High Pressure study of Bromine Shimizu Lab M

High Pressure study of Bromine Shimizu Lab M 2 Hayashi Yuma

Contents • Introduction Iodine • Motivation • Experiments • Summary • Next plan

Introduction room –temperature superconductor? http: //www. cqst. osaka-u. ac. jp
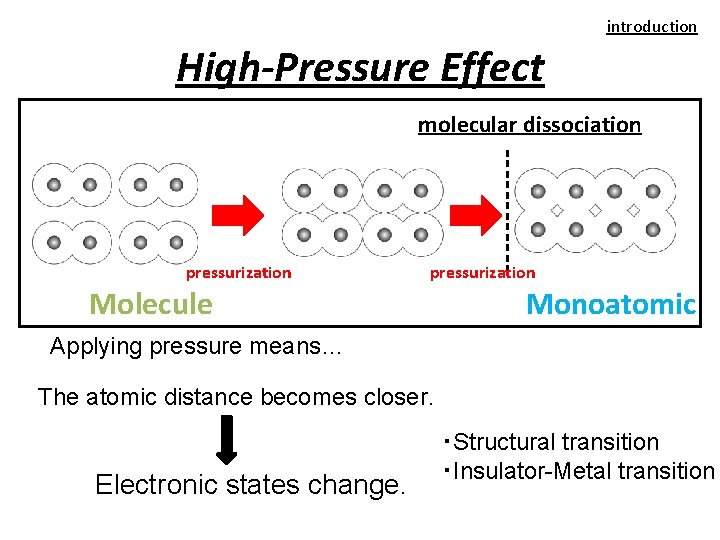
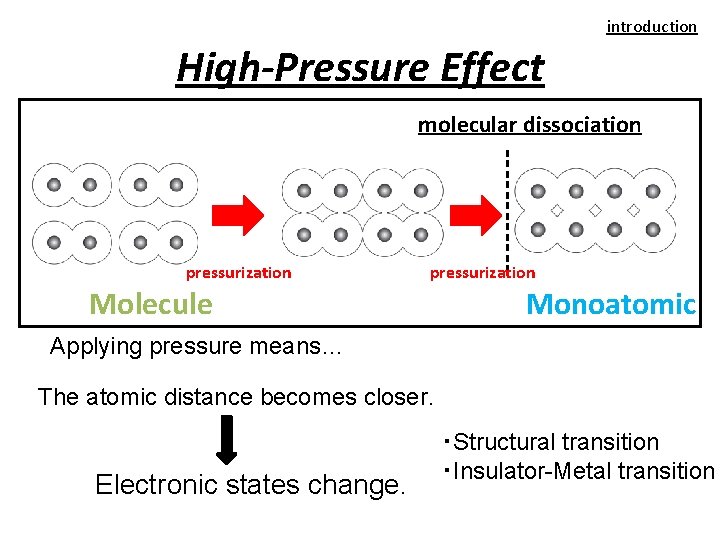
introduction High-Pressure Effect molecular dissociation pressurization Molecule Monoatomic Applying pressure means… The atomic distance becomes closer. Electronic states change. ・Structural transition ・Insulator-Metal transition
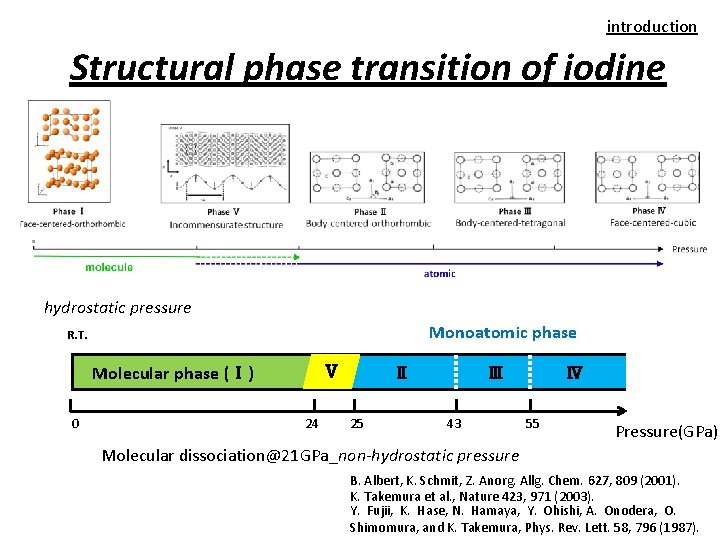
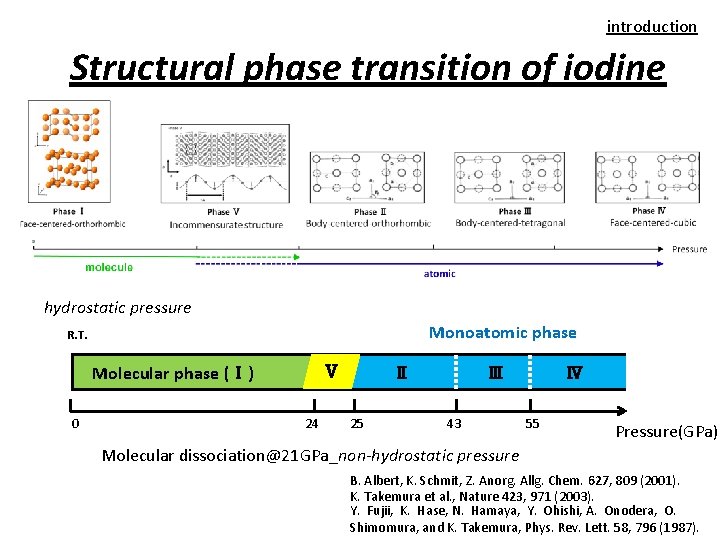
