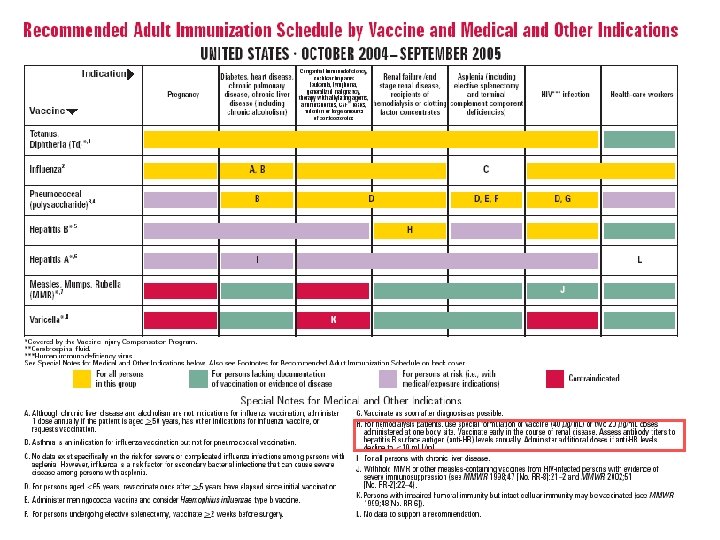
Hepatitis B vaccination indications Medical indications hemodialysis patients







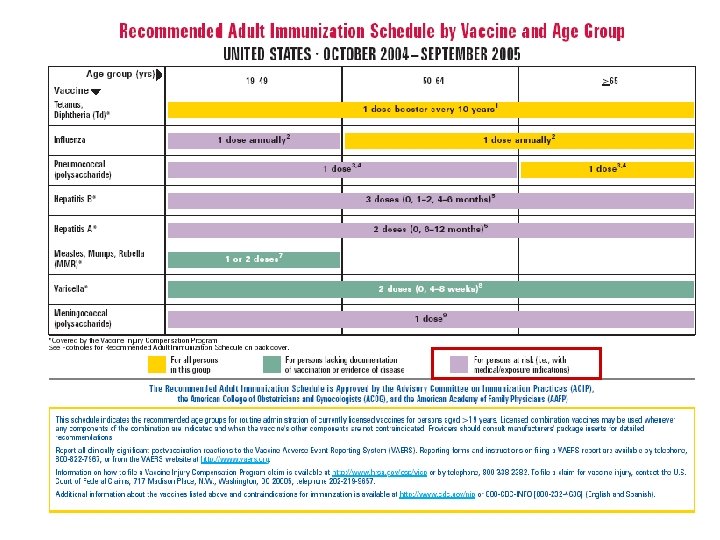

Hepatitis B vaccination indications Medical indications: hemodialysis patients or patients who receive clotting factor concentrates. o Occupational indications: health-care workers and public-safety workers who have exposure to blood in the workplace; and persons in training in schools of medicine, dentistry, nursing, laboratory technology, and other allied health professions. o Behavioral indications: injection drug users; persons with more than one sex partner during the previous 6 months; persons with a recently acquired sexually transmitted disease (STD); all clients in STD clinics; and men who have sex with men. o

Hepatitis B vaccination indications o Other indications: n n n household contacts and sex partners of persons with chronic hepatitis B virus (HBV) infection; clients and staff members of institutions for the developmentally disabled; inmates of correctional facilities; international travelers who will be in countries with high or intermediate prevalence of chronic HBV infection for >6 months



