HIGH RESOLUTION LASER SPECTROSCOPY OF THE A 3







- Slides: 16

HIGH – RESOLUTION LASER SPECTROSCOPY OF THE A 3Π 1 ← X 1Σ+ SYSTEM OF ICl IN 0. 8 mm REGION N. NISHIMIYA and T. YUKIYA Tokyo Polytechnic University, Kanagawa, JAPAN. 1

Our Research l l Laser spectroscopy of electronic band system of halide molecules. Target: ICl, IBr, I 2, and Br 2 For establishing the frequency standard in the near infrared region. In this time we have measured the A-X spectra of ICl in the region of 11300 -14450 cm-1 region. p p p l ICl spectrum is weak comparing to other halogen molecules. It is easy to assign, because of its wide spectrum intervals. One spectrum at least can be found within a continuous tuning range of a titanium sapphire laser in the 0. 8 mm region. The constants have determined by taking into consideration isotopic reduced mass ratio. 2

References (1) J. A. Coxon, et. al. , ``The A 3 P 1 u X 1 S+ Absorption Spectrum of ICl. '', J. Mol. Spectrosc. 79, 363 (1980) (2) J. A. Coxon and M. A. Wickramaaratchi, ``The A 3 P 1 X 1 S+ Emission Spectrum of ICl in the Near Infrared. '', J. Mol. Spectrosc. 79, 380 (1980) (3) J. C. DBrand A. R. Hoy, ``High Vibrational Level of the X state of ICl, and the Electronic -Coriolis Coupling of the X and A states. '', J. Mol. Spectroscopy, 114, 197 (1985) (4) J. C. D. Brand, et. al. ``The A’(3 P 2) of ICl”, J. Mol. Spectrosc. , 113, 388 (1985) (5) H. G. Hedderich, et. al. , ``The High-Resolution Infrared Spectrum of Iodine Monochloride. '', J. Mol. Spectrosc. , 155, 384 (1992). (6) C. M. Western, ``Variation of the electronic wave function with internuclear separation: Highresolution spectroscopy of the A 3 P(1) state of I 35 Cl near the dissociation limit. ’’, J. Chem. Phys. , 98, 1826 (1993) (7) T. J. Slotterback, et. al. , ``Hyperfine measurements in the X and B electronic states of I 35, 37 Cl: Probing the ionic character of the chemical bond. '', J. Chem. Phys. , 101, 7221(1994) (8) T. J. Slotterback, et. al. , ``Hyperfine analysis of the mixed A 3 P 1 v=28 and X 1 S+ v=69 states of I 35 Cl”, J. Chem. Phys. , 103, 9125(1994) 3

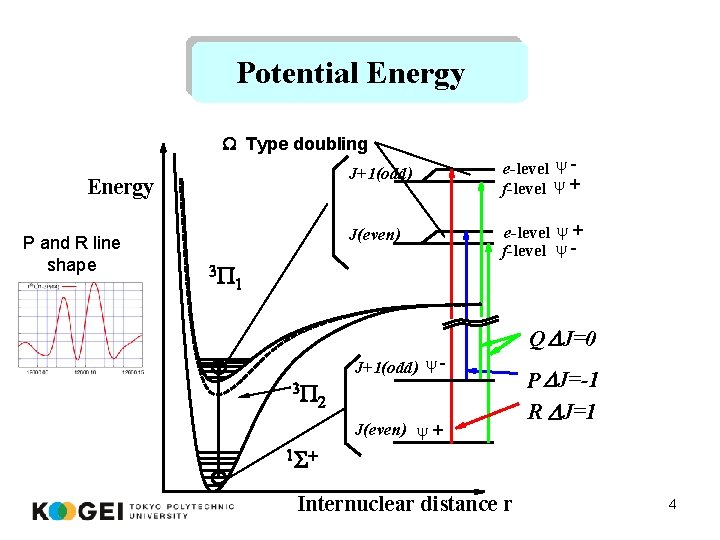
Potential Energy W Type doubling Energy P and R line shape 3 P J+1(odd) e-level y f-level y + J(even) e-level y + f-level y - 1 Q DJ=0 3 P J+1(odd) y 2 J(even) y + P DJ=-1 R DJ=1 1 S + Internuclear distance r 4

