Hereditary Colorectal Cancer Prepared by June C Carroll

Hereditary Colorectal Cancer Prepared by: June C Carroll MD, CCFP, FCFP Sydney G. Frankfort Chair in Family Medicine Mount Sinai Hospital, University of Toronto Andrea Rideout MS, CGC, CCGC Certified Genetic Counsellor Project Manager – The Genetics Education Project Sean Blaine BSc, MD, CCFP Mount Sinai Hospital, University of Toronto Stratford, Ontario Funded by: Ontario Women’s Health Council Version: January 2010

Acknowledgments n Reviewers: Members of The Genetics Education Project (see slide 51) + Kara M. Semotiuk, MS, (C)CGC Genetic Counsellor Heidi Rothenmund, MS, (C)CGC Genetic Counsellor Familial GI Cancer Registry, Mount Sinai Hospital n Funded by: Ontario Women’s Health Council as part of its funding to The Genetics Education Project * Health care providers must use their own clinical judgment in addition to the information presented herein. The authors assume no responsibility or liability resulting from the use of information in this presentation.

Outline Sporadic verses familial cancer n Hereditary colorectal cancer syndromes n Referral guidelines n Benefits, risks and limitations of genetic testing n Management n Case examples n

Cancer All cancer involves changes in genes…. Threshold effect: n During mitosis & DNA replication ¨ mutations occur in the cell’s genetic code Mutations are normally corrected by DNA repair mechanisms n If repair mechanism or cell cycle regulation is damaged n ¨ Cell accumulates too → reaches ‘threshold’ → tumour development many mutations

Sporadic Cancer n All cancer arises from changes in genes…. ¨ But NOT all cancer is inherited ¨ Most CRC is sporadic ~75 – 80% n Due to acquired mutations throughout a person’s lifetime: Causes unknown – multifactorial ¨ Interaction of many factors: age, environment, lifestyle, chance, unknown factors ¨ ¨ Sporadic cancer generally has a later onset

Clustering of Cancer in Families n ~6% lifetime risk of CRC in general population ~20% of people with CRC have a family history: n ~15% of CRC is familial: n ¨ ¨ ¨ Environmental factors Chance Undiscovered gene mutation ¨ Generally n not eligible for genetic testing ~5% of CRC cancer is hereditary ¨ Caused by an inherited gene mutation that puts them at increased risk for cancer n n Majority is Lynch syndrome/HNPCC (Hereditary Non-Polyposis Colorectal Cancer) Small fraction is Familial Adenomatous Polyposis (FAP) or other rare cancer syndromes ¨ May be eligible for genetic testing
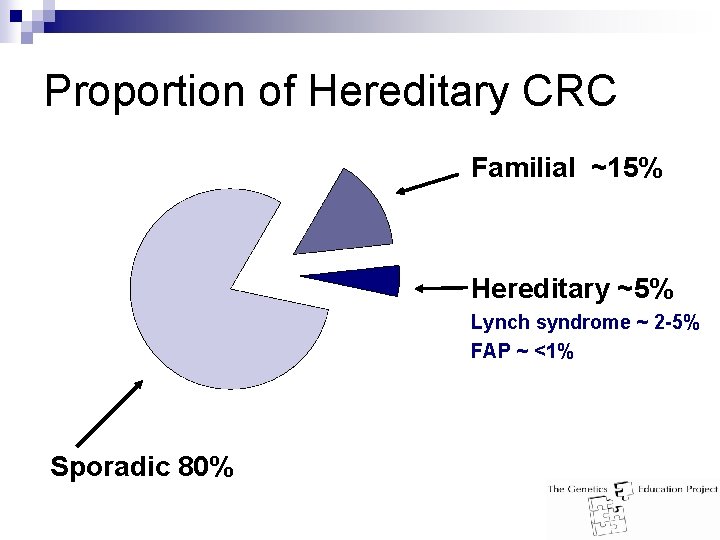
Proportion of Hereditary CRC Familial ~15% Hereditary ~5% Lynch syndrome ~ 2 -5% FAP ~ <1% Sporadic 80%
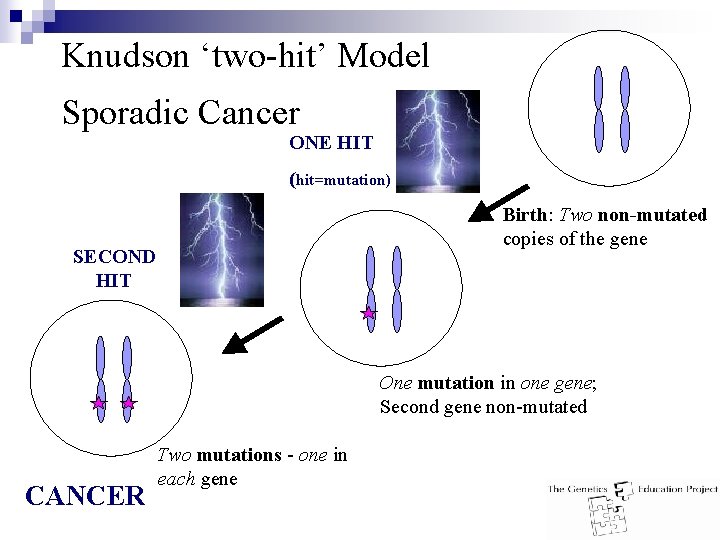
Knudson ‘two-hit’ Model Sporadic Cancer ONE HIT (hit=mutation) Birth: Two non-mutated copies of the gene SECOND HIT One mutation in one gene; Second gene non-mutated CANCER Two mutations - one in each gene
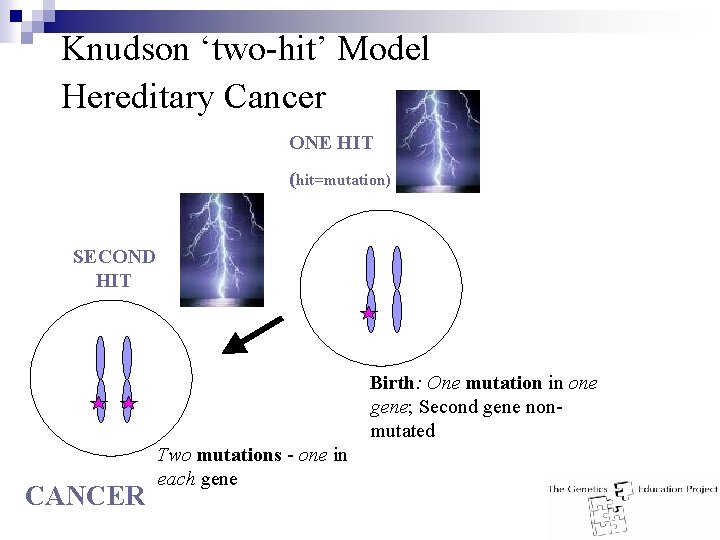
Knudson ‘two-hit’ Model Hereditary Cancer ONE HIT (hit=mutation) SECOND HIT Birth: One mutation in one gene; Second gene nonmutated CANCER Two mutations - one in each gene