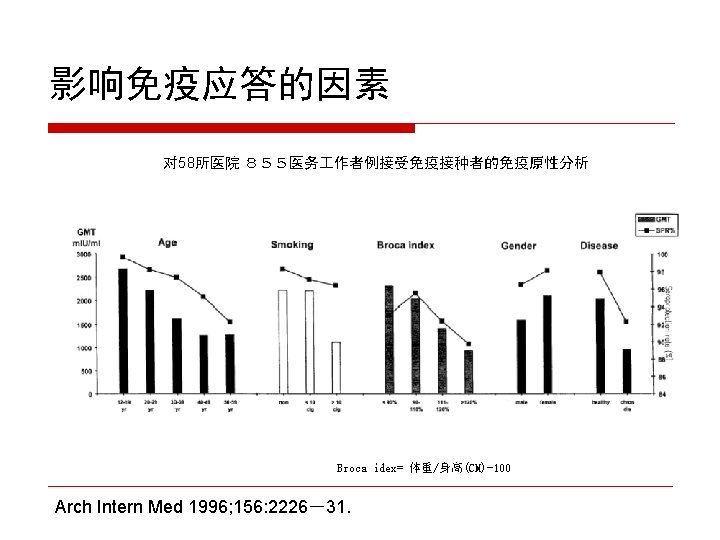

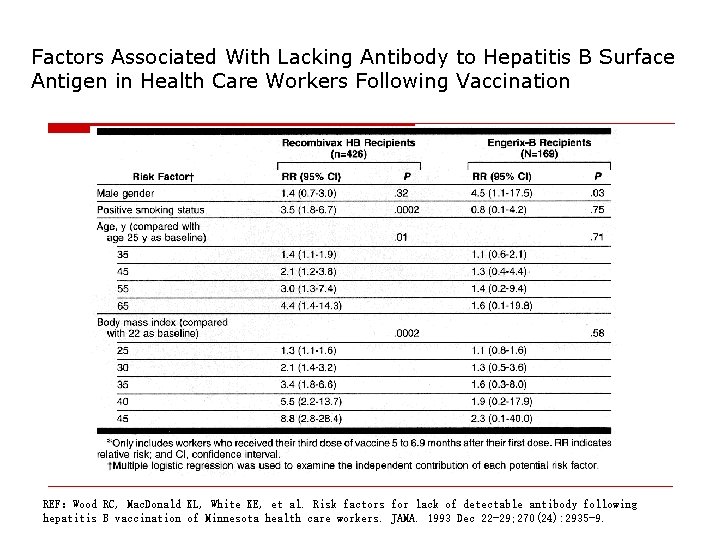
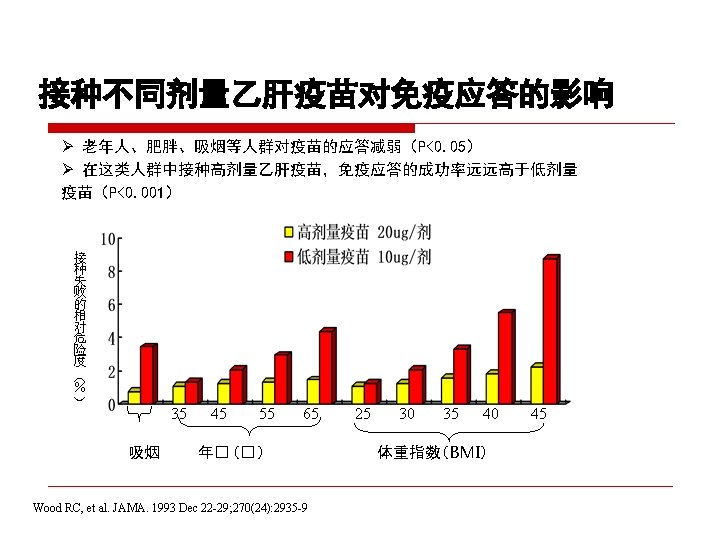
Factors Associated With Lacking Antibody to Hepatitis B Surface Antigen in Health Care Workers Following Vaccination REF:Wood RC, Mac. Donald KL, White KE, et al. Risk factors for lack of detectable antibody following hepatitis B vaccination of Minnesota health care workers. JAMA. 1993 Dec 22 -29; 270(24): 2935 -9.





