HEXAVAC A new liquid DTac PIPVHibHB hexavalent vaccine

HEXAVAC® A new liquid DTac. P-IPV-Hib-HB hexavalent vaccine Overview of its clinical profile Benoît Soubeyrand M. D. (Courtesy L. Hessel) B. Soubeyrand - VHPB October 2001 Malte 1

HEXAVAC®: A NEW LIQUID DTa. P-IPV-HIB-HB HEXAVALENT COMBINATION VACCINE l What is Hexavac® ? l Clinical development plan l Safety profile l Immunogenicity l Conclusions B. Soubeyrand - VHPB October 2001 Malte . 2

WHAT IS HEXAVAC® (1): Composition and Presentation l A unique ready to use, preservative-free, liquid formulation, combining all 6 antigens in 0. 5 ml Ø Ø Ø Ø Ø purified diphtheria toxoid, purified tetanus toxoid, adsorbed purified pertussis toxoid, adsorbed purified FHA, Polio type 1 (Mahoney strain), Polio type 2 (MEF 1 strain), Polio type 3 (Saukett strain), PRP-T, adsorbed, purified HBs. Ag, B. Soubeyrand - VHPB October 2001 Malte 20 IU (30 Lf) 40 IU (10 Lf) 25 g 25 g 40 D units 8 D units 32 D units 12 g (PRP) 5 g 3

WHAT IS HEXAVAC® (2): Critical timelines l l l Start of the project 1 st clinical lot available 1 st inclusion phase 1 1 st inclusion phase 2 1 st consistency batch (development scale) 1 st inclusion phase 3 1 st consistency batch (industrial scale) 1 st industrial lot EMEA submission CPMP positive opinion Market autorisation Launch in Germany Feb. 94 Jan. 95 Feb. 95 May 95 July 95 June 96 Dec. 98 April 99 July 99 June 00 Oct. 00 i. e. - 5 years and 5 months: time to develop - 6 years and 8 months: time to register / market B. Soubeyrand - VHPB October 2001 Malte 4

