Highresolution IR absorption spectroscopy of Polycyclic Aromatic Hydrocarbons

High-resolution IR absorption spectroscopy of Polycyclic Aromatic Hydrocarbons (PAHs): shining light on the interstellar 3 micron emission bands… Elena Maltseva University of Amsterdam

PAHs in interstellar space? www. spitzer. caltech. edu
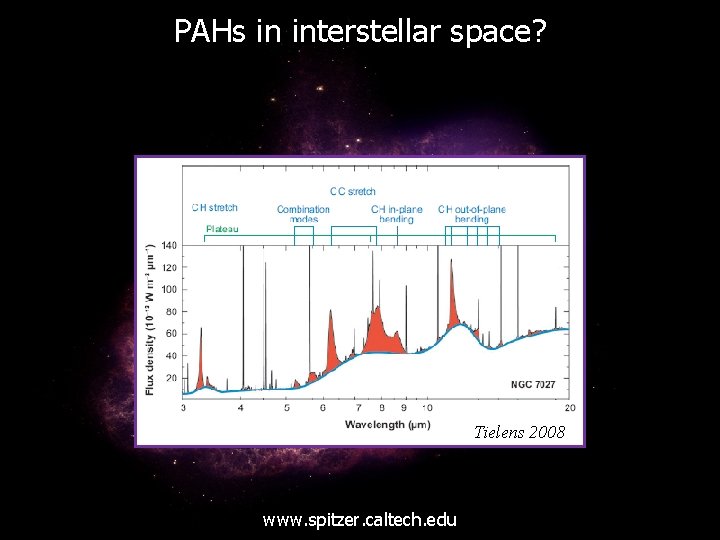
PAHs in interstellar space? Tielens 2008 www. spitzer. caltech. edu
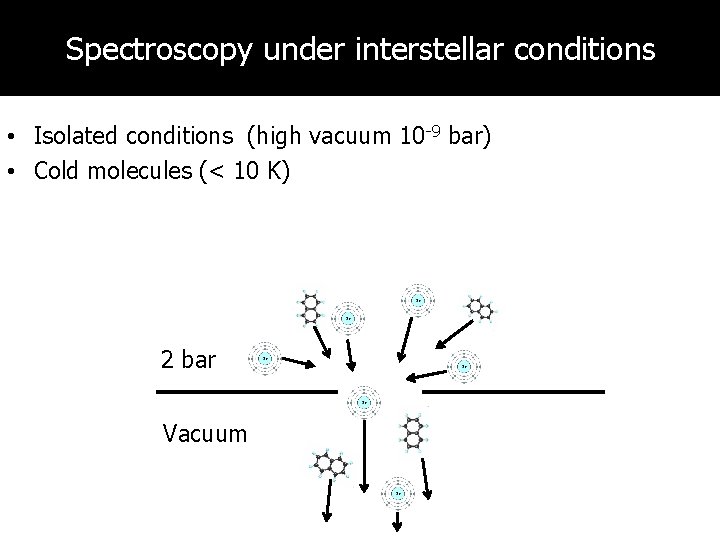
Spectroscopy under interstellar conditions • Isolated conditions (high vacuum 10 -9 bar) • Cold molecules (< 10 K) 2 bar Vacuum
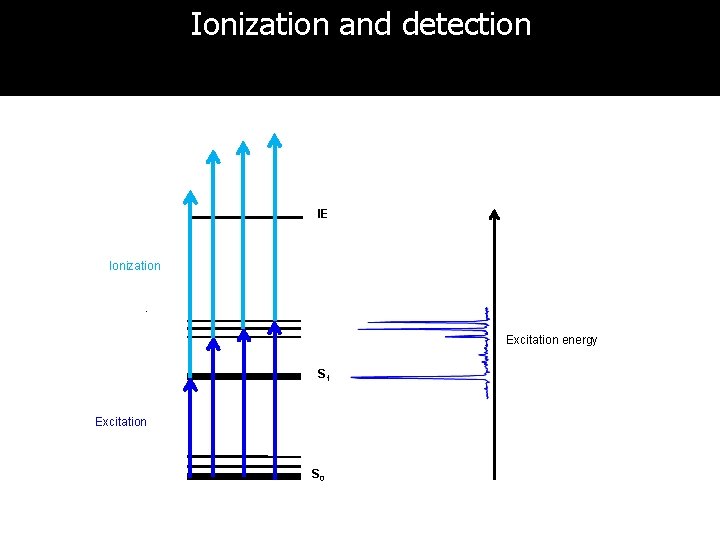
Ionization and detection IE Ionization Excitation energy S 1 Excitation S 0
