HEPATITIS WEB STUDY HEPATITIS C ONLINE Simeprevir Olysio
HEPATITIS WEB STUDY HEPATITIS C ONLINE Simeprevir (Olysio) Prepared by: David Spach, MD & H. Nina Kim, MD Last Updated: July 14, 2015 Hepatitis web study

SIMEPREVIR (OLYSIO) Background and Dosing Hepatitis web study

Simeprevir (Olysio) Summary • Approval Status: FDA approved December 6, 2013 • Indication for HCV Monoinfection - GT 1: Simeprevir (12 weeks) + peginterferon + ribavirin (12 or 36 weeks) - Poor response to Simeprevir + Peginteferon + Ribavirin with GT 1 a and NS 3 Q 80 K polymorphism at baseline • Class & Mechanism - NS 3/4 A protease inhibitor - Activity against GT 1, 2, 4, 5, 6 (strongest activity against GT 1 a, 1 b) • Simeprevir Dosing - 150 mg PO once daily with food - In combination with peginterferon + ribavirin (triple therapy) • Adverse Effects (AE) attributable to Simeprevir - Rash (including a photosensitivity reaction), pruritus, and nausea • Wholesaler Acquisition Cost in United States - 28 tablet bottle = $22, 120; estimated 12 -week cost = $66, 360 Hepatitis web study
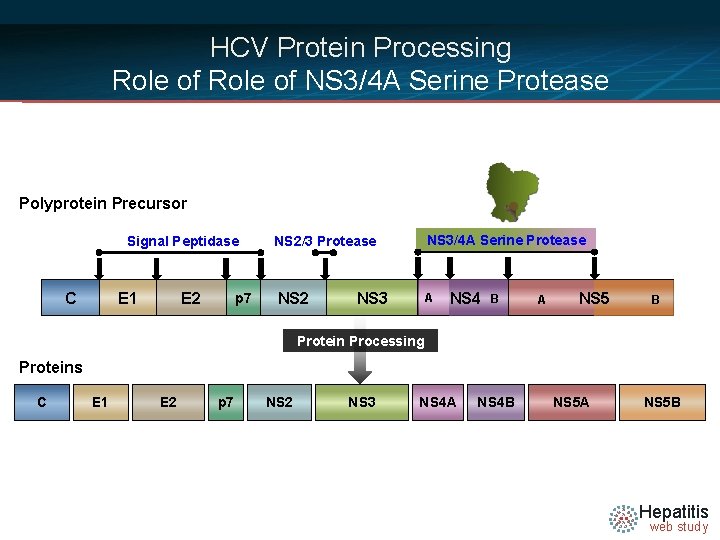
HCV Protein Processing Role of NS 3/4 A Serine Protease Polyprotein Precursor Signal Peptidase C E 1 E 2 p 7 NS 2/3 Protease NS 3/4 A Serine Protease NS 2 A NS 3 NS 4 B A NS 5 B Protein Processing Proteins C E 1 E 2 p 7 NS 2 NS 3 NS 4 A NS 4 B NS 5 A NS 5 B Hepatitis web study
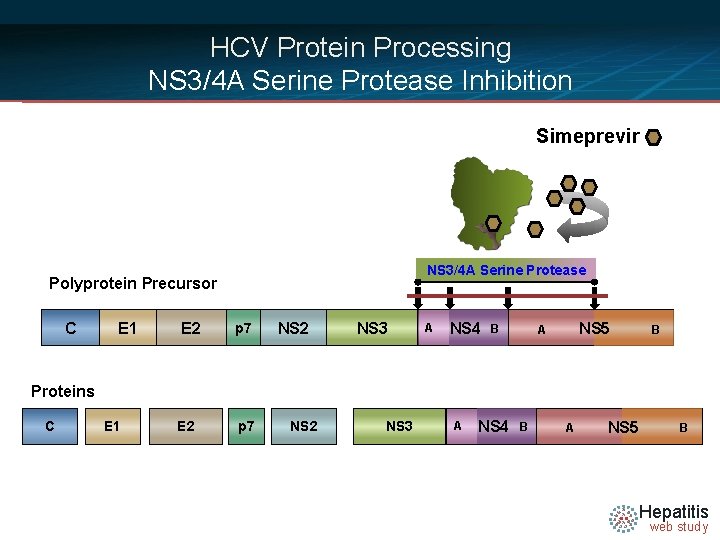
HCV Protein Processing NS 3/4 A Serine Protease Inhibition Simeprevir NS 3/4 A Serine Protease Polyprotein Precursor C E 1 E 2 p 7 NS 2 NS 3 A NS 4 B NS 5 A B Proteins C E 1 E 2 p 7 NS 2 NS 3 A NS 4 B A NS 5 B Hepatitis web study

SIMEPREVIR (OLYSIO) Clinical Trials Hepatitis web study

Simeprevir: Summary of Key Studies • Phase 3 Trials in Treatment Naive - QUEST 1: Simeprevir-PEG-RBV vs. PEG-RBV in GT 1 - QUEST 2: Simeprevir-PEG-RBV vs. PEG-RBV in GT 1 • Phase 3 Trials in Treatment Experienced - PROMISE: Simeprevir-PEG-RBV vs. PEG-RBV in GT 1, prior relapse - ATTAIN: Simeprevir vs. Telaprevir in prior null or partial responders • Phase 2 Trials in Treatment Experienced - ASPIRE: Simeprevir + PR in GT 1 treatment experienced Hepatitis web study

Simeprevir: Summary of Key Studies • Phase 3 Trials in Treatment Naïve and Experienced - RESTORE: Simeprevir in HCV genotype 4 (naïve and experienced) • Simeprevir + Sofosbuvir - COSMOS: Simeprevir + Sofosbuvir +/- RBV in GT 1 - OPTIMIST-1: Simeprevir + Sofosbuvir in GT 1 without cirrhosis - OPTIMIST-2: Simeprevir + Sofosbuvir in GT 1 with cirrhosis • HIV Coinfection - C 212: Simeprevir in HIV-HCV coinfected in GT 1 Hepatitis web study

Simeprevir in Treatment-Naïve Patients Hepatitis web study

Phase 3 Treatment Naïve Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-1 Trial Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study

Simeprevir + PEG + Ribavirin for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Features § Design: Randomized, double-blind, placebo-controlled, phase 3 trial with simeprevir + PEG + RBV versus PEG + RBV in treatment-naïve GT 1 § Setting: Multicenter at 71 sites in 13 countries § Entry Criteria - Treatment-naïve, chronic HCV monoinfection - HCV Genotype 1 (1 a or 1 b) § Patient Characteristics - N = 394 - HCV Genotype: 1 a (56%); 1 b (44%) - IL 28 B Genotype: 71% non-CC - Age: median age 48 - Sex: 56% male - Race: 89% white, 8% black - Liver disease: F 3 = 18%; F 4 = 12% § Primary end-points: Efficacy (SVR 12) and safety Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
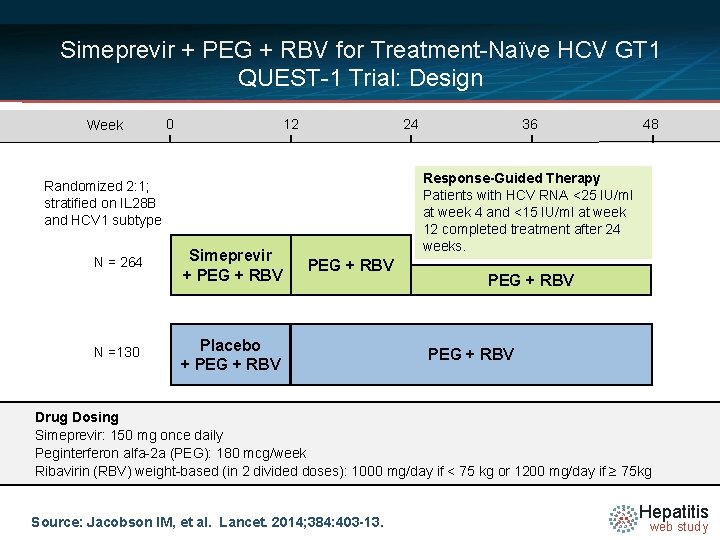
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Design Week 0 12 24 N =130 Simeprevir + PEG + RBV 48 Response-Guided Therapy Patients with HCV RNA <25 IU/ml at week 4 and <15 IU/ml at week 12 completed treatment after 24 weeks. Randomized 2: 1; stratified on IL 28 B and HCV 1 subtype N = 264 36 PEG + RBV Placebo + PEG + RBV Drug Dosing Simeprevir: 150 mg once daily Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
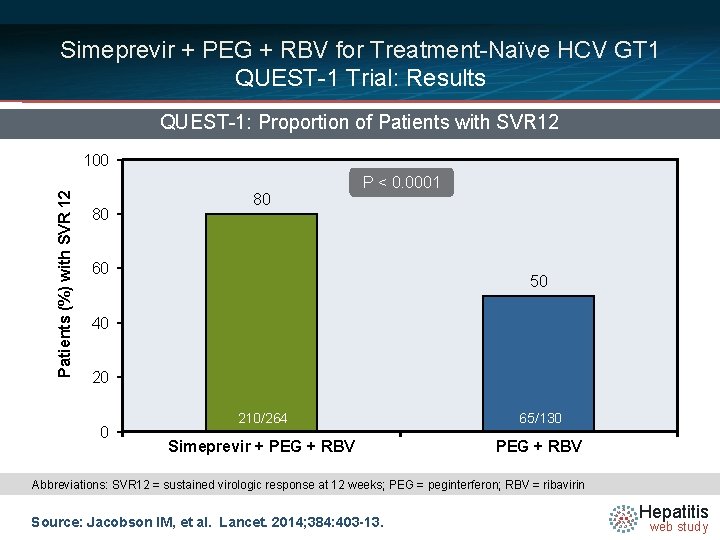
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results QUEST-1: Proportion of Patients with SVR 12 Patients (%) with SVR 12 100 80 80 P < 0. 0001 60 50 40 20 0 210/264 65/130 Simeprevir + PEG + RBV Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
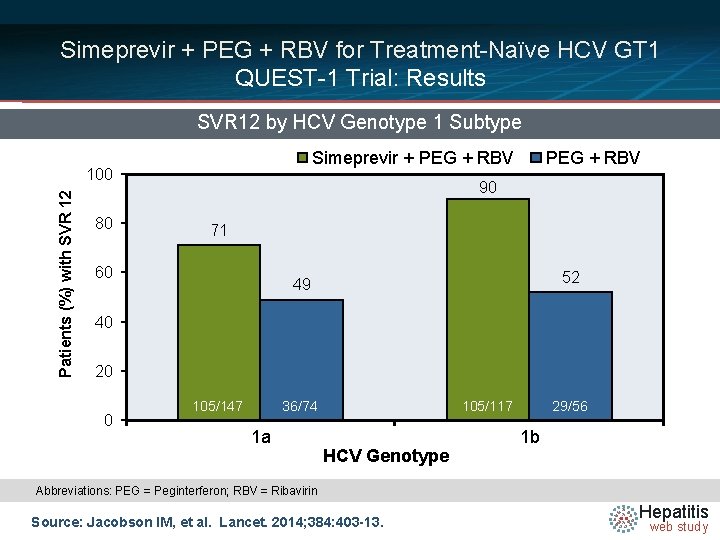
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results SVR 12 by HCV Genotype 1 Subtype Simeprevir + PEG + RBV Patients (%) with SVR 12 100 80 PEG + RBV 90 71 60 52 49 40 20 0 105/147 36/74 1 a 105/117 HCV Genotype 29/56 1 b Abbreviations: PEG = Peginterferon; RBV = Ribavirin Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
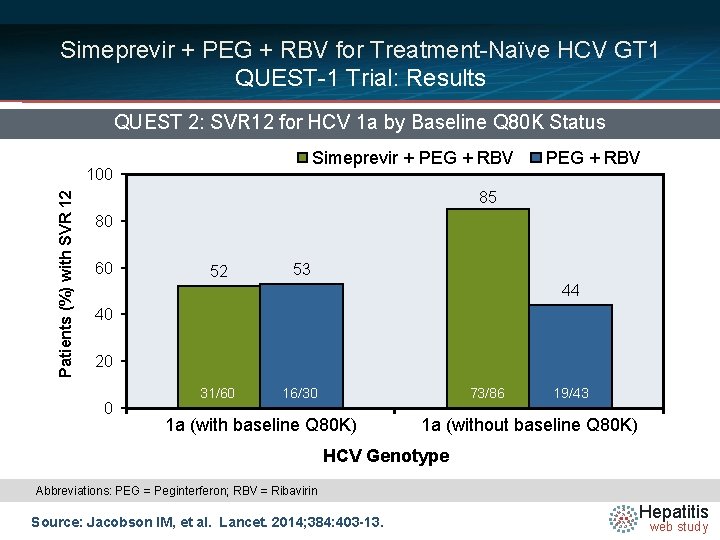
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results QUEST 2: SVR 12 for HCV 1 a by Baseline Q 80 K Status Simeprevir + PEG + RBV Patients (%) with SVR 12 100 PEG + RBV 85 80 60 52 53 44 40 20 0 31/60 16/30 73/86 1 a (with baseline Q 80 K) 19/43 1 a (without baseline Q 80 K) HCV Genotype Abbreviations: PEG = Peginterferon; RBV = Ribavirin Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
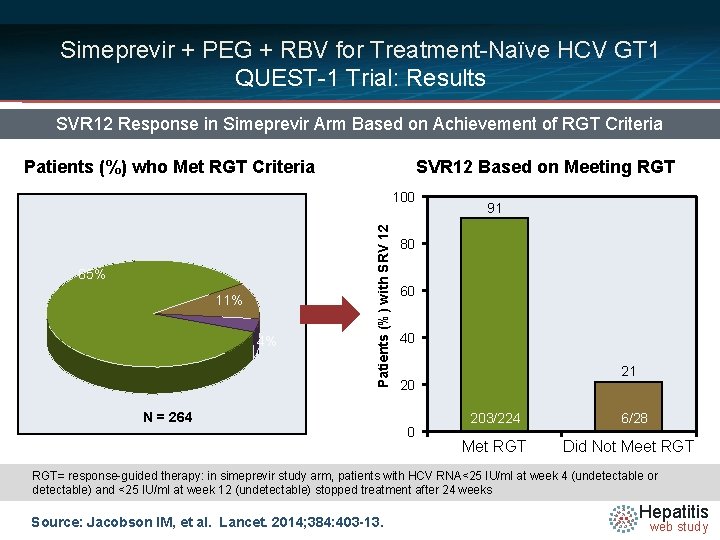
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results SVR 12 Response in Simeprevir Arm Based on Achievement of RGT Criteria Patients (%) who Met RGT Criteria SVR 12 Based on Meeting RGT 85% 11% 4% Patients (%) with SRV 12 100 N = 264 91 80 60 40 21 20 0 203/224 Met RGT 6/28 Did Not Meet RGT= response-guided therapy: in simeprevir study arm, patients with HCV RNA<25 IU/ml at week 4 (undetectable or detectable) and <25 IU/ml at week 12 (undetectable) stopped treatment after 24 weeks Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
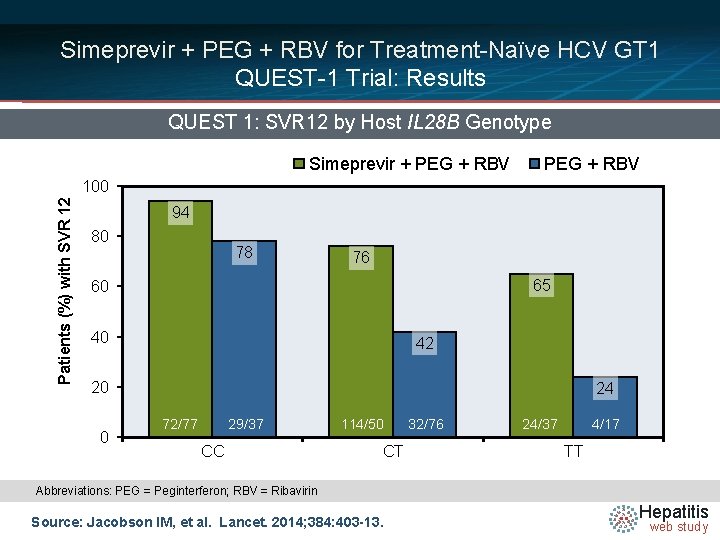
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results QUEST 1: SVR 12 by Host IL 28 B Genotype Simeprevir + PEG + RBV Patients (%) with SVR 12 100 94 80 78 76 65 60 40 42 20 0 24 72/77 29/37 CC 114/50 CT 32/76 24/37 4/17 TT Abbreviations: PEG = Peginterferon; RBV = Ribavirin Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
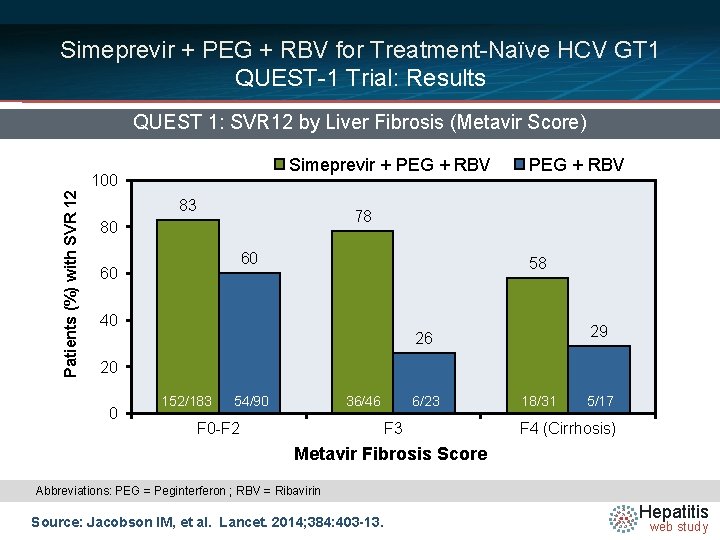
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results QUEST 1: SVR 12 by Liver Fibrosis (Metavir Score) Simeprevir + PEG + RBV Patients (%) with SVR 12 100 83 PEG + RBV 78 80 60 60 58 40 29 26 20 0 152/183 54/90 36/46 F 0 -F 2 6/23 F 3 18/31 5/17 F 4 (Cirrhosis) Metavir Fibrosis Score Abbreviations: PEG = Peginterferon ; RBV = Ribavirin Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
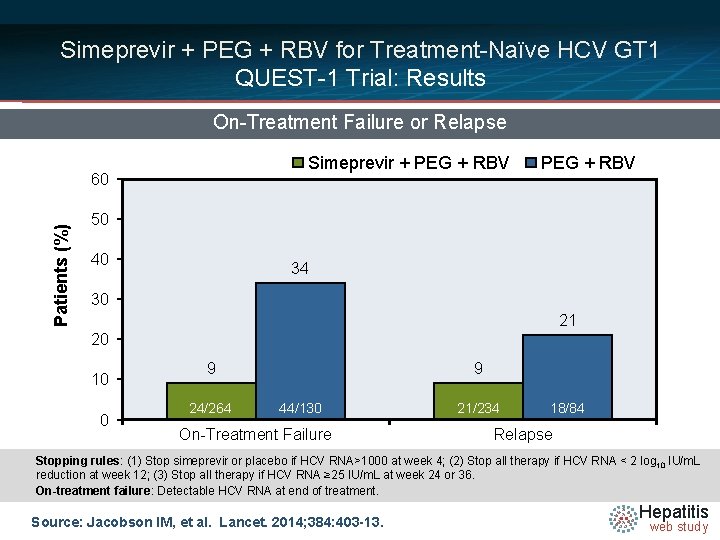
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results On-Treatment Failure or Relapse Simeprevir + PEG + RBV Patients (%) 60 PEG + RBV 50 40 34 30 21 20 10 0 9 24/264 9 44/130 On-Treatment Failure 21/234 18/84 Relapse Stopping rules: (1) Stop simeprevir or placebo if HCV RNA>1000 at week 4; (2) Stop all therapy if HCV RNA < 2 log 10 IU/m. L reduction at week 12; (3) Stop all therapy if HCV RNA ≥ 25 IU/m. L at week 24 or 36. On-treatment failure: Detectable HCV RNA at end of treatment. Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study

Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Results Emergent Protease Resistance in Patients who Failed to Achieve SVR 12 § Among simeprevir-treated patients who failed to achieve SVR 12, emergent mutations in NS 3 protease domain detected in 35 (92%) of 38 § Genotype 1 A: Most common mutation = R 155 K alone or in combination with mutations at codons 80 and/or 168 § Genotype 1 B: Most common mutation = D 168 V Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study
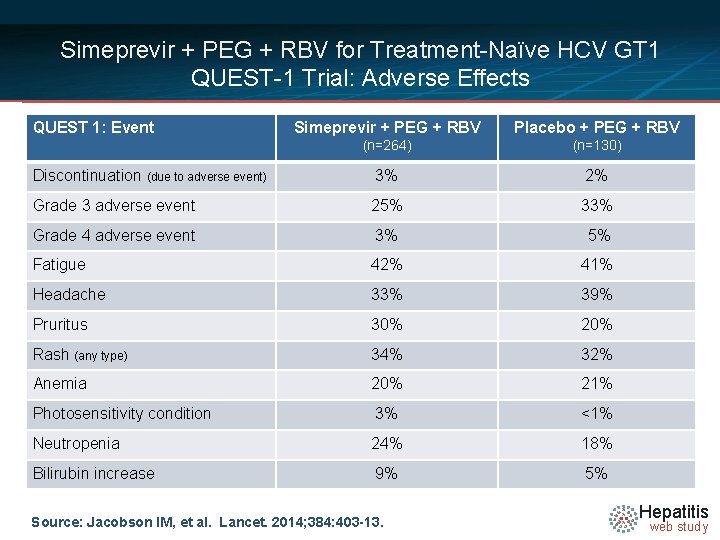
Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Adverse Effects QUEST 1: Event Simeprevir + PEG + RBV Placebo + PEG + RBV (n=264) (n=130) Discontinuation (due to adverse event) 3% 2% Grade 3 adverse event 25% 33% Grade 4 adverse event 3% 5% Fatigue 42% 41% Headache 33% 39% Pruritus 30% 20% Rash (any type) 34% 32% Anemia 20% 21% Photosensitivity condition 3% <1% Neutropenia 24% 18% Bilirubin increase 9% 5% Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study

Simeprevir + PEG + RBV for Treatment-Naïve HCV GT 1 QUEST-1 Trial: Conclusions Interpretation: “Simeprevir once daily with peginterferon alfa 2 a and ribavirin shortens therapy in treatment-naive patients with HCV genotype 1 infection without worsening the adverse event profiles associated with peginterferon alfa 2 a plus ribavirin. ” Source: Jacobson IM, et al. Lancet. 2014; 384: 403 -13. Hepatitis web study

Phase 3 Treatment Naïve Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study

Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Background QUEST-2 Trial: Features § Design: Randomized, double-blind, placebo-controlled, phase 3 trial of simeprevir + PEG + RBV versus PEG + RBV in HCV GT 1 § Setting: Multicenter at 76 sites in 14 countries § Entry Criteria - Treatment-naïve, chronic HCV monoinfection - HCV Genotypes 1 a or 1 b § Patient Characteristics - N = 391 - HCV Subtype: 1 a (41%); 1 b (58%); other (<1%) - IL 28 B Genotype: 30% CC - Age and Sex: median age 46; 55% male - Race: 92% white - Liver disease: 14% with F 3; 6% with F 4 § Primary end-points: Efficacy (SVR 12) and safety Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
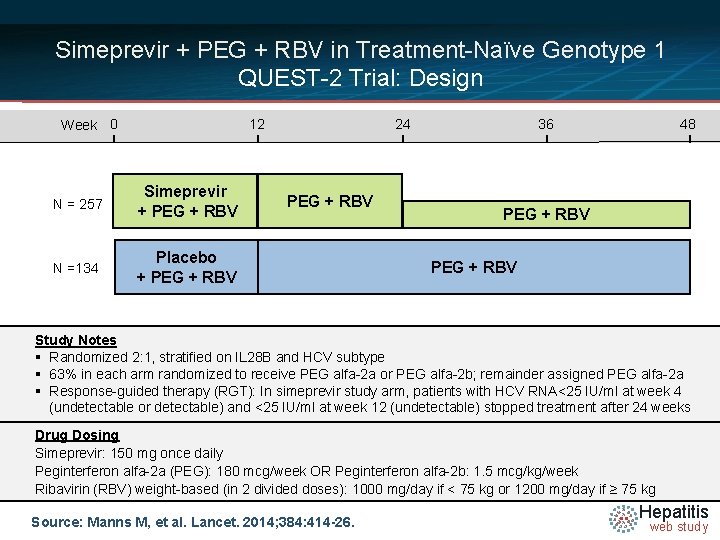
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Design Week 0 12 N = 257 Simeprevir + PEG + RBV N =134 Placebo + PEG + RBV 24 PEG + RBV 36 48 PEG + RBV Study Notes § Randomized 2: 1, stratified on IL 28 B and HCV subtype § 63% in each arm randomized to receive PEG alfa-2 a or PEG alfa-2 b; remainder assigned PEG alfa-2 a § Response-guided therapy (RGT): In simeprevir study arm, patients with HCV RNA<25 IU/ml at week 4 (undetectable or detectable) and <25 IU/ml at week 12 (undetectable) stopped treatment after 24 weeks Drug Dosing Simeprevir: 150 mg once daily Peginterferon alfa-2 a (PEG): 180 mcg/week OR Peginterferon alfa-2 b: 1. 5 mcg/kg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
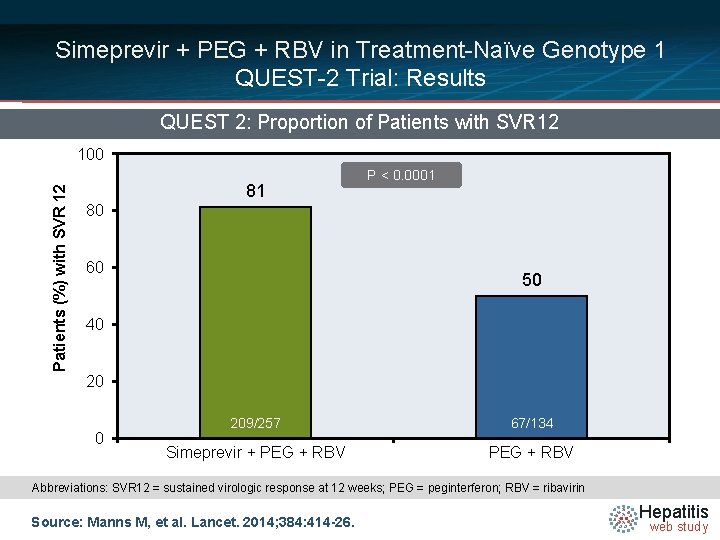
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: Proportion of Patients with SVR 12 Patients (%) with SVR 12 100 81 P < 0. 0001 80 60 50 40 209/257 67/134 Simeprevir + PEG + RBV Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
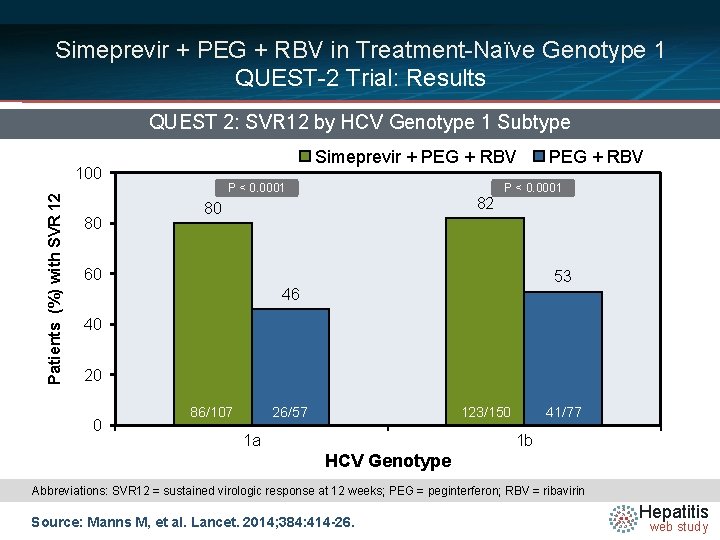
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: SVR 12 by HCV Genotype 1 Subtype Simeprevir + PEG + RBV Patients (%) with SVR 12 100 80 P < 0. 0001 82 80 PEG + RBV P < 0. 0001 60 53 46 40 20 0 86/107 26/57 123/150 1 a 41/47 41/77 1 b HCV Genotype Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
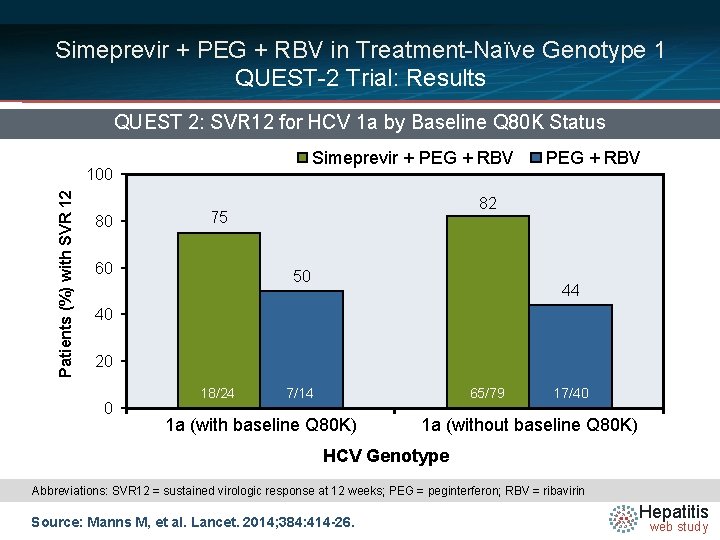
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: SVR 12 for HCV 1 a by Baseline Q 80 K Status Simeprevir + PEG + RBV Patients (%) with SVR 12 100 80 82 75 60 PEG + RBV 50 44 40 20 0 18/24 7/14 65/79 1 a (with baseline Q 80 K) 17/40 1 a (without baseline Q 80 K) HCV Genotype Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
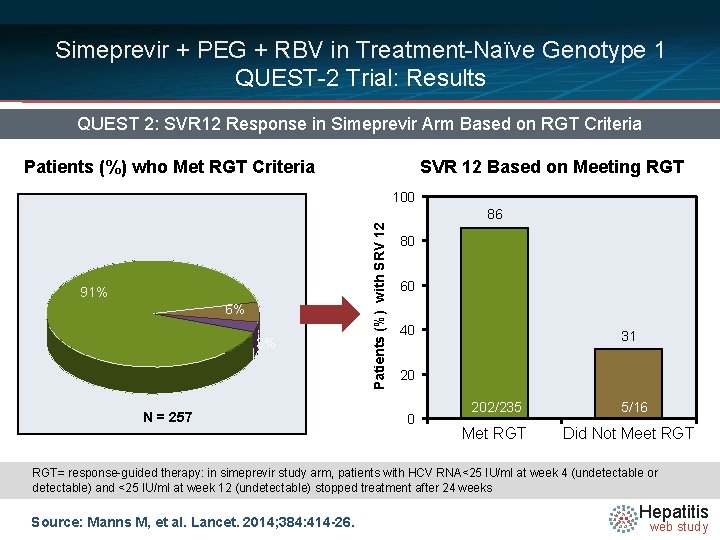
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: SVR 12 Response in Simeprevir Arm Based on RGT Criteria Patients (%) who Met RGT Criteria SVR 12 Based on Meeting RGT 100 91% 6% 3% N = 257 Patients (%) with SRV 12 86 80 60 40 31 20 0 202/235 Met RGT 5/16 Did Not Meet RGT= response-guided therapy: in simeprevir study arm, patients with HCV RNA<25 IU/ml at week 4 (undetectable or detectable) and <25 IU/ml at week 12 (undetectable) stopped treatment after 24 weeks Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
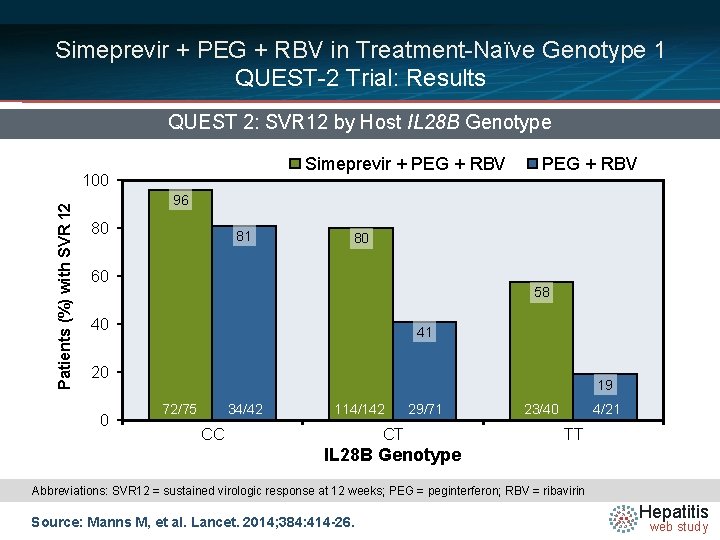
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: SVR 12 by Host IL 28 B Genotype Simeprevir + PEG + RBV Patients (%) with SVR 12 100 PEG + RBV 96 80 81 80 60 58 40 41 20 0 19 72/75 34/42 114/142 CC 29/71 CT 23/40 4/21 TT IL 28 B Genotype Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
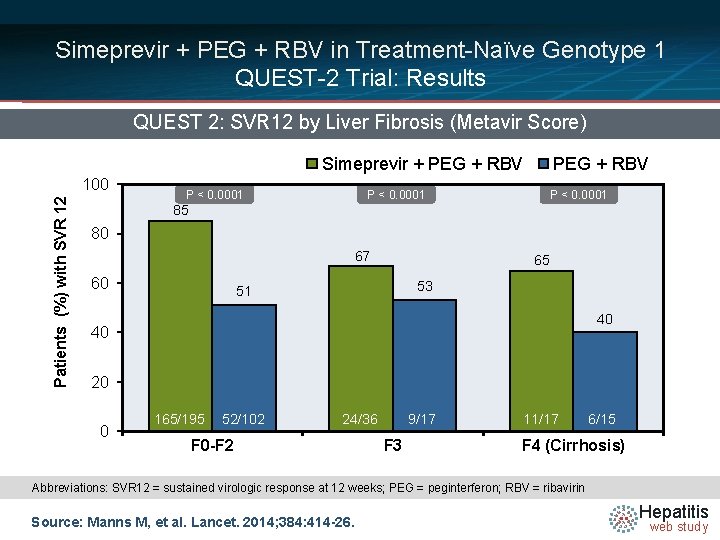
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: SVR 12 by Liver Fibrosis (Metavir Score) Simeprevir + PEG + RBV Patients (%) with SVR 12 100 P < 0. 0001 PEG + RBV P < 0. 0001 85 80 67 60 65 53 51 40 40 20 0 165/195 52/102 24/36 F 0 -F 2 9/17 F 3 11/17 6/15 F 4 (Cirrhosis) Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
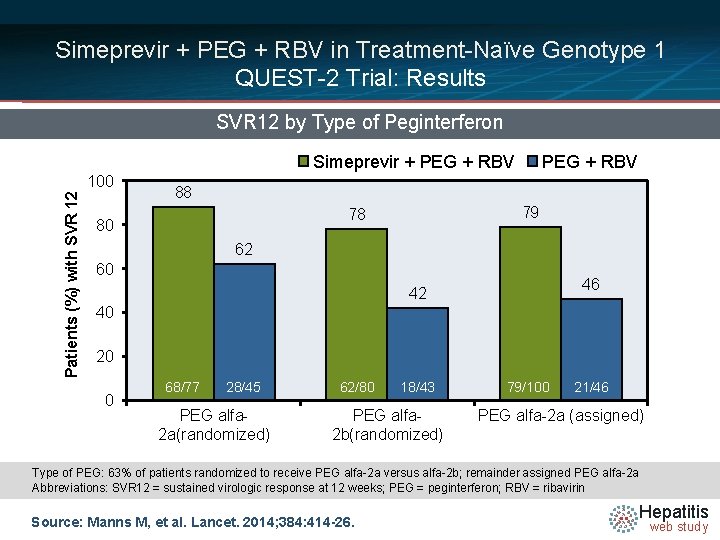
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results SVR 12 by Type of Peginterferon Simeprevir + PEG + RBV Patients (%) with SVR 12 100 PEG + RBV 88 79 78 80 62 60 46 42 40 20 0 68/77 28/45 PEG alfa 2 a(randomized) 62/80 18/43 PEG alfa 2 b(randomized) 79/100 21/46 PEG alfa-2 a (assigned) Type of PEG: 63% of patients randomized to receive PEG alfa-2 a versus alfa-2 b; remainder assigned PEG alfa-2 a Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
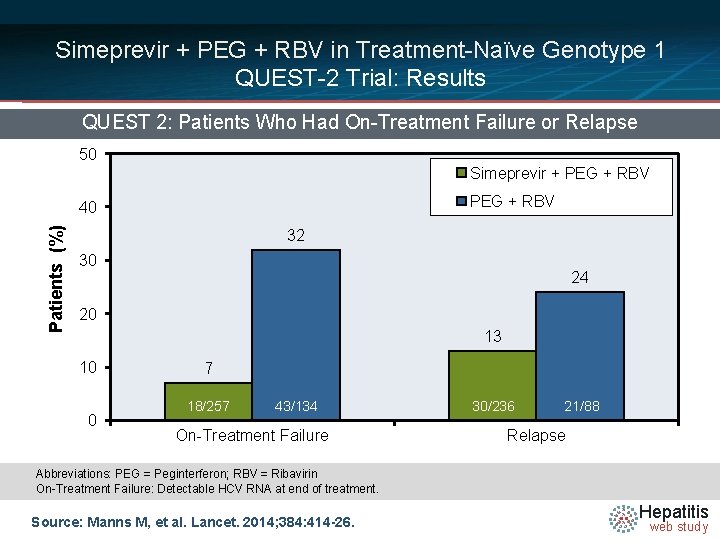
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: Patients Who Had On-Treatment Failure or Relapse 50 Simeprevir + PEG + RBV Patients (%) 40 32 30 24 20 13 10 0 7 18/257 43/134 On-Treatment Failure 30/236 21/88 Relapse Abbreviations: PEG = Peginterferon; RBV = Ribavirin On-Treatment Failure: Detectable HCV RNA at end of treatment. Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study
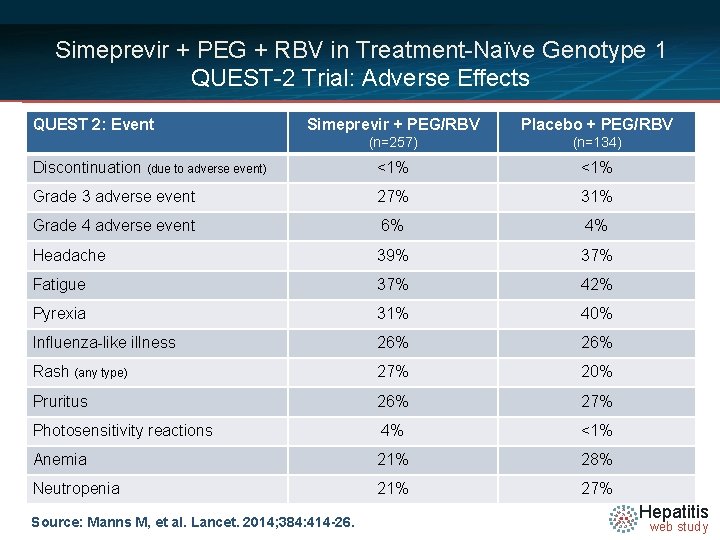
Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Adverse Effects QUEST 2: Event Simeprevir + PEG/RBV Placebo + PEG/RBV (n=257) (n=134) Discontinuation (due to adverse event) <1% Grade 3 adverse event 27% 31% Grade 4 adverse event 6% 4% Headache 39% 37% Fatigue 37% 42% Pyrexia 31% 40% Influenza-like illness 26% Rash (any type) 27% 20% Pruritus 26% 27% Photosensitivity reactions 4% <1% Anemia 21% 28% Neutropenia 21% 27% Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study

Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Results QUEST 2: Emergent Resistance in Patients who Failed to Achieve SVR 12 § Among simeprevir-treated patients who failed to achieve SVR 12, emergent mutations in NS 3 protease domain detected in 98% § Genotype 1 A: Most common mutation = R 155 K alone or in combination with mutations at codons 80 and/or 168 § Genotype 1 B: Most common mutation = D 168 V and Q 80 R + D 168 E Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study

Simeprevir + PEG + RBV in Treatment-Naïve Genotype 1 QUEST-2 Trial: Conclusions Interpretation: “Addition of simeprevir to either peginterferon alfa 2 a or peginterferon alfa 2 b plus ribavirin improved SVR in treatment-naive patients with HCV genotype 1 infection, without worsening the known adverse events associated with peginterferon alfa plus ribavirin. ” Source: Manns M, et al. Lancet. 2014; 384: 414 -26. Hepatitis web study

Simeprevir in Treatment-Experienced Patients Hepatitis web study

Phase 3 Treatment Experienced Simeprevir in Genotype 1 (Viral Relapsers) PROMISE Trial Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study