Compared to sporadic cancer people with hereditary cancer have… A higher risk of developing cancer n A younger age of onset of cancer n ¨ Generally < 50 years of age Multiple primary cancers n Generally have a family history of cancer Hereditary cancer is less common in the general population than sporadic cancer n

Inherited Colorectal Cancer Two common syndromes: n Lynch syndrome ¨ Also known as Hereditary Non Polyposis Colorectal Cancer or HNPCC ¨ ~2 - 5% of colorectal cancer ¨ Prevalence of 1 in 200 - 2, 000* n Familial Adenomatous Polyposis (FAP) ¨ <1% of colorectal cancer ¨ Prevalence of 1 in 8, 000 – 14, 000* n Autosomal dominant inheritance *Prevalence depends on population
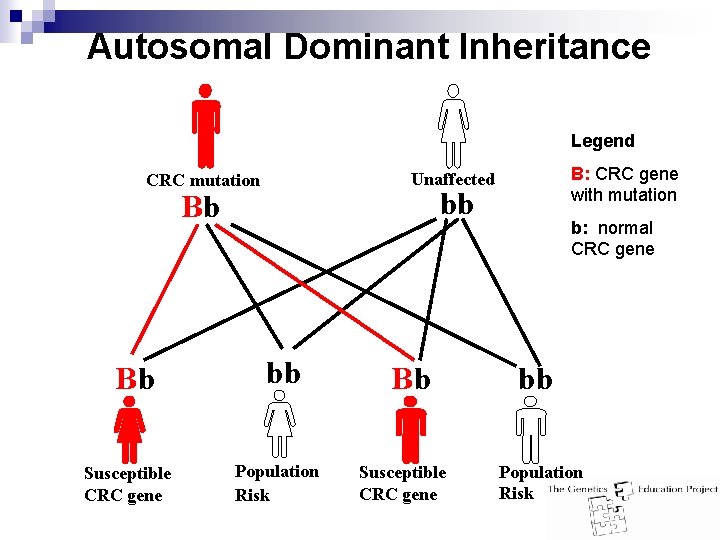
Autosomal Dominant Inheritance Legend bb Bb Bb Susceptible CRC gene B: CRC gene with mutation Unaffected CRC mutation bb Population Risk Bb Susceptible CRC gene b: normal CRC gene bb Population Risk

Colorectal cancer genes… when mutated n Lynch syndrome (HNPCC): ¨Mutations in DNA repair genes lead to an accumulation of mutations which may result in malignancy. n FAP: ¨Mutations in a tumour suppressor gene cause an increase in cell proliferation and a decrease in cell death.

Lynch syndrome (HNPCC) n Lynch syndrome is genetically heterogeneous ¨ Clinical testing available for 4 genes: MLH 1 & MSH 2 (most common), MSH 6 & PMS 2 ¨ Research testing may be available for other genes High penetrance n Characterized by: n ¨ Earlier onset than sporadic cancer ¨ More aggressive, proximal, right sided tumours ¨ Risk for extra-colonic tumours ¨ Distinct tumour pathology
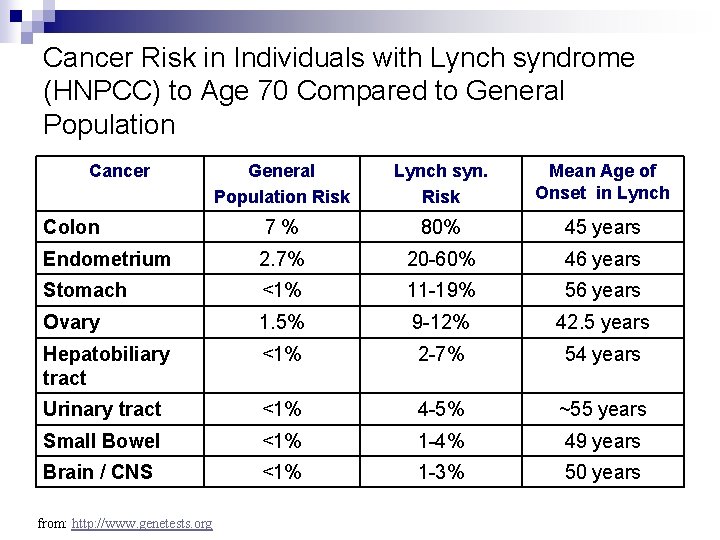
Cancer Risk in Individuals with Lynch syndrome (HNPCC) to Age 70 Compared to General Population Cancer General Population Risk Lynch syn. Risk Mean Age of Onset in Lynch Colon 7% 80% 45 years Endometrium 2. 7% 20 -60% 46 years Stomach <1% 11 -19% 56 years Ovary 1. 5% 9 -12% 42. 5 years Hepatobiliary tract <1% 2 -7% 54 years Urinary tract <1% 4 -5% ~55 years Small Bowel <1% 1 -4% 49 years Brain / CNS <1% 1 -3% 50 years from: http: //www. genetests. org

Familial Adenomatous Polyposis Chromosome 5, APC gene n High penetrance n Characterized by: n ¨ Early onset ¨ >100 adenomatous polyps ¨ Variant form: n Attenuated FAP may occur with >10 but <100 polyps.
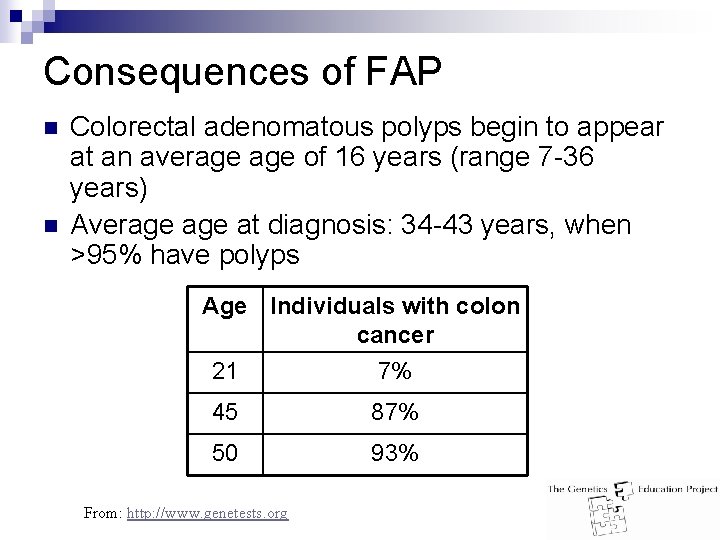
Consequences of FAP n n Colorectal adenomatous polyps begin to appear at an average of 16 years (range 7 -36 years) Average at diagnosis: 34 -43 years, when >95% have polyps Age Individuals with colon cancer 21 7% 45 87% 50 93% From: http: //www. genetests. org

Consequences of FAP n ~50 -90% develop small bowel polyps ¨lifetime risk of small bowel malignancy is 4 -12% n ~50% develop gastric polyps ¨~10% n ~10% gastric cancer develop desmoid tumours

Red Flags for hereditary colorectal cancer – consider referral to genetics n n n Multiple cases in family with Lynch syndrome/HNPCC spectrum of cancers with at least 1 relative with CRC or endometrial CA CRC at <45 years Multiple Lynch syndrome cancers in 1 family member Family member with FAP or >10 adenomatous polyps Family member with known mutation Family member with colonic adenoma or cancer with high microsatellite instability (MSI) ¨ n See extra slides following references for more information about MSI Not all who are referred will have genetic testing