introduction Structural phase transition of iodine hydrostatic pressure Monoatomic phase R. T. Molecular phase (Ⅰ) 0 Ⅴ 24 Ⅱ 25 Ⅲ 43 Ⅳ 55 Pressure(GPa) Molecular dissociation@21 GPa_non-hydrostatic pressure B. Albert, K. Schmit, Z. Anorg. Allg. Chem. 627, 809 (2001). K. Takemura et al. , Nature 423, 971 (2003). Y. Fujii, K. Hase, N. Hamaya, Y. Ohishi, A. Onodera, O. Shimomura, and K. Takemura, Phys. Rev. Lett. 58, 796 (1987).
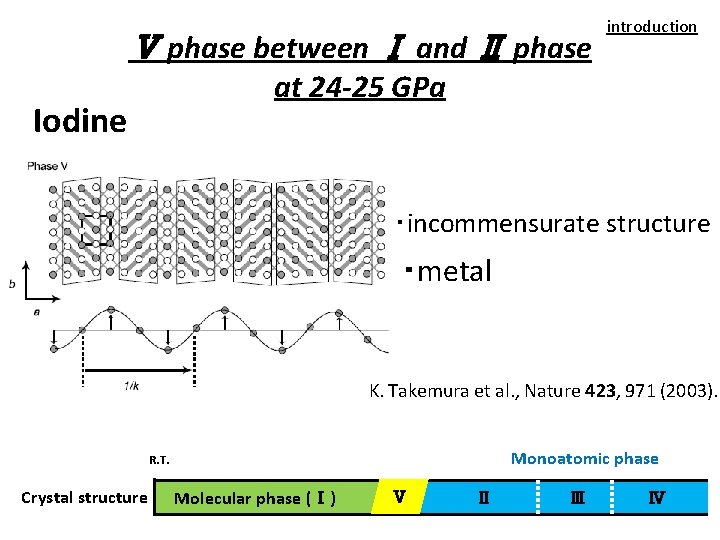
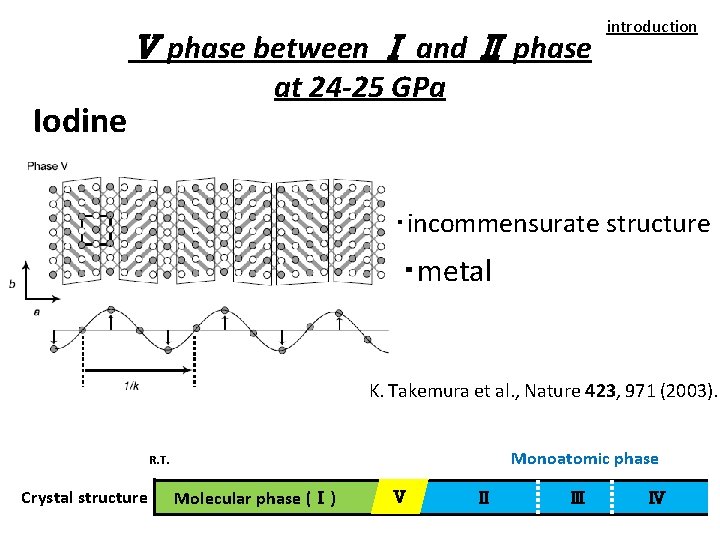
Ⅴ phase between Ⅰ and Ⅱ phase introduction at 24 -25 GPa Iodine ・incommensurate structure ・metal K. Takemura et al. , Nature 423, 971 (2003). Monoatomic phase R. T. Crystal structure Molecular phase (Ⅰ) Ⅴ Ⅱ Ⅲ Ⅳ
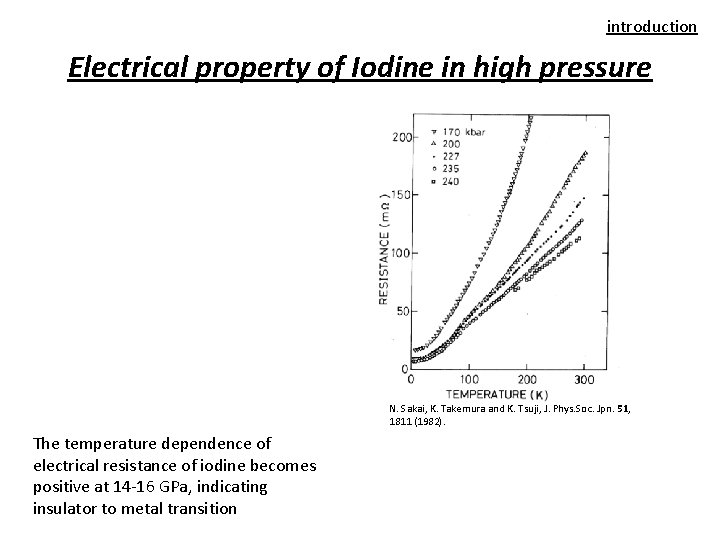