WHAT IS HEXAVAC® (3): Formulation Process l 200 different experimental formulations prepared, varying : Ø Ø Ø p. H ionic strength of different buffers (C 032 -, P 043 -) batches of antigens batches of adjuvant from different manufacturers concentration of antigens order of addition of antigens, adjuvant, buffer as well as conditions of blending l 50 different hexavalent formulations physico-chemically and immunologically tested l 7 optimized formulations extensively tested at 4°C, RT, 37°C l 2 semi-final formulations tested, over time, at + 4°C 1 final formulation for clinical development and industrial production B. Soubeyrand - VHPB October 2001 Malte 5

HEXAVAC : Clinical development Plan (1) l Goals of clinical development : Ø To show that the hexavalent combination vaccine is safe and induces protective immune responses equivalent (non-inferior) to previously licensed combination vaccines PENTAVAC™ and RECOMBIVAX® Ø To support clinically the consistency of manufacture by comparing the immunogenicity of three consecutively batches Ø To support the use of the vaccine with various vaccination regimens : 2, 3, 4 / 12 -18 months or 2, 4, 6 / 12 -18 months ; 3, 5 / 12 months B. Soubeyrand - VHPB October 2001 Malte 6
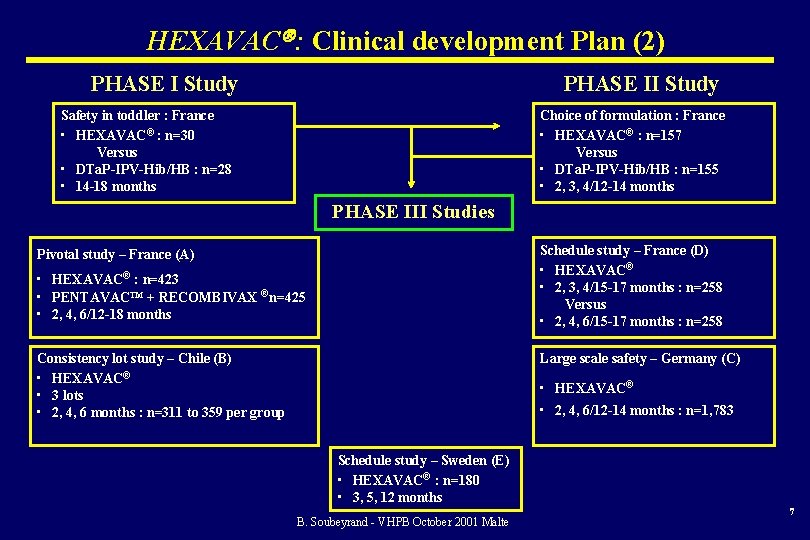
HEXAVAC : Clinical development Plan (2) PHASE I Study PHASE II Study Safety in toddler : France • HEXAVAC® : n=30 Versus • DTa. P-IPV-Hib/HB : n=28 • 14 -18 months Choice of formulation : France • HEXAVAC® : n=157 Versus • DTa. P-IPV-Hib/HB : n=155 • 2, 3, 4/12 -14 