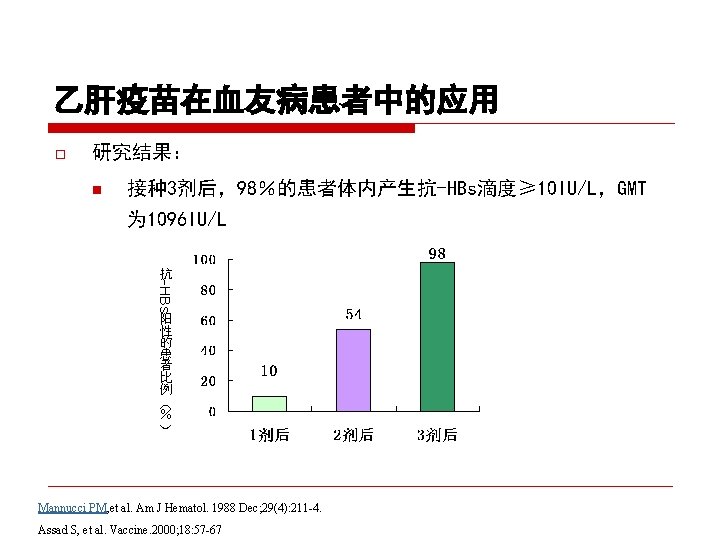


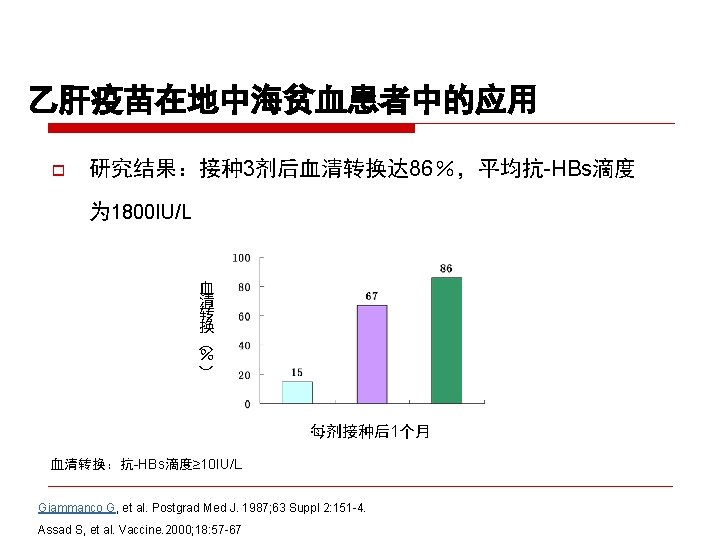

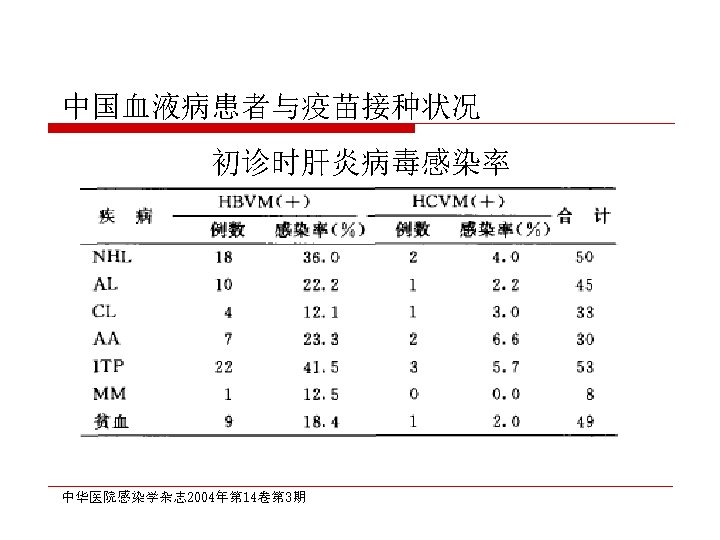