introduction Electrical property of Iodine in high pressure N. Sakai, K. Takemura and K. Tsuji, J. Phys. Soc. Jpn. 51, 1811 (1982). The temperature dependence of electrical resistance of iodine becomes positive at 14 -16 GPa, indicating insulator to metal transition
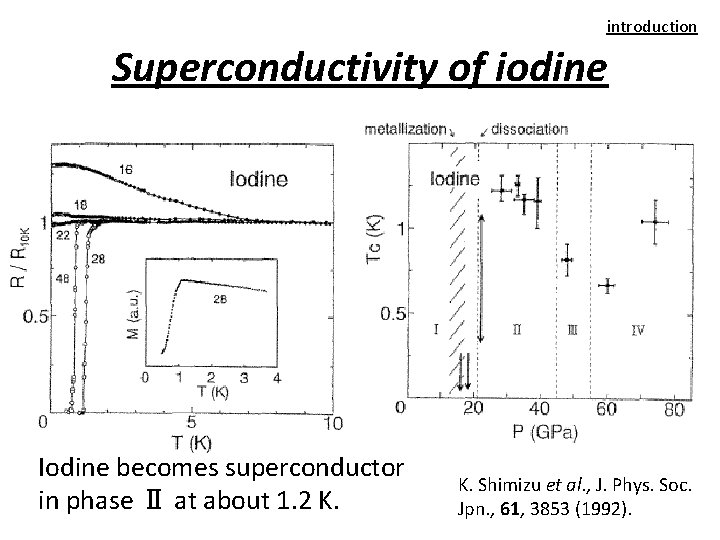
introduction Superconductivity of iodine Iodine becomes superconductor in phase Ⅱ at about 1. 2 K. Shimizu et al. , J. Phys. Soc. Jpn. , 61, 3853 (1992).

introduction Phase diagram of iodine Molecular dissociation Monatomic phase R. T. Molecular phase (Ⅰ) Crystal structure Electrical characteristic Ⅴ Ⅱ Ⅲ Ⅳ metal Insulator Superconductor 0 16 21 43 55 Pressure [GPa] ・Iodine becomes superconductor in phase Ⅱ at about 1. 2 K.