months PHASE III Studies Schedule study – France (D) • HEXAVAC® • 2, 3, 4/15 -17 months : n=258 Versus • 2, 4, 6/15 -17 months : n=258 Pivotal study – France (A) • HEXAVAC® : n=423 • PENTAVAC™ + RECOMBIVAX ®n=425 • 2, 4, 6/12 -18 months Consistency lot study – Chile (B) • HEXAVAC® • 3 lots • 2, 4, 6 months : n=311 to 359 per group Large scale safety – Germany (C) • HEXAVAC® • 2, 4, 6/12 -14 months : n=1, 783 Schedule study – Sweden (E) • HEXAVAC® : n=180 • 3, 5, 12 months B. Soubeyrand - VHPB October 2001 Malte 7

HEXAVAC : Clinical development Plan (3) PHASE III studies : booster vaccination Safety of HEXAVAC® in children primed with DTw. P-IPV-Hib (PENTACOQ®), France (F) • n = 1, 304 • 2, 3, 4/16 -20 months Safety & Immunogenicity of HEXAVAC® in children primed with DTw. P-IPV-Hib (PENTACOQ®) + RECOMBIVAX®, France (G) • n = 313 • 2, 3, 4/14 -20 months Safety & Immunogenicity of HEXAVAC® in children primed with DTa. P-IPV-Hib (PENTAVACTM) + RECOMBIVAX®, Turkey (H) • n = 129 • 2, 3, 4/14 -18 months B. Soubeyrand - VHPB October 2001 Malte . 8

HEXAVAC : Clinical development Plan (4) l Database submitted in the application file to CPMP Ø 11 507 doses of vaccines administered to 3 905 infants in primary series. Ø 4 437 booster doses given to toddlers primed with either wholecell or acellular Pertussis combination vaccines : - Pentacoq™ ( DTw. P-IPV//PRP-T) - Pentavac™ ( DTa. P-IPV//PRP-T) l European license received on October 24 th 2000 B. Soubeyrand - VHPB October 2001 Malte 9

