High Performance Liquid Chromatography by Dr Gohar Taqi
High Performance Liquid Chromatography by Dr Gohar Taqi Kazimi

High Performance Liquid Chromatography (HPLC) formerly referred to as high-pressure liquid chromatography is a form of column chromatography that pumps a sample mixture or analyte in a solvent (mobile phase) at high pressure through a column with chromatographic packing material (stationary phase). The sample is carried by a moving carrier gas stream of helium or nitrogen. Each component in the sample interacts slightly differently with the adsorbent material, causing different flow rates for the different components and leading to the separation of the components as they flow out of the column.

HPLC has the ability to separate, and identify compounds that are present in any sample that can be dissolved in a liquid in trace concentrations as low as parts per trillion. Because of this versatility, HPLC is used in a variety of industrial and scientific applications, such as pharmaceutical, environmental, forensics, and chemicals.

Sample retention time will vary depending on the interaction between the stationary phase, the molecules being analyzed, and the solvent, or solvents used. As the sample passes through the column it interacts between the two phases at different rate, primarily due to different polarities in the analytes. Analytes that have the least amount of interaction with the stationary phase or the most amount of interaction with the mobile phase will exit the column faster.

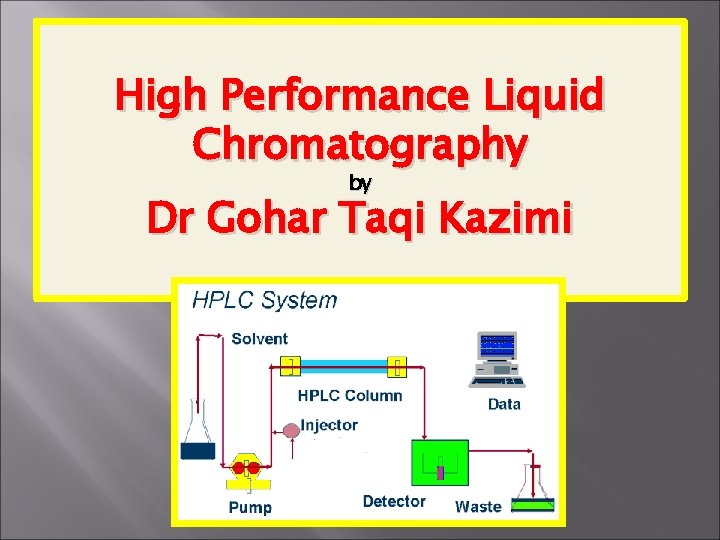
Instrumentation Main components in an HPLC system include the solvent reservoir, or multiple reservoirs, a highpressure pump, a column, injector system and the detector.

Reservoir The reservoir holds the solvent, which is referred to as the mobile phase because it moves. There are usually a minimum of two reservoirs in a system, with each holding up to 1000 cc of solvent and usually fitted with a gas diffuser through which helium can be bubbled. A pump is used to generate a specified flow of the mobile phase. Although manual injection of samples is still possible, most HPLCs are now fully automated and controlled by computer.

Pump: The role of the pump is to force a liquid (mobile phase) through the liquid chromatograph at a specific flow rate A pump can deliver a constant mobile phase composition (Isocratic)where the mobile phase composition remains unchanged during the analysis. or (Gradient) where the mobile phase changed during the analysis. The mobile phase is pumped through the column by a pump. Solvents must be degassed to eliminate formation of bubbles.

Injector: The injector introduces the liquid sample into the flow stream of the mobile phase that carries the sample into the high pressure (up to 400 bar) column. The injector may be auto-sampler or manual Column: The heart of a HPLC system is the column. The column contains the particles that contains the stationary phase needed to effect separation.

Detector: A detector is needed to see the separated bands as they elute from the high pressure column. The information is sent from the detector to a computer which generates the chromatogram. The mobile phase exits the detector and is either sent to a waste, or collected, as desired. Helium sparying is an effective method of degassing the mobile phase to avoid unstable baselines caused by dissolved air. Nitrogen is used as a nebulisation gas in Evaporative Light Scattering Detector (ELSD) where the solvent is evaporated from the sample leaving a mist as is measured.

There a wide variety of stationary phases available for HPLC : * Normal Phase. - Polar stationary phase and non-polar solvent. E. g. silica gel * Reverse Phase. - Non-polar stationary phase and a polar solvent. E. g. silica gel -C 18

Ion exchange: Stationary phase contains ionic groups and the mobile phase is an aqueous buffer Size Exclusion There is no interaction between the sample compounds and the column. Large molecules elute first. Smaller molecules elute later

Chromatogram

Parameters of HPLC: 1 - Retention time: The most common parameter for compound is retention time The time it takes for that specific compound to elute from the column after injection. Injection time

Capacity Factor (k’): Is a measure for the position of a sample peak in the chromatogram. k’ = (t. R 1 -to)/to Selectivity Factor (a ( ): Also called separation or selectivity coefficient is defined as a = k 2’/k 1’ = (t. R 2 -to) / (t. R 1 -to)

Quantitative Analysis The measurement of the amount of compound in a sample (concentration) 1. Determination of the peak height 2. Determination of the peak area

Resolution (RS) It is a quantitative measure of its ability to separate two analytes Rs = 2(TR 2 - TR 1 ) / W 2+W 1 Theoretical Plates (N): The number of theoretical plates characterizes the efficiency of a column. N = 16 (t. R/W)2
- Slides: 16