Heterogeneous catalysis Basic principles of heterogeneous catalysis This

Heterogeneous catalysis Basic principles of heterogeneous catalysis ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Heterogena kataliza i adsorpcija �Podrazumeva promenu brzine hem reakcije pod uticajem supstance koja se hemijski ne menja u toku odigravanja katalitičke reakcije, a mehanizam odigravanja kat r-cije se zasniva na adsorpciji reaktanata na površini katalizatora. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

�Povećanje koncentracije neke komponente na graničnoj površini faza je adsorpcija. �Adsorpcijom se smanjuje Gibsova površinska energija, a smanjivaće se entropija sistema. Faza na čijoj se površini adsorbuje neka komponenta je ADSORBENS (katalizator) ili adsorbovana supstanca-ADSORBAT ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Fizička i hemijska adsorpcija • Osobine katalizatora bitne za adsorpciju odnose se na njegovo površinsko stanje. To su : hemijski sastav, struktura, razvijenost i energetrski reljef površine, veličina, oblik, raspored pora na čijoj se površini odigrava adsorpcija. • Osobine reakatanata koji je ADSORBAT su vezane za hemijski sastav molekula, veličinu, prostornu građu, polarnost i energ stanje molekula. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

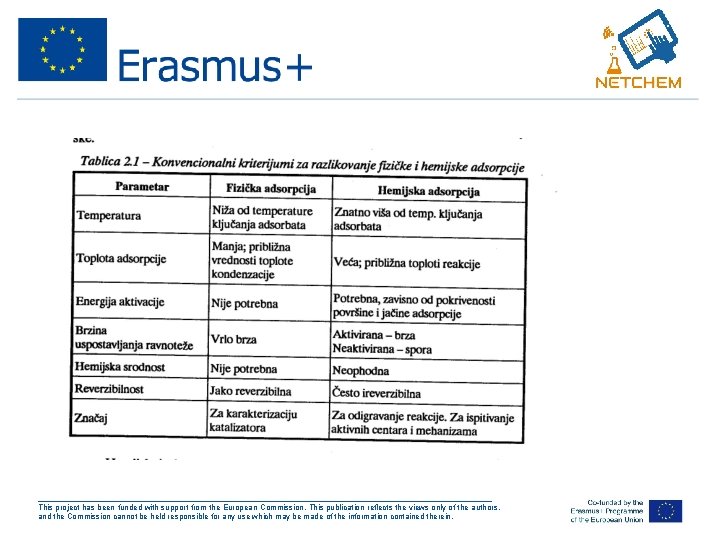
• Fizička adsorpcija se uspostavlja putem Van der Valsovih sila, relativno je slaba adsorpcija. • Pr: adsorpcija N 2 na koksu, njena toplota je mala oko 40 k. J/mol. • U slučaju fizičke adsorpcije može nastati višemolekulski sloj adsorbovane supstance na površ adsorbensa što predstavlja slučaj VIŠESLOJNE ADSORPCIJE. • Fizička adsorpcija je reverzibilan proces, uspostavlja se ravnoteža između adsorbensa i adsorbata ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• Ukoliko između molekula adsorbovane supstance i adsorbensa dolazi do građenja neke vrste jedinjenja u tom slučaju govorimo o HEMISORPCIJI. • Adsorpciona veza se uspostavlja uz učešće valentnih e- pa je slična hem vezi. Toplota hemisorpcije je reda veličine 200 -500 k. J/mol ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
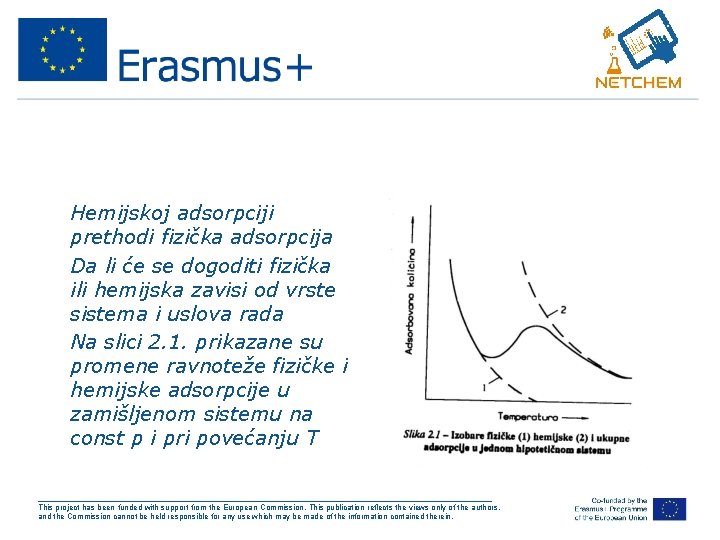
• Hemijskoj adsorpciji prethodi fizička adsorpcija • Da li će se dogoditi fizička ili hemijska zavisi od vrste sistema i uslova rada • Na slici 2. 