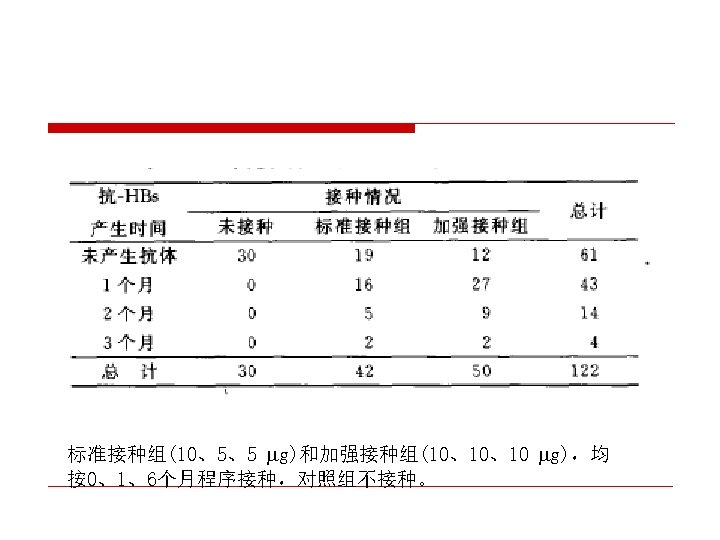



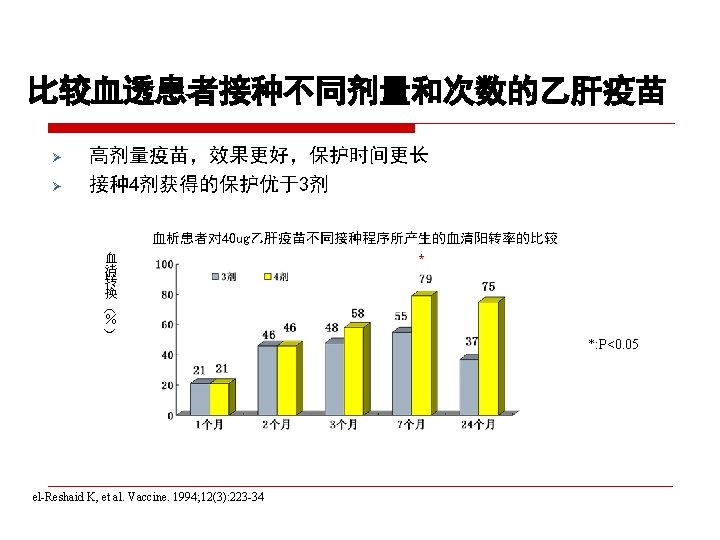

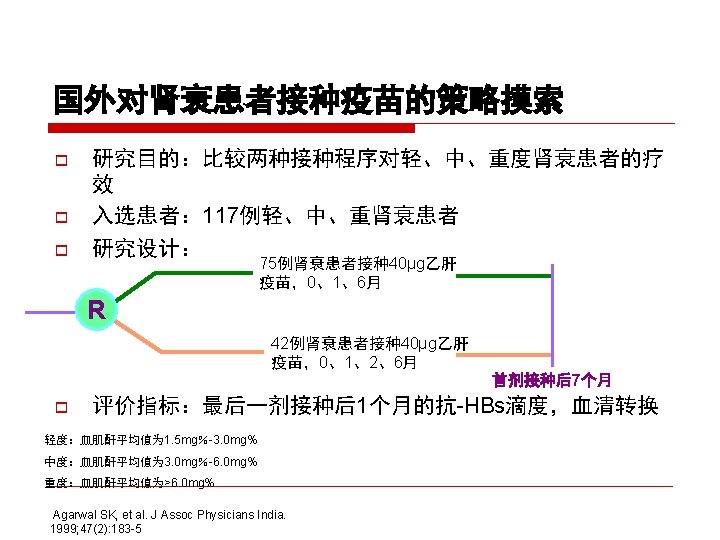
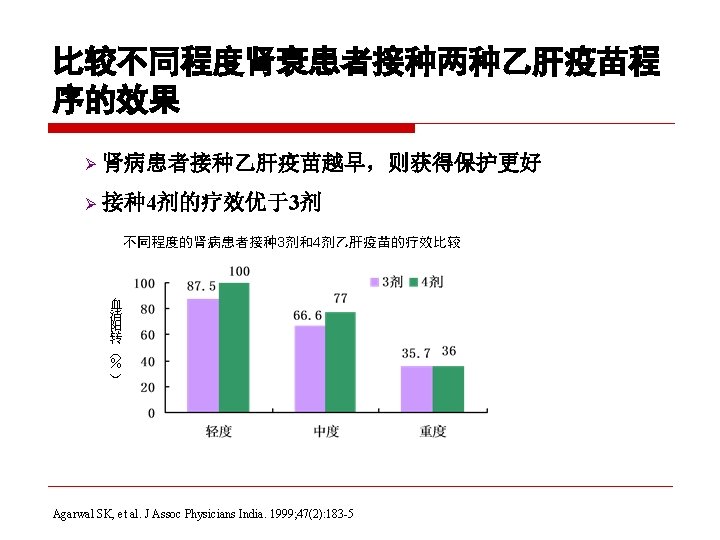