Introduction room –temperature superconductor? http: //www. cqst. osaka-u. ac. jp
introduction High-Pressure Effect molecular dissociation pressurization Molecule Monoatomic Applying pressure means… The atomic distance becomes closer. Electronic states change. ・Structural transition ・Insulator-Metal transition
introduction Structural phase transition of iodine hydrostatic pressure Monoatomic phase R. T. Molecular phase (Ⅰ) 0 Ⅴ 24 Ⅱ 25 Ⅲ 43 Ⅳ 55 Pressure(GPa) Molecular dissociation@21 GPa_non-hydrostatic pressure B. Albert, K. Schmit, Z. Anorg. Allg. Chem. 627, 809 (2001). K. Takemura et al. , Nature 423, 971 (2003). Y. Fujii, K. Hase, N. Hamaya, Y. Ohishi, A. Onodera, O. Shimomura, and K. Takemura, Phys. Rev. Lett. 58, 796 (1987).
Ⅴ phase between Ⅰ and Ⅱ phase introduction at 24 -25 GPa Iodine ・incommensurate structure ・metal K. Takemura et al. , Nature 423, 971 (2003). Monoatomic phase R. T. Crystal structure Molecular phase (Ⅰ) Ⅴ Ⅱ Ⅲ Ⅳ
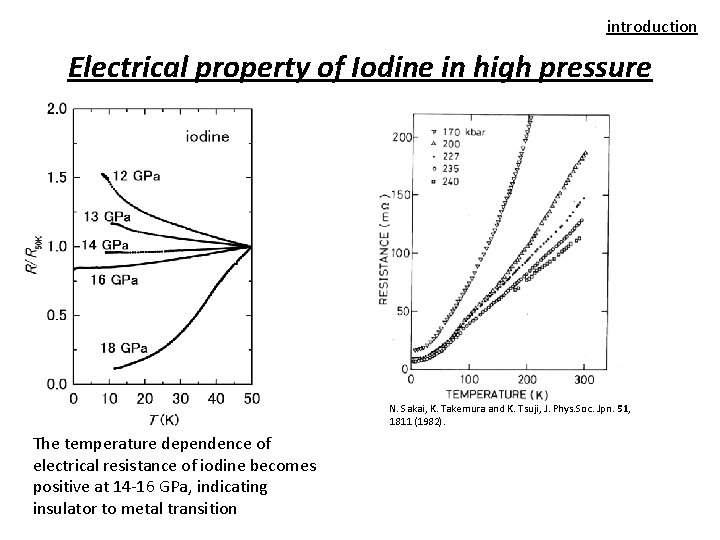
introduction Electrical property of Iodine in high pressure N. Sakai, K. Takemura and K. Tsuji, J. Phys. Soc. Jpn. 51, 1811 (1982). The temperature dependence of electrical resistance of iodine becomes positive at 14 -16 GPa, indicating insulator to metal transition
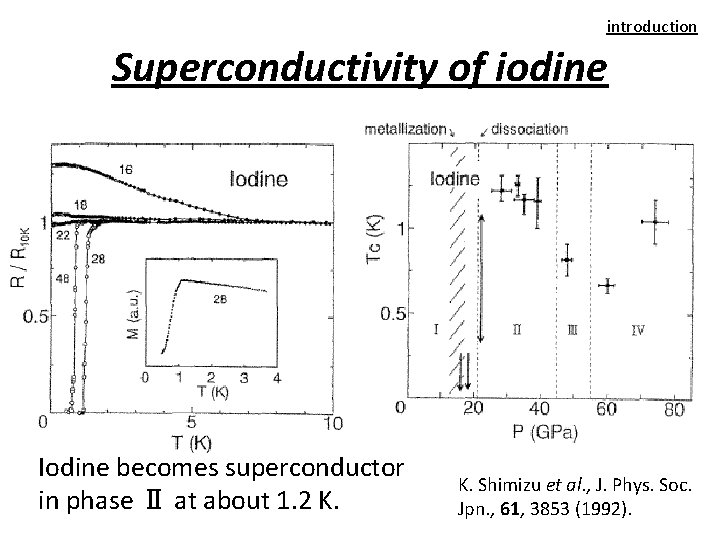
introduction Superconductivity of iodine Iodine becomes superconductor in phase Ⅱ at about 1. 2 K. Shimizu et al. , J. Phys. Soc. Jpn. , 61, 3853 (1992).