HEXAVAC® : Safety profile (1) In all studies, evaluation of safety included: 1. Immediate reactions within the first 30 minutes after each dose 2. Local reactions and systemic events from 30 minutes to 72 hours following each dose 3. Any adverse event that resulted in a visit to a physician 4 and 30 days after each injection 4. Any serious adverse event (SAE) occurring throughout the trial. B. Soubeyrand - VHPB October 2001 Malte 10
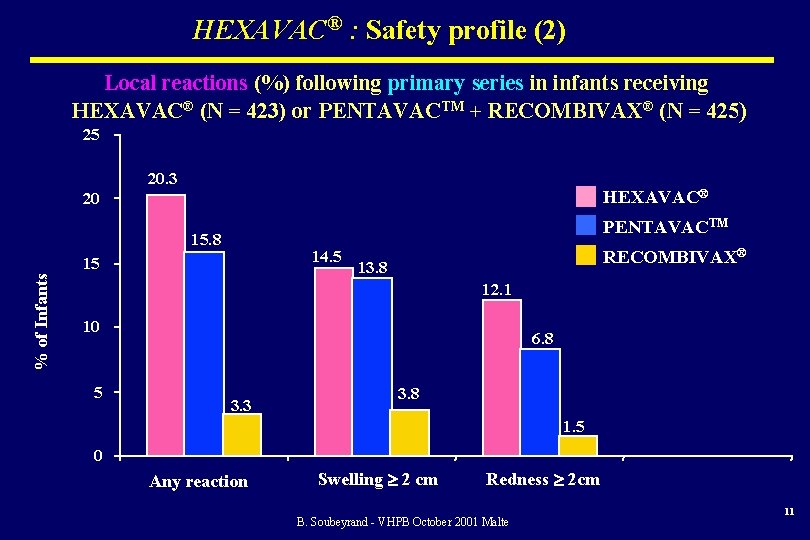
HEXAVAC® : Safety profile (2) Local reactions (%) following primary series in infants receiving HEXAVAC® (N = 423) or PENTAVACTM + RECOMBIVAX® (N = 425) 25 20. 3 HEXAVAC® 20 PENTAVACTM 15. 8 14. 5 % of Infants 15 RECOMBIVAX® 13. 8 12. 1 10 5 6. 8 3. 3 3. 8 1. 5 0 Any reaction Swelling 2 cm Redness 2 cm B. Soubeyrand - VHPB October 2001 Malte 11
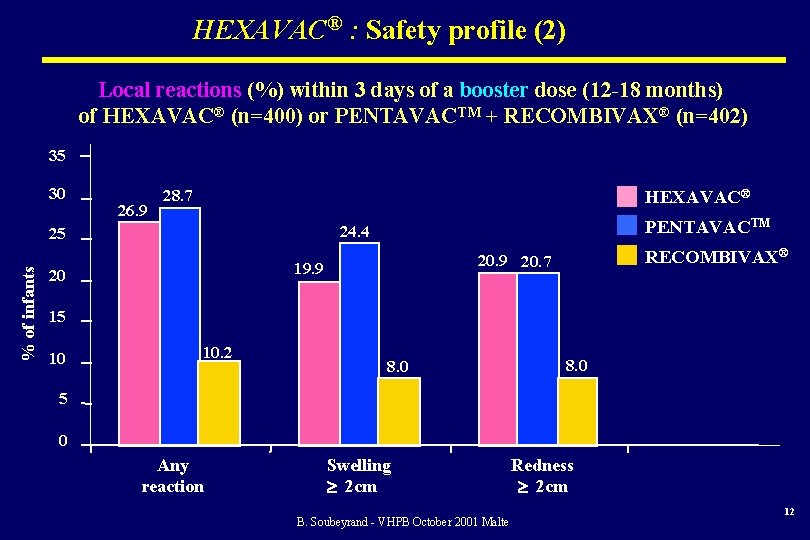
HEXAVAC® : Safety profile (2) Local reactions (%) within 3 days of a booster dose (12 -18 months) of HEXAVAC® (n=400) or PENTAVACTM + RECOMBIVAX® (n=402) 35 30 26. 9 28. 7 HEXAVAC® % of infants PENTAVACTM 24. 4 25 19. 9 20 RECOMBIVAX® 20. 9 20. 7 15 10 10. 2 8. 0 5 0 Any reaction Swelling 2 cm B. Soubeyrand - VHPB October 2001 Malte Redness 2 cm 12
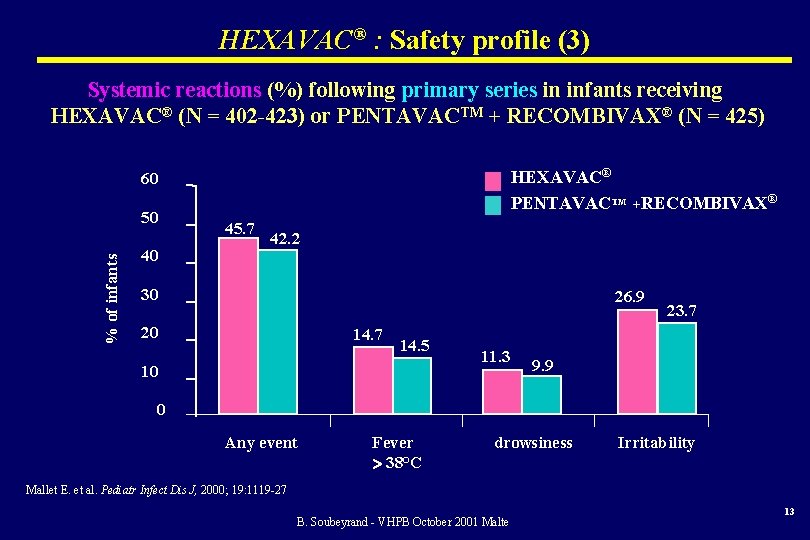
HEXAVAC® : Safety profile (3) Systemic reactions (%) following primary series in infants receiving HEXAVAC® (N = 402 -423) or PENTAVACTM + RECOMBIVAX® (N = 425) HEXAVAC® PENTAVACTM +RECOMBIVAX® 60 % of infants 50 40 45. 7 42. 2 30 26. 9 20 14. 7 14. 5 10 11. 3 23. 7 9. 9 0 Any event Fever 38°C drowsiness Irritability Mallet E. et al. Pediatr Infect Dis J, 2000; 19: 1119 -27 B. Soubeyrand - VHPB October 2001 Malte 13
