HEPATITIS B VIRUS WHATS NEW EASL treatment guidelines

HEPATITIS B VIRUS ; WHAT`S NEW ? EASL treatment guidelines 2017 AASLD treatment guidelines 2018 Dr. Ahmed Zeid Prof. Internal Medicine- Hepatology Unit Alexandria Faculty of Medicine 1
2

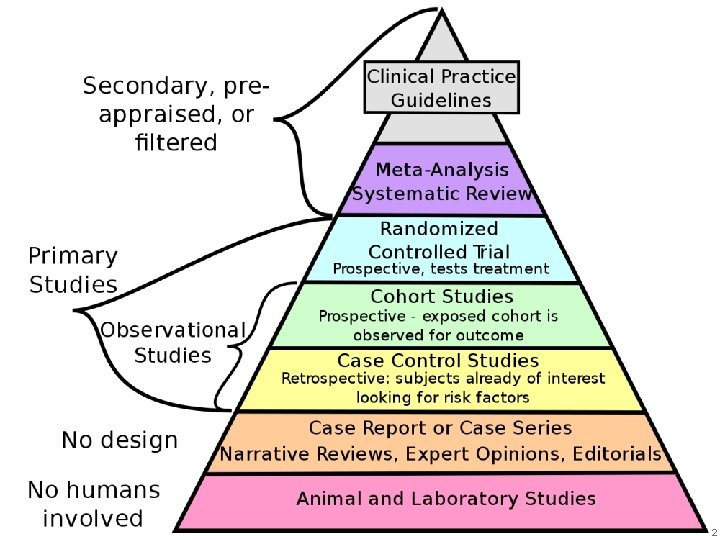
Agenda • Basic data. • Who to treat ? • How to treat ? • When to stop treatment ? 3
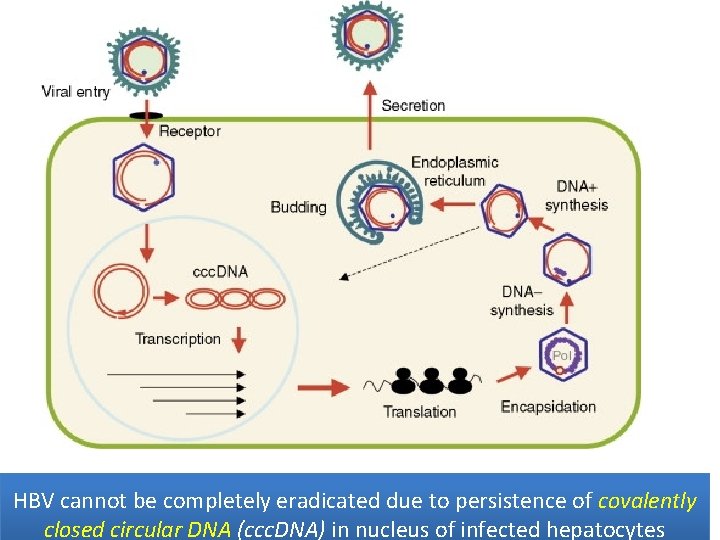
HBV cannot be completely eradicated due to persistence of covalently closed circular DNA (ccc. DNA) in nucleus of infected hepatocytes
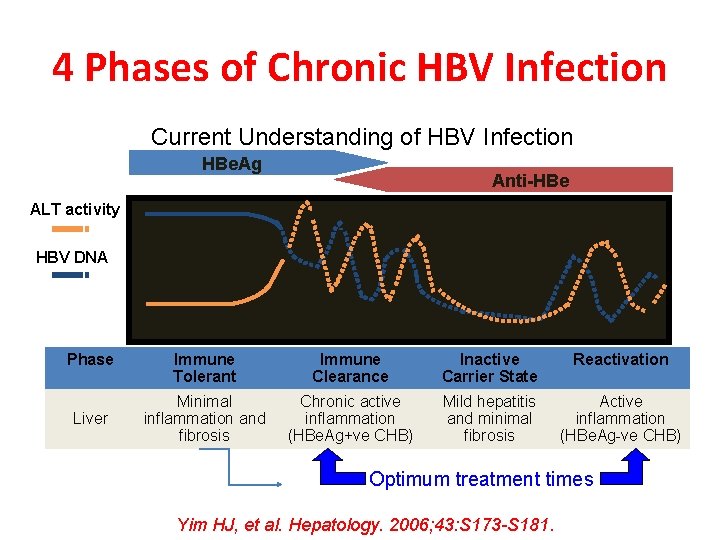
4 Phases of Chronic HBV Infection Current Understanding of HBV Infection HBe. Ag Anti-HBe ALT activity HBV DNA Phase Liver Immune Tolerant Immune Clearance Inactive Carrier State Reactivation Minimal inflammation and fibrosis Chronic active inflammation (HBe. Ag+ve CHB) Mild hepatitis and minimal fibrosis Active inflammation (HBe. Ag-ve CHB) Optimum treatment times Yim HJ, et al. Hepatology. 2006; 43: S 173 -S 181.