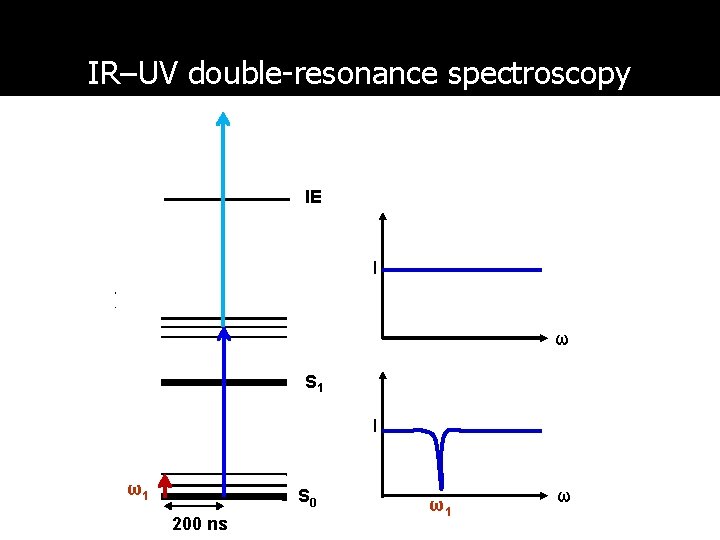
IR–UV double-resonance spectroscopy IE I ω S 1 I ω1 S 0 200 ns ω1 ω
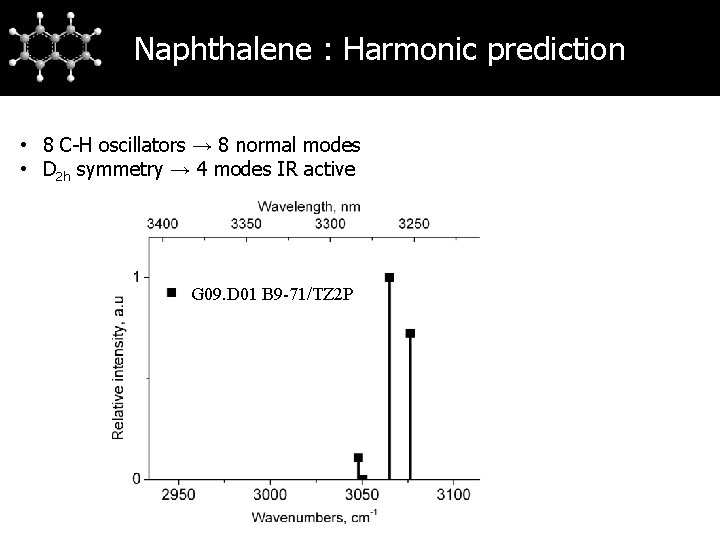
Naphthalene : Harmonic prediction • 8 C-H oscillators → 8 normal modes • D 2 h symmetry → 4 modes IR active G 09. D 01 B 9 -71/TZ 2 P
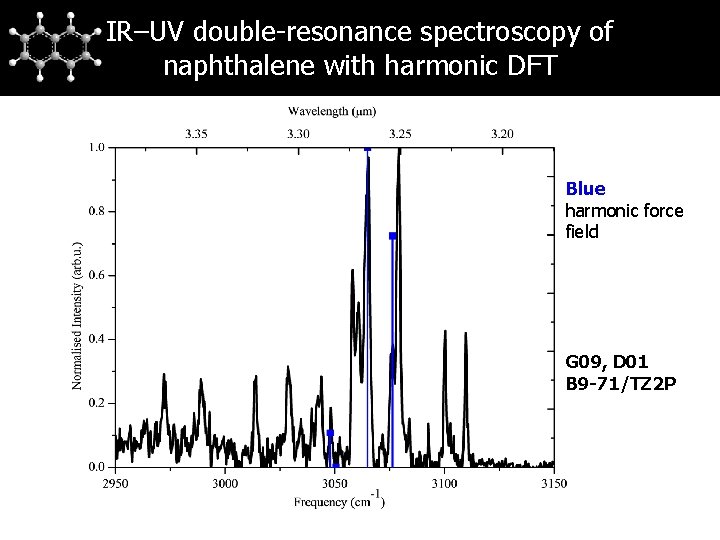
IR–UV double-resonance spectroscopy of naphthalene with harmonic DFT Blue harmonic force field G 09, D 01 B 9 -71/TZ 2 P
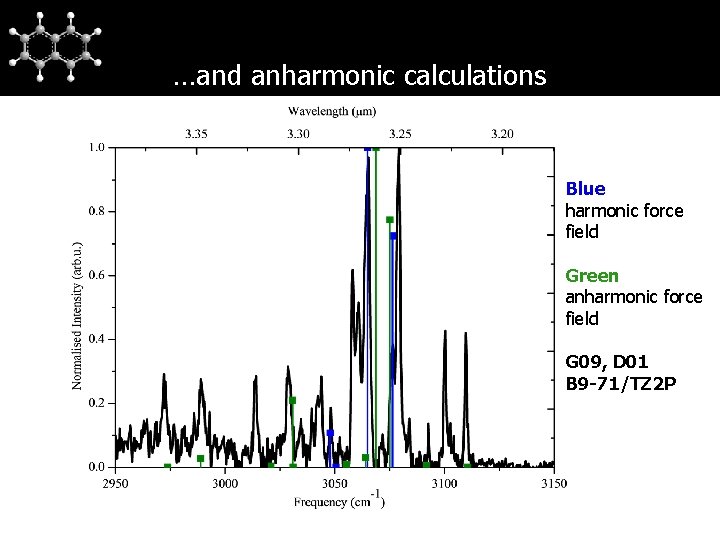
…and anharmonic calculations Blue harmonic force field Green anharmonic force field G 09, D 01 B 9 -71/TZ 2 P