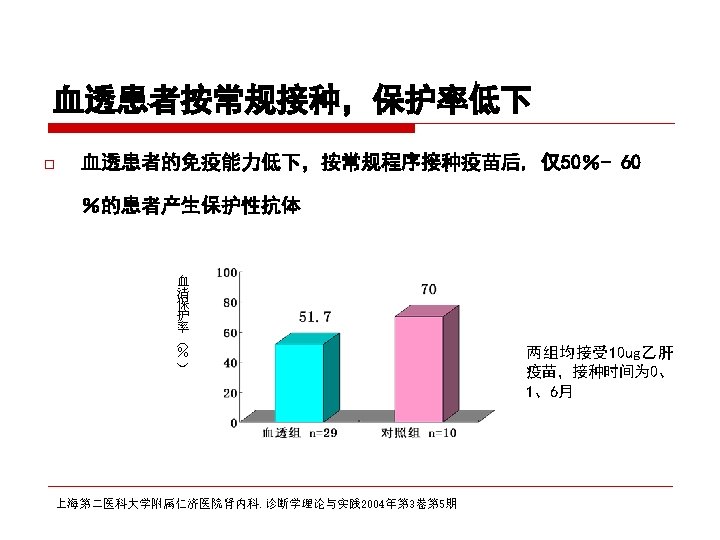
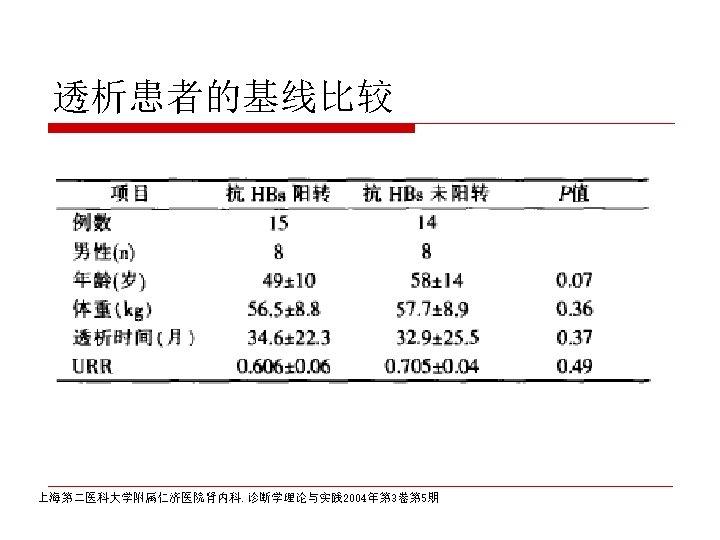
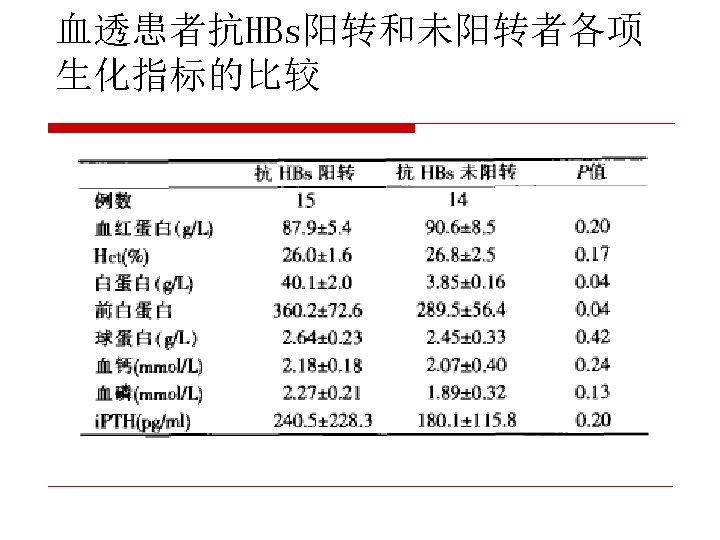
CDC recommendation for hemodialysis patients ouse special formulation of vaccine (40 µg/m. L) or two 20 µg/m. L doses administered at one body site. o. Vaccinate o. Assess early in the course of renal disease. antibody titers to hepatitis B surface antigen (anti-HB) levels annually. Administer additional doses if anti-HB levels decline to <10 m. IU/m. L.