Risk of Developing Colorectal Cancer Family History Relative Risk for CRC Absolute Risk of CRC by age 79 No family history 1 4% 1 FDR with CRC 2 9% >1 FDR with CRC 4 16% 1 FDR Dx <45 yrs 4 15% 1 FDR Dx CRC adenoma 2 8% From: http: //www. cancer. gov

Case n Jane - healthy 26 y. o. n Office visit for a routine pap smear and renewal of birth control pills n History: ¨Any cancer in the family? n Mother with breast cancer at 66

Case continued… n Father’s side of the family: uncle - CA ureter age 72 – uncle - CA colon age 56 – aunt - double primary: endometrial CA age 45, colon CA age 68 – 1 cousin - endometrial CA age 40 – 2 cousins - both have colon CA –
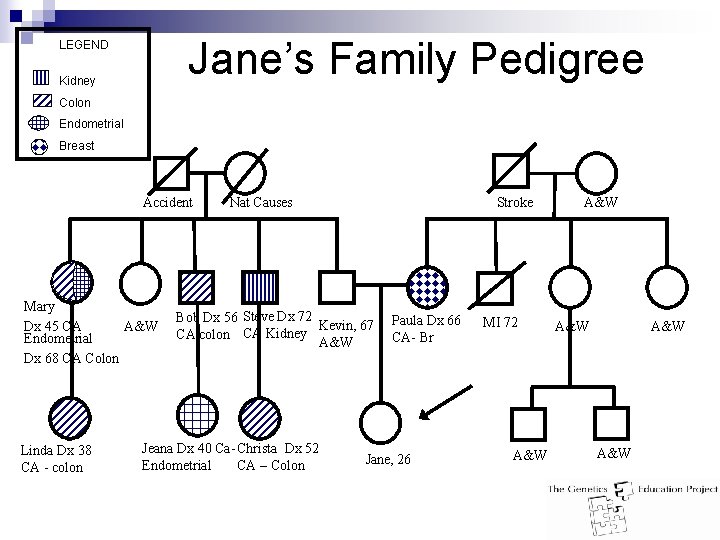
Jane’s Family Pedigree LEGEND Kidney Colon Endometrial Breast Accident Mary A&W Dx 45 CA Endometrial Dx 68 CA Colon Linda Dx 38 CA - colon Stroke Nat Causes Bob Dx 56 Steve Dx 72 Kevin, 67 CA colon CA Kidney A&W Jeana Dx 40 Ca- Christa Dx 52 Endometrial CA – Colon Paula Dx 66 CA- Br Jane, 26 MI 72 A&W A&W A&W

Jane was referred to genetics… A genetics consultation involves: n n Detailed family history information Pedigree documentation ¨ Confirmation of cancer history: pathology reports/death certificates n n Medical & exposure history Empiric risk assessment Hereditary cancer / genetic risk assessment Psychological assessment

…A genetics consultation involves: n Assessment of eligibility for genetic testing ¨ Availability of living affected relative to be tested first n Discussion of risks, benefits & limitations of test n Testing and disclosure of genetic test results ¨ May n n be months before results are available Determining patient’s thoughts about colorectal cancer - motivations for testing Screening/management recommendations

Recommendations for Jane’s family n Jane’s paternal family history is suggestive of Lynch syndrome/HNPCC. n Jane was asked to discuss genetic testing with her family members diagnosed with cancer. n Appropriate to test an affected member first. n If a mutation found in one of the Lynch syndrome genes then sequential testing of the family can be performed. n If Jane’s family declines genetic testing then family members should follow high risk screening recommendations for CRC. Colonoscopy q 1 -2 years; consider referral to a GYN to discuss endometrial cancer screening n

Results from Genetic Testing n Positive ¨ Deleterious n mutation identified Negative ¨ Interpretation differs if a mutation has previously been identified in the family n n n Mutation known – true negative Mutation unknown – uninformative Variant of unknown significance ¨ Significance will depend on how variant tracks through family, i. e. is variant present in people with disease? ¨ Can use software to predict functional significance ¨ Check with lab: ? reported previously

Risks/Benefits/Limitations of genetic testing Positive test result Potential Benefits: n n Clinical intervention may improve outcome Family members at risk can be identified Positive health behaviour can be reinforced Reduction of uncertainty Potential Risks: n n n Adverse psychological reaction Family issues/distress Uncertainty -incomplete penetrance Insurance/job discrimination Confidentiality issues Intervention may carry risk

Risks/Benefits/Limitations of genetic testing? True Negative test result Potential Benefits: n Avoidance of unnecessary clinical interventions n Emotional - relief n Children can be reassured Potential Risks: n Adverse psychological reaction (i. e. survivor guilt) n Dysfunctional family dynamics n Complacent attitude to health

Risks/Benefits/Limitations of genetic testing? Uninformative test result Potential Risks: Potential Benefits: n Continue clinical n Future research may inventions which may clarify test results carry risks n Importance of positive n Complacent attitude to health behaviour can health be reinforced n Uncertainty n Some relief n Continued anxiety

What is the benefit of genetic testing? Can anything be done to change risk /outcome? n Patients with Lynch syndrome/HNPCC: ¨ Colonoscopy beginning age 20 -25 or 10 years younger than youngest CRC or adenomatous polyp diagnosis, whichever comes first ¨ Subsequent n colonoscopy every 1 -2 years Category of evidence III, grade C Vasen et al. J Med Genet. 2007; 44: 353 -362.

What is the benefit of genetic testing? Can anything be done to change risk /outcome? n Evidence for screening in Lynch syndrome/HNPCC: Cohort study of CRC screening – 15 yr F/U n Subgroup of Lynch syndrome carriers n CRC in 8/44 with colonoscopy q 3 years vs. 19/46 controls ( p=0. 02) n RR of CRC = 0. 44 (95% CI 0. 2 -0. 9) n RR of death = 0. 35 (95% CI 0. 1 -0. 99) n 15 yr survival 92% vs. 74% n Jarvinin et al Gastroenterology 2000

What is the benefit of genetic testing? Can anything be done to change risk /outcome? n Lynch syndrome/HNPCC gynecological cancers: ¨ Little evidence re GYN cancer screening ¨ Educate re symptoms of endometrial & ovarian cancer ¨ Beginning age 30 -35 consider 1 -2 years: n n Gynecological examination Trans-vaginal ultrasound +/- aspiration biopsy ¨ n Category of evidence III, grade C CA 125 ¨ Consider prophylactic hysterectomy and bilateral salpingooophorectomy (BSO) n Grade C Vasen et al. J Med Genet. 2007; 44: 353 -362

n n n Lynch syndrome - Evidence for screening for endometrial cancer (EC): Finnish HNPCC registry – chart review for 10 years N=175 EC screening; N=83 no EC screening Screening consisted of: GYN exam (100%), trans-vaginal U/S (94%), endometrial biopsy (74%) Median screening interval 3 years/ Median age 52 years Screening group: 14 cases of EC detected ¨ ¨ n No screening group: ¨ ¨ n n 11 cases by screening alone 2 cases by manifesting symptoms (interval cancers) 1 case occult cancer found at the time of hysterectomy 0 EC deaths Number of EC cases not reported 6 EC deaths Survival curves: 100% screening group; 92% no screening Differences b/w survival curves not significant (P=0. 4) Renkonen-Sinisalo Int J Cancer 2006: 120: 821 -824

What is the benefit of genetic testing? Can anything be done to change risk /outcome? n Lynch syndrome – evidence for risk reducing surgery ¨ ¨ Chart Review of HNPCC mutation positive women Hysterectomy N = 61 n ¨ No hysterectomy N = 254 n n ¨ No cases of ovarian cancer No BSO N=223 n n ¨ 69 cases of endometrial cancer – 33% P<0. 001 Bilateral salpingo-oophorectomy (BSO) N=47 n ¨ No cases of endometrial cancer 12 cases of ovarian cancer – 5. 5% P=0. 09 No peritoneal cancers in the study period Schmeler et al. NEJM 2006; 354261 -269.