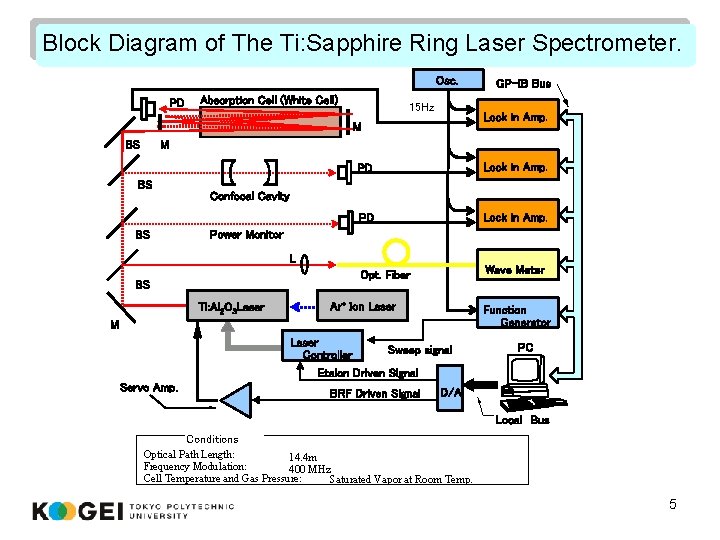
Block Diagram of The Ti: Sapphire Ring Laser Spectrometer. Osc. PD Absorption Cell (White Cell) 15 Hz Lock in Amp. M BS GP-IB Bus M BS BS PD Lock in Amp. Opt. Fiber Wave Meter Confocal Cavity Power Monitor L BS Ti: Al 2 O 3 Laser Ar+ Ion Laser Function Generator M Laser Controller Sweep signal PC Etalon Driven Signal Servo Amp. BRF Driven Signal D/A Local Bus Conditions Optical Path Length: 14. 4 m Frequency Modulation: 400 MHz Cell Temperature and Gas Pressure: Saturated Vapor at Room Temp. 5

Recorder Trace of the Absorption Lines in 12600 cm-1 Region 6

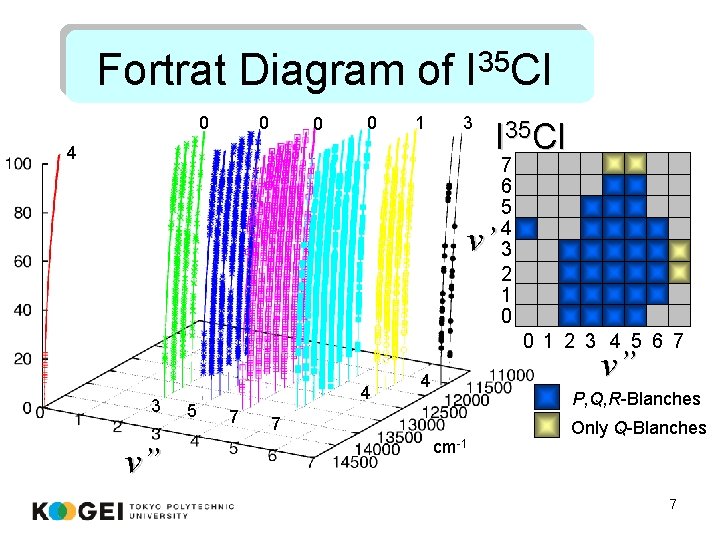
Fortrat Diagram of I 35 Cl 0 0 1 3 4 I 35 Cl v’ 7 6 5 4 3 2 1 0 0 1 2 3 4 5 6 7 3 v” 5 4 7 v” 4 P, Q, R-Blanches 7 cm-1 Only Q-Blanches 7

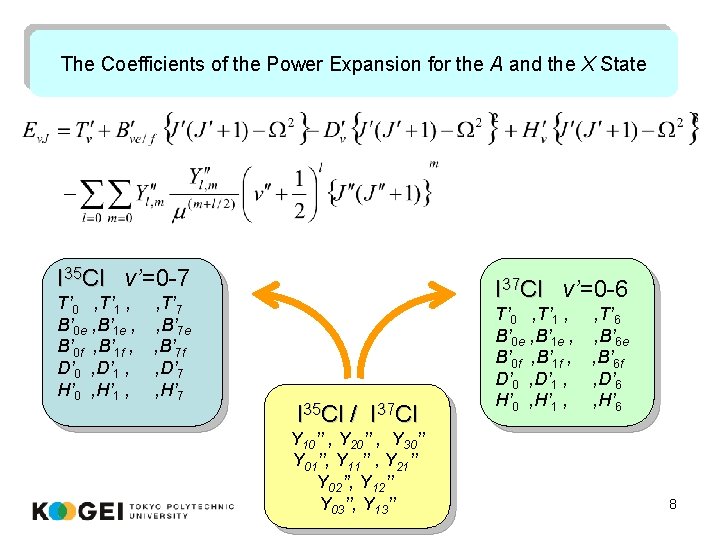
The Coefficients of the Power Expansion for the A and the X State I 35 Cl v’=0 -7 T’ 0 , T’ 1 , B’ 0 e , B’ 1 e , B’ 0 f , B’ 1 f , D’ 0 , D’ 1 , H’ 0 , H’ 1 , , T’ 7 , B’ 7 e , B’ 7 f , D’ 7 , H’ 7 I 37 Cl v’=0 -6 I 35 Cl / I 37 Cl Y 10’’ , Y 20’’ , Y 30’’ Y 01’’, Y 11’’ , Y 21’’ Y 02’’, Y 12’’ Y 03’’, Y 13’’ T’ 0 , T’ 1 , B’ 0 e , B’ 1 e , B’ 0 f , B’ 1 f , D’ 0 , D’ 1 , H’ 0 , H’ 1 , , T’ 6 , B’ 6 e , B’ 6 f , D’ 6 , H’ 6 8