Simeprevir + PEG + Ribavirin for Chronic HCV PROMISE Trial: Study Features § Design: Randomized, double-blind, placebo-controlled phase 3 trial of triple therapy with simeprevir, peginterferon alfa-2 a, and ribavirin in treatment-experienced patients with HCV GT 1 infection § Entry Criteria - Treatment-experienced, chronic HCV monoinfection - Viral relapse with prior (≥ 24 weeks) of peginterferon-based therapy - HCV Genotype 1 § Patient Characteristics - N = 393 - HCV Genotype: 1 a (42%); 1 b (58%) - IL 28 B Genotype: 76% non-CC - Age and Sex: median age 52; 66% male - Race: 94% white - Liver disease: 15% had METAVIR F 3; 15% F 4 § Primary end-points: Efficacy (SVR 12) Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
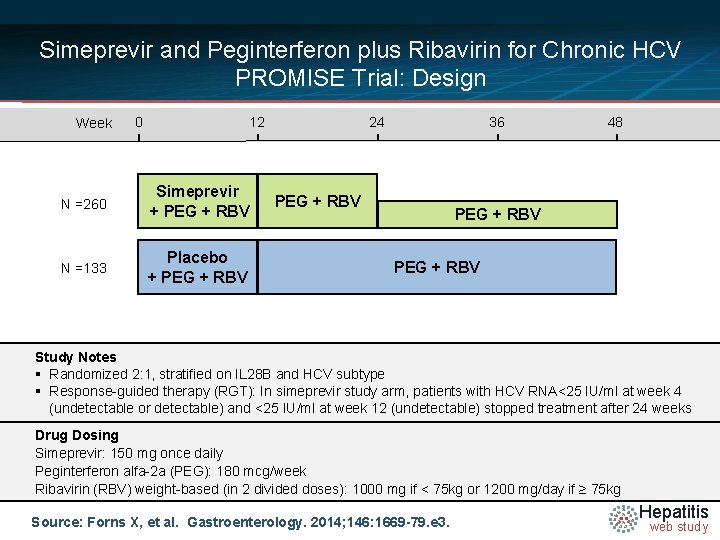
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Trial: Design Week 0 12 N =260 Simeprevir + PEG + RBV N =133 Placebo + PEG + RBV 24 36 PEG + RBV 48 PEG + RBV Study Notes § Randomized 2: 1, stratified on IL 28 B and HCV subtype § Response-guided therapy (RGT): In simeprevir study arm, patients with HCV RNA<25 IU/ml at week 4 (undetectable or detectable) and <25 IU/ml at week 12 (undetectable) stopped treatment after 24 weeks Drug Dosing Simeprevir: 150 mg once daily Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
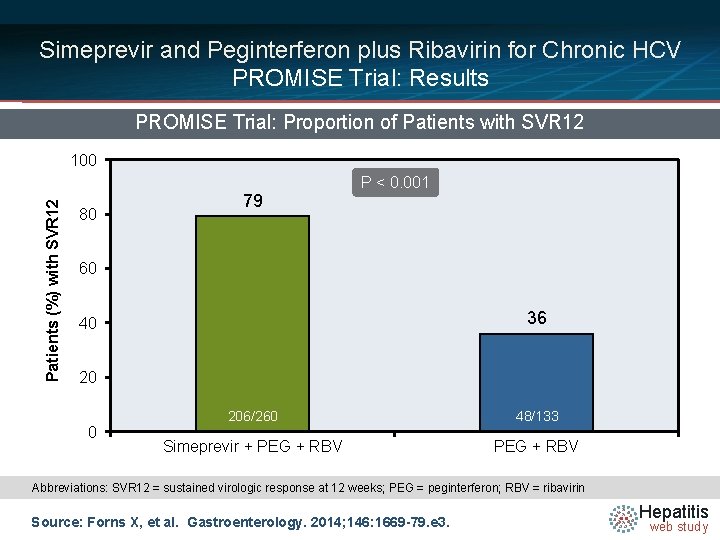
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Trial: Results PROMISE Trial: Proportion of Patients with SVR 12 Patients (%) with SVR 12 100 80 79 P < 0. 001 60 36 40 206/260 48/133 Simeprevir + PEG + RBV Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
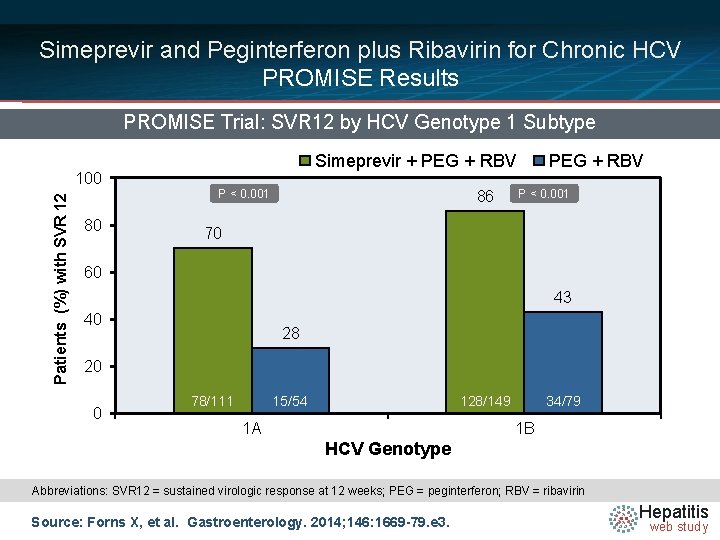
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Results PROMISE Trial: SVR 12 by HCV Genotype 1 Subtype Patients (%) with SVR 12 100 80 Simeprevir + PEG + RBV P < 0. 001 86 PEG + RBV P < 0. 001 70 60 43 40 28 20 0 78/111 15/54 128/149 1 A 34/79 1 B HCV Genotype Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
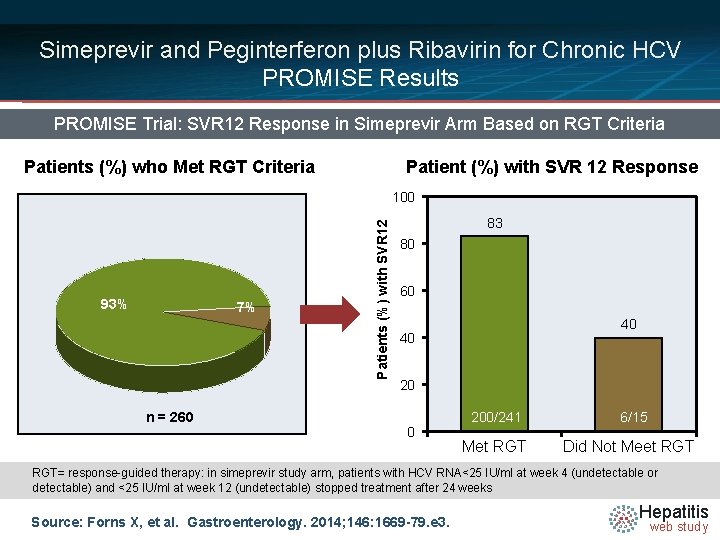
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Results PROMISE Trial: SVR 12 Response in Simeprevir Arm Based on RGT Criteria Patients (%) who Met RGT Criteria Patient (%) with SVR 12 Response 93% 7% n = 260 Patients (%) with SVR 12 100 83 80 60 40 40 200/241 Met RGT 6/15 Did Not Meet RGT= response-guided therapy: in simeprevir study arm, patients with HCV RNA<25 IU/ml at week 4 (undetectable or detectable) and <25 IU/ml at week 12 (undetectable) stopped treatment after 24 weeks Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
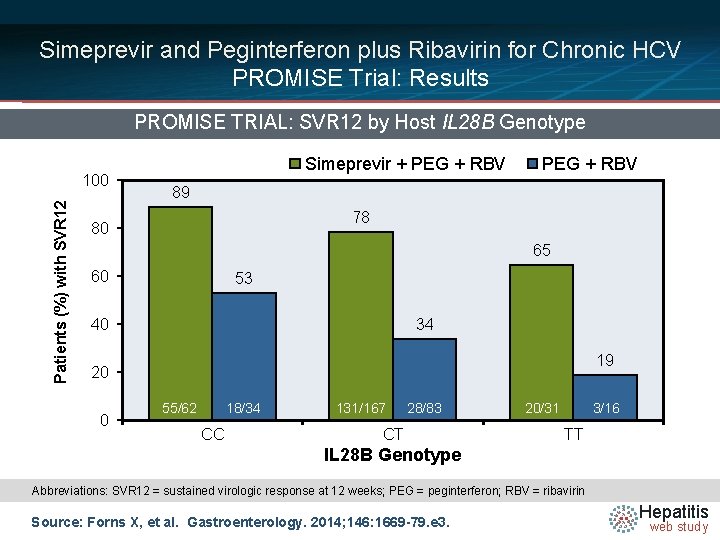
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Trial: Results PROMISE TRIAL: SVR 12 by Host IL 28 B Genotype Patients (%) with SVR 12 100 Simeprevir + PEG + RBV 89 78 80 65 60 53 40 34 19 20 0 55/62 18/34 CC 131/167 28/83 CT 20/31 3/16 TT IL 28 B Genotype Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
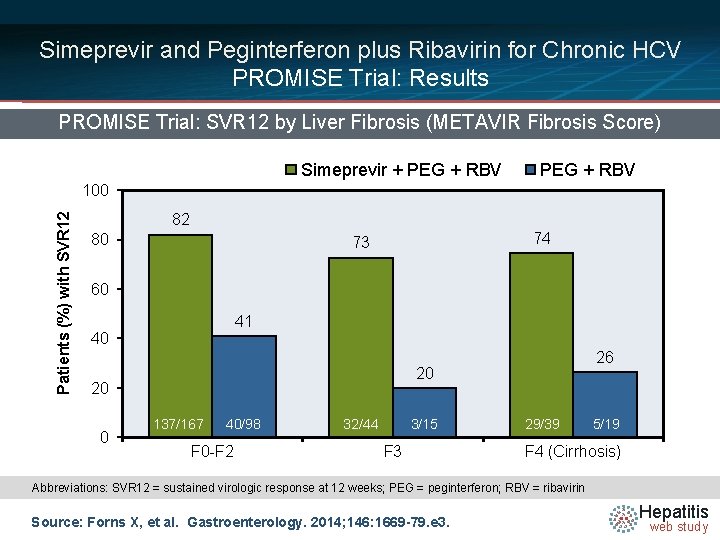
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Trial: Results PROMISE Trial: SVR 12 by Liver Fibrosis (METAVIR Fibrosis Score) Simeprevir + PEG + RBV Patients (%) with SVR 12 100 82 80 74 73 60 41 40 20 20 0 26 137/167 40/98 F 0 -F 2 32/44 3/15 F 3 29/39 5/19 F 4 (Cirrhosis) Abbreviations: SVR 12 = sustained virologic response at 12 weeks; PEG = peginterferon; RBV = ribavirin Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
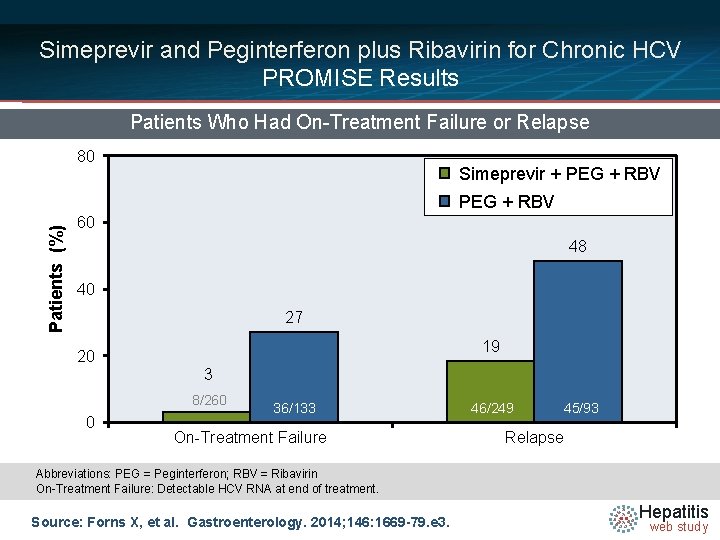
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Results Patients Who Had On-Treatment Failure or Relapse 80 Simeprevir + PEG + RBV Patients (%) PEG + RBV 60 48 40 27 19 20 3 8/260 0 36/133 On-Treatment Failure 46/249 45/93 Relapse Abbreviations: PEG = Peginterferon; RBV = Ribavirin On-Treatment Failure: Detectable HCV RNA at end of treatment. Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
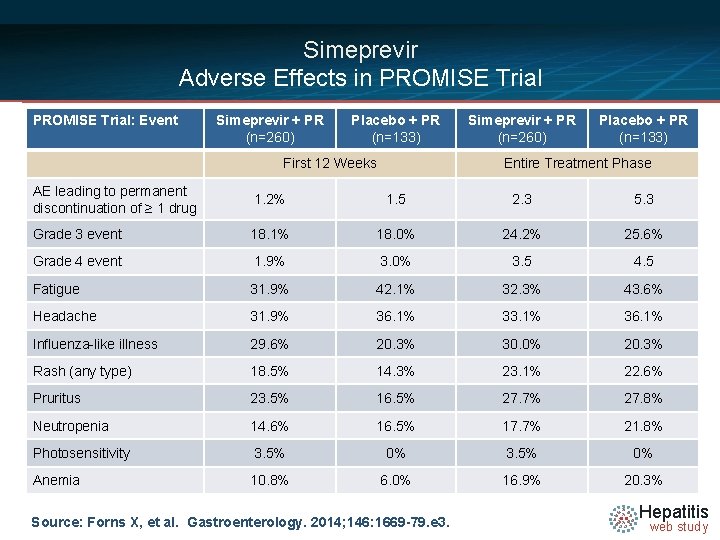
Simeprevir Adverse Effects in PROMISE Trial: Event Simeprevir + PR (n=260) Placebo + PR (n=133) First 12 Weeks Simeprevir + PR (n=260) Placebo + PR (n=133) Entire Treatment Phase AE leading to permanent discontinuation of ≥ 1 drug 1. 2% 1. 5 2. 3 5. 3 Grade 3 event 18. 1% 18. 0% 24. 2% 25. 6% Grade 4 event 1. 9% 3. 0% 3. 5 4. 5 Fatigue 31. 9% 42. 1% 32. 3% 43. 6% Headache 31. 9% 36. 1% 33. 1% 36. 1% Influenza-like illness 29. 6% 20. 3% 30. 0% 20. 3% Rash (any type) 18. 5% 14. 3% 23. 1% 22. 6% Pruritus 23. 5% 16. 5% 27. 7% 27. 8% Neutropenia 14. 6% 16. 5% 17. 7% 21. 8% Photosensitivity 3. 5% 0% Anemia 10. 8% 6. 0% 16. 9% 20. 3% Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study

Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Results Emergent Protease Resistance in Patients who Failed to Achieve SVR 12 § Most (90. 4%) of simeprevir-treated patients who failed to achieve SVR 12 developed emerging mutations in the NS 3 protease domain § Genotype 1 A: Most common mutation = R 155 K or D 168 E, or combination of R 155 K and mutations at codons 80 and/or 168 § Genotype 1 B: Most common mutations = D 168 V or D 168 A, E, T or E/V or the combinations Q 80 R + D 168 E/V, or Q 80 R + S 122 T + D 168 E Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study
Simeprevir and Peginterferon plus Ribavirin for Chronic HCV PROMISE Conclusions: “In a Phase 3 trial of patients who had relapsed following interferon-based therapy, addition of simeprevir to PR was generally well tolerated, with an SVR 12 rate of 79. 2%. Most patients (92. 7%) receiving simeprevir were able to shorten therapy to 24 weeks. ” Source: Forns X, et al. Gastroenterology. 2014; 146: 1669 -79. e 3. Hepatitis web study

Phase 3 Treatment Experienced Simeprevir versus Telaprevir with PR in GT 1 ATTAIN Trial Reddy KR, et al. Lancet Infect Dis. 2015; 15: 27 -35. Hepatitis web study