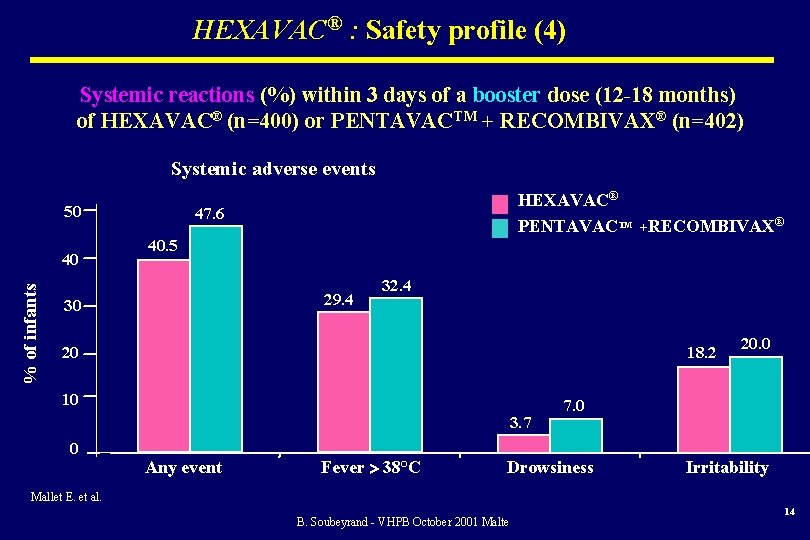
HEXAVAC® : Safety profile (4) Systemic reactions (%) within 3 days of a booster dose (12 -18 months) of HEXAVAC® (n=400) or PENTAVACTM + RECOMBIVAX® (n=402) Systemic adverse events 50 % of infants 40 HEXAVAC® PENTAVACTM +RECOMBIVAX® 47. 6 40. 5 29. 4 30 32. 4 20 18. 2 10 3. 7 0 Any event Fever 38°C 20. 0 7. 0 Drowsiness Irritability Mallet E. et al. B. Soubeyrand - VHPB October 2001 Malte 14
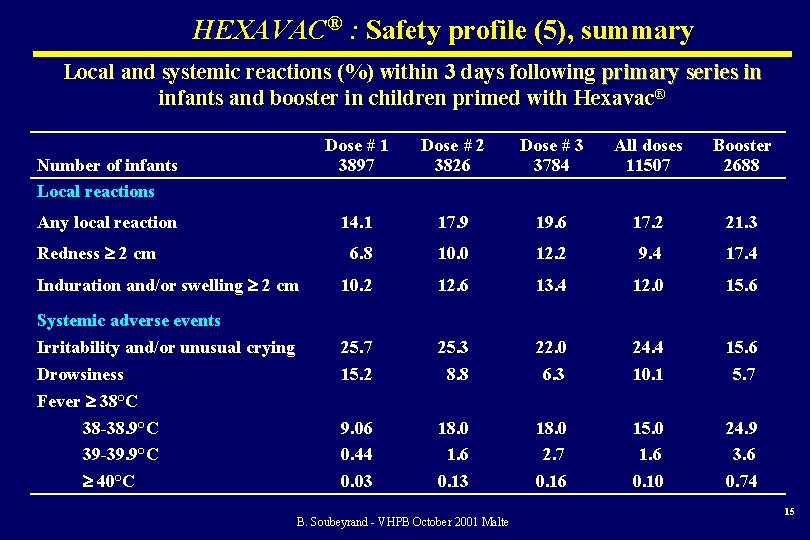
HEXAVAC® : Safety profile (5), summary Local and systemic reactions (%) within 3 days following primary series in infants and booster in children primed with Hexavac® Number of infants Local reactions Any local reaction Redness 2 cm Induration and/or swelling 2 cm Dose # 1 3897 Dose # 2 3826 Dose # 3 3784 All doses 11507 Booster 2688 14. 1 17. 9 19. 6 17. 2 21. 3 6. 8 10. 0 12. 2 9. 4 17. 4 10. 2 12. 6 13. 4 12. 0 15. 6 25. 7 15. 2 25. 3 8. 8 22. 0 6. 3 24. 4 10. 1 15. 6 5. 7 9. 06 0. 44 0. 03 18. 0 1. 6 0. 13 18. 0 2. 7 0. 16 15. 0 1. 6 0. 10 24. 9 3. 6 0. 74 Systemic adverse events Irritability and/or unusual crying Drowsiness Fever 38°C 38 -38. 9°C 39 -39. 9°C 40°C B. Soubeyrand - VHPB October 2001 Malte 15
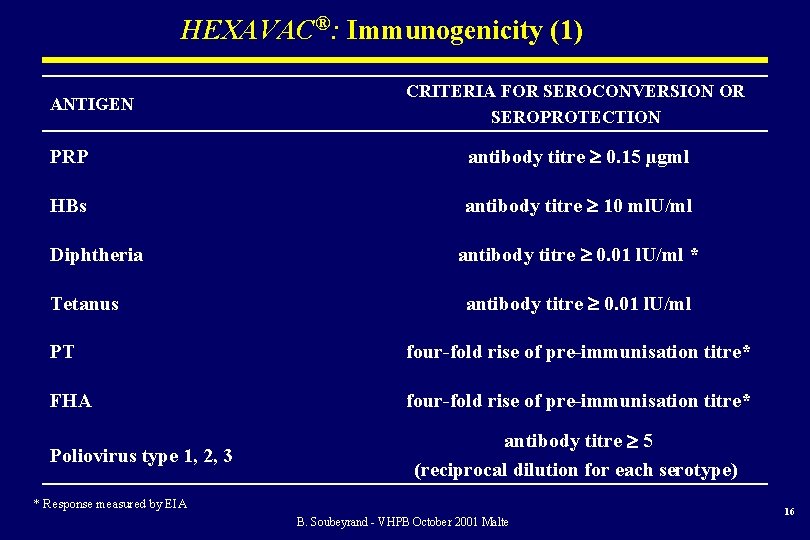
