HIGHRESOLUTION LASER SPECTROSCOPY OF S 1 S 0

![Fluorescence excitation spectrum of naphthalene [1] D. L. Joo et al. , J. Mol. Fluorescence excitation spectrum of naphthalene [1] D. L. Joo et al. , J. Mol.](https://slidetodoc.com/presentation_image_h2/329b56f95faa2e97321bc66e7dad6bbe/image-4.jpg)




- Slides: 26

HIGH-RESOLUTION LASER SPECTROSCOPY OF S 1 -S 0 TRANSITION OF NAPHTHALENE: MEASUREMENT OF VIBRATIONALLY EXCITED STATE (Kobe Univ. ) Takumi Nakano, Ryo Yamamoto, Shunji Kasahara

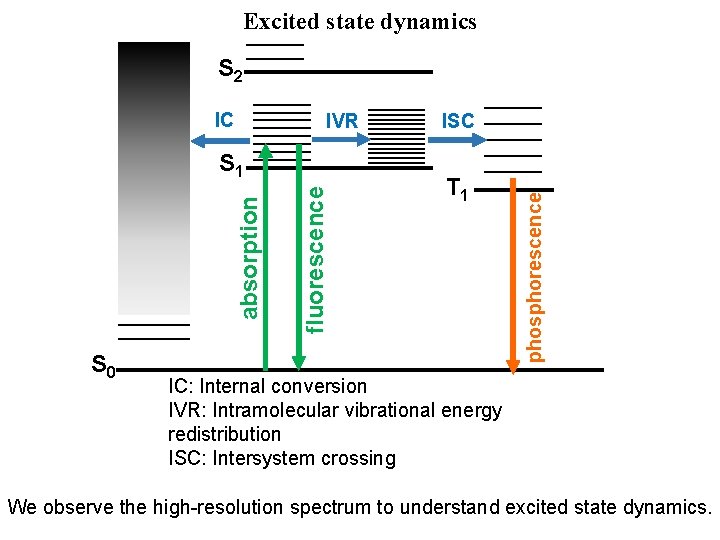
Excited state dynamics S 2 IVR S 0 fluorescence absorption S 1 ISC T 1 phosphorescence IC IC: Internal conversion IVR: Intramolecular vibrational energy redistribution ISC: Intersystem crossing We observe the high-resolution spectrum to understand excited state dynamics.