introduction Phase diagram of iodine Molecular dissociation Monatomic phase R. T. Molecular phase (Ⅰ) Crystal structure Electrical characteristic Ⅴ Ⅱ Ⅲ Ⅳ metal Insulator Superconductor 0 16 21 43 55 Pressure [GPa] ・Iodine becomes superconductor in phase Ⅱ at about 1. 2 K.

Motivation Perform investigation of the crystal structure and electrical property of bromine under pressure. Clarifying the mechanism of the pressureinduced metallization in halogen elements and molecular crystals.
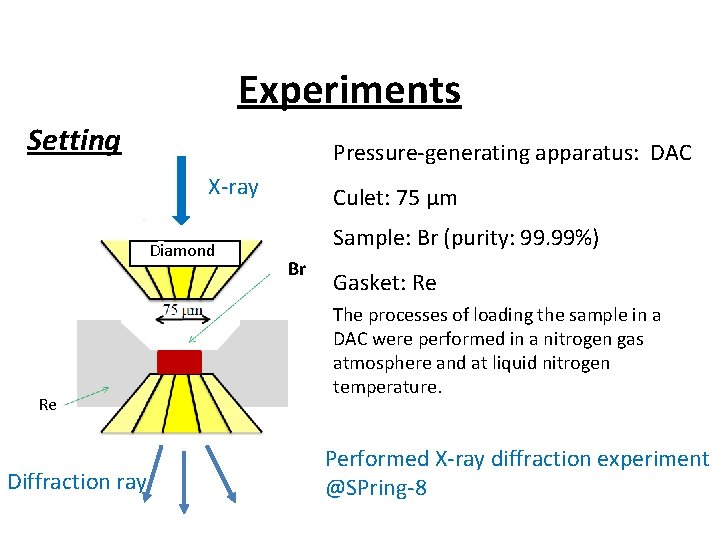
Experiments Setting Pressure-generating apparatus: DAC X-ray Diamond Re Diffraction ray Culet: 75 μm Sample: Br (purity: 99. 99%) Br Gasket: Re The processes of loading the sample in a DAC were performed in a nitrogen gas atmosphere and at liquid nitrogen temperature. Performed X-ray diffraction experiment @SPring-8
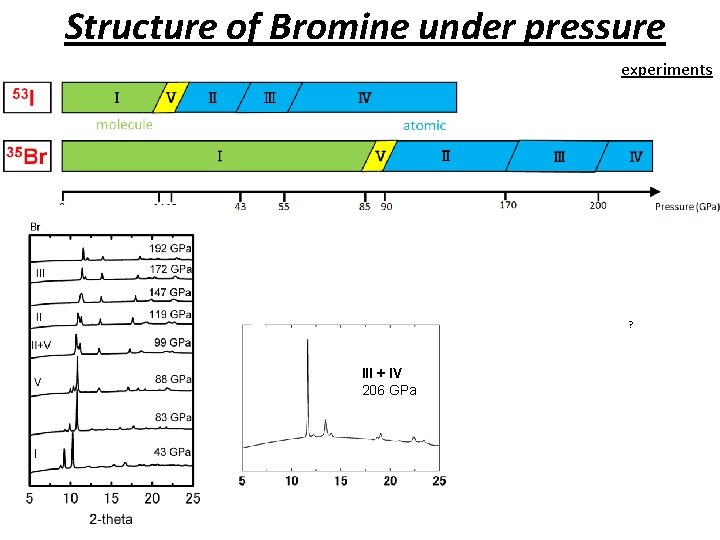
Structure of Bromine under pressure experiments ? III + IV 206 GPa
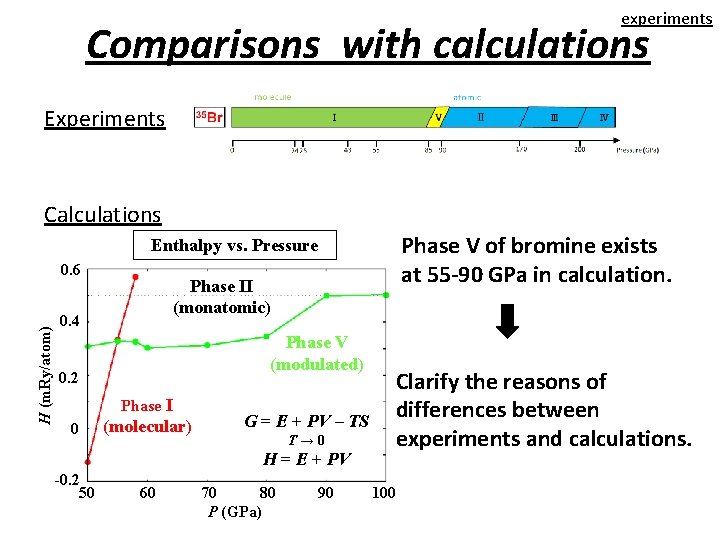