Hepatitis B Immunoglobulin Discontinuation Followed by Hepatitis B Virus Vaccination: A New Strategy in the Prophylaxis of Hepatitis B Virus Recurrence After Liver ransplantation ? HEPATOLOGY February 2000

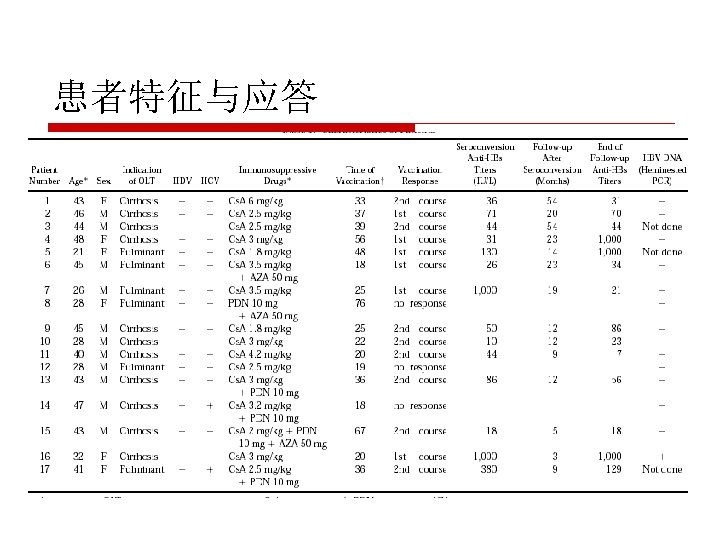
入选病例 o o o liver transplantation for conditions related to nonreplicative HBV infection (hepatitis B surface antigen [HBs. Ag] positive, hepatitis B e antigen [HBe. Ag] negative, and HBV DNA negative); at least 18 months of HBIG administration; no HBV infection recurrence, normal or slightly altered liver graft function, and low-grade immunosuppression at the time of vaccination. HEPATOLOGY February 2000 University of Barcelona, Spain.


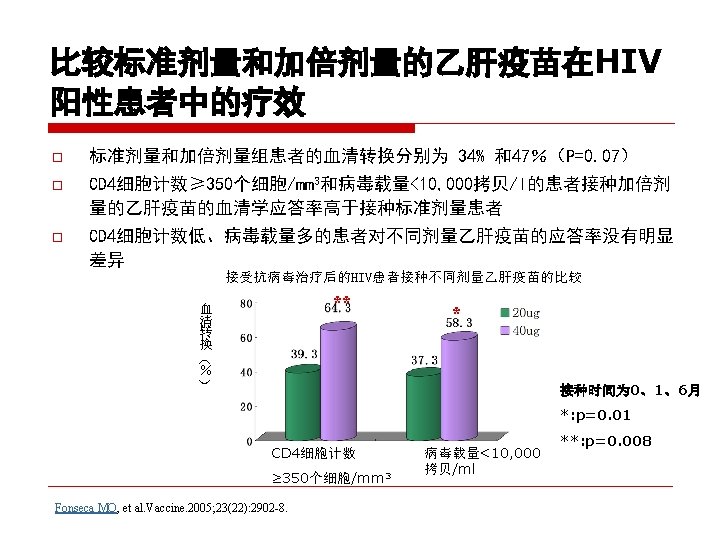
Challenge:really? Fifty-two patients on lamivudine prophylaxis at a median of 412 days (median, 370 -2040 days) after transplantation for chronic HBV-related liver disease received two courses of an accelerated schedule of double-dose recombinant HBV vaccine. o Before vaccination, all patients were seronegative for HBs. Ag, anti-HBs and HBV DNA (by q. PCR). o Three intramuscular doses of vaccine (40 microg each) were administered monthly and another identical course was repeated after 3 months. o Lamivudine (100 mg/day) was continued Journal of Hepatology 43 (2005) 283– 287 Centre for the Study of Liver Disease, Department of Surgery, The University of Hong Kong throughout the study. o