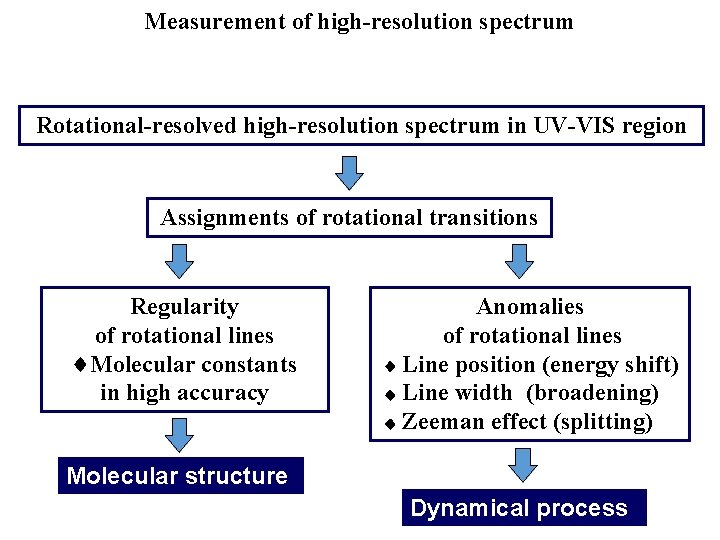
Measurement of high-resolution spectrum Rotational-resolved high-resolution spectrum in UV-VIS region Assignments of rotational transitions Regularity of rotational lines Molecular constants in high accuracy Anomalies of rotational lines Line position (energy shift) Line width (broadening) Zeeman effect (splitting) Molecular structure Dynamical process
![Fluorescence excitation spectrum of naphthalene 1 D L Joo et al J Mol Fluorescence excitation spectrum of naphthalene [1] D. L. Joo et al. , J. Mol.](https://slidetodoc.com/presentation_image_h2/329b56f95faa2e97321bc66e7dad6bbe/image-4.jpg)
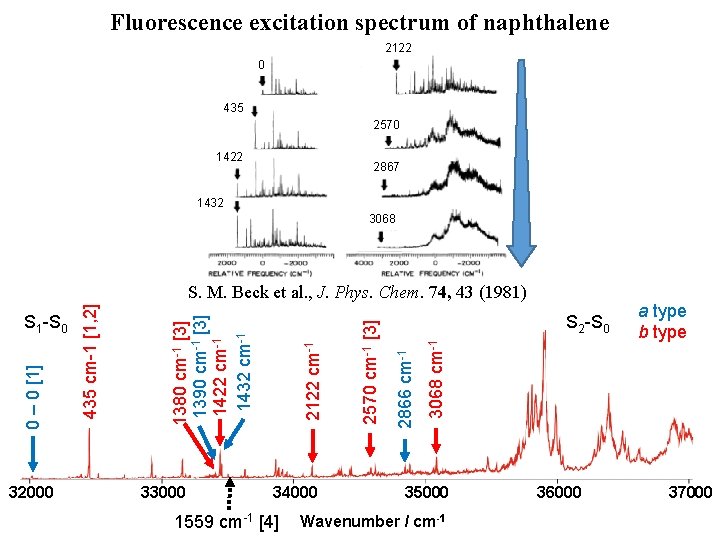
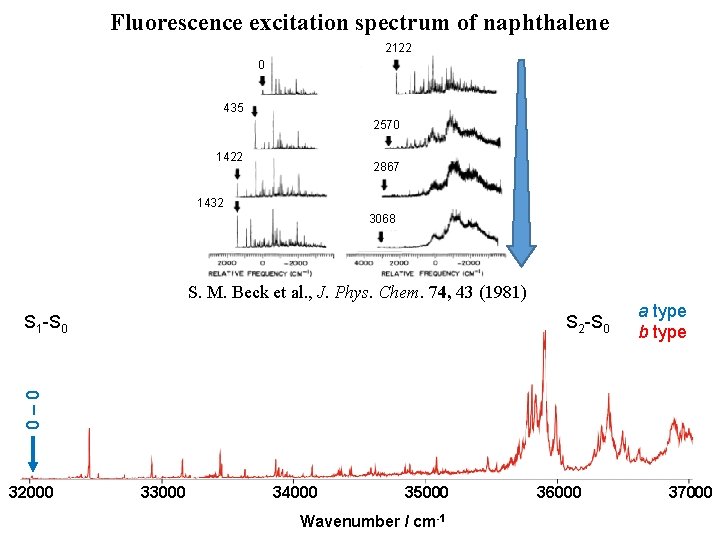
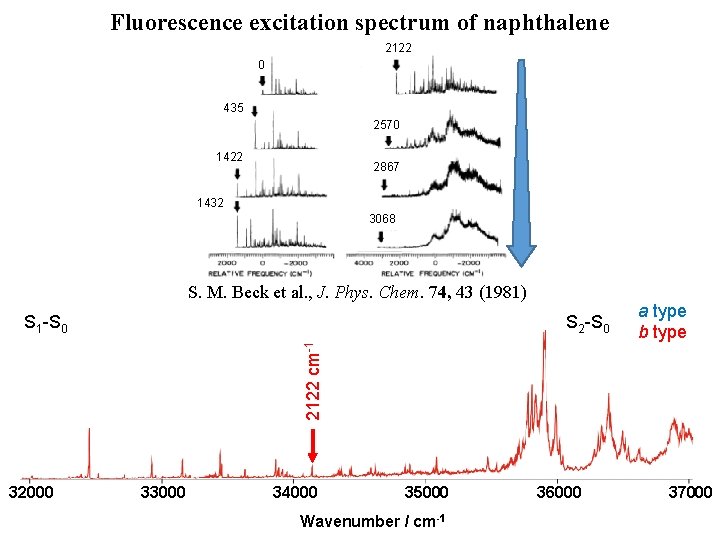
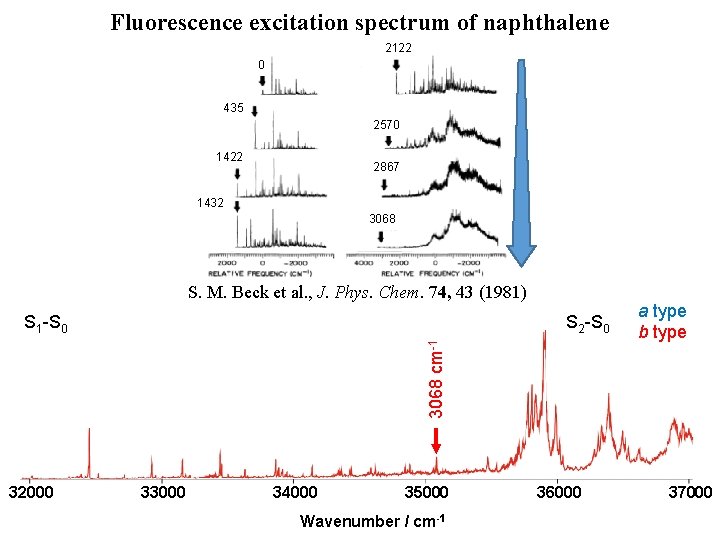
Fluorescence excitation spectrum of naphthalene [1] D. L. Joo et al. , J. Mol. Spectrosc. 215, 155 (2011) [2] M. H. Kabir et al. , J. Chem. Phys. 119, 3691 (2003) [3] K. Yoshida et al. , J. Chem. Phys. 130, 194304 (2009) 32000 33000 S 2 -S 0 2570 cm-1 [3] 1380 cm-1 [3] 1390 cm-1 [3] 0 – 0 [1] S 1 -S 0 435 cm-1 [1, 2] [4] M. Okubo et al. , J. Chem. Phys. 116, 9293 (2002) H. Kato et al. , Bull. Chem. Soc. Jpn. 80, 456 (2007) 34000 1559 cm-1 [4] 35000 Wavenumber / cm-1 36000 a type b type 37000