EASL 2017 Phase 5: ‘‘occult HBV infection” • HBs. Ag-negative ± HBs. Ab positive. • HBc. Ab positive. • ± Serum HBV DNA. • HBV DNA (ccc. DNA) can be detected in liver.

WHO TO TREAT ? ? 7
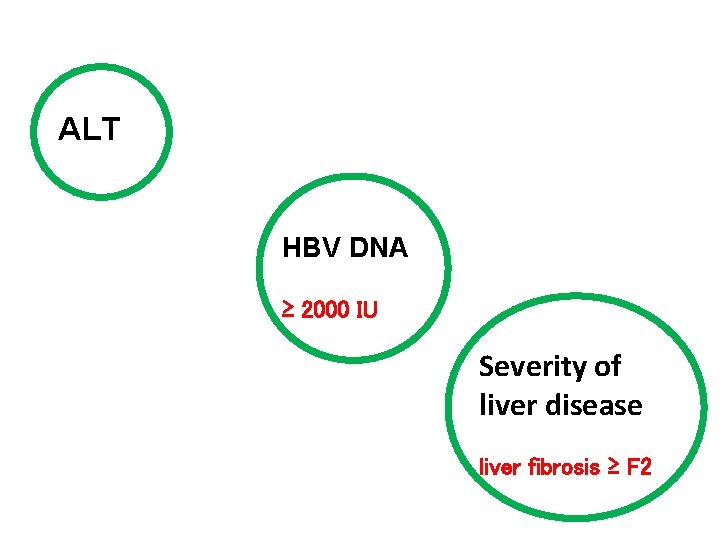
ALT HBV DNA ≥ 2000 IU Severity of liver disease liver fibrosis ≥ F 2
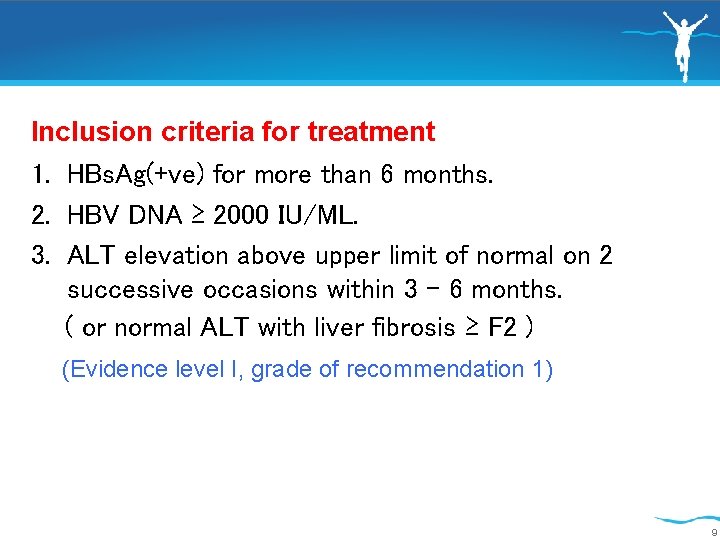
Inclusion criteria for treatment 1. HBs. Ag(+ve) for more than 6 months. 2. HBV DNA ≥ 2000 IU/ML. 3. ALT elevation above upper limit of normal on 2 successive occasions within 3 - 6 months. ( or normal ALT with liver fibrosis ≥ F 2 ) (Evidence level I, grade of recommendation 1) 9