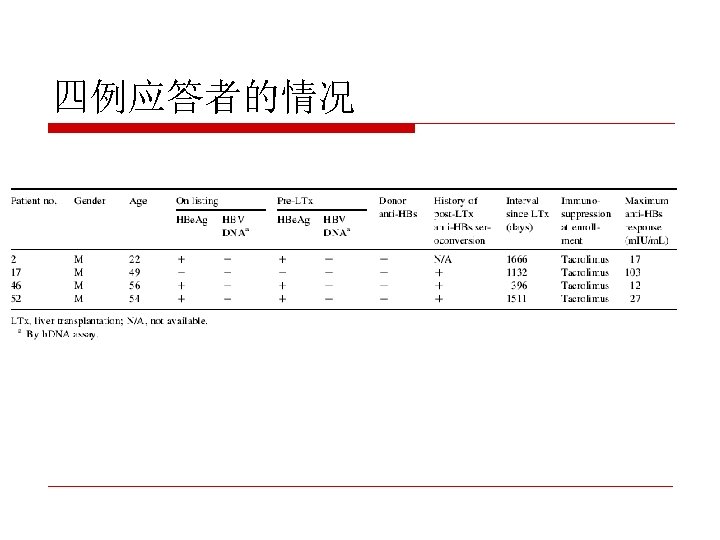
似乎不行 o o o After the first course, two patients developed a weak response (anti-HBs titre of 12 m. IU/m. L) that disappeared rapidly. One early responder developed anti-HBs (27 m. IU/m. L) again after the second course but the other did not. Two other patients developed response (anti. HBs titre of 17 and 103 m. IU/m. L, respectively) giving an overall response rate of 7. 7%. The antibody level declined rapidly. At the end of the study, one patient who did not respond had developed viral breakthrough which was treated with adefovir dipivoxil therapy. CONCLUSIONS: Active immunization with two courses of double-dose recombinant HBV


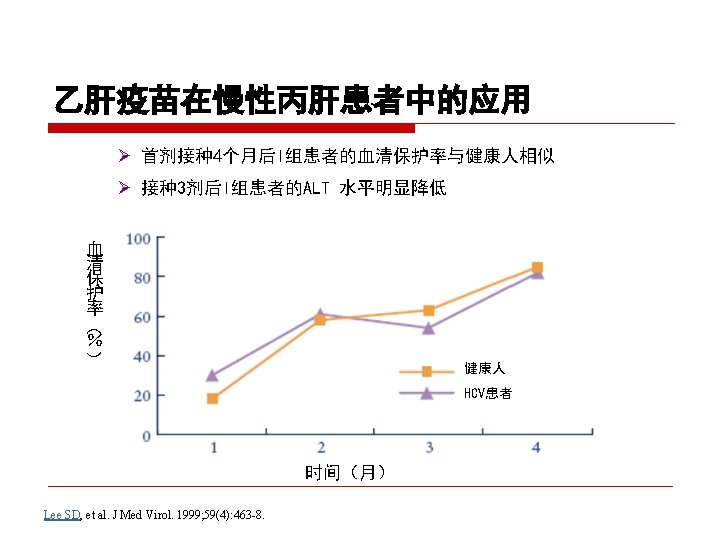
在HBIg联合拉米夫定治疗的病人接种 o o o double course of double dose recombinant HBV vaccine. Vaccination has been started 1 month after HBIg discontinuation, and lamivudine (100 mg/day) was given throughout the study. The first cycle consisted of 0, 1 - and 6 -month schedule, and, in nonresponders, second cycle 0, 1 -, 2 -month schedule. Fourteen patients included into the study. Only one patient seroconverted (an anti-HBs titre of 37 IU/L) after the first cycle. No other patient responded to second cycle. J Viral Hepat. 2005 Mar; 12(2): 212 -5. Department of Gastroenterology, Ege University Medical School, Izmir, Turkey







- Slides: 75