experiments Comparisons with calculations Experiments Calculations Phase V of bromine exists at 55 -90 GPa in calculation. Enthalpy vs. Pressure H (m. Ry/atom) 0. 6 Phase II (monatomic) 0. 4 Phase V (modulated) 0. 2 Phase I 0 (molecular) G = E + PV – TS T→ 0 H = E + PV -0. 2 50 60 70 80 P (GPa) 90 Clarify the reasons of differences between experiments and calculations. 100
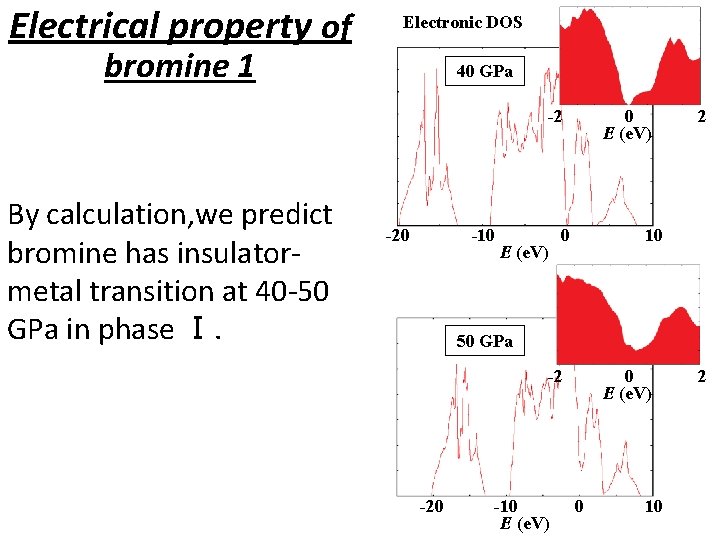
Electrical property of Electronic DOS bromine 1 40 GPa -2 By calculation, we predict bromine has insulatormetal transition at 40 -50 GPa in phase Ⅰ. -20 -10 E (e. V) 0 2 10 50 GPa -2 -20 -10 E (e. V) 0 10 2
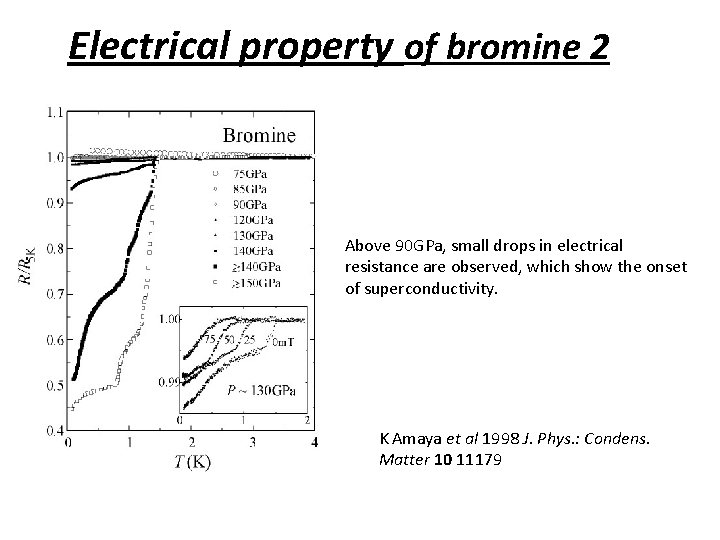
Electrical property of bromine 2 Above 90 GPa, small drops in electrical resistance are observed, which show the onset of superconductivity. K Amaya et al 1998 J. Phys. : Condens. Matter 10 11179

Summary ・Under high pressure, bromine has the same structure changes as iodine without transition pressures. ・It is predicted bromine has insulator-metal transition at 40 -50 GPa by calculations, and at pressure higher than 90 GPa, small drops in electrical resistance are observed, which show the onset of superconductivity.