• Patients with immune tolerant phase (HBe. Ag-positive chronic HBV infection) may be treated if they are > 30 years regardless of severity of liver disease. • Patients with HBe. Ag-positive or HBe. Ag-negative chronic HBV infection and family history of HCC or cirrhosis and extrahepatic manifestations can be treated even if typical treatment indications are not fulfilled. (Evidence level III, grade of recommendation 2). 10

HOW TO TREAT ? ? 11

FDA-Approved HBV Therapies Peginterferon alfa-2 a Entecavir Lamivudine 1990 Interferon alfa-2 b 1998 TAF 2002 Adefovir 2005 Tenofovir 2006 Telbivudine 2008

Interferon Advantages of interferon • A finite course of therapy for naïve patients. • No viral resistance. • High rate of HBe. Ag loss with in 1 year. • High rate of HBs. Ag loss with short duration The response rate is 20 -30% 13

Interferon Pre-treatment predictors of response • In HBe. Ag positive CHB, predictors of anti-HBe seroconversion 1) low viral load. 2) high serum ALT levels (above 2– 5 times ULN). 3) HBV genotypes A and B. 4) high activity scores on liver biopsy (at least A 2). • In HBe. Ag negative CHB, There are no strong pre-treatment predictors of virological response. 14

First line therapy for all naïve patients is: Entecavir 0. 5 mg once daily Or Tenofovir 300 mg once daily 15

In HBV patients with liver cirrhosis: All cirrhotic patients should receive oral therapy if HBV DNA is detectable irrespective of viral load. Compensated Cirrhosis: Entecavir 0. 5 mg or Tenofovir 300 mg once daily. Decompensated Cirrhosis: Entecavir 1 mg once daily. Note: The dose of antiviral needs to be adjusted in patients with low creatinine clearance (< 50 ml/min). 16

Treatment of HBV in special populations 17

Acute HBV infection Spontaneous recovery in more than 95% of cases and seroconversion to anti HBs without antiviral therapy. Supportive management and close monitoring for early detection of fulminant hepatitis. Acute severe hepatitis and Fulminant hepatitis : Entecavir 0. 5 mg for at least 6 months after seroconversion to anti HBs or for at least 12 months after seroconversion to anti HBe without HBs Ag loss. 18

HCV co-infected patients

HBV and pregnancy • In pregnant women already on NA therapy, TDF should be continued while ETV or other NA switched to TDF • In all pregnant women with high HBV DNA levels (200, 000 IU/ml), antiviral prophylaxis with TDF should start at week 24– 28 of gestation and continue for up to 12 weeks after delivery 20

New born • Newborns for chronic HBV mothers should receive HBIG and the first dose of HBV vaccine at birth (612 hours after delivery). • Breast feeding is not contraindicated in HBs. Agpositive untreated women or on TDF treatment 21

Healthcare workers
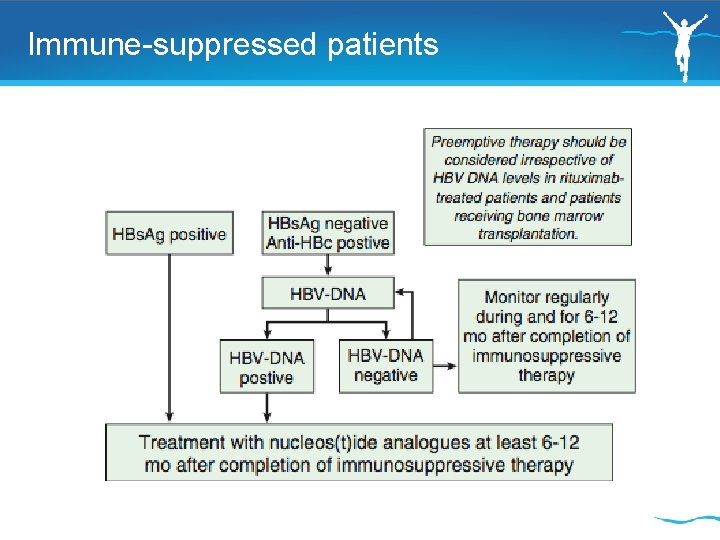
Immune-suppressed patients