What is the benefit of genetic testing? Can anything be done to change risk /outcome? n n Lynch syndrome/HNPCC screening for other cancers: ONLY if there is a family history of the type of cancer listed below - controversial ¨ Gastric cancer n Gastroduodenoscopy q 1 -2 years beginning age 30 – 35 years ¨ Urinary tract cancer n Renal U/S + urine cytology q 1 -2 years beginning age 30 to 35 years ¨ Other cancers n Screen as per family history of skin, small bowel, pancreaticobiliary cancers

What is the benefit of genetic testing? Can anything be done to change risk/outcome? n Patients with FAP: ¨Sigmoidoscopy every 1 -2 years beginning at age 10 to 12 n subsequent colonoscopy every 1 -2 years ¨Colonoscopy once polyps are detected ¨Colectomy ¨Annual colonoscopy if colectomy is delayed more than 1 year after polyps emerge

Management of Mutation Carriers Consider… n Psychosocial support to assist with: ¨ Adjusting to new information most adjust within 3 -6 months n subset remain psychologically distressed n ¨ Making decisions regarding management ¨ Addressing family issues, self concept, body image ¨ Dealing with future concerns n Referral to support groups

Management of Mutation Carriers Consider… n Additional psychosocial support may be needed for high risk individuals such as those with: ¨ History ¨ Poor of depression/anxiety coping skills ¨ Inadequate ¨ Multiple ¨ Loss social support / conflict in the family losses in the family of parent at a young age ¨ Recent ¨ Multiple loss surgical procedures

Resources n The National Cancer Institute: ¨ http: //www. cancer. gov/ Gene Tests: http: //www. genetests. org n Colon Cancer Alliance: n ¨ http: //www. ccalliance. org/ Canadian Cancer Society: www. cancer. ca n Cancer Genetics Support Group of Canada (CHGSGC): n Contact Name: Nancy Schofield, President 16 Redford Road Canada London, ON N 5 X 3 V 5 Email: wschofield@odyssey. on. ca

Case Examples

Assessing the Risk for Hereditary CRC Using the Canadian Cancer Society triage card (below), what category of risk do the following family histories fit into?
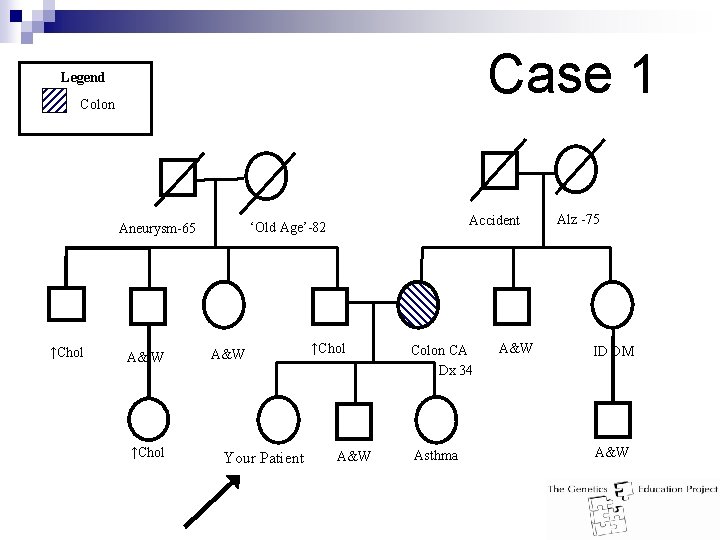
Case 1 Legend Colon ↑Chol A& W ↑Chol Accident ‘Old Age’-82 Aneurysm-65 A&W Your Patient ↑Chol A&W Colon CA Dx 34 Asthma A&W Alz -75 ID DM A&W
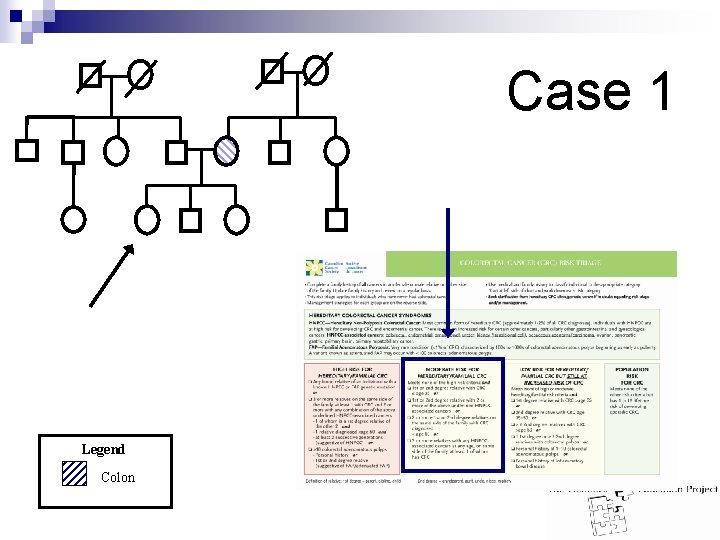
Case 1 Legend Colon

