Hess Law and enthalpy cycles using enthalpy of
























- Slides: 29

Hess’ Law and enthalpy cycles using enthalpy of formation Monday, June 14, 2021

Hess’ Law using enthalpy of formation Objective: to recall and use Hess’s Law for enthalpy of formation data Success criteria: • Recall Hess’ Law • Interpret an enthalpy cycle using Hess’ Law • Construct an enthalpy cycle using Hess’ Law, formation data • To calculate an enthalpy change from formation data using Hess’s Law • Know reasons why the enthalpy change cannot always be measured directly

Starter 1. Define Δf. H 2. What are standard conditions? 3. What is the value of Δf. H for any element?

Using Δf Ө H As well as burning things, chemists love making things! So we also have huge tables of Δ Δf. H Ө including very many of the most common chemicals. In a reaction which we cannot measure directly, Δf. HӨ can also give us a link between reactants and products.

• Δ Δf. HӨ is the energy to make something from its elements, so the arrows will be going in the opposite direction to enthalpies of combustion. • Route 1: B + A Actually you • Route 2: C might see it • Hess’ law tells us that B + A = C as: • We are trying to find A: A = C - B A = -B + C

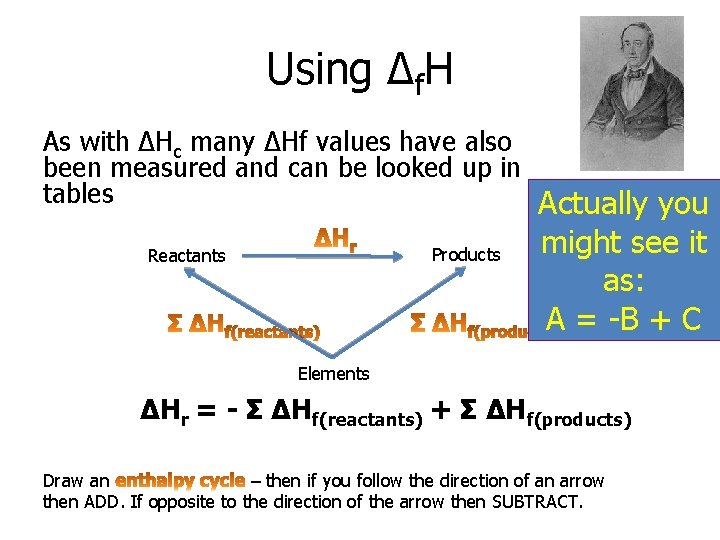
Using Δf. H As with ΔHc many ΔHf values have also been measured and can be looked up in tables Actually you Products Reactants might see it as: A = -B + C Elements ΔHr = - Σ ΔHf(reactants) + Σ ΔHf(products) Draw an – then if you follow the direction of an arrow then ADD. If opposite to the direction of the arrow then SUBTRACT.

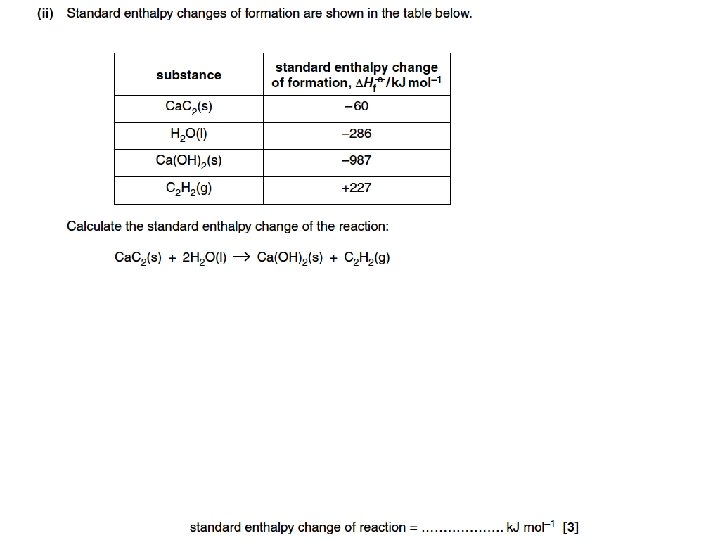
Example on worksheet

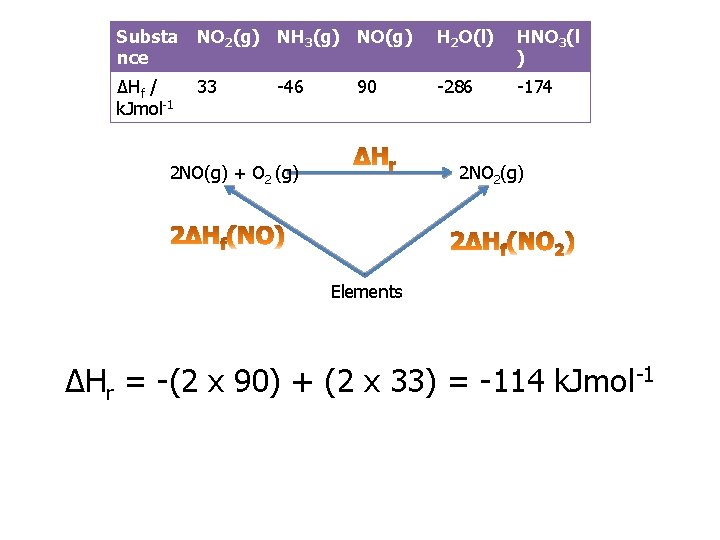
Practice question 1 a You are provided with the following enthalpy changes of formation Substa nce NO 2(g) NH 3(g) NO(g) H 2 O(l) HNO 3(l ) ΔHf / k. Jmol-1 33 -286 -174 -46 90 Determine the enthalpy change for the following reaction: 2 NO (g) + O 2(g) 2 NO 2(g)