Next plan Careful investigation of the crystal structure and electrical property of Bromine in the phase I , and comparisons of experiments with calculations. Clarifying the mechanism of the pressureinduced metallization in bromine.




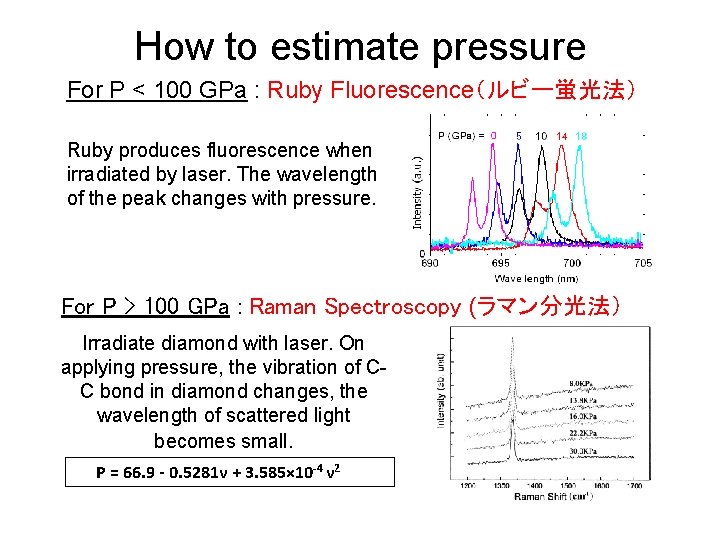
How to estimate pressure For P < 100 GPa : Ruby Fluorescence(ルビー蛍光法) Ruby produces fluorescence when irradiated by laser. The wavelength of the peak changes with pressure. For P > 100 GPa : Raman Spectroscopy (ラマン分光法) Irradiate diamond with laser. On applying pressure, the vibration of CC bond in diamond changes, the wavelength of scattered light becomes small. P = 66. 9 - 0. 5281ν + 3. 585× 10 -4 ν 2
![introduction Hall Effect of Iodine V- V+ I- I+ V 0 [top view] ・The introduction Hall Effect of Iodine V- V+ I- I+ V 0 [top view] ・The](http://slidetodoc.com/presentation_image/1396f0cb10e8785b8686f95d8684f044/image-30.jpg)