Naphthalene Assignment of naphthalene Experiments on naphthalene-d 8 do not show any structure in 3 μm region → additional bands not solely due to anharmonicity → Fermi resonance
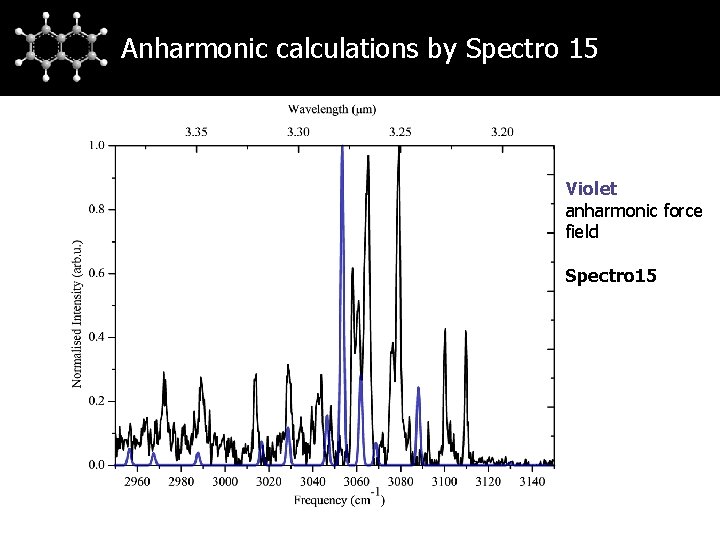
Anharmonic calculations by Spectro 15 Violet anharmonic force field Spectro 15
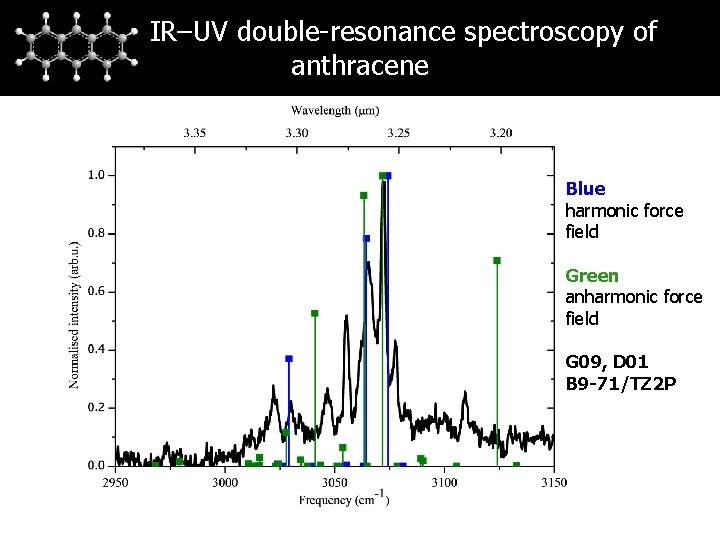
IR–UV double-resonance spectroscopy of anthracene Blue harmonic force field Green anharmonic force field G 09, D 01 B 9 -71/TZ 2 P
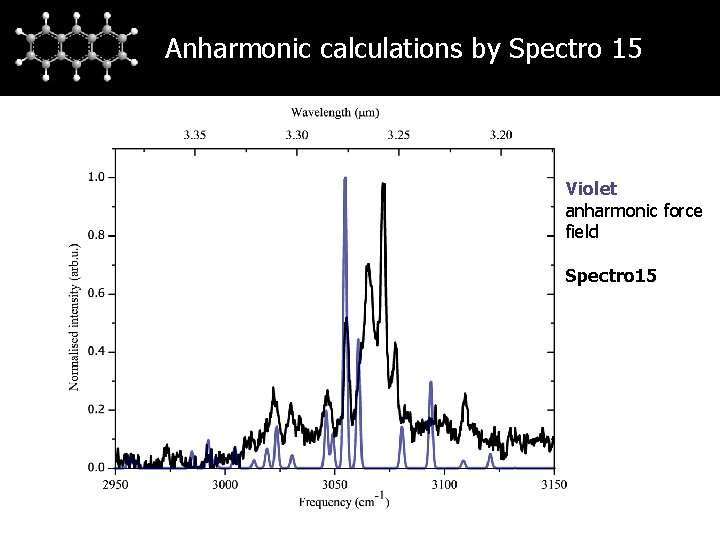
Anharmonic calculations by Spectro 15 Violet anharmonic force field Spectro 15
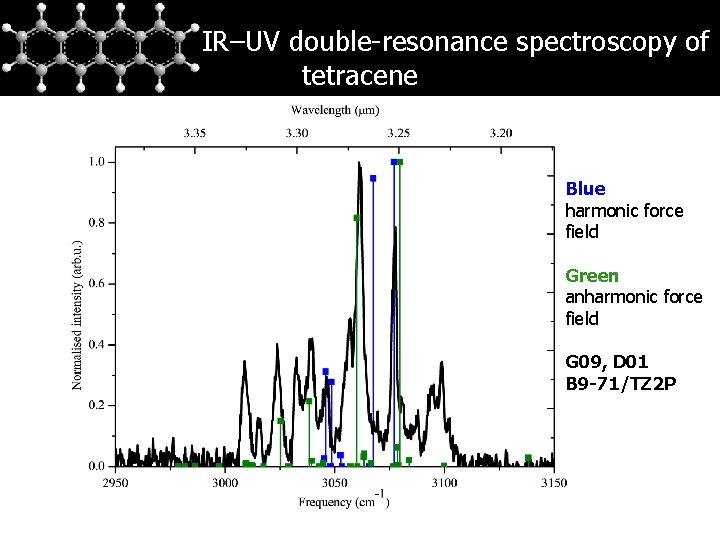
IR–UV double-resonance spectroscopy of tetracene Blue harmonic force field Green anharmonic force field G 09, D 01 B 9 -71/TZ 2 P