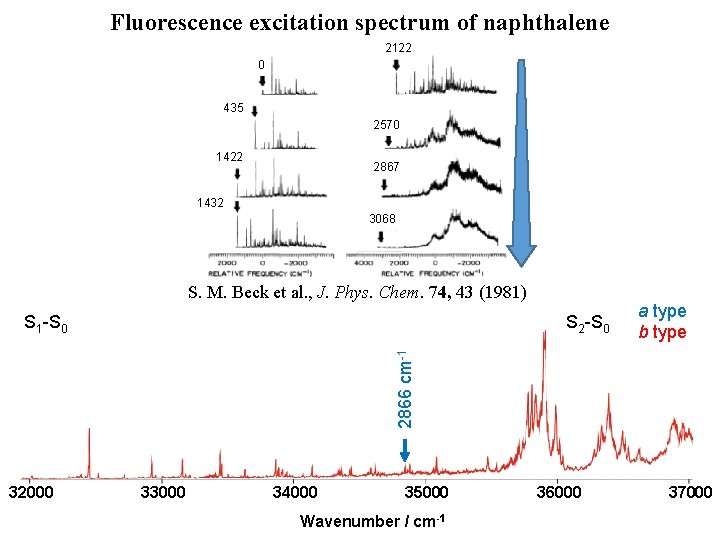
Fluorescence excitation spectrum of naphthalene 2122 0 435 2570 1422 2867 1432 3068 32000 33000 34000 1559 cm-1 [4] 3068 cm-1 S 2 -S 0 2866 cm-1 2570 cm-1 [3] 2122 cm-1 1380 cm-1 [3] 1390 cm-1 [3] 1422 cm-1 1432 cm-1 0 – 0 [1] S 1 -S 0 435 cm-1 [1, 2] S. M. Beck et al. , J. Phys. Chem. 74, 43 (1981) 35000 Wavenumber / cm-1 36000 a type b type 37000

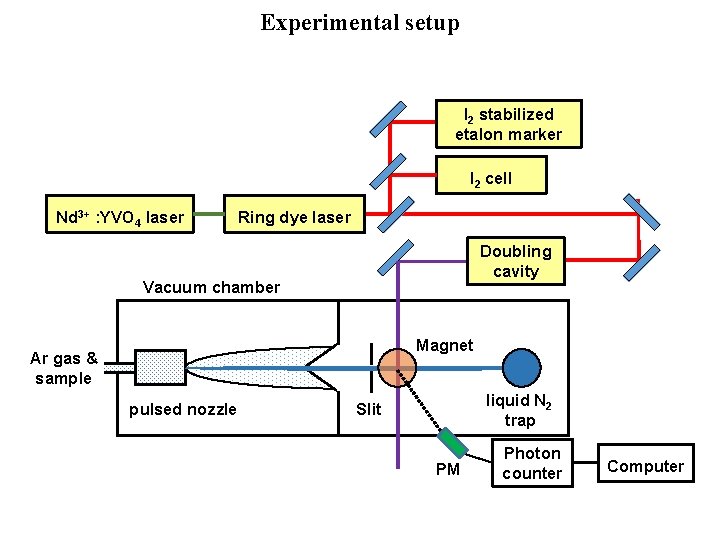
Experimental setup I 2 stabilized etalon marker I 2 cell Nd 3+ : YVO 4 laser Ring dye laser Doubling cavity Vacuum chamber Magnet Ar gas & sample pulsed nozzle liquid N 2 trap Slit PM Photon counter Computer

Fluorescence excitation spectrum of naphthalene 2122 0 435 2570 1422 2867 1432 3068 S. M. Beck et al. , J. Phys. Chem. 74, 43 (1981) S 2 -S 0 a type b type 0– 0 S 1 -S 0 32000 33000 34000 35000 Wavenumber / cm-1 36000 37000