HEXAVAC®: Immunogenicity (1) ANTIGEN CRITERIA FOR SEROCONVERSION OR SEROPROTECTION PRP antibody titre 0. 15 µgml HBs antibody titre 10 ml. U/ml Diphtheria Tetanus antibody titre 0. 01 l. U/ml * antibody titre 0. 01 l. U/ml PT four-fold rise of pre-immunisation titre* FHA four-fold rise of pre-immunisation titre* Poliovirus type 1, 2, 3 antibody titre 5 (reciprocal dilution for each serotype) * Response measured by EIA B. Soubeyrand - VHPB October 2001 Malte 16
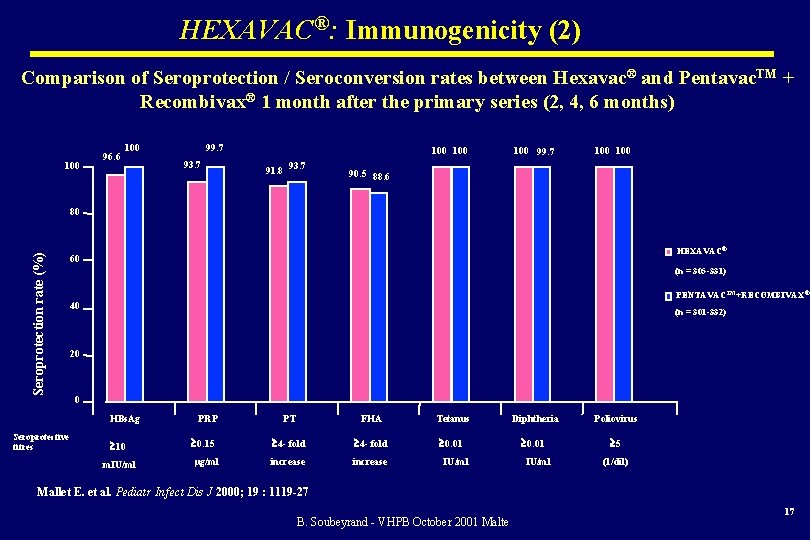
HEXAVAC®: Immunogenicity (2) Comparison of Seroprotection / Seroconversion rates between Hexavac® and Pentavac. TM + Recombivax® 1 month after the primary series (2, 4, 6 months) 100 96. 6 100 99. 7 93. 7 100 91. 8 93. 7 100 99. 7 100 90. 5 88. 6 Seroprotection rate (%) 80 HEXAVAC® 60 (n = 305 -331) PENTAVACTM+RECOMBIVAX® 40 (n = 301 -332) 20 0 HBs. Ag Seroprotective titres 10 m. IU/ml PRP PT FHA Tetanus Diphtheria Poliovirus 0. 15 4 - fold 0. 01 5 µg/ml increase IU/ml (1/dil) Mallet E. et al. Pediatr Infect Dis J 2000; 19 : 1119 -27 B. Soubeyrand - VHPB October 2001 Malte 17
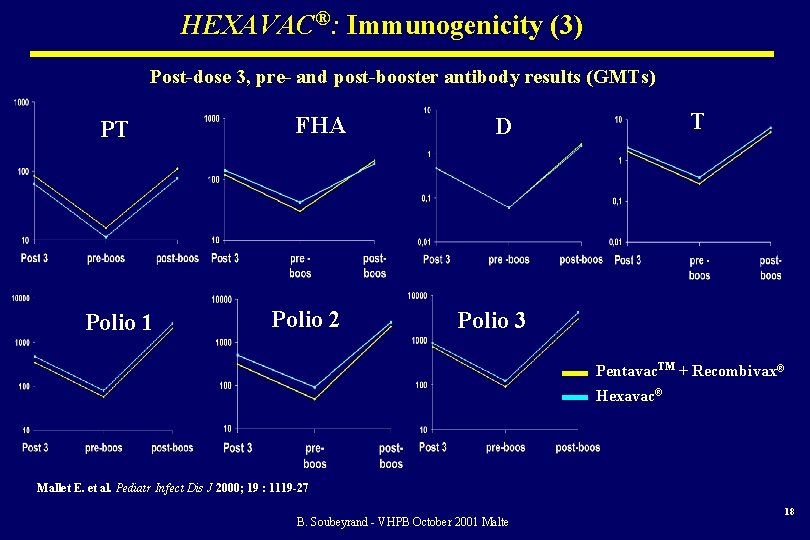
HEXAVAC®: Immunogenicity (3) Post-dose 3, pre- and post-booster antibody results (GMTs) PT Polio 1 FHA Polio 2 T D Polio 3 Pentavac. TM + Recombivax® Hexavac® Mallet E. et al. Pediatr Infect Dis J 2000; 19 : 1119 -27 B. Soubeyrand - VHPB October 2001 Malte 18
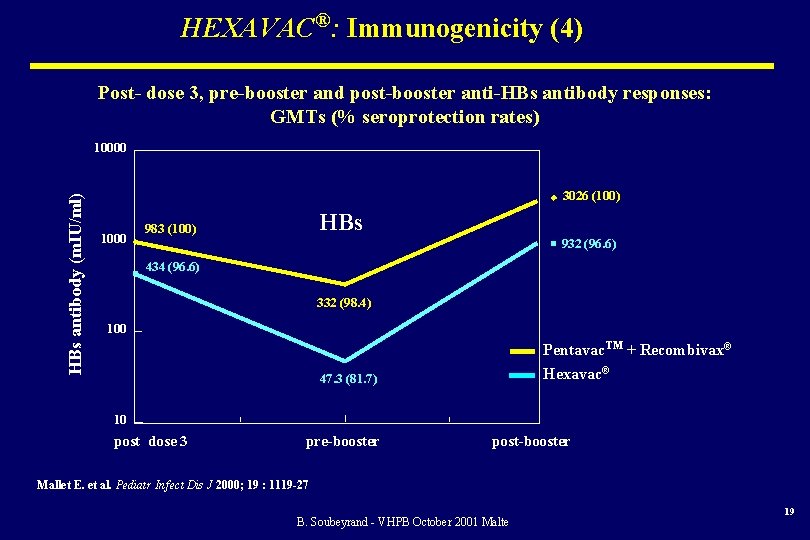
HEXAVAC®: Immunogenicity (4) Post- dose 3, pre-booster and post-booster anti-HBs antibody responses: GMTs (% seroprotection rates) HBs antibody (m. IU/ml) 10000 3026 (100) 1000 HBs 983 (100) 932 (96. 6) 434 (96. 6) 332 (98. 4) 100 Pentavac. TM + Recombivax® Hexavac® 47. 3 (81. 7) 10 post dose 3 pre-booster post-booster Mallet E. et al. Pediatr Infect Dis J 2000; 19 : 1119 -27 B. Soubeyrand - VHPB October 2001 Malte 19