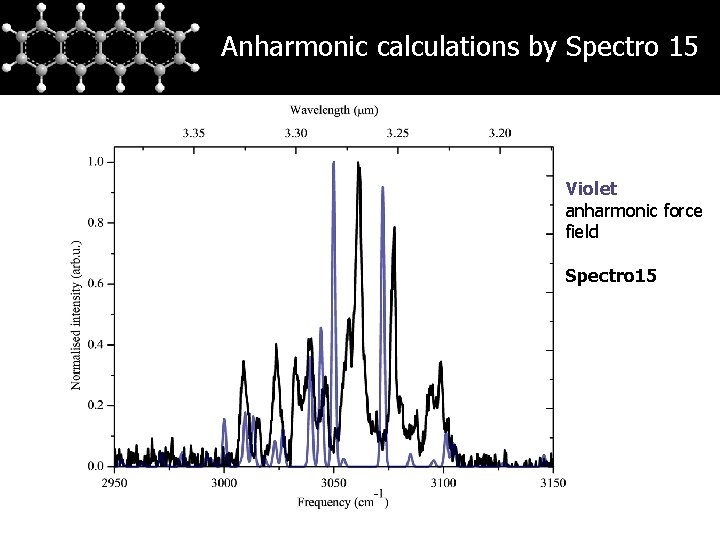
Anharmonic calculations by Spectro 15 Violet anharmonic force field Spectro 15
![IR–UV double-resonance spectroscopy of 3 - and 4 -ring systems phenanthrene Benz[a]anthracene pyrene chrysene IR–UV double-resonance spectroscopy of 3 - and 4 -ring systems phenanthrene Benz[a]anthracene pyrene chrysene](http://slidetodoc.com/presentation_image_h2/595c472ee3316f5fdde8d68cbfa3b5fc/image-16.jpg)
IR–UV double-resonance spectroscopy of 3 - and 4 -ring systems phenanthrene Benz[a]anthracene pyrene chrysene Triphenylene
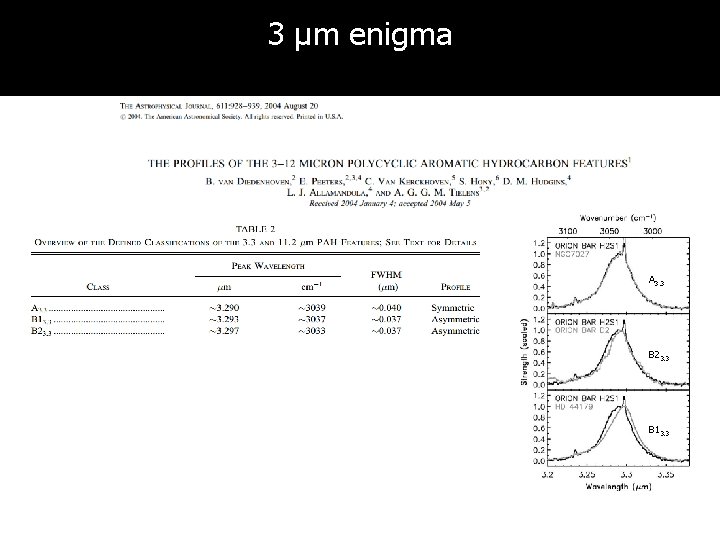
3 µm enigma A 3. 3 B 23. 3 B 13. 3
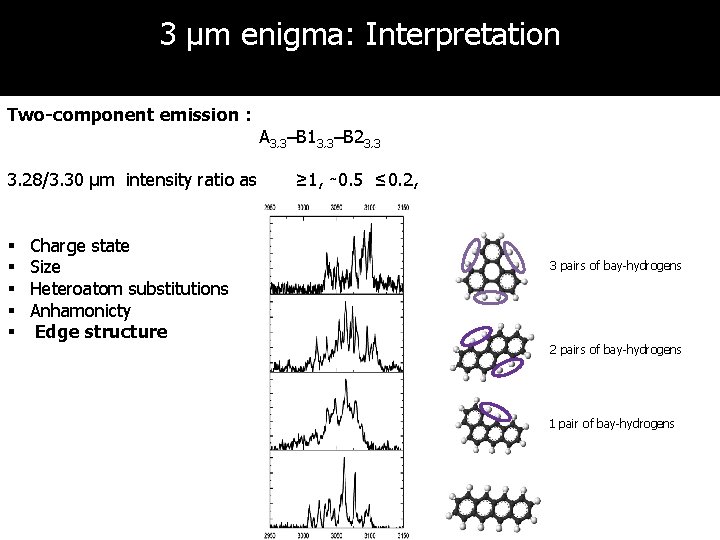