Case 1 Answer: n Moderate risk for hereditary CRC n 1 st or 2 nd degree relative with CRC ≤ 35 n Management: ¨ Offer referral to hereditary CRC/Genetics Clinic ¨ Colonoscopy q 3 -5 years starting 10 years younger than youngest CRC diagnosis ¨ Educate patient about symptoms of endometrial cancer
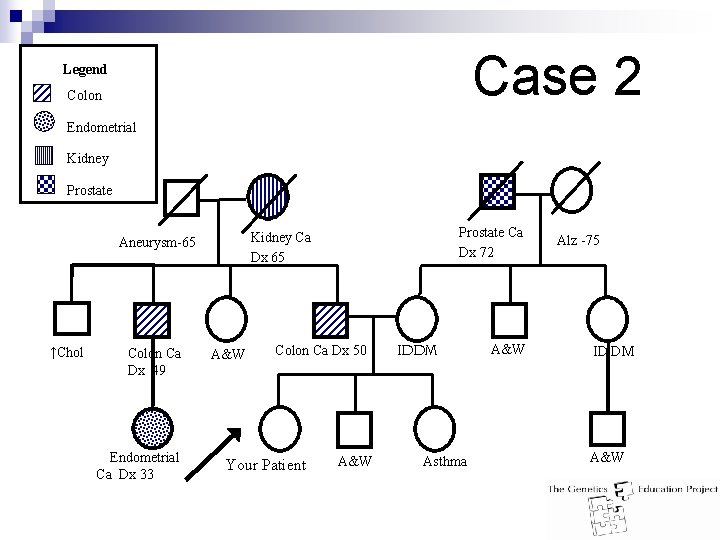
Case 2 Legend Colon Endometrial Kidney Prostate ↑Chol Colon Ca Dx 49 Endometrial Ca Dx 33 Prostate Ca Dx 72 Kidney Ca Dx 65 Aneurysm-65 A&W Colon Ca Dx 50 Your Patient A&W IDDM Asthma A&W Alz -75 ID DM A&W
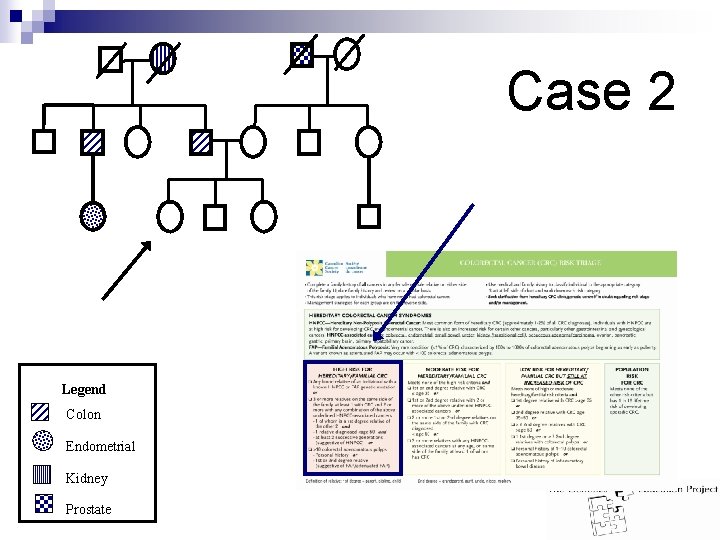
Case 2 Legend Colon Endometrial Kidney Prostate

Case 2 Answer: n n High risk for hereditary CRC ≥ 3 relatives on the same side of the family, at least 1 CRC and ≥ 2 with any combination of Lynch syndrome-associated cancer AND ¨ 1 is a 1 st degree relative of the other 2 and ¨ 1 relative diagnosed <50 and ¨ At least 2 successive generations (suggestive of Lynch syndrome) n Management: ¨ Offer referral to hereditary CRC/genetics clinic ¨ Colonoscopy q 1 -2 years beginning age 20 or 10 years younger than youngest CRC diagnosis ¨ Educate patient about symptoms of endometrial cancer
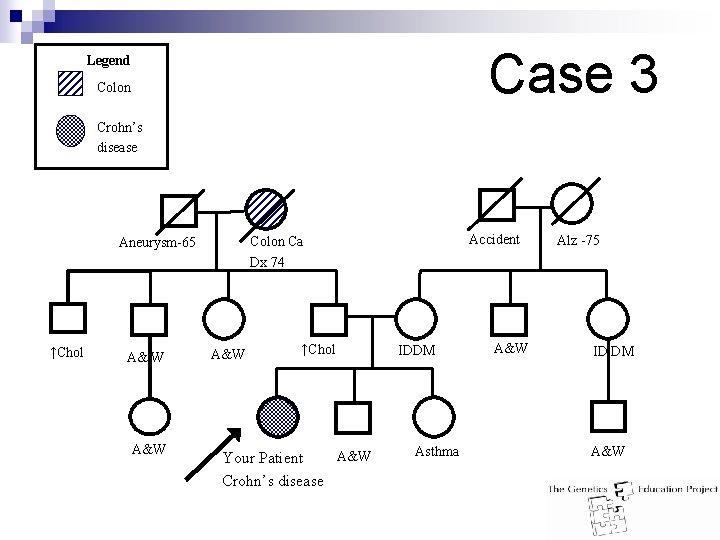
Case 3 Legend Colon Crohn’s disease ↑Chol A& W A&W Accident Colon Ca Dx 74 Aneurysm-65 A&W ↑Chol A&W Your Patient Crohn’s disease IDDM Asthma A&W Alz -75 ID DM A&W
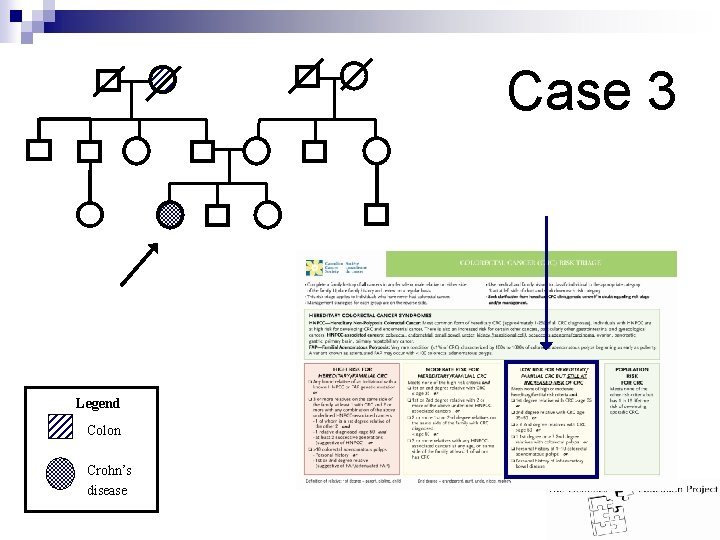
Case 3 Legend Colon Crohn’s disease