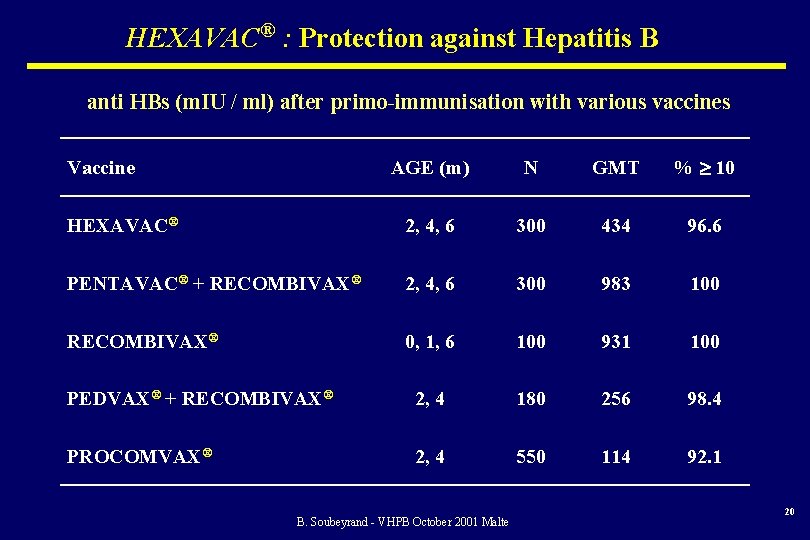
HEXAVAC® : Protection against Hepatitis B anti HBs (m. IU / ml) after primo-immunisation with various vaccines AGE (m) N GMT % 10 HEXAVAC® 2, 4, 6 300 434 96. 6 PENTAVAC® + RECOMBIVAX® 2, 4, 6 300 983 100 RECOMBIVAX® 0, 1, 6 100 931 100 PEDVAX® + RECOMBIVAX® 2, 4 180 256 98. 4 PROCOMVAX® 2, 4 550 114 92. 1 Vaccine B. Soubeyrand - VHPB October 2001 Malte 20
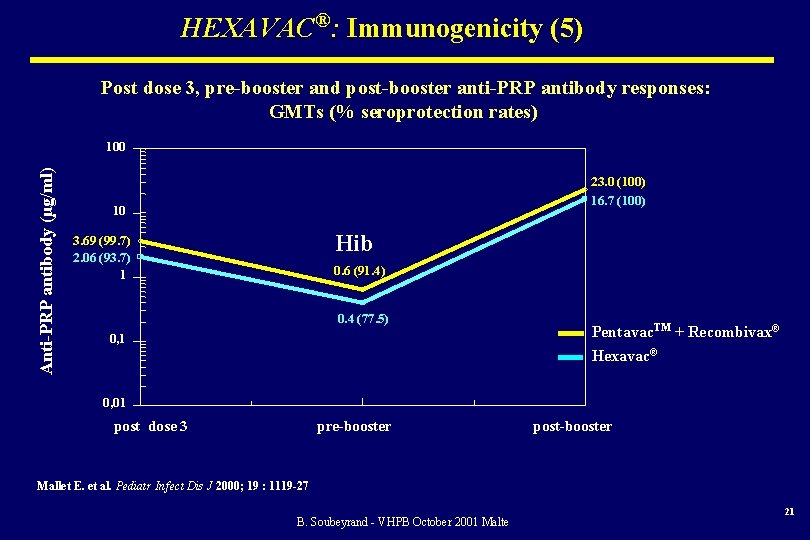
HEXAVAC®: Immunogenicity (5) Post dose 3, pre-booster and post-booster anti-PRP antibody responses: GMTs (% seroprotection rates) Anti-PRP antibody (µg/ml) 100 23. 0 (100) 16. 7 (100) 10 Hib 3. 69 (99. 7) 2. 06 (93. 7) 1 0. 6 (91. 4) 0. 4 (77. 5) 0, 1 Pentavac. TM + Recombivax® Hexavac® 0, 01 post dose 3 pre-booster post-booster Mallet E. et al. Pediatr Infect Dis J 2000; 19 : 1119 -27 B. Soubeyrand - VHPB October 2001 Malte 21
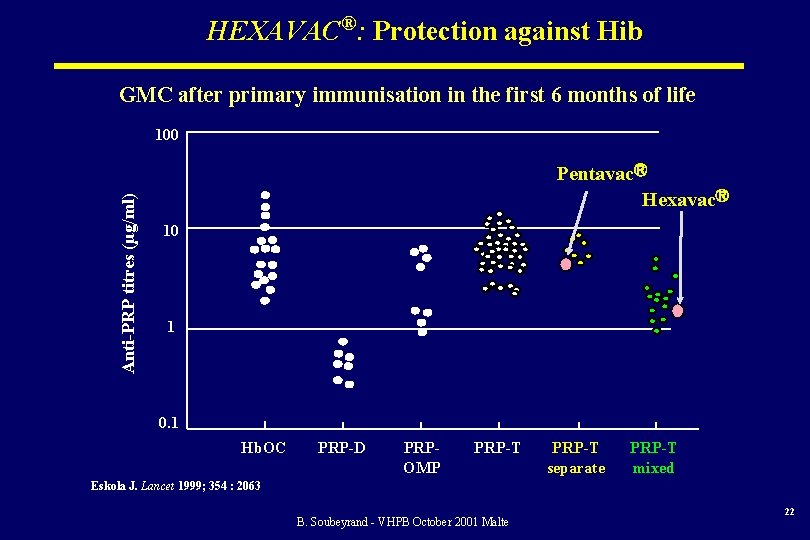
HEXAVAC®: Protection against Hib GMC after primary immunisation in the first 6 months of life Anti-PRP titres (µg/ml) 100 PentavacⓇ HexavacⓇ 10 1 0. 1 Hb. OC PRP-D PRPOMP PRP-T separate PRP-T mixed Eskola J. Lancet 1999; 354 : 2063 B. Soubeyrand - VHPB October 2001 Malte 22
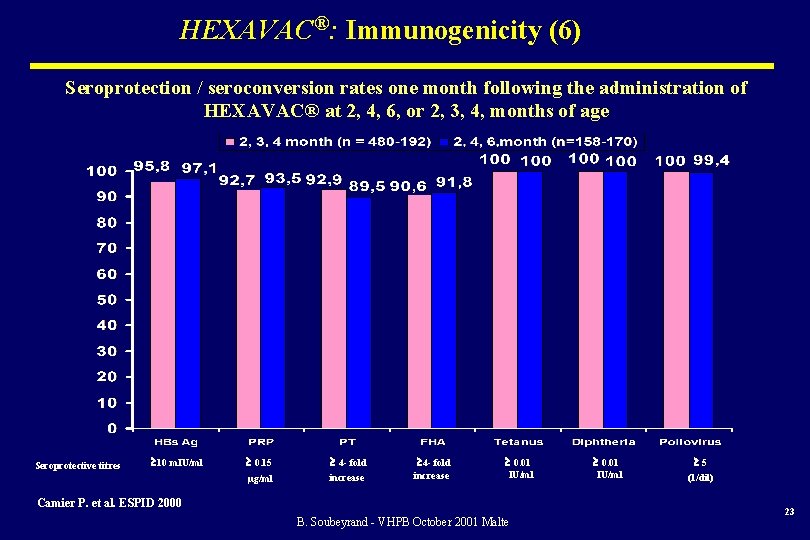
HEXAVAC®: Immunogenicity (6) Seroprotection / seroconversion rates one month following the administration of HEXAVAC® at 2, 4, 6, or 2, 3, 4, months of age Seroprotective titres 10 m. IU/ml 0. 15 4 - fold µg/ml increase 4 - fold increase 0. 01 IU/ml Camier P. et al. ESPID 2000 B. Soubeyrand - VHPB October 2001 Malte 0. 01 IU/ml 5 (1/dil) 23