3 µm enigma: Interpretation Two-component emission : 3. 28/3. 30 µm intensity ratio as § § § Charge state Size Heteroatom substitutions Anhamonicty Edge structure A 3. 3–B 13. 3–B 23. 3 ≥ 1, ∼ 0. 5 ≤ 0. 2, 3 pairs of bay-hydrogens 2 pairs of bay-hydrogens 1 pair of bay-hydrogens
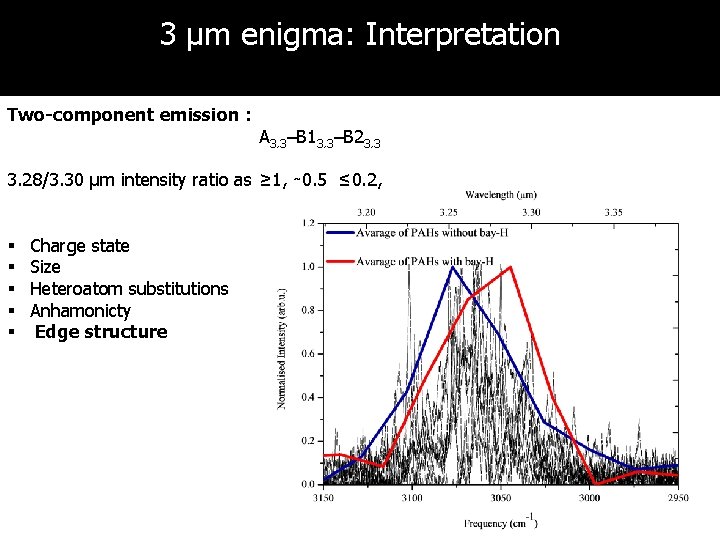
3 µm enigma: Interpretation Two-component emission : A 3. 3–B 13. 3–B 23. 3 3. 28/3. 30 µm intensity ratio as ≥ 1, ∼ 0. 5 ≤ 0. 2, § § § Charge state Size Heteroatom substitutions Anhamonicty Edge structure A 3. 3
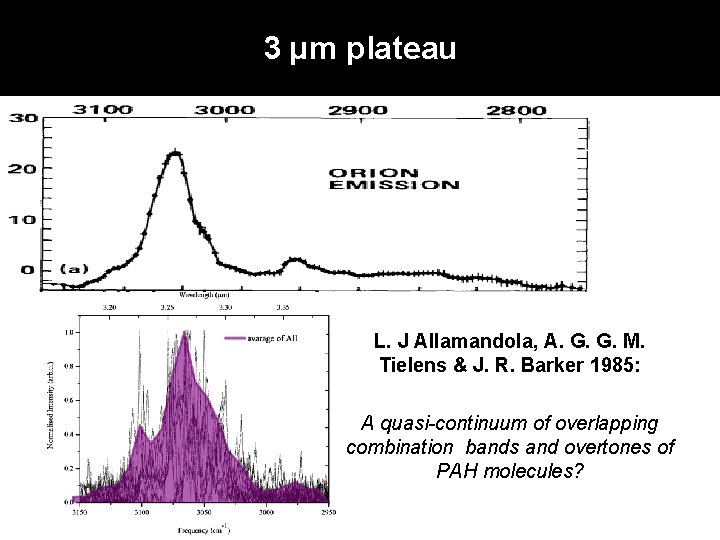
3 µm plateau L. J Allamandola, A. G. G. M. Tielens & J. R. Barker 1985: A quasi-continuum of overlapping combination bands and overtones of PAH molecules?