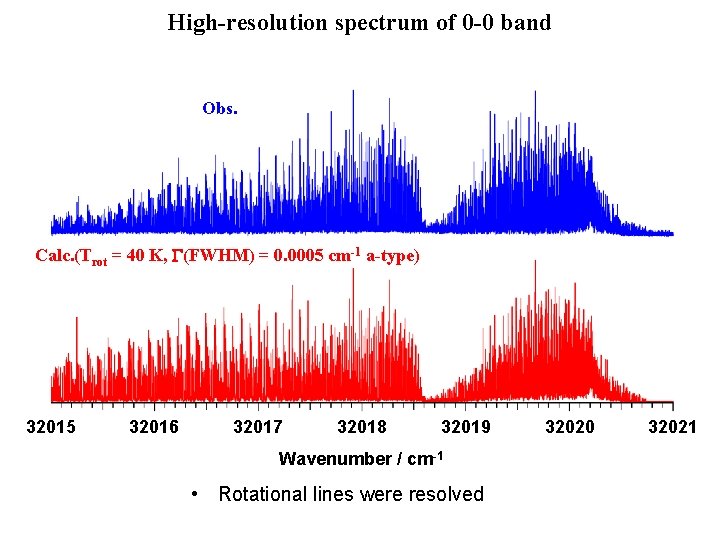
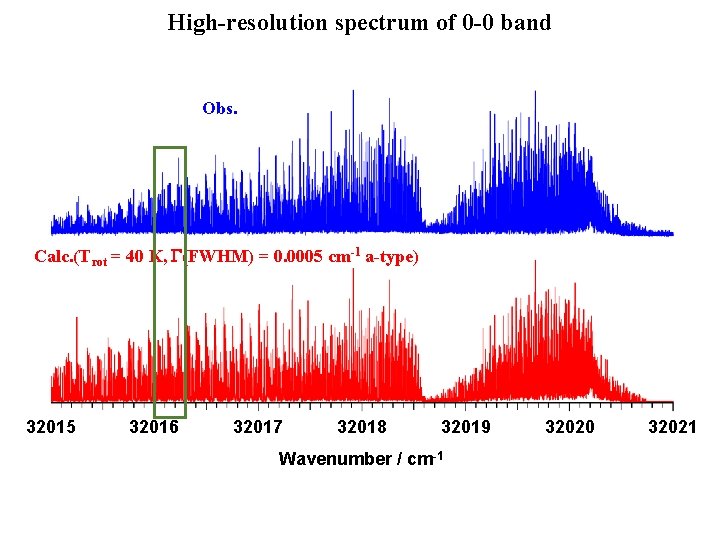
High-resolution spectrum of 0 -0 band Obs. Calc. (Trot = 40 K, G(FWHM) = 0. 0005 cm-1 a-type) 32015 32016 32017 32018 32019 Wavenumber / cm-1 • Rotational lines were resolved 32020 32021

Molecular constants of 0 -0 band S 0 (v = 0) 0 -0 band (previous work[1]) 0 -0 band A / cm-1 0. 10405207(16) 0. 101470325(51) 0. 101462295(60) B 0. 041126892(27) 0. 040498566(74) 0. 040498715(31) C 0. 0294838072(80) 0. 028955573(79) 0. 028955165(22) ΔK 1. 87(32)× 10 -9 ― 1. 82(11)× 10 -9 ΔJK 1. 18(22)× 10 -9 ― 1. 751(55)× 10 -9 ΔJ 5. 80(21)× 10 -10 ― 5. 704(80)× 10 -9 d. K 1. 37(17)× 10 -9 ― 3. 87(11)× 10 -9 d. J 1. 59(10)× 10 -10 ― 5. 696(53)× 10 -10 T 0 ― 32018. 5967(3) 32018. 5870(2) band type ― A A Assigned line ― 1043 3083 Std. Dev. ― 0. 00020 0. 00016 [1] D. L. Joo et al. , J. Mol. Spectrosc. 215, 155 (2011)

High-resolution spectrum of 0 -0 band Obs. Calc. (Trot = 40 K, G(FWHM) = 0. 0005 cm-1 a-type) 32015 32016 32017 32018 32019 Wavenumber / cm-1 32020 32021