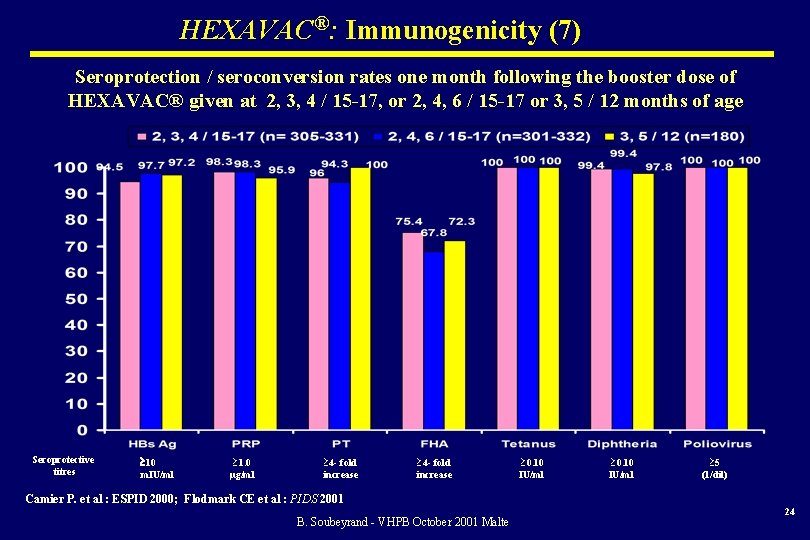
HEXAVAC®: Immunogenicity (7) Seroprotection / seroconversion rates one month following the booster dose of HEXAVAC® given at 2, 3, 4 / 15 -17, or 2, 4, 6 / 15 -17 or 3, 5 / 12 months of age Seroprotective titres 10 m. IU/ml ³ 1. 0 µg/ml ³ 4 - fold increase ³ 0. 10 IU/ml ³ 5 (1/dil) Camier P. et al : ESPID 2000; Flodmark CE et al : PIDS 2001 B. Soubeyrand - VHPB October 2001 Malte 24

CONCLUSIONS Hexavac®, the liquid combined hexavalent vaccine : l is well tolerated l provides protection and seroconversion in the range of other licensed vaccines containing a. P l immune responses to PRP and HBs are in the range of those reported with vaccines proven to be efficacious l confers excellent priming for all antigens as evidenced by strong booster responses B. Soubeyrand - VHPB October 2001 Malte 25

HEXAVAC®: Clinical development team l Investigators : E. Mallet, J. Lang, P. Camier, P. Reinert, F. Undreiner, F. Roussel, R. Lagos, M. Levine B. Belohradsky, J. Liese, S. Stojanov, G. Kanra, P. Carriere, M. Girard, M. Muller and all participating pediatricians l l Hexavac project team Aventis Pasteur Merck & Co. Aventis Pasteur MSD A. Hoffenbach E. Pines P. Fabre E. Vidor H. Salomon M. Dupuy C. Blondeau P. Mendelman J. Boslego F. Schödel H. Matthews T. Staub G. Chryssomalis M. Garnier L. Hessel And all participating babies and parents B. Soubeyrand - VHPB October 2001 Malte 26
- Slides: 26