Simeprevir vs Telaprevir with Peginterferon + Ribavirin in GT 1 ATTAIN: Study Features ATTAIN Trial: Features § Design: Randomized, double-blind, phase 3, study evaluating simeprevir versus telaprevir with peginterferon alfa-2 a plus ribavirin for treatmentexperienced patients with genotype 1 chronic HCV § Setting: International at 169 sites in 24 countries § Entry Criteria - Chronic HCV genotype 1 - HCV RNA > 10, 000 IU/m. L - Adults ≥ 18 - Prior null or partial responder with prior peginterferon + ribavirin - Compensated liver disease § Exclusion Criteria - Non-HCV-related liver disease, including hepatocellular carcinoma - Prior HCV treatment with medication other than peginterferon + ribavirin - Coinfection with HAV, HBV, HIV, or non-genotype 1 HCV § Primary End-Points: Efficacy (SVR 12) Source: Reddy KR, et al. Lancet Infect Dis. 2015; 15: 27 -35. Hepatitis web study
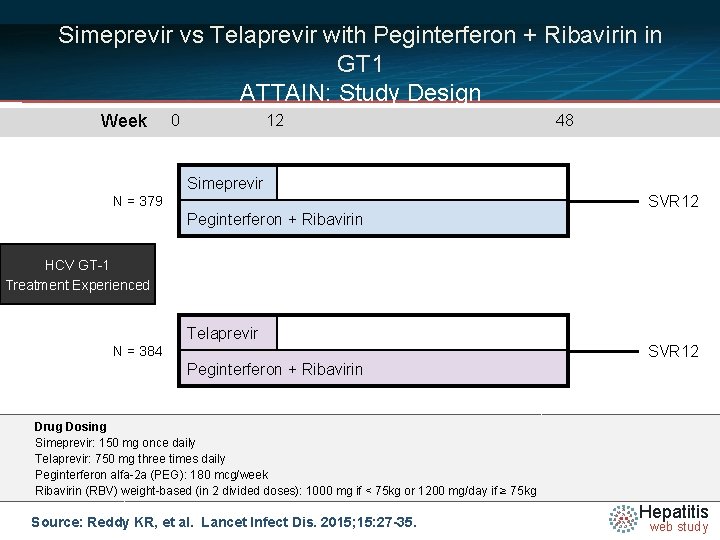
Simeprevir vs Telaprevir with Peginterferon + Ribavirin in GT 1 ATTAIN: Study Design Week 0 12 Simeprevir N = 379 Peginterferon + Ribavirin 48 SVR 12 HCV GT-1 Treatment Experienced Telaprevir N = 384 Peginterferon + Ribavirin SVR 12 Drug Dosing Simeprevir: 150 mg once daily Telaprevir: 750 mg three times daily Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Reddy KR, et al. Lancet Infect Dis. 2015; 15: 27 -35. Hepatitis web study
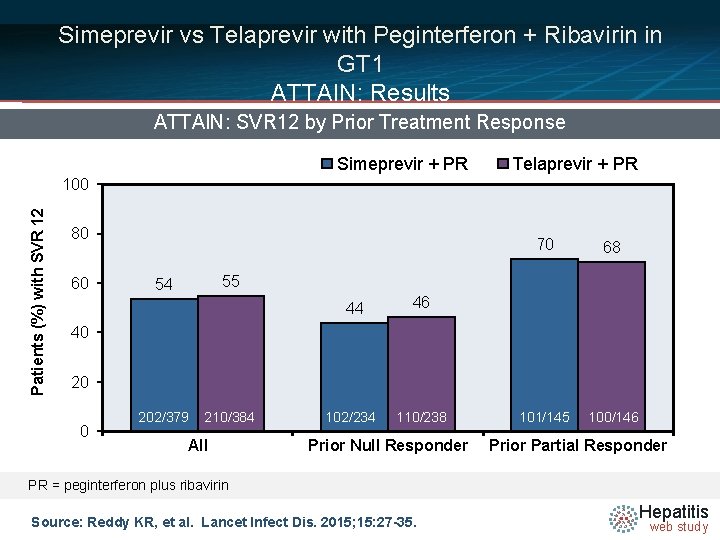
Simeprevir vs Telaprevir with Peginterferon + Ribavirin in GT 1 ATTAIN: Results ATTAIN: SVR 12 by Prior Treatment Response Simeprevir + PR Telaprevir + PR Patients (%) with SVR 12 100 80 60 70 68 101/145 100/146 55 54 44 46 102/234 110/238 40 202/379 210/384 All Prior Null Responder Prior Partial Responder PR = peginterferon plus ribavirin Source: Reddy KR, et al. Lancet Infect Dis. 2015; 15: 27 -35. Hepatitis web study

Simeprevir vs Telaprevir with Peginterferon + Ribavirin in GT 1 ATTAIN: Conclusions Interpretation: “Simeprevir once a day with peginterferon alfa-2 a and ribavirin was well tolerated in HCV genotype 1 -infected previous nonresponders and was non-inferior to telaprevir, thus providing an alternative treatment in areas of the world where all-oral HCV regimens are not available or accessible. ” Source: Reddy KR, et al. Lancet Infect Dis. 2015; 15: 27 -35. Hepatitis web study

Phase 3 Treatment Experienced Simeprevir in Treatment –Experienced Genotype 1 ASPIRE Trial Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Hepatitis web study

Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Features ASPIRE Trial: Study Features § Design: Randomized, double-blind, placebo-controlled, 7 arm, phase 2 b trial of PEG and RBV with and without simeprevir in HCV GT 1 for prior treatment failures with PEG and RBV § Setting: Europe, North America, Australia, and New Zealand § Entry Criteria - Treatment-experienced, chronic HCV GT-1 monoinfection - Prior failure with (≥ 12 weeks) of peginterferon-alfa plus ribavirin - HCV RNA > 10, 000 IU/m. L § Patient Characteristics - N = 462 - HCV Genotype: 1 a (41%); 1 b (58%); other (1%) - IL 28 B Genotype: 82% non-CC - Demographics: median age 50; 67% male; 93% white - Metavir Fibrosis: F 3 = 19%; F 4 = 18% § Primary end-points: Efficacy (SVR 24) Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Hepatitis web study
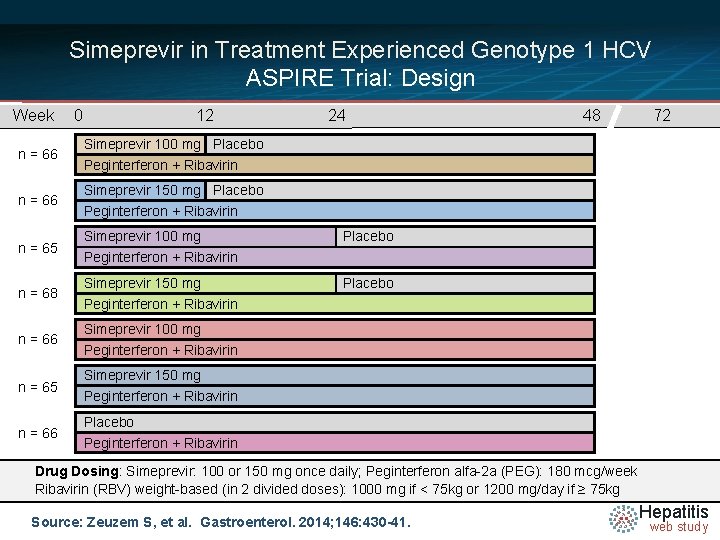
Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Design Week n = 66 n = 65 n = 68 n = 66 n = 65 n = 66 0 12 24 48 72 Simeprevir 100 mg Placebo Peginterferon + Ribavirin Simeprevir 150 mg Placebo Peginterferon + Ribavirin Simeprevir 100 mg Peginterferon + Ribavirin Simeprevir 150 mg Peginterferon + Ribavirin Placebo Peginterferon + Ribavirin Drug Dosing: Simeprevir: 100 or 150 mg once daily; Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Hepatitis web study

Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Results Week n = 66 n = 65 n = 68 n = 66 n = 65 n = 66 0 12 24 Simeprevir 100 mg Placebo Simeprevir 150 mg Placebo SVR 24 = 62% Peginterferon + Ribavirin Placebo Peginterferon + Ribavirin Simeprevir 150 mg 72 SVR 24 = 70% Peginterferon + Ribavirin Simeprevir 100 mg 48 Placebo Peginterferon + Ribavirin Simeprevir 100 mg Peginterferon + Ribavirin Simeprevir 150 mg Peginterferon + Ribavirin Placebo Peginterferon + Ribavirin SVR 24 = 66% SVR 24 = 72% SVR 24 = 61% SVR 24 = 80% SVR 24 = 23% Drug Dosing: Simeprevir: 100 or 150 mg once daily; Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Hepatitis web study
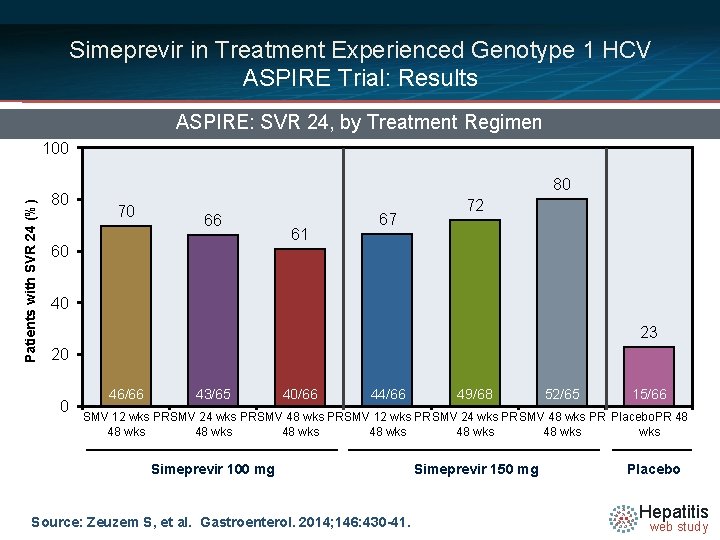
Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Results ASPIRE: SVR 24, by Treatment Regimen Patients with SVR 24 (%) 100 80 80 70 66 60 61 67 72 40 23 20 0 46/66 43/65 40/66 44/66 49/68 52/65 15/66 SMV 12 wks PR SMV 24 wks PR SMV 48 wks PR Placebo. PR 48 48 wks 48 wks wks Simeprevir 100 mg Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Simeprevir 150 mg Placebo Hepatitis web study
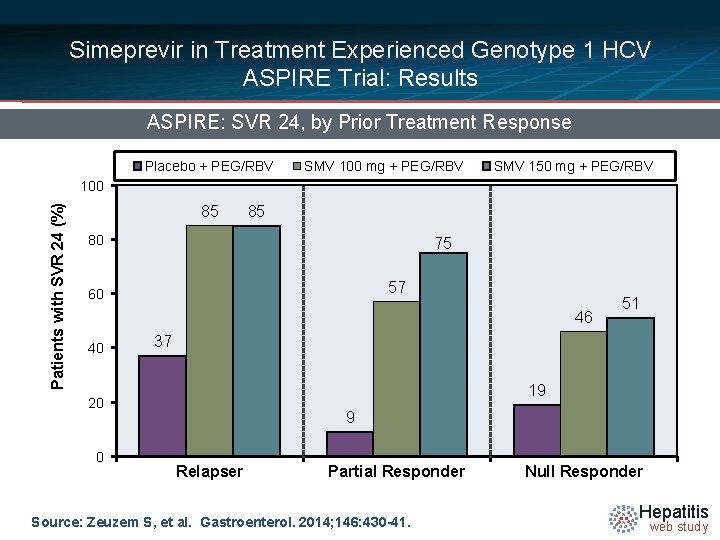
Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Results ASPIRE: SVR 24, by Prior Treatment Response Placebo + PEG/RBV SMV 100 mg + PEG/RBV SMV 150 mg + PEG/RBV Patients with SVR 24 (%) 100 85 85 80 75 57 60 46 40 20 46/66 0 51 37 43/65 40/66 44/66 49/68 19 52/65 15/66 9 Relapser Partial Responder Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Null Responder Hepatitis web study
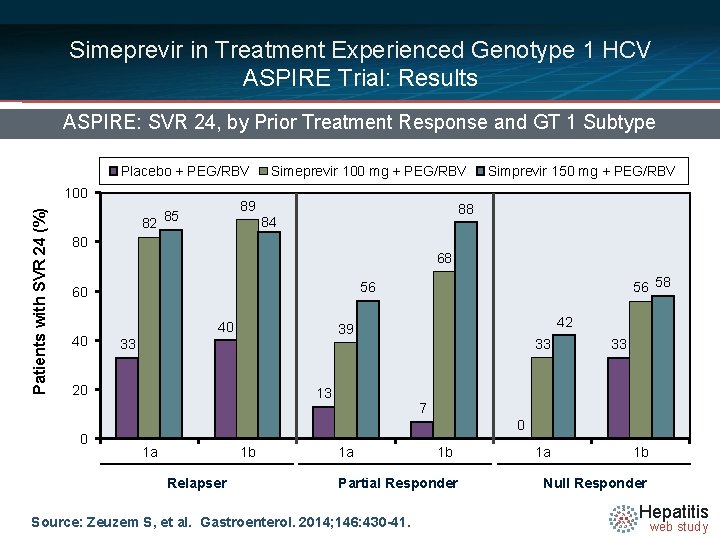
Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Results ASPIRE: SVR 24, by Prior Treatment Response and GT 1 Subtype Placebo + PEG/RBV Patients with SVR 24 (%) 100 82 Simeprevir 100 mg + PEG/RBV 89 85 88 84 80 68 40 33 46/66 42 39 20 0 56 58 56 60 40 Simprevir 150 mg + PEG/RBV 13 40/66 43/65 33 44/66 7 49/68 52/65 33 15/66 0 1 a 1 b Relapser 1 a 1 b Partial Responder Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. 1 a 1 b Null Responder Hepatitis web study

Simeprevir in Treatment Experienced Genotype 1 HCV ASPIRE Trial: Conclusions Conclusion: “In treatment-experienced patients, 12, 24, or 48 weeks simeprevir (100 mg or 150 mg once daily) in combination with 48 weeks peginterferon and ribavirin significantly increased rates of SVR at 24 weeks compared with patients given placebo, peginterferon, and ribavirin, and was generally well tolerated. ” Source: Zeuzem S, et al. Gastroenterol. 2014; 146: 430 -41. Hepatitis web study

