor, treatment options include antiandrogens or direct hair removal. In menopausal women with ovarian stromal hyperthecosis and severe hirsutism, bilateral oophorectomy may be considered. (See "Evaluation and management of postmenopausal hyperandrogenism" and "Ovarian hyperthecosis". )

77 SUMMARY AND RECOMMENDATIONS ●Hirsutism, defined as excessive male-pattern hair growth in a woman, is common (affects between 5 and 10 percent of women of reproductive age) and is associated with significant emotional distress and depression. It is usually an indication of an underlying endocrine disorder (most commonly polycystic ovary syndrome [PCOS]) (table 1). (See 'Initial visit' above. ) ●We consider any woman with patient-important hirsutism to be a candidate for treatment (pharmacologic, direct hair removal, or both). (See 'Baseline Ferriman-Gallwey score' above and 'Assess degree of emotional distress' above and 'Initial visit' above. ) ●For the majority of women with hirsutism who choose pharmacologic therapy, we suggest combined estrogen-progestin oral contraceptives (COCs) as initial therapy (Grade 2 C). COCs and antiandrogens have similar efficacy, but we prefer COCs because antiandrogen monotherapy has potential adverse effects on a developing male fetus. (See 'Combined estrogen-progestin oral contraceptives' above and 'Monotherapy' above. ) Although we do not suggest one particular COC over another, it is reasonable to avoid preparations with the most androgenic progestin, levonorgestrel, which has less desirable metabolic effects. (See 'Combined estrogen-progestin oral contraceptives' above. ) ●For women who have started pharmacologic therapy, it is important to wait at least six months before making any changes in dose, adding a medication, or switching to a new medication. This is because the growth phase of a hair follicle is approximately six months; a significant reduction in hair growth may not occur before then. (See 'Six-month assessment' above. )

78 ●For women with a suboptimal cosmetic result after six months of COC monotherapy, we suggest adding an antiandrogen (Grade 2 C). We suggest spironolactone as the antiandrogen of choice (Grade 2 C). (See 'Antiandrogens' above. ) ●In occasional women with either severe hirsutism or hirsutism causing severe emotional distress, we start with combination therapy (a COC and an antiandrogen). However, we do not use combination therapy as a standard approach. (See 'Combined with COCs' above. ) ●We suggest against the use of flutamide for hirsutism because of its potential hepatotoxicity (Grade 2 C). (See 'Choice of antiandrogen' above. ) ●We suggest not using insulin-lowering drugs for hirsutism (Grade 2 B). (See 'Treatments not routinely recommended' above. ) ●For women with nonclassic congenital adrenal hyperplasia due to 21 -hydroxylase deficiency (NCCAH), we suggest COCs rather than glucocorticoids as initial therapy for hirsutism. (Grade 2 C). (See 'Women with NCCAH' above. ) ●For women with hyperandrogenism undergoing direct hair removal methods, we suggest that pharmacologic therapy be added or continued to minimize hair regrowth (Grade 2 C). (See 'Role of direct hair removal methods' above. ) Use of Up. To. Date is subject to the Subscription and License Agreement

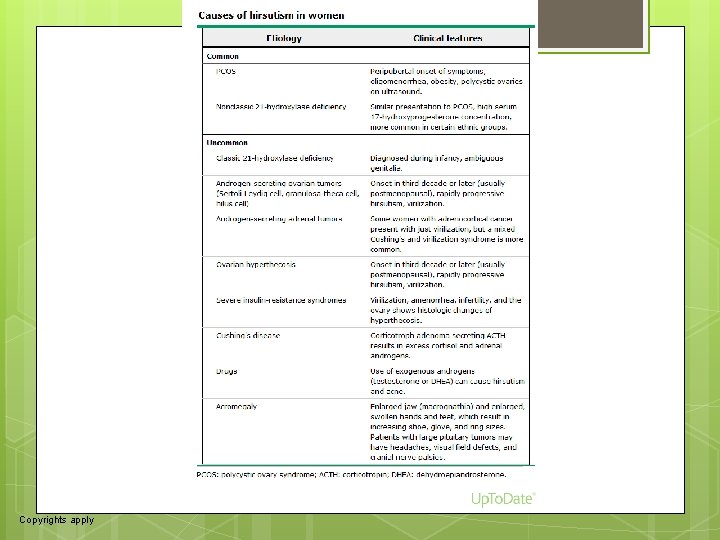
79 Etiology Clinical features Common PCOS Peripubertal onset of symptoms, oligomenorrhea, obesity, polycystic ovaries on ultrasound. Nonclassic 21 -hydroxylase deficiency Similar presentation to PCOS, high serum 17 -hydroxyprogesterone concentration, more common in certain ethnic groups. Uncommon Classic 21 -hydroxylase deficiency Diagnosed during infancy, ambiguous genitalia. Androgen-secreting ovarian tumors (Sertoli-Leydig cell, granulosa-theca cell, hilus cell) Onset in third decade or later (usually postmenopausal), rapidly progressive hirsutism, virilization. Androgen-secreting adrenal tumors Some women with adrenocortical cancer present with just virilization, but a mixed Cushing's and virilization syndrome is more common. Ovarian hyperthecosis Onset in third decade or later (usually postmenopausal), rapidly progressive hirsutism, virilization. Severe insulin-resistance syndromes Virilization, amenorrhea, infertility, and the ovary shows histologic changes of hyperthecosis. Cushing's disease Corticotroph adenoma secreting ACTH results in excess cortisol and adrenal androgens. Drugs Use of exogenous androgens (testosterone or DHEA) can cause hirsutism and acne. Acromegaly Enlarged jaw (macrognathia) and enlarged, swollen hands and feet, which result in increasing shoe, glove, and ring sizes. Patients with large pituitary tumors may have headaches, visual field defects, and cranial nerve palsies.

Thank you

81 Thank you
- Slides: 81