Case 3 Answer: n Low risk for Hereditary CRC but still at increased risk of CRC n Personal history of inflammatory bowel disease n Management: ¨ Seek advice from gastroenterologist or surgeon for individuals with inflammatory bowel disease.
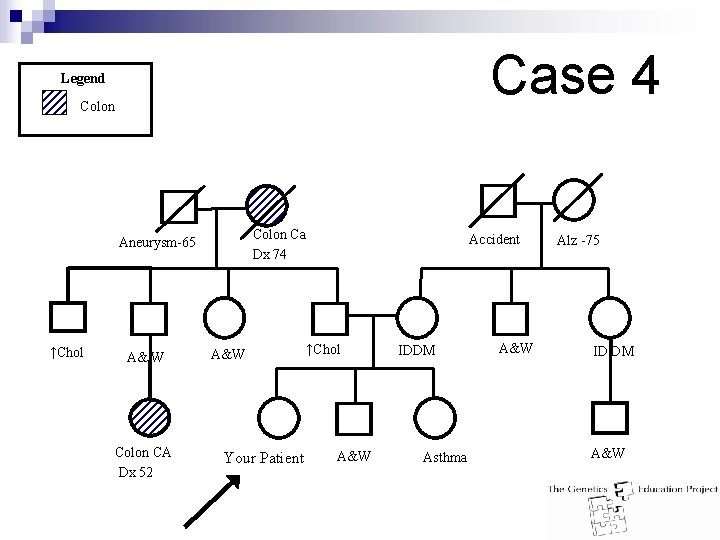
Case 4 Legend Colon Ca Dx 74 Aneurysm-65 ↑Chol A& W Colon CA Dx 52 A&W Your Patient Accident ↑Chol A&W IDDM Asthma A&W Alz -75 ID DM A&W
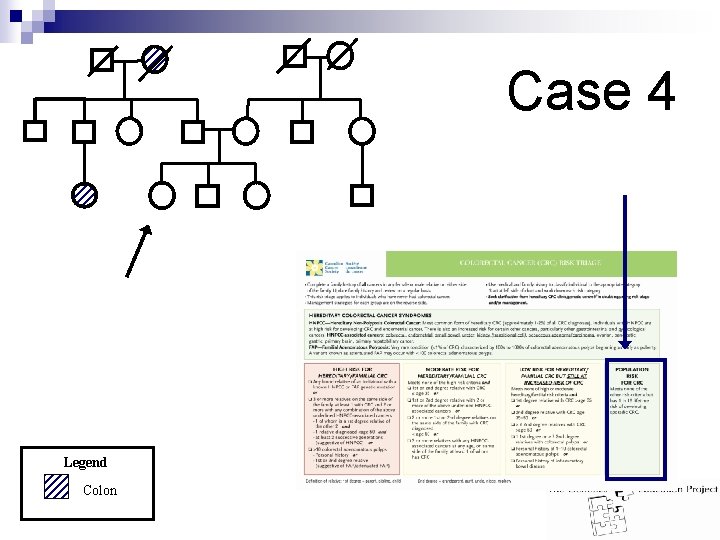
Case 4 Legend Colon

Case 4 Answer: n n A = Good evidence B = Fair evidence I = Insufficient evidence Population risk Meets none of the other risk criteria Still has a 1 in 16 lifetime risk of sporadic CRC Management: ¨ Beginning at age 50: n Annual or biennial fecal occult blood testing (FOBT)A OR n Flexible sigmoidoscopy q 5 years. B OR n FOBT + flexible sigmoidoscopy q 5 years. I OR n Double contrast barium enema q 5 years OR n Colonoscopy q 10 years. I
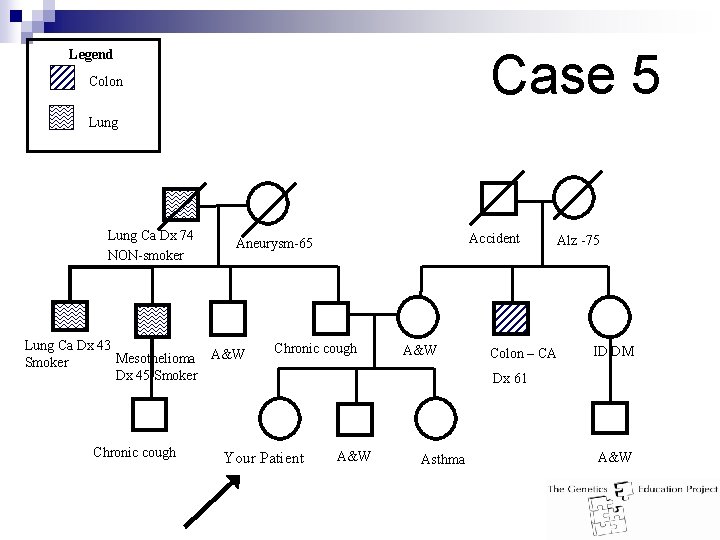
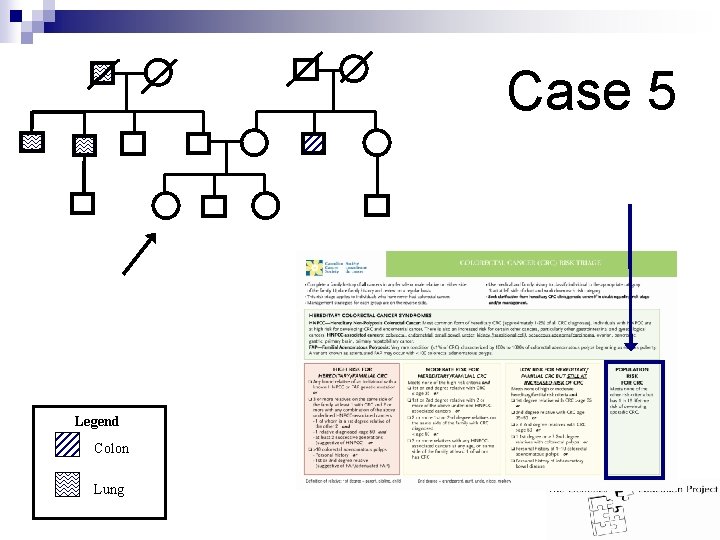
Case 5 Legend Colon Lung Ca Dx 74 NON-smoker Lung Ca Dx 43 Mesothelioma A&W Smoker Dx 45 Smoker Chronic cough Accident Aneurysm-65 Chronic cough Your Patient A&W Alz -75 Colon – CA ID DM Dx 61 A&W Asthma A&W
Case 5 Legend Colon Lung

Case 5 Answer: n n A = Good evidence B = Fair evidence I = Insufficient evidence Population risk for CRC Patient’s family worked in a shipyard insulating pipes ¨ Asbestos exposure increases risk of lung and mesothelioma cancers ¨ High incidence of lung cancer due to common environment exposures n Management: ¨ Beginning at Age 50: n n n Annual or biennial FOBTA OR Flexible sigmoidoscopy q 5 years. B OR FOBT + flexible sigmoidoscopy q 5 years. I OR Double contrast barium enema q 5 years OR Colonoscopy q 10 years. I
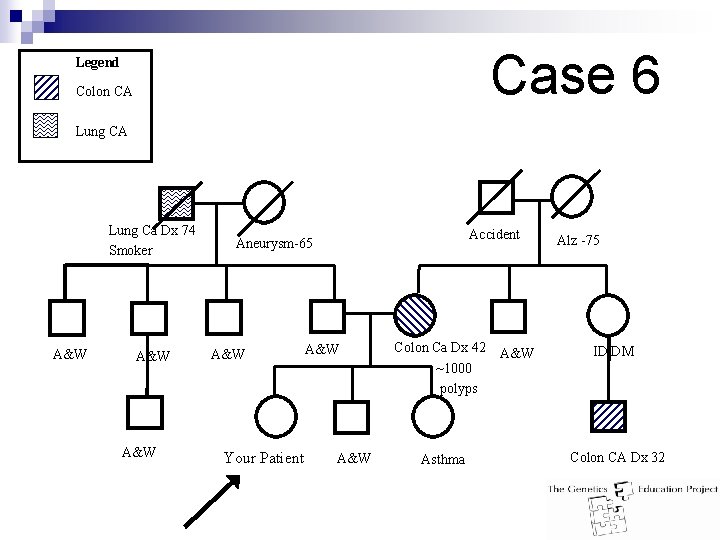
Case 6 Legend Colon CA Lung Ca Dx 74 Smoker A&W A&W Accident Aneurysm-65 A&W Your Patient A&W Colon Ca Dx 42 ~1000 polyps Asthma A&W Alz -75 ID DM Colon CA Dx 32
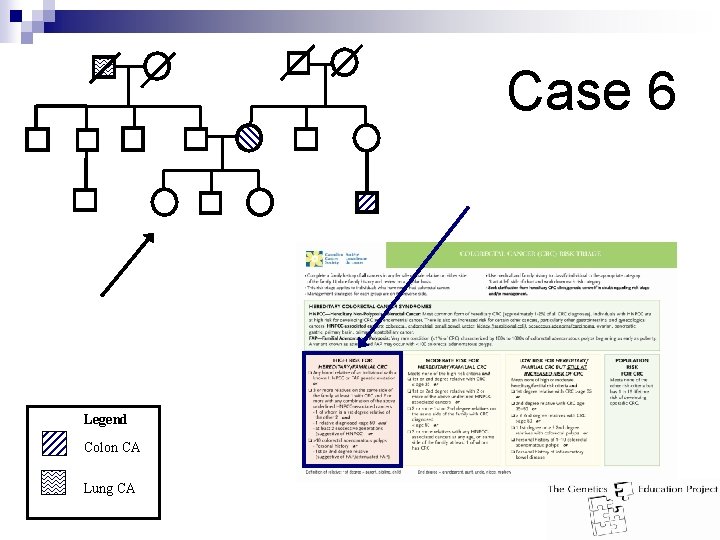
Case 6 Legend Colon CA Lung CA