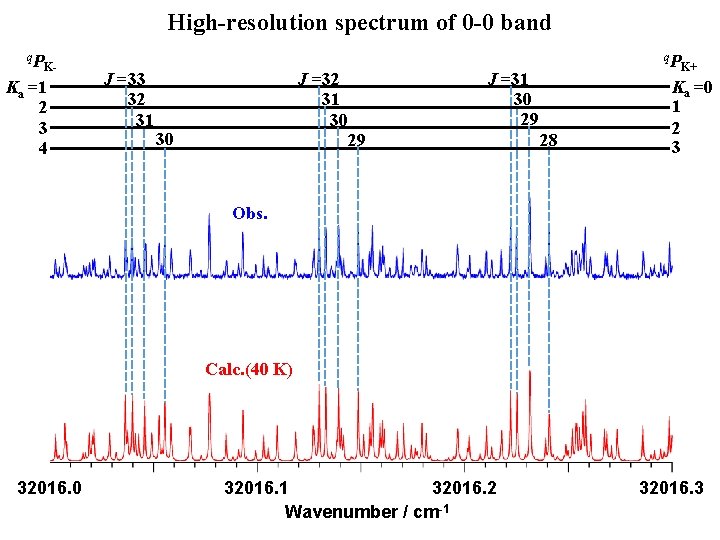
High-resolution spectrum of 0 -0 band q. P K- Ka =1 2 3 4 J =33 32 31 J =32 31 30 30 q. P J =31 30 29 29 28 K+ Ka =0 1 2 3 Obs. Calc. (40 K) 32016. 0 32016. 1 32016. 2 Wavenumber / cm-1 32016. 3

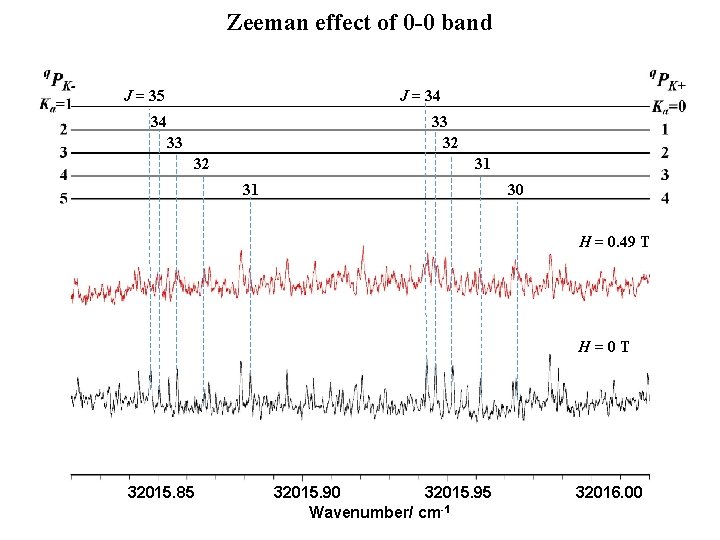
Zeeman effect of 0 -0 band J = 35 J = 34 34 33 32 31 31 30 H = 0. 49 T H=0 T 32015. 85 32015. 90 32015. 95 Wavenumber/ cm-1 32016. 00

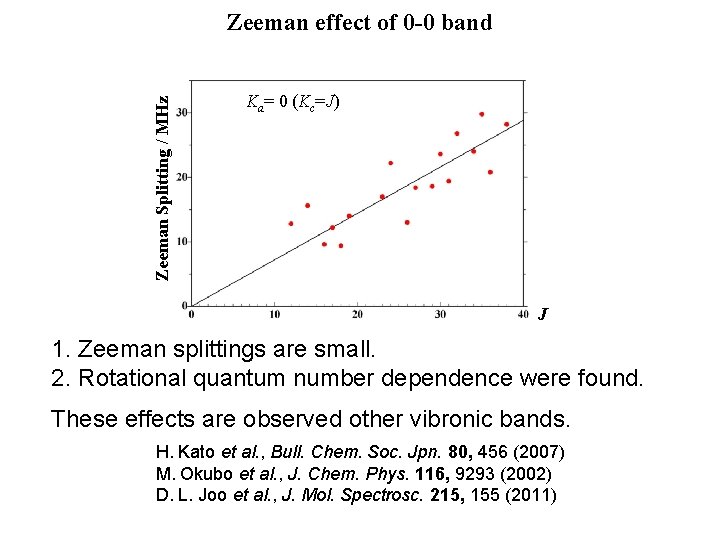
Zeeman Splitting / MHz Zeeman effect of 0 -0 band Ka= 0 (Kc=J) J 1. Zeeman splittings are small. 2. Rotational quantum number dependence were found. These effects are observed other vibronic bands. H. Kato et al. , Bull. Chem. Soc. Jpn. 80, 456 (2007) M. Okubo et al. , J. Chem. Phys. 116, 9293 (2002) D. L. Joo et al. , J. Mol. Spectrosc. 215, 155 (2011)