The Spectroscopic Constants of The X State Type B Type C Type A (All parameter is variable. ) ( constrain Y’’ 10 and Y’’ 01) ( constrain Y’’ 10, Y’’ 01 and Y’’ 03) s=0. 0021 cm-1 s=0. 0022 cm-1 In cm-1 and 2. 5 s in parentheses 1) Y’’ 10 384. 29416 Y’’ 01 0. 114157656 H. G. Heddrich and P. F. Bernath , J. Mol. Spectrosc. 155. 384 -392(1992) 2) Y’’ 03 is calculated using 9

The Spectroscopic Constants of The A State I 35 Cl I 37 Cl 10

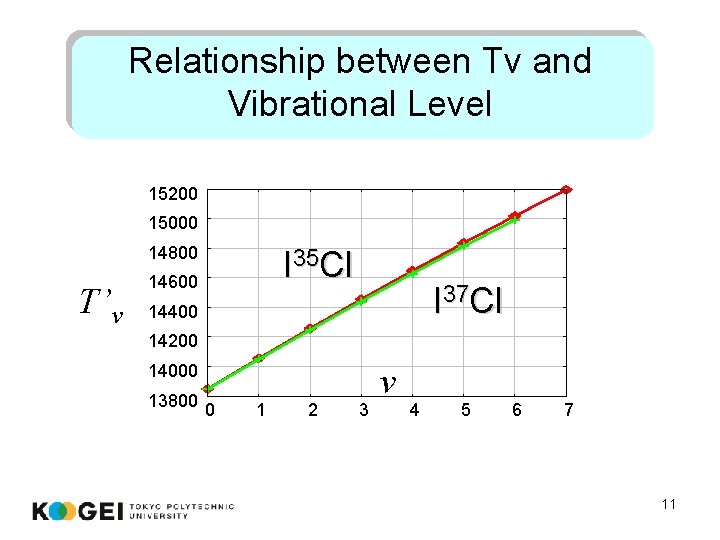
Relationship between Tv and Vibrational Level 15200 15000 14800 T’v I 35 Cl 14600 I 37 Cl 14400 14200 14000 13800 0 1 2 3 v 4 5 6 7 11

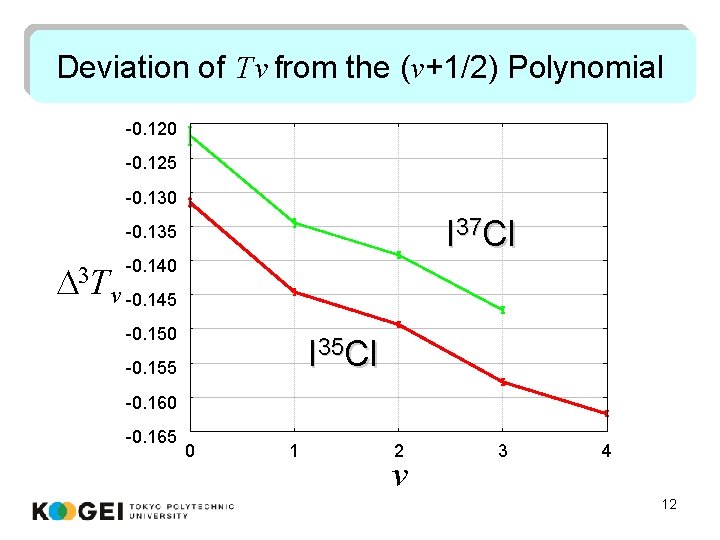
Deviation of Tv from the (v+1/2) Polynomial -0. 120 -0. 125 -0. 130 I 37 Cl -0. 135 D 3 T -0. 140 v -0. 145 -0. 150 I 35 Cl -0. 155 -0. 160 -0. 165 0 1 2 v 3 4 12