Discussion & conclusions 1. High-resolution IR spectra of naphthalene, anthracene, tetracene, phenanthrene, pyrene, chrysene, triphenylene, 1, 2 -benzanthracene have been obtained 2. Experiments well-suited for studies of the molecular physics of PAHs • • Isolated and cold molecules Conformation- and mass-selective method 3. Harmonic calculations do not work for 3μm region! 4. Anharmonic calculations with properly treated Fermi resonance must be used in characterization of 3 μm features in interstellar space 5. 6. Two-component emission can be explained by presence of bay-hydrogen sites 3 µm plateau can originate from overlapping of combination bands

Acknowledgment Wybren Jan Buma and Molecular Photonics group (Uv. A) Cameron Mackie, Alessandra Candian, Annemieke Petrignani, Xander Tielens (Leiden Observatory) Jos Oomens (Molecular Structure and Dynamics, FELIX Facility) Timothy Lee and Xinchuan Huang (NASA Ames Research Center) Dutch Astrochemistry Network Spinoza prize Prof. Tielens

Thank you for your attention!
Gas-phase study of PAHs • Done only for Naphthalene, Anthracene, Phenanthrene, Pyrene, Perylene • Is it only ‘the right’ absorption? • Signal/noise • Assignment
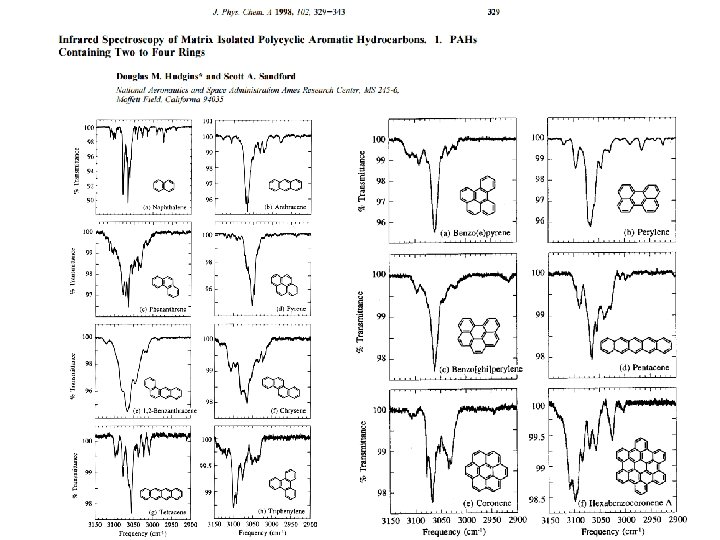
Infrared Spectroscopy of Matrix Isolated Polycyclic Aromatic Hydrocarbons
- Slides: 25