Substa nce NO 2(g) NH 3(g) NO(g) H 2 O(l) HNO 3(l ) ΔHf / k. Jmol-1 33 -286 -174 -46 90 2 NO(g) + O 2 (g) 2 NO 2(g) Elements ΔHr = -(2 x 90) + (2 x 33) = -114 k. Jmol-1

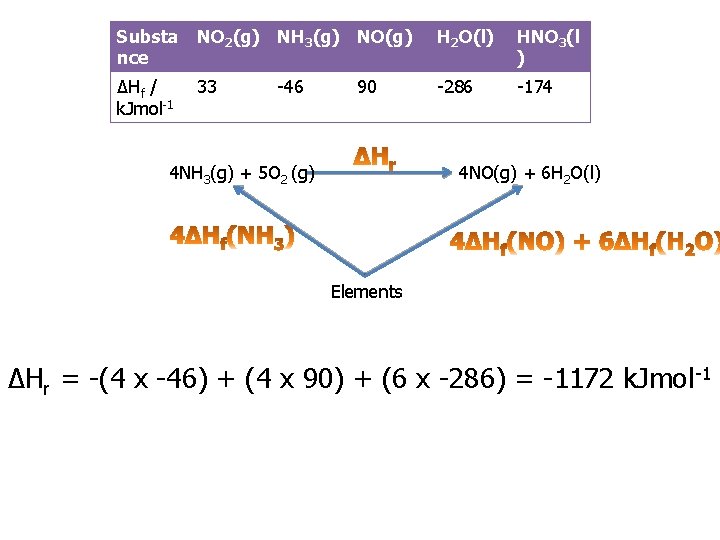
Practice question 1 b You are provided with the following enthalpy changes of formation Substa nce NO 2(g) NH 3(g) NO(g) H 2 O(l) HNO 3(l ) ΔHf / k. Jmol-1 33 -286 -174 -46 90 Determine the enthalpy change for the following reaction: 4 NH 3 (g) + 5 O 2(g) 4 NO(g) + 6 H 2 O(l)

Substa nce NO 2(g) NH 3(g) NO(g) H 2 O(l) HNO 3(l ) ΔHf / k. Jmol-1 33 -286 -174 -46 90 4 NH 3(g) + 5 O 2 (g) 4 NO(g) + 6 H 2 O(l) Elements ΔHr = -(4 x -46) + (4 x 90) + (6 x -286) = -1172 k. Jmol -1

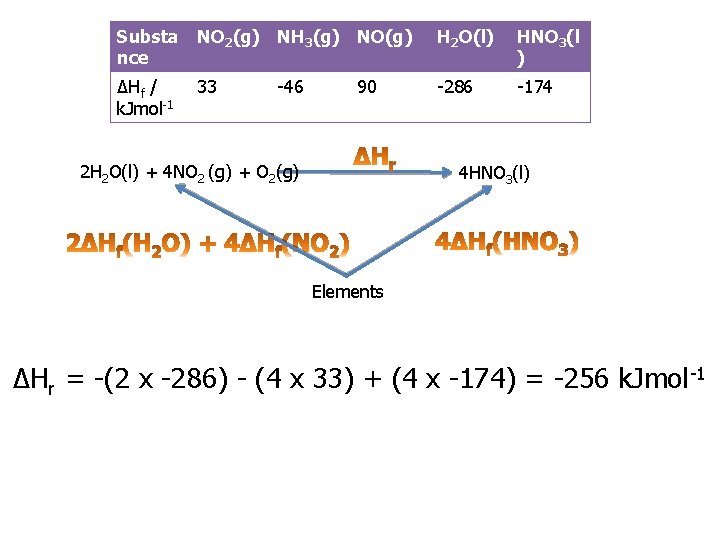
Practice question 1 c You are provided with the following enthalpy changes of formation Substa nce NO 2(g) NH 3(g) NO(g) H 2 O(l) HNO 3(l ) ΔHf / k. Jmol-1 33 -286 -174 -46 90 Determine the enthalpy change for the following reaction: 2 H 2 O (l) + 4 NO 2(g) + O 2(g) 4 HNO 3(l)

Substa nce NO 2(g) NH 3(g) NO(g) H 2 O(l) HNO 3(l ) ΔHf / k. Jmol-1 33 -286 -174 -46 90 2 H 2 O(l) + 4 NO 2 (g) + O 2(g) 4 HNO 3(l) Elements ΔHr = -(2 x -286) - (4 x 33) + (4 x -174) = -256 k. Jmol -1

Other enthalpy cycles • In your AS course you have to be able to construct and use cycles that use and data. • The same principles can be applied to any energy cycle provided it is constructed correctly. , and you can’t go wrong.

Exam questions (Edexcel) printed as booklet Hess Cycles both and enthalpy of combustion to finish for homework

Question 1 from old OCR January and June 2010





Question 2




Mark schemes

Mark scheme Q 1 – cover until you have answered

Mark scheme

Mark scheme Q 2

Mark scheme