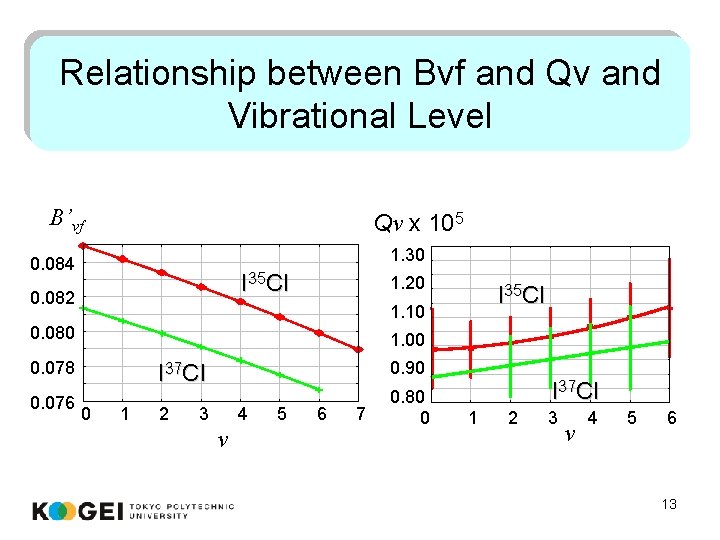
Relationship between Bvf and Qv and Vibrational Level B’vf Qv x 105 1. 30 0. 084 I 35 Cl 0. 082 1. 20 1. 10 0. 080 1. 00 0. 078 0. 076 I 35 Cl 0. 90 I 37 Cl 0 1 2 3 4 v 5 6 7 0. 80 0 I 37 Cl 1 2 3 v 4 5 6 13

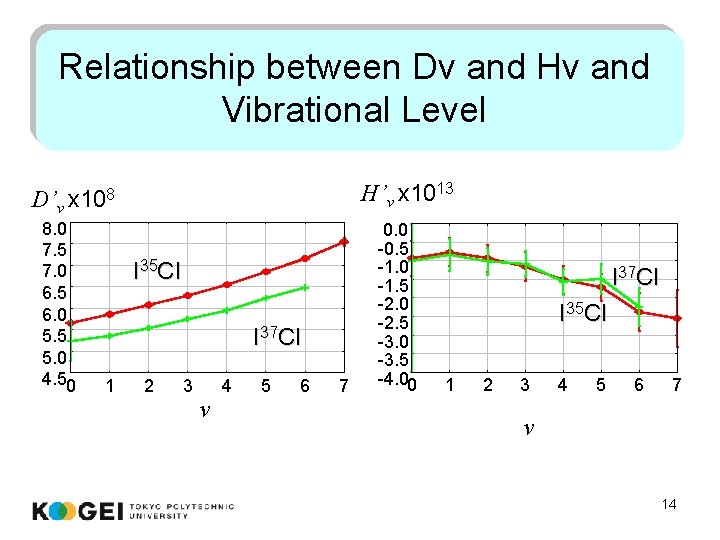
Relationship between Dv and Hv and Vibrational Level H’v x 1013 D’v x 108 8. 0 7. 5 7. 0 6. 5 6. 0 5. 5 5. 0 4. 50 I 35 Cl I 37 Cl 1 2 3 v 4 5 6 7 0. 0 -0. 5 -1. 0 -1. 5 -2. 0 -2. 5 -3. 0 -3. 5 -4. 00 I 37 Cl I 35 Cl 1 2 3 4 5 6 7 v 14

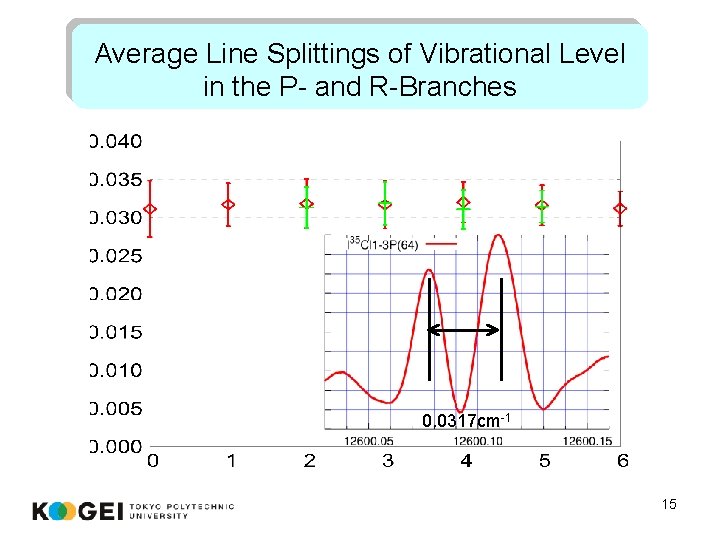
Average Line Splittings of Vibrational Level in the P- and R-Branches 0. 0317 cm-1 15

Summary l A – X system of P, Q, R-branch lines were assigned. 4900 lines l X-state of Dunham coefficients were determined by using a mass-reduced least square fitting procedure. 7 parameters l Spectroscopic constants of A-state were calculated. Tv, Bvf, qv, Dv, Hv for each vibrational levels (Dunham coefficients are not suitable for A- state. ) 16