When to stop Treatment ? The ideal endpoint is HBs. Ag loss with or without seroconversion to anti-HBs; however, this is rarely feasible. 24

When not to stop treatment ? • All cirrhotic patients (compensated and decompensated) should receive oral therapy if HBV DNA is detectable and should not stop treatment. 25

In HBe. Ag positive CHB patients, NA (nucleoside/nucleotide analogue) therapy can be stopped 12 ms after Hbe seroconversion or idealy continued till HBs. Ag loss particularly in patients with advanced fibrosis. (Evidence level II-2, grade of recommendation 2)

In HBe. Ag negative CHB patients Discontinuation of NA in selected non-cirrhotic HBe. Ag-negative patients who have achieved long-term (3 years) virological suppression may be considered if close monitoring can be guaranteed (Evidence level II-2, grade of recommendation 2)

28

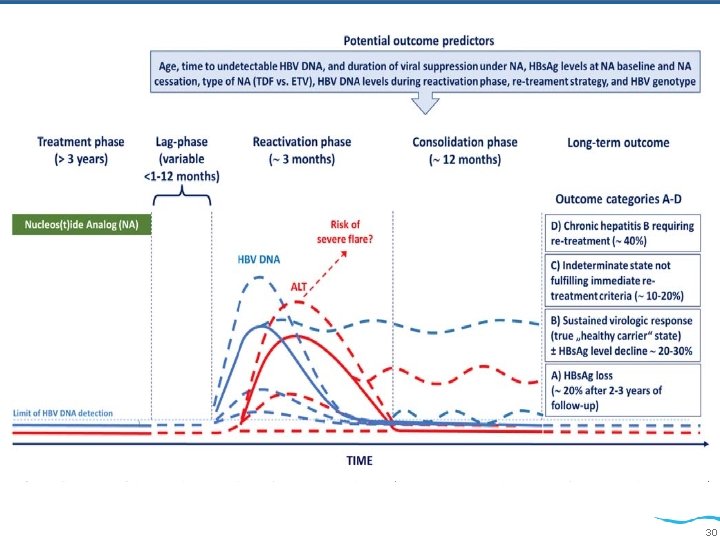
• The first study, a collaboration between two expert centers in Greece and Taiwan. The study included 130 HBe. Ag- negative patients without cirrhosis. • The second study, by Jeng et al, reviewed the incidence and predictors of HBs. Ag seroclearance after NA cessation in 691 Taiwanese patients with HBe. Ag-negative chronic hepatitis B with (45%) and without (55%) cirrhosis who discontinued oral therapy according to the APASL. • During the off-therapy follow-up of approximately 3 years, the cumulative virologic and clinical relapse rates were assessed 29
30

Authors recommendations: • CHB without cirrhosis. • Persistently normal ALT and negative HBV DNA over 3 y • Close monitoring is guaranteed. • End-of-treatment HBs. Ag levels <100 IU/m. L. 31

CONCLUSIONS • HBV infection is characterized by persistence owing to the presence of ccc. DNA. • Not all HBV infected patients are candidates for treatment. • Identification of the patient HBV status ( acute, chronic, carrier, past infection, …. ) is essential before the decision making to start therapy.

CONCLUSIONS • A finite NA treatment approach has an increasing evidence and leads to high HBs. Ag loss rates in selected HBe. Ag-negative patients without cirrhosis who are willing to comply with close follow-up. 33

THANK YOU 34
- Slides: 34