HHS Smallpox Vaccine Program ACAM 2000 Briefing for

HHS Smallpox Vaccine Program & ACAM 2000 Briefing for VRBPAC 17 May 2007 Dr. Gerald Parker Principal Deputy Assistant Secretary for Preparedness and Response FOUO PROCUREMENT SENSITIVE DRAFT

Briefing Overview DHHS Smallpox Program • Requirement for Smallpox Vaccine • ACAM 2000 • Smallpox Vaccine Utilization Policy • Public Health Response Planning at Federal, State, and Local Levels • Training and Education programs • Summary 1

Requirement for Smallpox Vaccine l Eradication of Smallpox – Vaccination and manufacturing programs halted ~25 years ago – Approximately one half of the US population has no immunity to smallpo l Pre 9/11 – USG Discussion of Potential Biological Warfare Threat Using Smallpox – Evaluated public health response capabilities – Evaluated existing stockpile of vaccine (15 M doses) – Consensus reached for new licensed smallpox vaccine l Post 9/11 – USG Requirement for Smallpox Vaccine Increased – Utilization policies and preparedness programs were put in place 2

ACAM 2000 l USG Requirements – Strain v New York City Board of Health strain of vaccinia (derived from licensed Dryvax® vaccine) v Used successfully during eradication period – Manufacturing and Immunogenicity v v l Grown in qualified cell substrate – Vero bioreactors Clonal isolate – well characterized Free of adventitious agents Elicitation of immune response analogous to current licensed Dryvax® vaccine Contractor – Acambis met all the public health, regulatory, and contractual requirements of the USG and was awarded its first contract (40 M doses) in 2000, second contract (250 M doses) in 2001 – Acambis has delivered 192 M doses to the Strategic National Stockpile 3
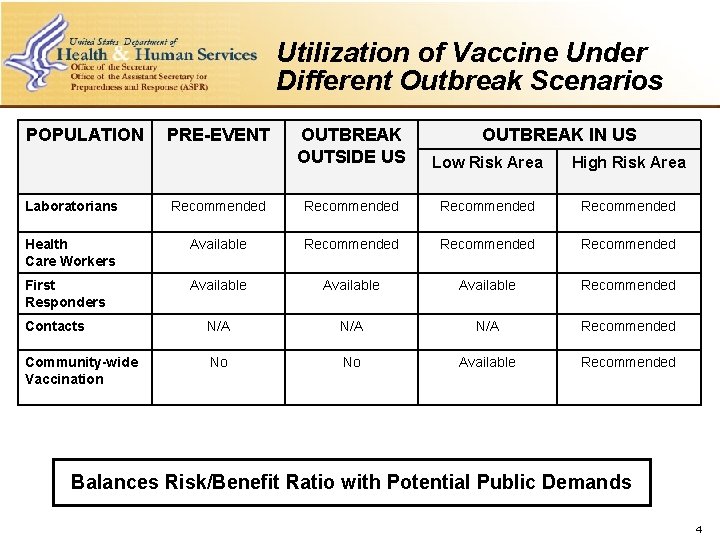
Utilization of Vaccine Under Different Outbreak Scenarios POPULATION PRE-EVENT OUTBREAK OUTSIDE US OUTBREAK IN US Low Risk Area High Risk Area Laboratorians Recommended Health Care Workers Available Recommended First Responders Available Recommended Contacts N/A N/A Recommended Community-wide Vaccination No No Available Recommended Balances Risk/Benefit Ratio with Potential Public Demands 4

HHS—Public Health Response Planning at the Federal Level l HHS/ASPR Participation in Preparedness Activities – Tabletop exercises and other activities v Catastrophic Assessment Task Force (2006) v Atlantic Storm (2005) v NJ TTX (Fierce Squall) (2005) v Global Mercury (2003) v Dark Winter (2001) l ASPR/OPEO – Playbooks for national level federal public health response planning l ASPR/BARDA – Developing next-generation vaccines and therapeutics for smallpox 5

CDC—Public Health Response Planning at Federal, State, and Local Levels l Working with HHS/ASPR to Coordinate Preparedness and Response l Enhancing Collaboration with State and Local Governments l Providing Grants for Preparedness Planning and Exercises l Conducting Exercises with State and Local Partners to Ensure Readiness and Response for Multiple Threat Scenarios Including a Smallpox Event l Managing the Strategic National Stockpile 6

CDC—Training for Smallpox Vaccination l Training of Health Care Workers, Clinicians, and Laboratorians v To recognize the disease and symptoms v To understand how the vaccine works and potential side effects v To effectively administer the vaccine using a bifurcated needle l Training for Smallpox Response Teams (Federal and State) l Training Products and Detailed Information Available in a Variety of Media Formats v v CDC website Video and CD Telephone Classroom setting 7

Summary l ACAM 2000 – NYCBH-derived vaccine – Delivered to Strategic National Stockpile – Replenishment/Warm-base l Response Planning and Utilization Policy – ASPR and CDC – Federal/State/Local plans – Pre-Event v Civilian—limited distribution and use v DOD program – to be discussed v Surveillance/reporting systems – Post-Event v Recommendations for low vs. high risk populations v AE/SAE reporting 8
- Slides: 9