Simeprevir in Treatment-Naïve and Treatment-Experienced Patients Hepatitis web study

Phase 3 Treatment Naïve and Treatment Experienced Simeprevir with Peginterferon and Ribavirin in GT-4 RESTORE Moreno C, et al. J Hepatol. 2015; 62: 1047 -55. Hepatitis web study

Simeprevir + Peginterferon + Ribavirin in Genotype 4 RESTORE: Study Features RESTORE Trial: Features § Design: Open-label, phase 3, study evaluating simeprevir plus peginterferon plus ribavirin for treatment naïve and experienced patients with genotype 4 chronic HCV § Setting: Multicenter and International § Entry Criteria - Chronic HCV genotype 4 (n = 107) - Treatment naïve (n = 35) or treatment experienced relapsers (n = 22) - Experienced (Nonresponder): partial (n = 10), null (n = 40) § Patient Characteristics - Sex: male 79% - Race: white (72%); black (28%) - Median age: 49 - IL genotype: 7. 5% CC - METAVIR Fibrosis Stage: F 4 = 29%; F 3 = 14% § Primary End-Points: Efficacy (SVR 12) Source: Moreno C, et al. J Hepatol. 2015; 62: 1047 -55. Hepatitis web study
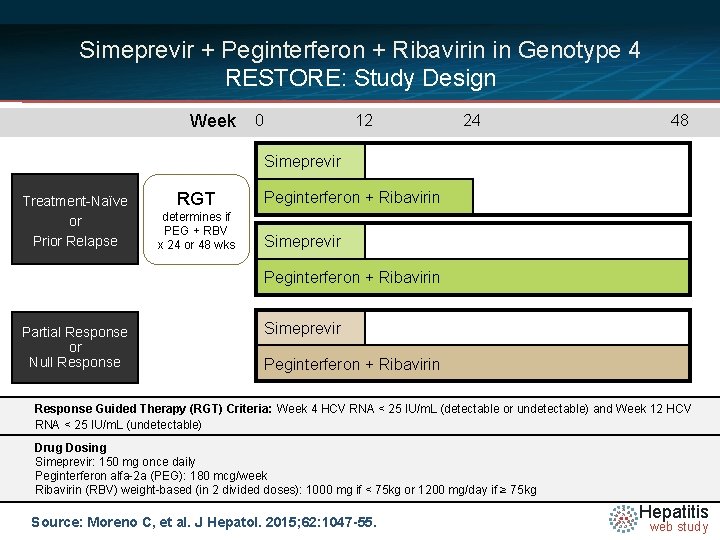
Simeprevir + Peginterferon + Ribavirin in Genotype 4 RESTORE: Study Design Week 0 12 24 48 Simeprevir Treatment-Naïve or Prior Relapse RGT determines if PEG + RBV x 24 or 48 wks Peginterferon + Ribavirin Simeprevir Peginterferon + Ribavirin Partial Response or Null Response Simeprevir Peginterferon + Ribavirin Response Guided Therapy (RGT) Criteria: Week 4 HCV RNA < 25 IU/m. L (detectable or undetectable) and Week 12 HCV RNA < 25 IU/m. L (undetectable) Drug Dosing Simeprevir: 150 mg once daily Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Moreno C, et al. J Hepatol. 2015; 62: 1047 -55. Hepatitis web study
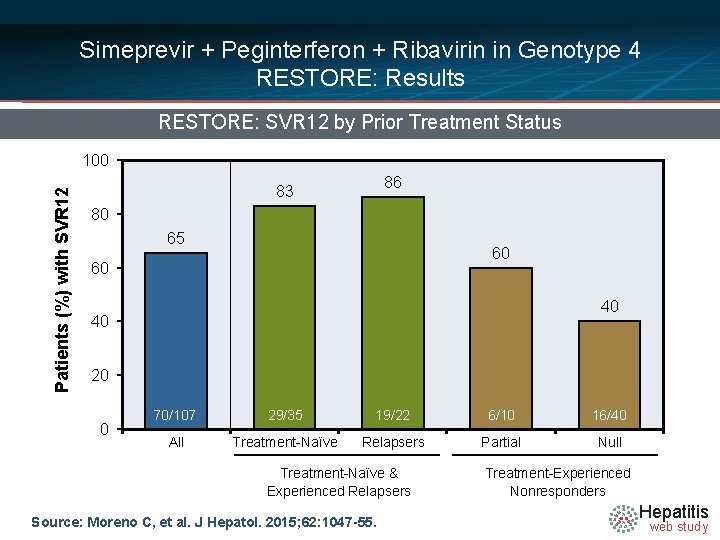
Simeprevir + Peginterferon + Ribavirin in Genotype 4 RESTORE: Results RESTORE: SVR 12 by Prior Treatment Status Patients (%) with SVR 12 100 86 83 80 65 60 60 40 40 20 0 70/107 29/35 19/22 6/10 16/40 All Treatment-Naïve Relapsers Partial Null Treatment-Naïve & Experienced Relapsers Source: Moreno C, et al. J Hepatol. 2015; 62: 1047 -55. Treatment-Experienced Nonresponders Hepatitis web study

Simeprevir + Peginterferon + Ribavirin in Genotype 4 RESTORE: Conclusions: “Efficacy and safety of simeprevir 150 mg QD for 12 weeks with peginterferon and ribavirin in treatment-naïve or -experienced patients with chronic HCV GT 4 infection were in line with previous reports for HCV GT 1 infection. ” Source: Moreno C, et al. J Hepatol. 2015; 62: 1047 -55. Hepatitis web study

Simeprevir + Sofosbuvir Hepatitis web study

Phase 2 a, Treatment Naïve and Treatment Experienced Simeprevir + Sofosbuvir +/- Ribavirin in Genotype 1 COSMOS Trial Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study

Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Study Features COSMOS Trial: Features § Design: Randomized, phase 2 a, open-label, using sofosbuvir + simeprevir +/- ribavirin in treatment naive or experienced, chronic HCV GT 1 § Setting: 23 centers in United States § Entry Criteria - Chronic HCV Genotype 1 - Age ≥ 18 - HCV RNA greater than 10, 000 IU/m. L - Cohort 1: prior nonresponders; Metavir F 0 -F 2 - Cohort 2: treatment naïve & prior nonresponders; Metavir F 3 -F 4 § Patient Characteristics (range in different treatment arms) - N = 167 (n = 80 in Cohort 1 and n = 87 in Cohort 2) - Baseline GT 1 a with Q 80 K: Cohort 1 = 50%; Cohort 2 = 40% - Non-CC IL 28 b Genotype: Cohort 1 = 94%; Cohort 2 = 79% § End-Points: Primary = SVR 12 Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study
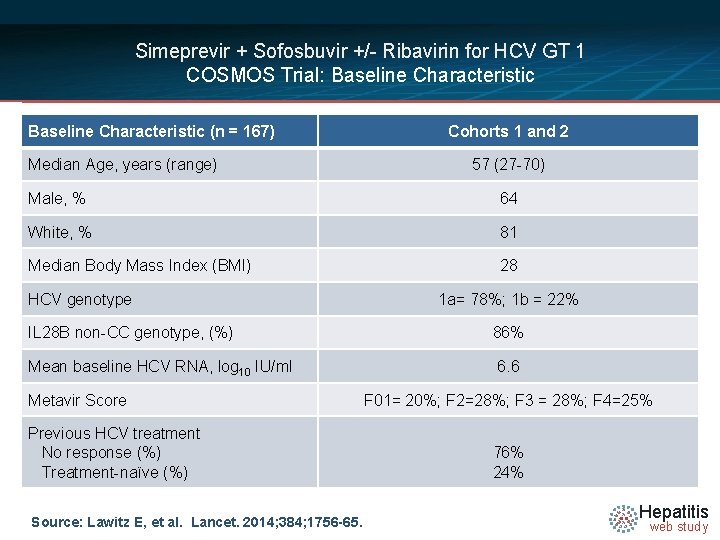
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Baseline Characteristic (n = 167) Median Age, years (range) Cohorts 1 and 2 57 (27 -70) Male, % 64 White, % 81 Median Body Mass Index (BMI) 28 HCV genotype IL 28 B non-CC genotype, (%) Mean baseline HCV RNA, log 10 IU/ml Metavir Score Previous HCV treatment No response (%) Treatment-naïve (%) Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. 1 a= 78%; 1 b = 22% 86% 6. 6 F 01= 20%; F 2=28%; F 3 = 28%; F 4=25% 76% 24% Hepatitis web study
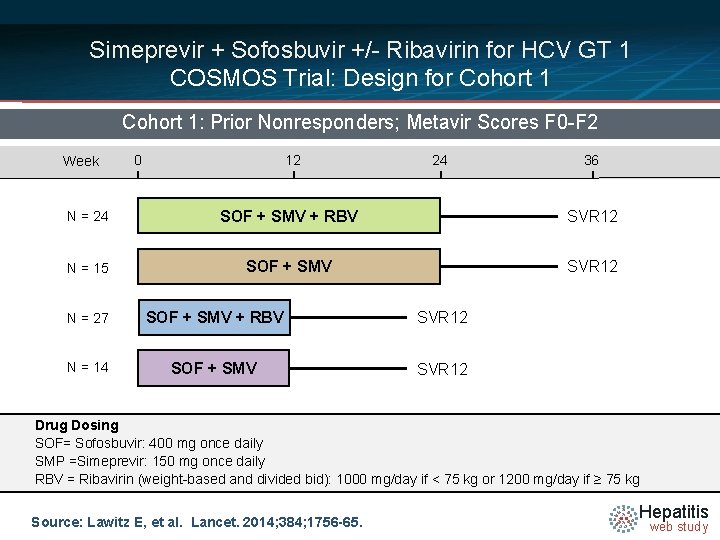
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Design for Cohort 1: Prior Nonresponders; Metavir Scores F 0 -F 2 Week 0 12 24 36 N = 24 SOF + SMV + RBV SVR 12 N = 15 SOF + SMV SVR 12 N = 27 SOF + SMV + RBV SVR 12 N = 14 SOF + SMV SVR 12 Drug Dosing SOF= Sofosbuvir: 400 mg once daily SMP =Simeprevir: 150 mg once daily RBV = Ribavirin (weight-based and divided bid): 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study
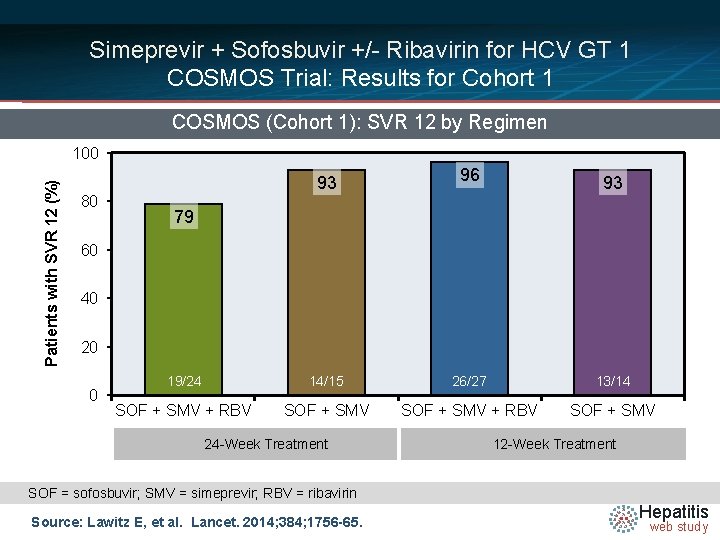
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Results for Cohort 1 COSMOS (Cohort 1): SVR 12 by Regimen Patients with SVR 12 (%) 100 93 80 96 93 79 60 40 20 0 19/24 14/15 26/27 13/14 SOF + SMV + RBV SOF + SMV 24 -Week Treatment 12 -Week Treatment SOF = sofosbuvir; SMV = simeprevir; RBV = ribavirin Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study
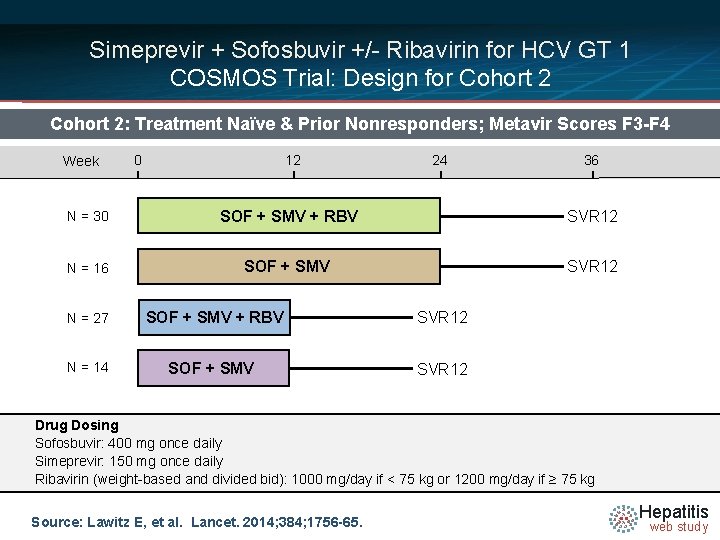
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Design for Cohort 2: Treatment Naïve & Prior Nonresponders; Metavir Scores F 3 -F 4 Week 0 12 24 36 N = 30 SOF + SMV + RBV SVR 12 N = 16 SOF + SMV SVR 12 N = 27 SOF + SMV + RBV SVR 12 N = 14 SOF + SMV SVR 12 Drug Dosing Sofosbuvir: 400 mg once daily Simeprevir: 150 mg once daily Ribavirin (weight-based and divided bid): 1000 mg/day if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study
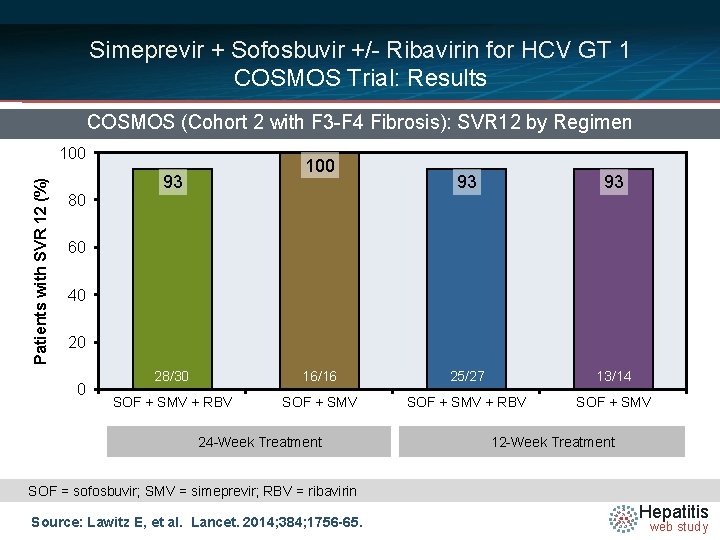
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Results COSMOS (Cohort 2 with F 3 -F 4 Fibrosis): SVR 12 by Regimen Patients with SVR 12 (%) 100 80 100 93 93 93 60 40 20 0 28/30 16/16 25/27 13/14 SOF + SMV + RBV SOF + SMV 24 -Week Treatment 12 -Week Treatment SOF = sofosbuvir; SMV = simeprevir; RBV = ribavirin Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study
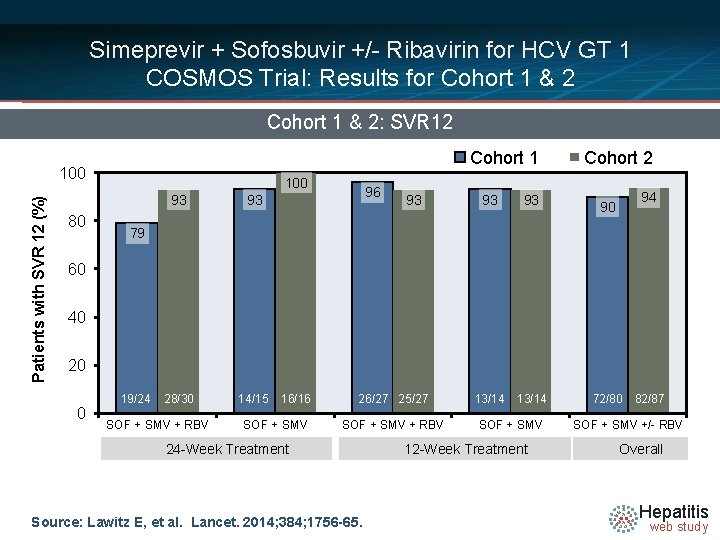
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Results for Cohort 1 & 2: SVR 12 Cohort 1 Patients with SVR 12 (%) 100 80 100 93 93 28/30 14/15 96 93 93 93 13/14 Cohort 2 90 94 79 60 40 20 0 19/24 SOF + SMV + RBV 16/16 SOF + SMV 26/27 25/27 SOF + SMV + RBV 24 -Week Treatment Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. SOF + SMV 12 -Week Treatment 72/80 82/87 SOF + SMV +/- RBV Overall Hepatitis web study
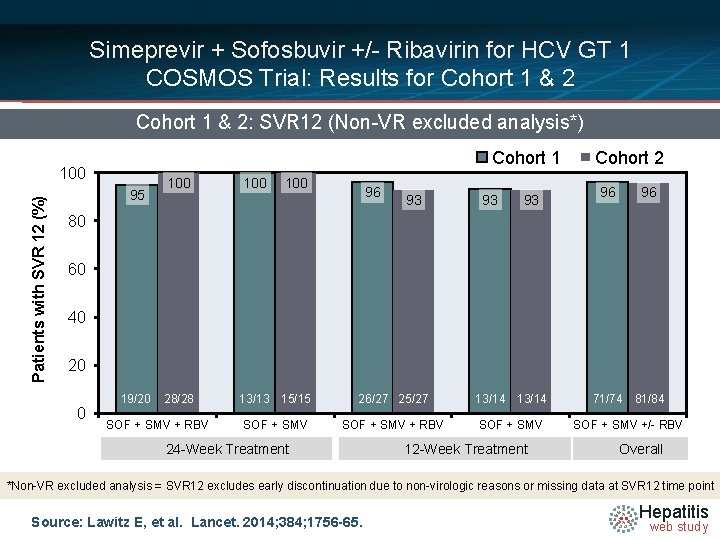
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Results for Cohort 1 & 2: SVR 12 (Non-VR excluded analysis*) Cohort 1 Patients with SVR 12 (%) 100 95 100 100 96 93 93 93 Cohort 2 96 96 80 60 40 20 0 19/20 28/28 SOF + SMV + RBV 13/13 15/15 26/27 25/27 13/14 71/74 81/84 SOF + SMV + RBV SOF + SMV +/- RBV 24 -Week Treatment 12 -Week Treatment Overall *Non-VR excluded analysis = SVR 12 excludes early discontinuation due to non-virologic reasons or missing data at SVR 12 time point Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study
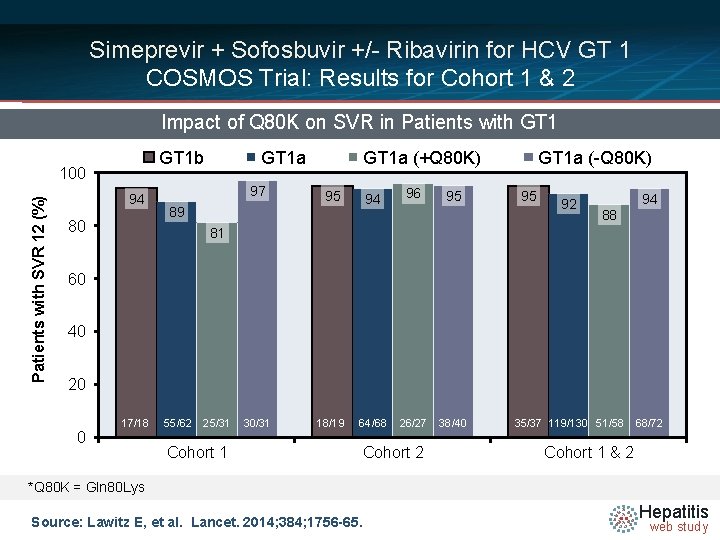
Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Results for Cohort 1 & 2 Impact of Q 80 K on SVR in Patients with GT 1 b Patients with SVR 12 (%) 100 94 80 GT 1 a 97 GT 1 a (+Q 80 K) 95 94 89 96 95 GT 1 a (-Q 80 K) 95 92 94 88 81 60 40 20 0 17/18 55/62 25/31 30/31 18/19 64/68 Cohort 1 26/27 Cohort 2 38/40 35/37 119/130 51/58 68/72 Cohort 1 & 2 *Q 80 K = Gln 80 Lys Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study