Fluorescence excitation spectrum of naphthalene 2122 0 435 2570 1422 2867 1432 3068 S. M. Beck et al. , J. Phys. Chem. 74, 43 (1981) S 2 -S 0 2122 cm-1 S 1 -S 0 32000 33000 34000 35000 Wavenumber / cm-1 36000 a type b type 37000

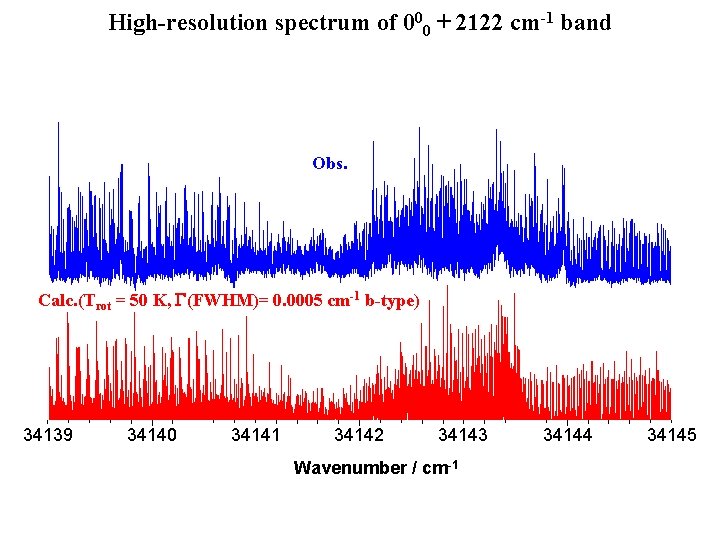
High-resolution spectrum of 000 + 2122 cm-1 band Obs. Calc. (Trot = 50 K, G(FWHM)= 0. 0005 cm-1 b-type) 34139 34140 34141 34142 34143 Wavenumber / cm-1 34144 34145

Molecular constants of vibronic bands S 0 (v = 0) 0 -0 band 000 + 2122 cm-1 A / cm-1 0. 10405207(16) 0. 101462295(60) 0. 10141594(47) B 0. 041126892(27) 0. 040498715(31) 0. 04047379(48) C 0. 0294838072(80) 0. 028955165(22) 0. 02894462(84) ΔK 1. 87(32)× 10 -9 1. 82(11)× 10 -9 ― ΔJK 1. 18(22)× 10 -9 1. 751(55)× 10 -9 ― ΔJ 5. 80(21)× 10 -10 5. 704(80)× 10 -9 ― d. K 1. 37(17)× 10 -9 3. 87(11)× 10 -9 ― d. J 1. 59(10)× 10 -10 5. 696(53)× 10 -10 ― T 0 ― 32018. 5870(2) 34141. 9434(2) band type ― A B Assigned line ― 3083 1232 Std. Dev. ― 0. 00015 0. 0008

Eob - Ecal Energy shift of 000 + 2122 cm-1 band

Eob - Ecal Energy shift of 000 + 2122 cm-1 band

Fluorescence excitation spectrum of naphthalene 2122 0 435 2570 1422 2867 1432 3068 S. M. Beck et al. , J. Phys. Chem. 74, 43 (1981) S 2 -S 0 a type b type 2866 cm-1 S 1 -S 0 32000 33000 34000 35000 Wavenumber / cm-1 36000 37000

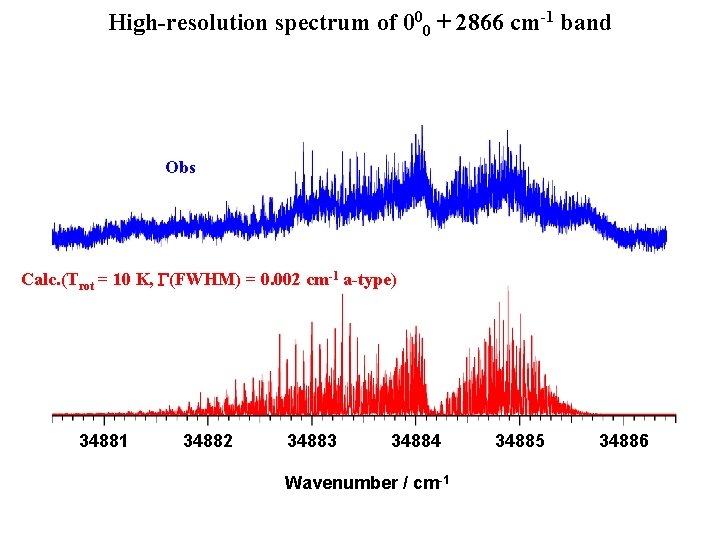
High-resolution spectrum of 000 + 2866 cm-1 band Obs Calc. (Trot = 10 K, G(FWHM) = 0. 002 cm-1 a-type) 34881 34882 34883 34884 Wavenumber / cm-1 34885 34886