introduction Hall Effect of Iodine V- V+ I- I+ V 0 [top view] ・The carrier of iodine is ‘hole’. B
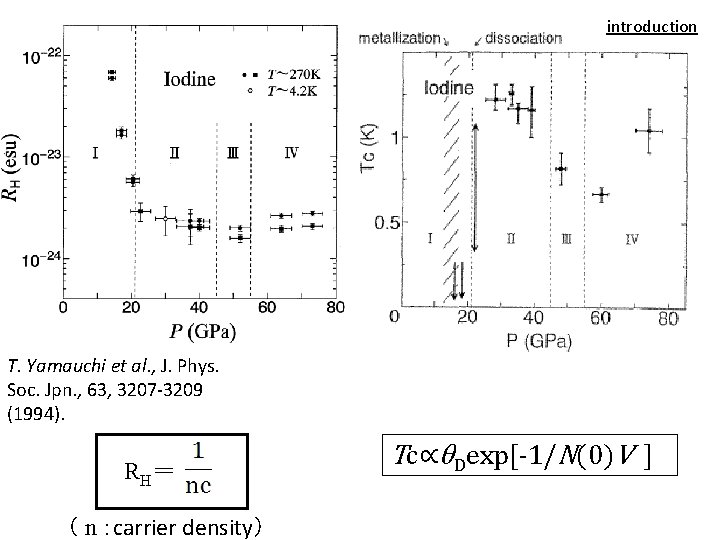
introduction T. Yamauchi et al. , J. Phys. Soc. Jpn. , 63, 3207 -3209 (1994). R H= ( n :carrier density) Tc∝θDexp[-1/N(0)V ]
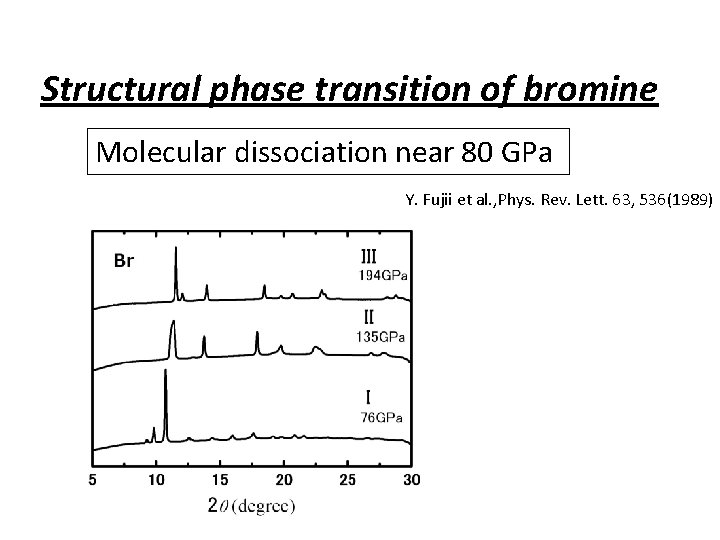
Structural phase transition of bromine Molecular dissociation near 80 GPa Y. Fujii et al. , Phys. Rev. Lett. 63, 536(1989)
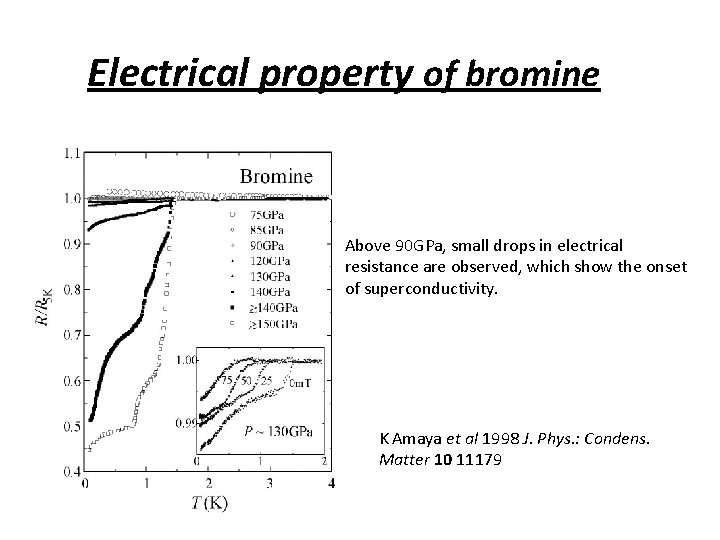
Electrical property of bromine Above 90 GPa, small drops in electrical resistance are observed, which show the onset of superconductivity. K Amaya et al 1998 J. Phys. : Condens. Matter 10 11179
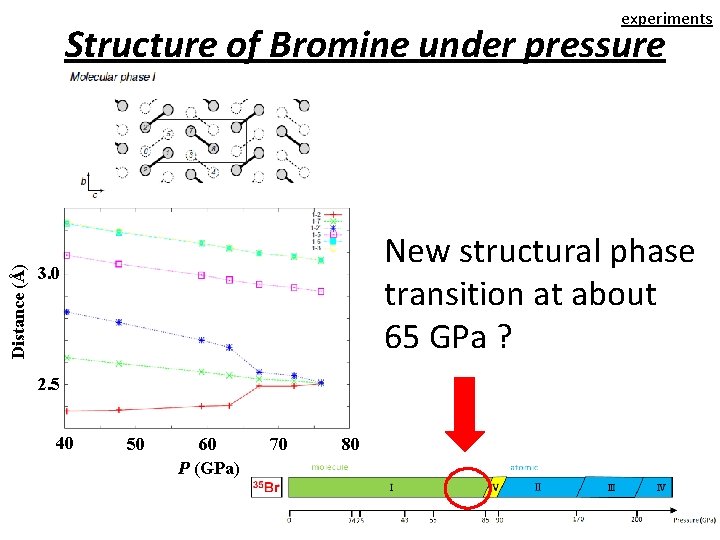
experiments Distance (Å) Structure of Bromine under pressure New structural phase transition at about 65 GPa ? 3. 0 2. 5 40 50 60 P (GPa) 70 80
- Slides: 34