Simeprevir + Sofosbuvir +/- Ribavirin for HCV GT 1 COSMOS Trial: Interpretation: “Combined simeprevir and sofosbuvir was efficacious and well tolerated. ” Source: Lawitz E, et al. Lancet. 2014; 384; 1756 -65. Hepatitis web study

Phase 3 Treatment Naïve and Treatment Experienced Simeprevir + Sofosbuvir in GT 1 without Cirrhosis OPTIMIST-1 Trial Kwo P, et al. 50 th EASL; 2015. Abstract LB 14. Hepatitis web study

Simeprevir + Sofosbuvir for HCV GT 1 without Cirrhosis OPTIMIST-1 Trial: Study Features OPTIMIST 1 Trial: Features § Design: Randomized, phase 3, open-label, using sofosbuvir plus simeprevir for 8 or 12 weeks in treatment naive or experienced patients with chronic HCV genotype 1 infection without cirrhosis § Setting: multicenter in United States and Canada § Entry Criteria - Chronic HCV Genotype 1 a or 1 b - Documented lack of cirrhosis - Age 18 -70 - HCV RNA greater than 10, 000 IU/m. L - Treatment experienced required to have ≥ 1 INF-based regimen +/- RBV § Exclusion - Cirrhosis, hepatic decompensation, or non-HCV-related liver disease - Coinfection with HBV or HIV § End-Points: Primary = SVR 12 by intent-to-treat analysis Source: Kwo P, et al. 50 th EASL; 2015. Abstract LB 14. Hepatitis web study
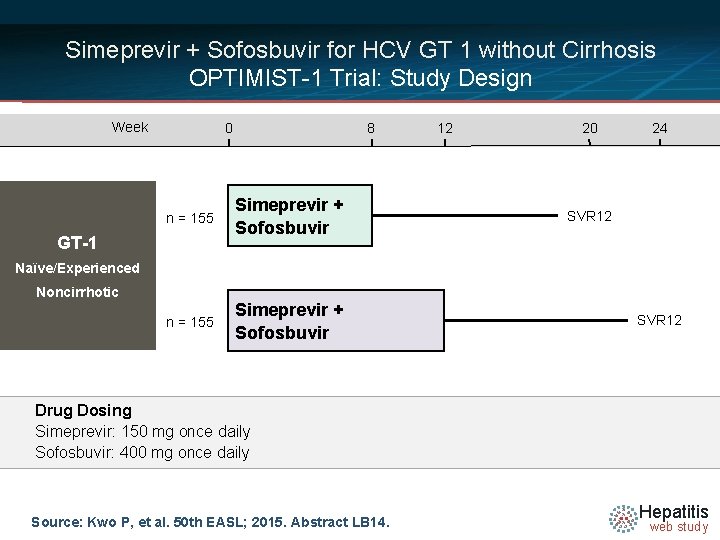
Simeprevir + Sofosbuvir for HCV GT 1 without Cirrhosis OPTIMIST-1 Trial: Study Design Week 0 8 n = 155 Simeprevir + Sofosbuvir GT-1 12 20 24 SVR 12 Naïve/Experienced Noncirrhotic SVR 12 Drug Dosing Simeprevir: 150 mg once daily Sofosbuvir: 400 mg once daily Source: Kwo P, et al. 50 th EASL; 2015. Abstract LB 14. Hepatitis web study
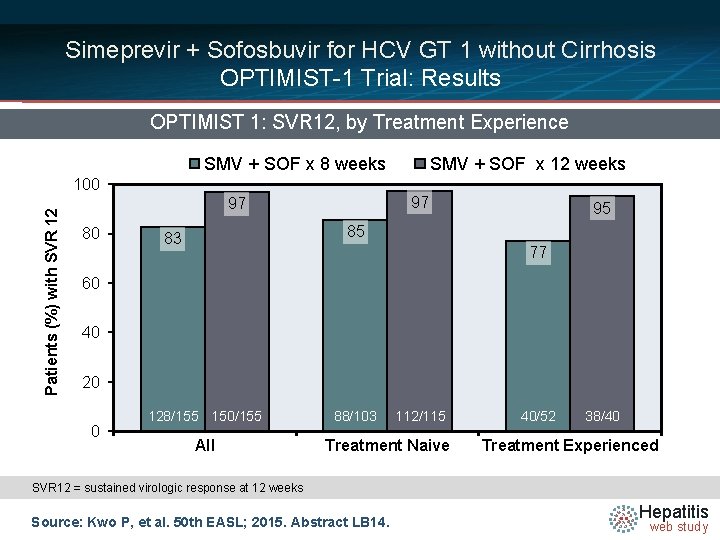
Simeprevir + Sofosbuvir for HCV GT 1 without Cirrhosis OPTIMIST-1 Trial: Results OPTIMIST 1: SVR 12, by Treatment Experience SMV + SOF x 8 weeks Patients (%) with SVR 12 100 97 97 80 SMV + SOF x 12 weeks 95 85 83 77 60 40 20 0 128/155 150/155 All 88/103 112/115 Treatment Naive 40/52 38/40 Treatment Experienced SVR 12 = sustained virologic response at 12 weeks Source: Kwo P, et al. 50 th EASL; 2015. Abstract LB 14. Hepatitis web study
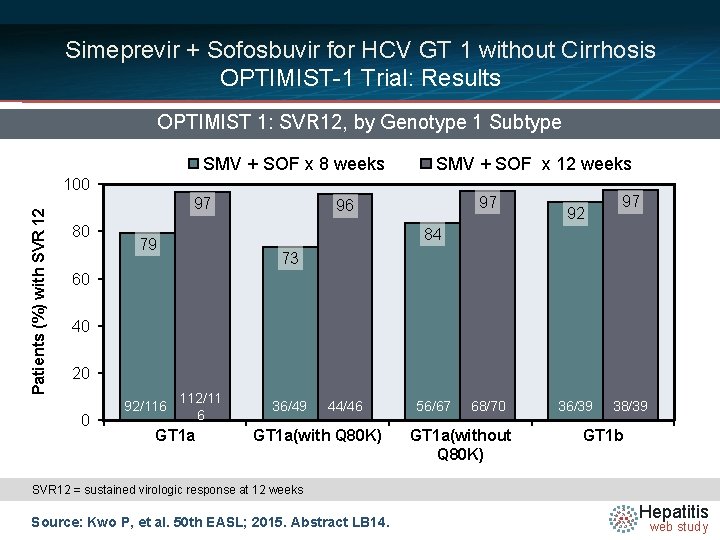
Simeprevir + Sofosbuvir for HCV GT 1 without Cirrhosis OPTIMIST-1 Trial: Results OPTIMIST 1: SVR 12, by Genotype 1 Subtype SMV + SOF x 8 weeks SMV + SOF x 12 weeks Patients (%) with SVR 12 100 97 80 97 96 92 97 84 79 73 60 40 20 0 92/116 112/11 6 GT 1 a 36/49 44/46 GT 1 a(with Q 80 K) 56/67 68/70 GT 1 a(without Q 80 K) 36/39 38/39 GT 1 b SVR 12 = sustained virologic response at 12 weeks Source: Kwo P, et al. 50 th EASL; 2015. Abstract LB 14. Hepatitis web study

Phase 3 Treatment Naïve and Treatment Experienced Simeprevir + Sofosbuvir in GT 1 with Cirrhosis OPTIMIST-2 Trial Lawitz E, et al. 50 th EASL; 2015. Abstract LP 04. Hepatitis web study