1. prikazane su promene ravnoteže fizičke i hemijske adsorpcije u zamišljenom sistemu na const p i pri povećanju T ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Fizička adsorpcija • Disperzione sile-zastupljene od Vandervalsovih sila • Privlačenje trenutno indukovanih dipola • Potencijalna E ovog privlačenja Ud zavisi od polarizabilnosti samih atoma (konstanta C) i od rastojanja r • Ud(r)= - C/r 6 ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• Sa približavanjem adsorbata površini raste privlačno dejstvo do optimalnog rastojanja r 0 • Posle dolazi dejstvo odbojnih sila • U slučaju višeslojne adsorpcije privlačno dejstvo najjače je u prvom sloju. • Kriva potencijalne E menja se u zavisnosti od rastojanja ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• Potencijalna energija adsorpcije zavisi i od položaja na površini adsorbensa na kome se odigrava adsorpcije i od prirode sila i rastojanja između adsorbata i adsorbensa ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• Mesta na površini adsorbensa mogu se razlikovati u energiji iz razloga: • - nehomogenosti hemijskog sastava površine • - defekata kristalne rešetke • - zbog naprezanja u materijalu nastalih tokom obrade adsorbensa • Nehomogenost izazivaju nečistoće i primese na adsobensu ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
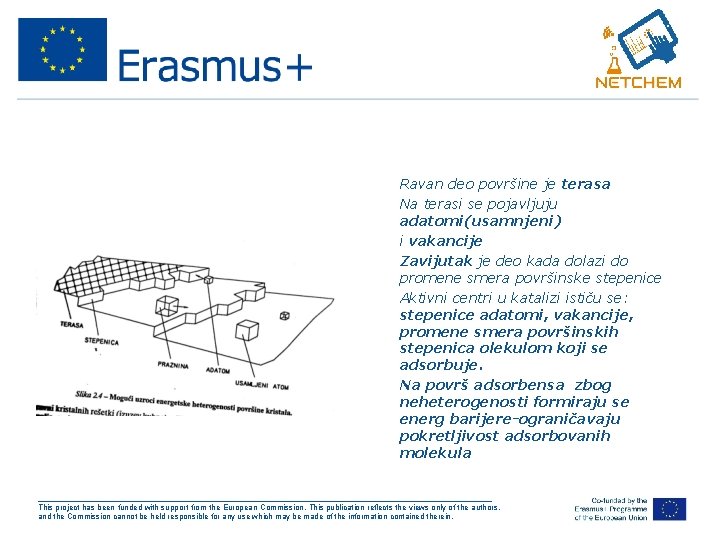
• • • Ravan deo površine je terasa Na terasi se pojavljuju adatomi(usamnjeni) i vakancije Zavijutak je deo kada dolazi do promene smera površinske stepenice Aktivni centri u katalizi ističu se: stepenice adatomi, vakancije, promene smera površinskih stepenica olekulom koji se adsorbuje. Na površ adsorbensa zbog neheterogenosti formiraju se energ barijere-ograničavaju pokretljivost adsorbovanih molekula ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Hemijska adsorpcija • Fizička adsorpcija prethodi hemijskoj • Ukoliko između aktivnih centara adsorpcije i molekula adsorbata postoji hem srodnost. • Hemisorpcija može biti: jonska, kovalentna, koordinativno kovalentna i vodonična. • Hemisorptivna veza slabija od hemijske veze. • Odigrava se između molekula adsorbata i cele površine kristala. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• U kovalentnoj vezi oba adsorbens i adsorbat učestvuju sa po jednim e- u formiranju el para. • U koordinativno-kovalentnoj vezi el par se nalazi u hibridizovanoj vezujućoj orbitali između adsorbata i površine • Potiče ili od adsorbata ili od površine • Jonska veza-prenosom e- između adsorbata i defekata kristalne rešetke adsorbensa. • Vodonična veza-preko protona koji dele među sobom po jedan O u adsorbatu i na površini adsorbensa ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• Joni adsorbensa kreću se normalno na ravanrelaksacija • Joni adsorbensa kreću se paralelno-rekonstrukcija • Relokacija-menjaju pravac. • Međusobno odbijanje adsorbovanih atoma utiče da se adsorpcija ostvaruje na svakom drugom aktivnom centru. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