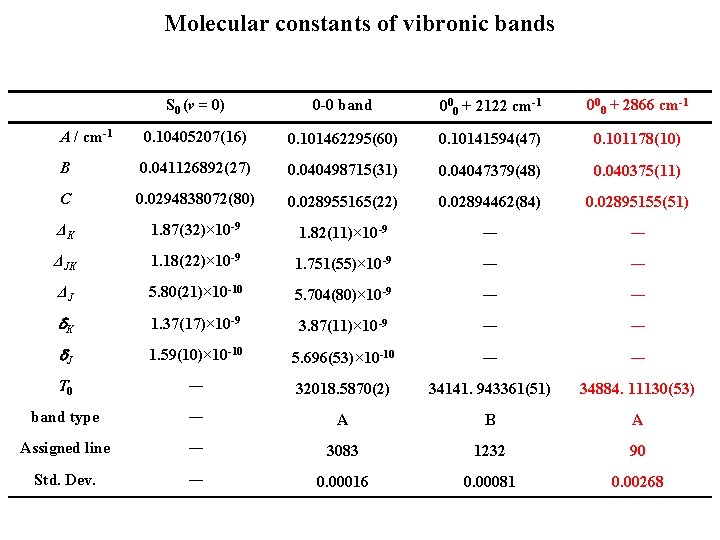
Molecular constants of vibronic bands S 0 (v = 0) 0 -0 band 000 + 2122 cm-1 000 + 2866 cm-1 A / cm-1 0. 10405207(16) 0. 101462295(60) 0. 10141594(47) 0. 101178(10) B 0. 041126892(27) 0. 040498715(31) 0. 04047379(48) 0. 040375(11) C 0. 0294838072(80) 0. 028955165(22) 0. 02894462(84) 0. 02895155(51) ΔK 1. 87(32)× 10 -9 1. 82(11)× 10 -9 ― ― ΔJK 1. 18(22)× 10 -9 1. 751(55)× 10 -9 ― ― ΔJ 5. 80(21)× 10 -10 5. 704(80)× 10 -9 ― ― d. K 1. 37(17)× 10 -9 3. 87(11)× 10 -9 ― ― d. J 1. 59(10)× 10 -10 5. 696(53)× 10 -10 ― ― T 0 ― 32018. 5870(2) 34141. 943361(51) 34884. 11130(53) band type ― A B A Assigned line ― 3083 1232 90 Std. Dev. ― 0. 00016 0. 00081 0. 00268

High-resolution spectrum of 000 + 2866 cm-1 band Obs Calc. (Trot = 10 K, G(FWHM) = 0. 002 cm-1 a-type) 34881 34882 34883 34884 Wavenumber / cm-1 34885 34886

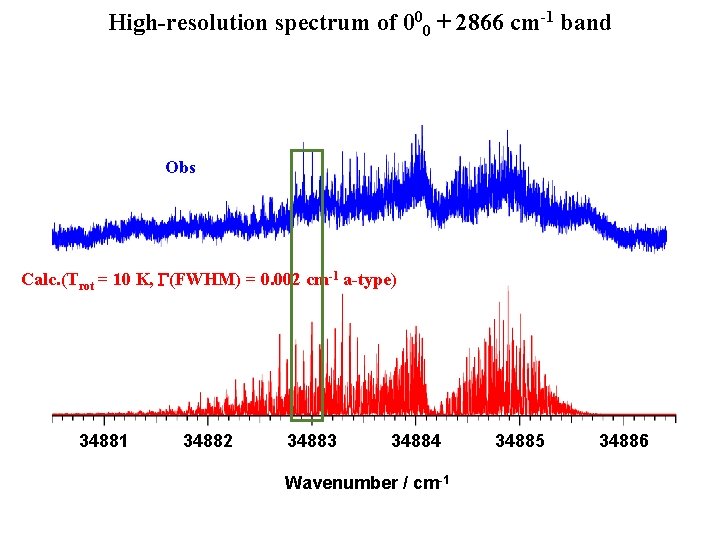
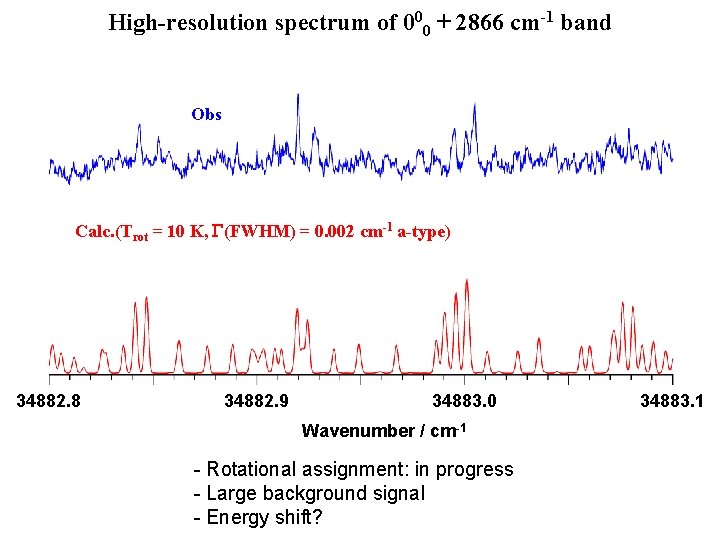
High-resolution spectrum of 000 + 2866 cm-1 band Obs Calc. (Trot = 10 K, G(FWHM) = 0. 002 cm-1 a-type) 34882. 8 34882. 9 34883. 0 Wavenumber / cm-1 - Rotational assignment: in progress - Large background signal - Energy shift? 34883. 1