Simeprevir + Sofosbuvir for HCV GT 1 with Cirrhosis OPTIMIST-2 Trial: Study Features OPTIMIST 2 Trial: Features § Design: Randomized, phase 3, open-label, single-arm trial using sofosbuvir plus simeprevir for 12 weeks in treatment naive or experienced patients with chronic HCV genotype 1 infection and compensated cirrhosis § Setting: multicenter in United States and Canada § Entry Criteria - Chronic HCV Genotype 1 infection - Studies indicating cirrhosis with compensation - Age 18 -70 - HCV RNA greater than 10, 000 IU/m. L - Any treatment history allowed § Exclusion - Hepatic decompensation, or non-HCV-related liver disease - Coinfection with HBV or HIV § End-Points: Primary = SVR 12 by intent-to-treat analysis Source: Lawitz E, et al. 50 th EASL; 2015. Abstract LP 04. Hepatitis web study
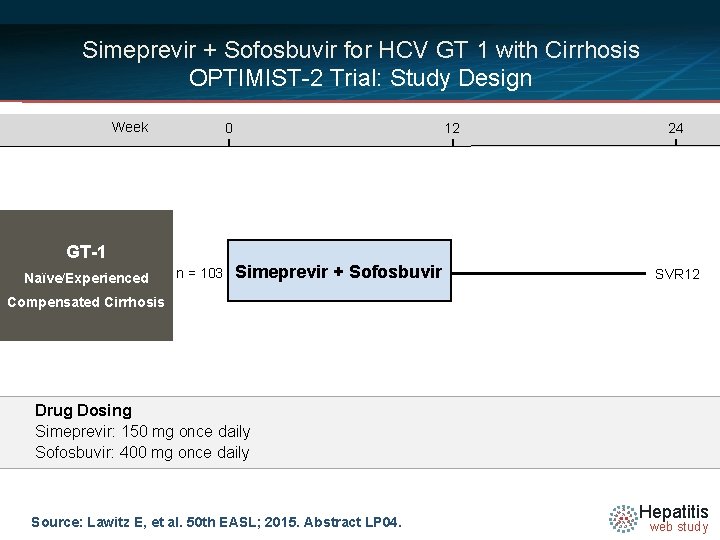
Simeprevir + Sofosbuvir for HCV GT 1 with Cirrhosis OPTIMIST-2 Trial: Study Design Week 0 GT-1 Naïve/Experienced n = 103 12 Simeprevir + Sofosbuvir 24 SVR 12 Compensated Cirrhosis Drug Dosing Simeprevir: 150 mg once daily Sofosbuvir: 400 mg once daily Source: Lawitz E, et al. 50 th EASL; 2015. Abstract LP 04. Hepatitis web study
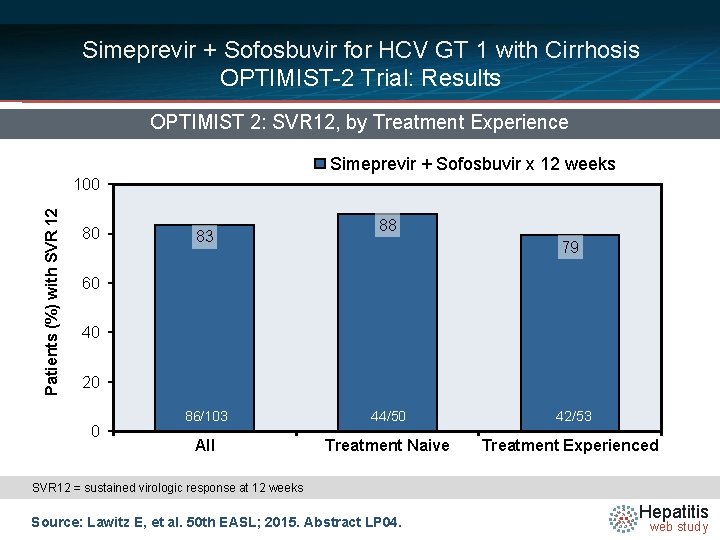
Simeprevir + Sofosbuvir for HCV GT 1 with Cirrhosis OPTIMIST-2 Trial: Results OPTIMIST 2: SVR 12, by Treatment Experience Simeprevir + Sofosbuvir x 12 weeks Patients (%) with SVR 12 100 80 83 88 79 60 40 20 0 86/103 44/50 42/53 All Treatment Naive Treatment Experienced SVR 12 = sustained virologic response at 12 weeks Source: Lawitz E, et al. 50 th EASL; 2015. Abstract LP 04. Hepatitis web study
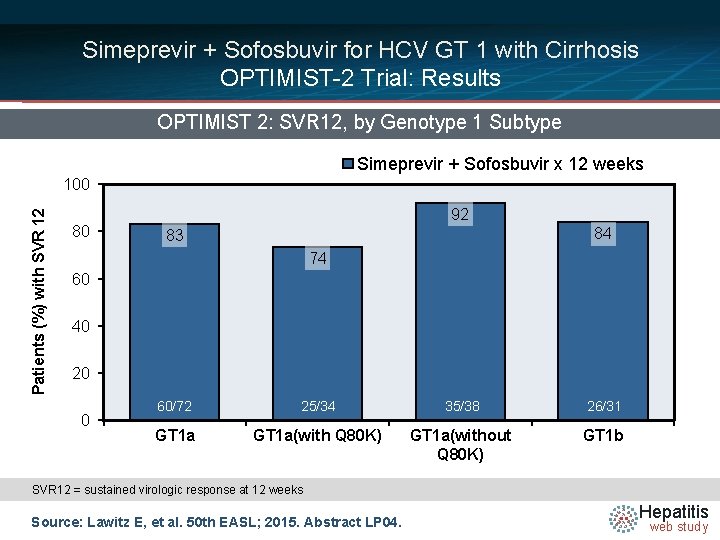
Simeprevir + Sofosbuvir for HCV GT 1 with Cirrhosis OPTIMIST-2 Trial: Results OPTIMIST 2: SVR 12, by Genotype 1 Subtype Simeprevir + Sofosbuvir x 12 weeks Patients (%) with SVR 12 100 80 92 84 83 74 60 40 20 0 60/72 25/34 35/38 26/31 GT 1 a(with Q 80 K) GT 1 a(without Q 80 K) GT 1 b SVR 12 = sustained virologic response at 12 weeks Source: Lawitz E, et al. 50 th EASL; 2015. Abstract LP 04. Hepatitis web study

Simeprevir in Patients with HCV-HIV Coinfection Hepatitis web study

Phase 3 Treatment Naïve and Treatment Experienced HIV Coinfection Simeprevir in HIV Coinfection, GT-1 C 212 Trial Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study

Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Study Features C 212 Trial: Features § Design: Open-label, phase 3, trial evaluating simeprevir + PEG + RBV in HCV-HIV and GT 1 (treatment naïve and experienced) § Setting: 39 sites in 7 countries § Entry Criteria - HIV coinfection; HCV genotype 1 - Treatment naïve or treatment experienced - Group 1: HCV treatment-naïve or prior relapse - Group 2: Prior partial or null response or cirrhosis - CD 4 ≥ 200 if on stable ARV therapy; CD 4 ≥ 500 if no ARV therapy - Stable antiretroviral therapy = HIV RNA < 50 copies/ml > 8 weeks § Patient Characteristics - N = 106 HCV-HIV coinfected patients - Race: white (82%); black (14%) - Baseline Median CD 4 (cells/mm 3): 629 cells/mm 3 § Primary End-Points: Efficacy (SVR 12), safety, and impact on HIV Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study

Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Design Week 0 12 24 48 Simeprevir Treatment-Naïve or Prior Relapse Response Guided Therapy Peginterferon + Ribavirin Simeprevir Peginterferon + Ribavirin Partial Response or Null Response Simeprevir Peginterferon + Ribavirin Drug Dosing Simeprevir: 150 mg once daily Peginterferon alfa-2 a (PEG): 180 mcg/week Ribavirin (RBV) weight-based (in 2 divided doses): 1000 mg if < 75 kg or 1200 mg/day if ≥ 75 kg Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study
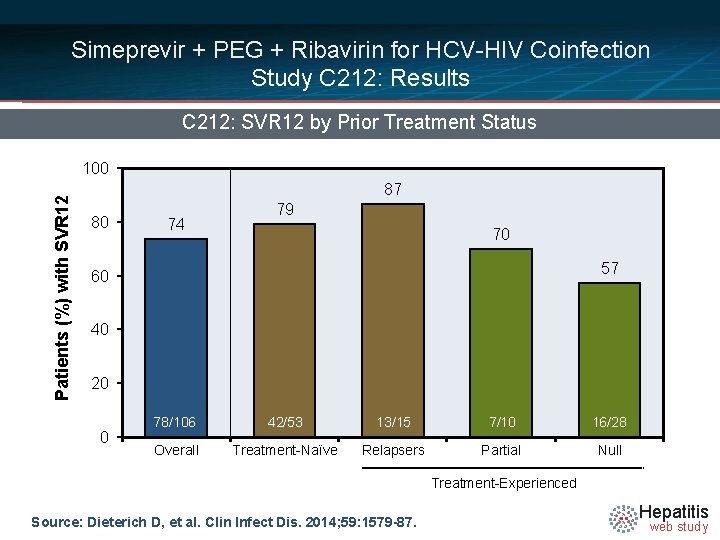
Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Results C 212: SVR 12 by Prior Treatment Status Patients (%) with SVR 12 100 87 80 74 79 70 57 60 40 20 0 78/106 42/53 13/15 7/10 16/28 Overall Treatment-Naïve Relapsers Partial Null Treatment-Experienced Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study
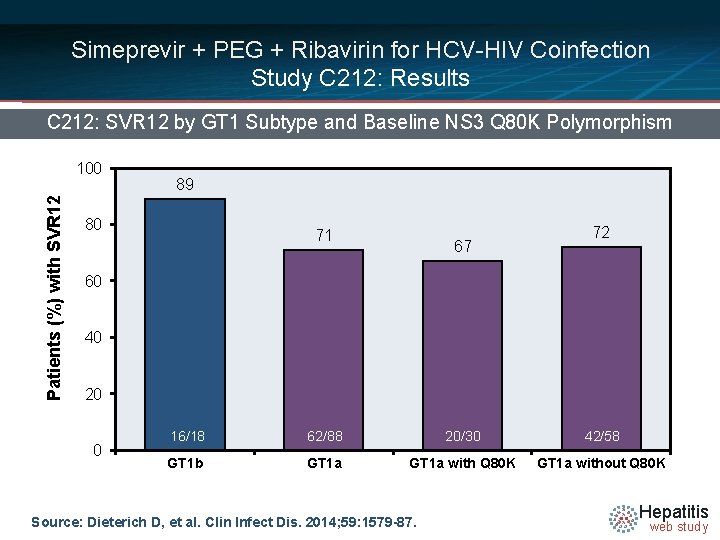
Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Results C 212: SVR 12 by GT 1 Subtype and Baseline NS 3 Q 80 K Polymorphism Patients (%) with SVR 12 100 89 80 71 67 72 60 40 20 0 16/18 62/88 20/30 42/58 GT 1 b GT 1 a with Q 80 K GT 1 a without Q 80 K Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study
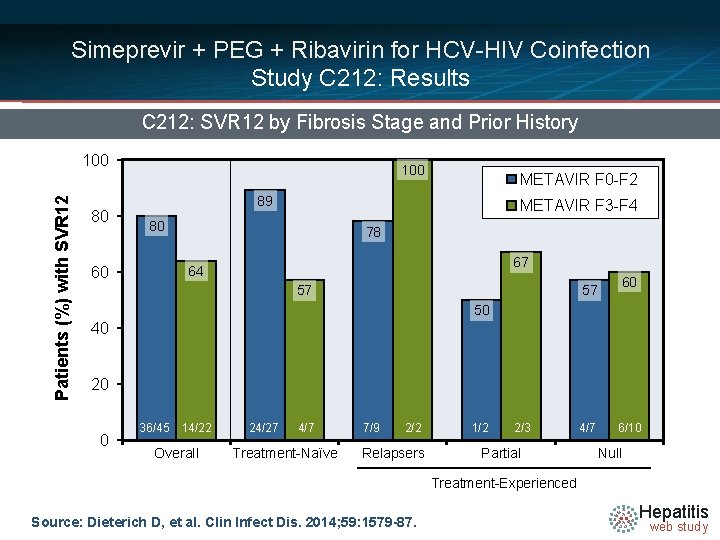
Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Results C 212: SVR 12 by Fibrosis Stage and Prior History Patients (%) with SVR 12 100 80 100 METAVIR F 0 -F 2 89 METAVIR F 3 -F 4 80 78 67 64 60 57 57 60 50 40 20 0 36/45 14/22 Overall 24/27 4/7 Treatment-Naïve 7/9 2/2 Relapsers 1/2 2/3 Partial 4/7 6/10 Null Treatment-Experienced Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study
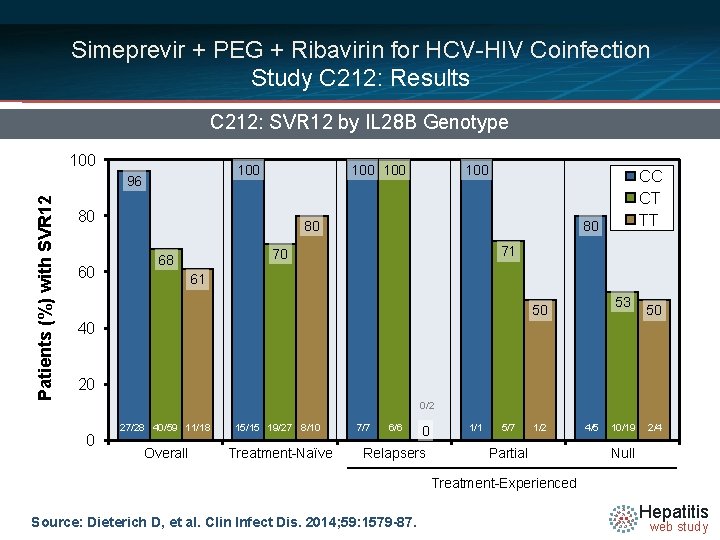
Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Results C 212: SVR 12 by IL 28 B Genotype 100 Patients (%) with SVR 12 96 100 80 60 100 80 80 71 70 68 CC CT TT 61 53 50 50 40 20 0/2 0 27/28 40/59 11/18 15/15 19/27 8/10 Overall Treatment-Naïve 7/7 6/6 0 Relapsers 1/1 5/7 1/2 Partial 4/5 10/19 2/4 Null Treatment-Experienced Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study

Simeprevir + PEG + Ribavirin for HCV-HIV Coinfection Study C 212: Conclusions: “Simeprevir was generally well tolerated with safety similar to that observed in HCV-monoinfected patients and high SVR 12 rates in HCV treatment-naive patients, prior relapsers, prior partial responders, and prior null responders with HIV-1 coinfection. ” Source: Dieterich D, et al. Clin Infect Dis. 2014; 59: 1579 -87. Hepatitis web study

This slide deck is from the University of Washington’s Hepatitis C Online and Hepatitis Web Study projects. Hepatitis C Online www. hepatitisc. uw. edu Hepatitis Web Study http: //depts. washington. edu/hepstudy/ Funded by a grant from the Centers for Disease Control and Prevention. Hepatitis web study
- Slides: 100