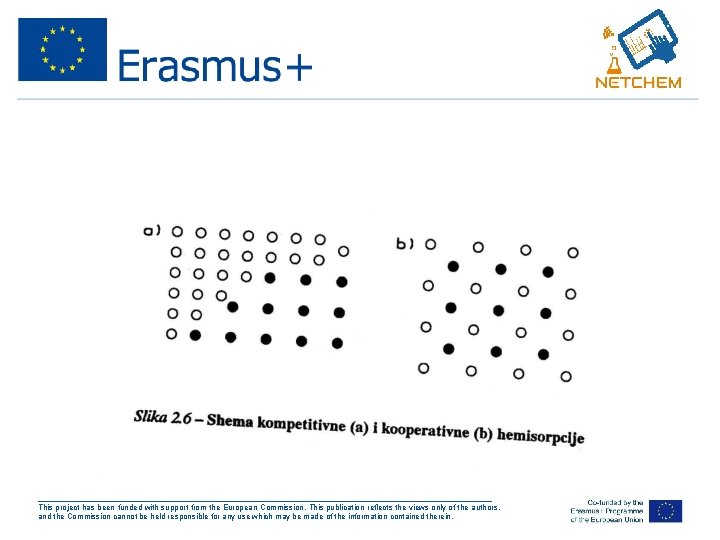
• Adsorpcijom raznih vrsta na površini-koadsorpcija (A i B): A-A, B-B i A-B-moguce interakcije • Kompetitivna adsorpcija čestice se odbiju • Kooperativna adsorpcija čestice se privlače (slika 2. 6 Putanov) ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Energija aktivacije adsorpcije • Adsorpcija-spontan proces teče uz oslobađanje E. • Ova E nije dovoljna za stvaranje jake hem veze na adsorpcionim centrima kao ni za pojave vezane za adsorpciju. • To je disocija-molekuli koji se hemisorbuju po disocijativnom mehanizmu. • Da bi mol disosovao u gasnoj fazi treba velika E. • Ako se istovremeno odigravaju adsorpcija i disocija onda se E adsorpcije nadoknađuje se E disocijacije. • Ovo se prikazuje Lenard-Džonsovim dijagramom ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
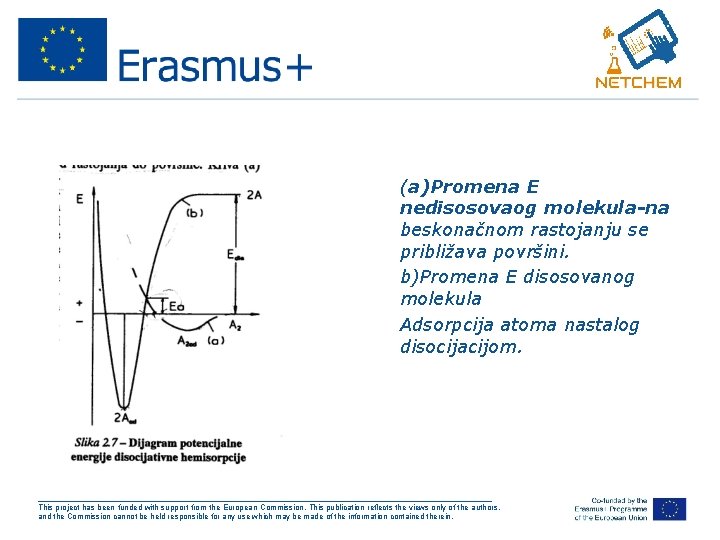
• (a)Promena E nedisosovaog molekula-na beskonačnom rastojanju se približava površini. • b)Promena E disosovanog molekula • Adsorpcija atoma nastalog disocijacijom. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Toplota adsorpcije • Količina toplote koja se oslobodi pri adsorpciji 1 mola posmatrane supstance. • Poznavajući je dolazi se do podataka o prirodi i jačini veza između a-bensa i a-bata, o osobinama površine a-bensa, o meh adsorpcije i o načinu odigravanja kat procesa. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

• Razlika između molarne entalpije neadsorbovanog gasa i parcijalne molarne entalpije adsorbovanog gasa qst je izosterna toplota adsorpcije. • (dlnp/d. T)=(Hq-Hs)/RT 2= qst/RT 2 ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Adsorpcione izoterme • Za izračunavanje količine adsorbovane supstance na graničnoj površini faza koriste se jednačine koje se nazivaju adsorpcione izoterme. • Postoji više izotermi i to: • 1. Lengmuirova– ova adsorpciona izoterma • 2. Freundlichova • 3. Gibbs • 4. Tjomkinova ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

Author, Editor and Referee References This remote access laboratory was created thanks to work done primarily at University of Niš. Contributors to this material were: ________ Refereeing of this material was done by: ___________ Editing into NETCHEM Format and onto NETCHEM platform was completed by: __________________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, ___________________________________________________ andhas the been Commission be held responsible for any use which may made of the information contained This project funded cannot with support from the European Commission. Thisbe publication reflects the views only of therein. the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.

References and Supplemental Material The NETCHEM platform was established at the University of Nis in 2016 -2019 through the Erasmus Programme. Please contact a NETCHEM representatives at your institution or visit our website for an expanded contact list. The work included had been led by the NETCHEM staff at your institution. ___________________________________________________ This project has been funded with support from the European Commission. This publication reflects the views only of the authors, ___________________________________________________ andhas the been Commission be held responsible for any use which may made of the information contained This project funded cannot with support from the European Commission. Thisbe publication reflects the views only of therein. the authors, and the Commission cannot be held responsible for any use which may be made of the information contained therein.
- Slides: 25