Case 6 Answer: n High risk for hereditary CRC n >10 colorectal adenomatous polyps ¨ Personal history or ¨ 1 st or 2 nd degree relative (suggestive of FAP) n Management: ¨ Suggestive n of FAP: Seek advice from a colorectal specialist ¨ Offer referral to hereditary CRC/genetics clinic

The Genetics Education Project Committee n n n n June C Carroll MD CCFP Judith Allanson MD FRCP(C) FCCMG FABMG Sean Blaine MD CCFP Mary Jane Esplen Ph. D RN Sandra Farrell MD FRCPC FCCMG Judy Fiddes Gail Graham MD FRCPC FCCMG Jennifer Mac. Kenzie MD FRCPC FAAP FCCMG n n n n Wendy Meschino MD FRCPC FCCMG Joanne Miyazaki Andrea L. Rideout MS CGC Cheryl Shuman MS CGC Anne Summers MD FCCMG FRCPC Sherry Taylor Ph. D FCCMG Brenda Wilson BSc MB Ch. B MSc MRCP(UK) FFPH

References 1. Offit K Clinical Cancer Genetics: Risk Counseling and Management. Wiley-Liss, New York. 1998. 2. Statistics from the Canadian Cancer Society: http: //www. ontario. cancer. ca/ccs/internet/standard/0, 3182, 3543_1 4447_371429_lang. Id-en, 00. html accessed on June 22, 2005. 3. Hampel H, Frankel WL, Martin E, Arnold M, Khanduja K, Kuebler P, Nakagawa H, Sotamaa K, Prior TW, Westman J, Panescu J, Fix D, Lockman J, Comeras I, de la Chapelle A. Screening for Lynch syndrome (hereditary nonpolyposis colorectal cancer). N Engl J Med 2005; 352: 1851 -1860. 4. Mitchell RJ, Farrington SM, Dunlop MG, Campbell H. Mismatch repair genes h. MLH 1 and h. MSH 2 and colorectal cancer: a huge review. Am J Epidemiol 2002; 156: 885 -902.

References 5. Ponz de Leon M, Sassatelli R, Benatti P, Roncucci L. Identification of hereditary nonpolyposis colorectal cancer in the general population. The 6 -year experience of a population-based registry. Cancer 1993; 71: 3493 -3501. 6. Lightning bolt photo credit: http: //www. ghouli. com/articles/sp/mainstream_4 b. htm 7. Dunlop MG, Farrington SM, Nicholl I, Aaltonen L, Petersen G, Porteous M, Carothers. Population carrier frequency of h. MSH 2 and h. MLH 1 mutations. Br J Cancer 2000; 83: 1643 -1645. 8. American Gastroenterological Association (The Clinical Practice and Practice Review Committee). AGA technical review on hereditary colorectal cancer and genetic testing. Gastroenterology 2001; 121: 198 -213.

References 9. Salovaara R, Loukola A, Kristo P, Kaariainen H, Ahtola H, Eskelinen M, Harkonen N, Julkunen R, Kangas E, Ojala S, Tulikoura J, Valkamo E, Jarvinen H, Jukka-Pekka M, Aaltonen L, de la Chapelle A. Populationbased molecular detection of hereditary nonpolyposis colorectal cancer. J Clin Oncol 2000; 18: 2193 -2200. 10. Wijnen JT Vassen HFA, Khan PM, Zwinderman AH, van der Klift H, Mulder A, Tops C, Moller P, Fodde R. Clinical findings with implications for genetic testing in families with clustering of colorectal cancer. N Engl J Med. 1998; 339: 511 -518. 11. Burke W, Petersen G, Lynch P, Botkin J, Daly M, Garber J, Khan MJE, Mc. Tiernan A, Offit K, Thompson E, Varricchio C for the Cancer Genetics Studies Consortium. Recommendations for follow-up care of individuals with an inherited predisposition to cancer. I. Hereditary nonpolyposis colon cancer. JAMA 1997; 277: 915 -918. 12. Lin K, Shashidaran M, Ternent C, Thorson AG, Blatchford GJ, Christensen MA, Lanspa SJ, Lemon SJ, Watson P, Lynch H. Colorectal and extracolonic cancer variations in MLH 1/MSH 2 hereditary nonpolyposis colorectal cancer kindreds and the general population. Dis Colon Rectum. 1998; 41: 428 -433.

References 13. Vasen HFA, Wijnen JT, Menko FH Kleibeuker JH, Taal BG, Griffioen G, Nagengast FM, Meijer-Heijboer EH, Bertario L, Varesco L, Bisgaard M_L, Mohr J, Fodde R, Khan PM. Cancer risk in families with hereditary colorectal cancer diagnosed by mutational analysis. Gastroenterology 1996; 110: 1020 -1027. 14. The Canadian Cancer Society, National Cancer Institute of Canada, Statistics Canada, Provincial/Territorial Cancer Registries, Public Health Agency of Canada. Canadian Cancer Statistics 2005 http: //www. cancer. ca/vgn/images/portal/cit_86751114/48/28/40159 4768 cw_2005 stats_en. pdf Accessed June 21, 2005. 15. Kohlmann W, Gruber SB Hereditary nonpolyposis colorectal cancer. Gene Tests Reviews www. genetests. org last updated November 29, 2006. Accessed June 21, 2009. 16. Watson P, Lynch HT Cancer risk in mismatch repair gene mutation carriers. Familial Cancer 2001; 1: 57 -60.