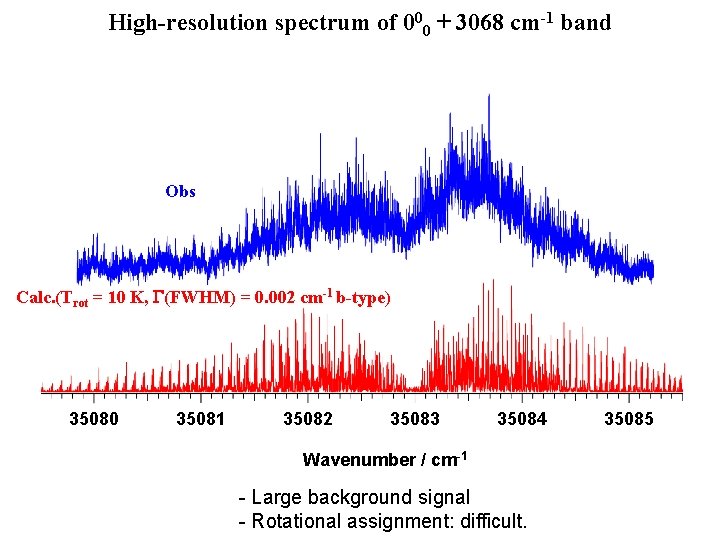
Fluorescence excitation spectrum of naphthalene 2122 0 435 2570 1422 2867 1432 3068 S. M. Beck et al. , J. Phys. Chem. 74, 43 (1981) S 2 -S 0 3068 cm-1 S 1 -S 0 32000 33000 34000 35000 Wavenumber / cm-1 36000 a type b type 37000

High-resolution spectrum of 000 + 3068 cm-1 band Obs Calc. (Trot = 10 K, G(FWHM) = 0. 002 cm-1 b-type) 35080 35081 35082 35083 35084 Wavenumber / cm-1 - Large background signal - Rotational assignment: difficult. 35085

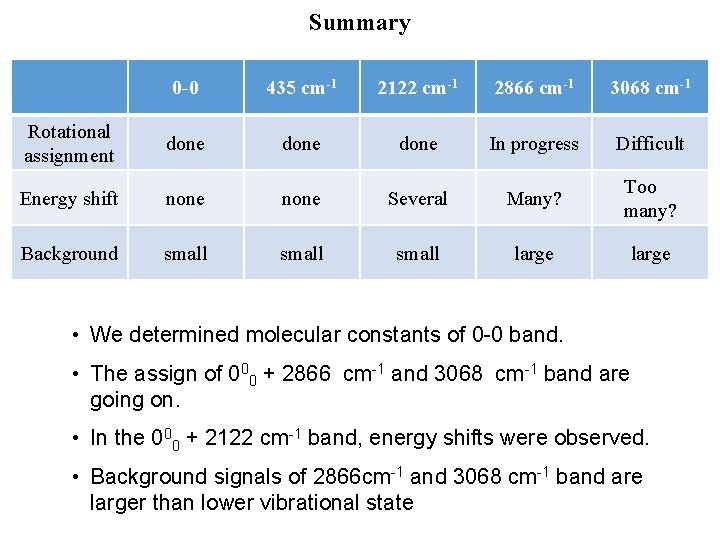
Summary 0 -0 435 cm-1 2122 cm-1 2866 cm-1 3068 cm-1 Rotational assignment done In progress Difficult Energy shift none Several Many? Too many? Background small large • We determined molecular constants of 0 -0 band. • The assign of 000 + 2866 cm-1 and 3068 cm-1 band are going on. • In the 000 + 2122 cm-1 band, energy shifts were observed. • Background signals of 2866 cm-1 and 3068 cm-1 band are larger than lower vibrational state