References 17. Rodriquez-Bigas MA, Vassen HF, Lynch HT, Waston P, Myrhoj T, Jarvinen HJ, Meckllin JP, Macrae F, St. John DJB, Bertario L, Fidalgo P, Madlensky L, Rozen P, and the International Collaborative Group on HNPCC. Characteristics if small bowel carcinoma in hereditary nonpolyposis colorectal carcinoma. Cancer 1998; 83: 240 -244. 18. Aarnio M, Sankila R, Pukkala E, Salovaara R, Aaltonen LA, de la Chapelle A, Peltomaki P, Mecklin J-P, Jarvinen HJ. Cancer risk in mutation carriers of DNA-mismatch-repair genes. Int J Cancer 1999; 81: 214 -218. 19. Aarnio M, Mecklin J-P, Aaltonen LA, Nystrom-Lahti M, Jarvinen HJ. Life-time risk of different cancer in the hereditary nonpolyposis colorectal cancer (HNPCC) syndrome. Int J Cancer 1995; 64: 430 -433. 20. Quehenberger F, Vasen HFA, van Houwelingen HC. Risk of colorectal and endometrial cancer for carriers of h. MLH 1 and h. MSH 2 gene: correction for ascertainment. J Med Genet 2005; 42: 491 -496.

References 21. Burt RW, Jasperson KW Familial adenomatous polyposis. www. genetests. org. Last updated July 24, 2008, accessed June 21, 2009. 22. Predictive Cancer Genetics Steering Committee. Ontario physicians’ guide to referral of patients with family history of cancer to a familial cancer genetics clinic or genetics clinic. Ontario Medical Review 2001; 68(10): 24 -29. 23. National Cancer Institute. Genetics of Colorectal Cancer (PDQ). http: //www. nci. nih. gov/cancertopics/pdq/genetics/colorectal/Health Professional/page 2 Assessed on June 21, 2009. Johns LE, Houlston RS A systematic review and meta-analysis of familial colorectal cancer risk. Am J Gastroenterol. 2001; 96: 2992 -3003. 24. Johns LE, Houlston RS A systematic review and meta-analysis of familial colorectal cancer risk. Am J Gastroenterol. 2001; 96: 2992 -3003.

References 25. Leddin D, Hunt R, Champion M, Cockeram A, Flook N, Gould M, Kim Y-I, Love J, Morgan D, Natsheh S, Sadowski D, for the Canadian Association of Gastroenterology and the Canadian Digestive Health Foundation committee on colorectal cancer screening. Can J Gastroenterologgy 2004; 18: 93 -99. 26. Vasen HFA, Moslein G, Alonso A, Bernstein I, Bertario L, Burn J, Capella G, Engel C, Frayling I, Friedl W, Hes FJ, Hodgson S, Mecklin J-P, Moller P, Nagengast F, Parc Y, Renkonen-Sinisalo L, Sampson JR, Stormorken A, Wijnen J. Guidelines for the clinical management of Lynch syndrome (hereditary non-polyposis cancer). J Med Genet 2007; 44: 353 -362. 27. Jarvinen HJ, Aarnio M, Mustonen H, Aktan-Collan K, Aaltonen LA, Peltomaki P, de la Chapelle A. Controlled 15 -year trial on screening for colorectal cancer in families with hereditary nonpolyposis colorectal cancer. Gastroenterology 2000; 118: 829834.

References 28. Dove-Edwin I, Sasieni P, Adams J, Thomas HJW Prevention of colorectal cancer by colonoscopic surveillance in individuals with a family history of colorectal cancer: a 16 year, prospective, follow-up study. BMJ. 2005; 331: 1047 -1049. 29. Yu H-JA, Lin KM, Ota DM, Lynch HT. Hereditary nonpolyposis colorectal cancer: preventive management. Cancer Treatment Rev 2003; 29: 461470. 30. Renkonen-Sinisalo L, Butzow R, Leminen A, Lehtovirta P, Mecklin J-P, Jarvinen HJ. Surveillance for endometrial cancer in hereditary nonpolyposis colorectal cancer syndrome. Int. J. Cancer. 2006; 821 -824. 31. Schmeler KM, Lynch HT, Chen L-M, Munsell MF, Soliman PT, Clark MB, Daniels MS, White KG, Boyd-Rodgers SG, Conrad PG, Yang KY, Rubin MM, Sun CC, Slomovitz BM, Gershenson DM, Lu KH. Prophylactic surgery to reduce the risk of gynaecologic cancers in Lynch syndrome. NEJM 2006; 345: 261 -269. 32. Koornstra JJ, Mourits MJE, Sijmons RH, Leliveld AM, Hollema H, Kleibeuker JH. Management of extracolonic tumours in patients with Lynch syndrome. Lancet Oncology 2009; 120: 400 -408.


Extra Slides
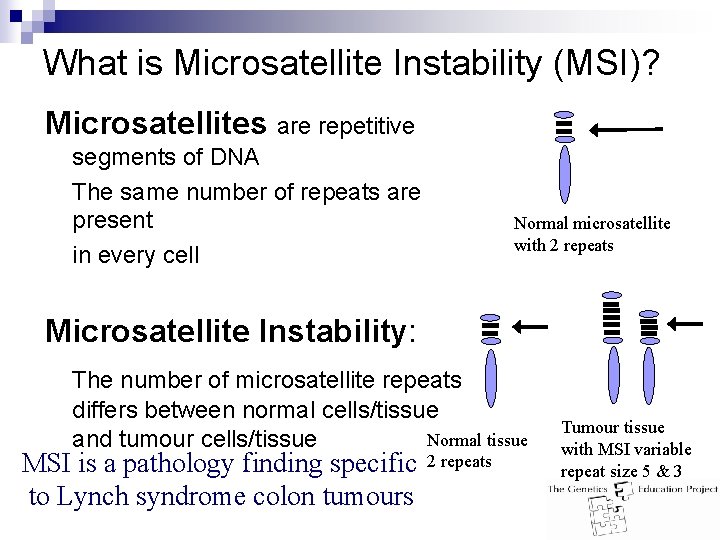
What is Microsatellite Instability (MSI)? Microsatellites are repetitive segments of DNA The same number of repeats are present in every cell Normal microsatellite with 2 repeats Microsatellite Instability: The number of microsatellite repeats differs between normal cells/tissue Normal tissue and tumour cells/tissue MSI is a pathology finding specific to Lynch syndrome colon tumours 2 repeats Tumour tissue with MSI variable repeat size 5 & 3

Pathology & Genetic Evidence for Increased Risk of Hereditary CRC n n n Principle: Mutations of the genes MSH 2, MLH 1, MSH 6 and PMS 2 increase the rate of genetic mutation in human cells. Small repetitive sequences (microsatellites) are very susceptible to increases in the mutation rate. These repetitive sequences can be surveyed to see if there are differences in their sequence between the normal and tumor tissues from an individual. If changes are seen the tumor can be referred to as showing “microsatellite instability”. Typically there is good concordance between seeing that a tumor is by immunohistochemistry immunodeficient for one of these gene products and the finding of microsatellite instability. Observing either one or both in a tumor increases the likelihood a familial mutation is present

Pathology and Genetic Evidence for Increased Risk of Hereditary Colorectal Cancer n n Colonic ademoma or other Lynch syndrome associated cancers can be found in the laboratory to have one or both of the following properties which increase the likelihood a familial mutation is responsible. The tumors: 1. Are deficient for immunohistochemical staining for the proteins n 2. MSH 2, MLH 1, MSH 6 and/or PMS 2 Show evidence of genetic instability of small repetitive DNA sequences (microsatellites) when compared to normal tissue.
- Slides: 74