Hepatitis A and B Vaccination and the New

Hepatitis A and B Vaccination and the New York State Adult Hepatitis Vaccination Program Elizabeth Rausch-Phung, MD MPH Medical Director, NYSDOH Bureau of Immunization 1

Hepatitis A Virus (HAV) • Transmission fecal-oral. • Symptoms include fatigue, abdominal pain, jaundice; children often asymptomatic. • Acute phase only, never chronic, complete recovery with lifelong immunity within 3 -6 months. • No specific treatment – supportive only. • Hepatitis A is vaccine preventable!!

Hepatitis B Virus (HBV) • Transmitted through blood/body fluids. • Symptoms similar to HAV; often asymptomatic. • Acute phase – 90% clear the virus & develop lifelong immunity. • 10% develop chronic infection – can lead to cirrhosis and liver cancer. • Treatment only for chronic infections on case-by-case basis. • ~5000 deaths annually in the U. S. from HBV. • Hepatitis B is vaccine preventable!!

Hepatitis A Vaccine Two inactivated whole virus vaccines available: • Vaqta ® (Merck) and Havrix ® (GSK) – Up to age 18: 0. 5 m. L – Age 19 and over: 1. 0 m. L Efficacy: • Highly immunogenic. – 94% to 100% seroconvert within a month after 2 doses. • No boosters are currently recommended. Administration: • Dose 1 followed by dose 2 at least 6 months after dose 1. • If dose 2 is late, it is NOT necessary to repeat dose 1.

Hepatitis B Vaccine Two inactivated vaccines available: • Engerix-B® (GSK) and Recombivax HB® (Merck) – Up to age 19: 0. 5 m. L – Age 20 and over: 1. 0 m. L Efficacy: • Protection ~ 50% after 1 dose; 85% after 2 doses; 98% after 3 doses. Administration: • 0, 1 -2 and 4 -6 months – Minimum spacing of 4 weeks between dose 1 and dose 2. – Minimum 8 weeks between dose 2 and dose 3. – Minimum of 16 weeks between dose 1 and dose 3.

Hepatitis A and B Vaccine • Twinrix ® – Combination hepatitis A & B vaccine (GSK). – Approved for persons aged 18 yrs and older in the US. – Indicated for persons at risk for both hepatitis A and hepatitis B. • Administered in a 3 dose series at 0, 1 and 6 months – Dose 1 and dose 2 should be separated by at least 4 weeks. – Dose 2 and dose 3 should be separated by at least 8 weeks. – Dose 1 and dose 3 should be separated by at least 6 months.
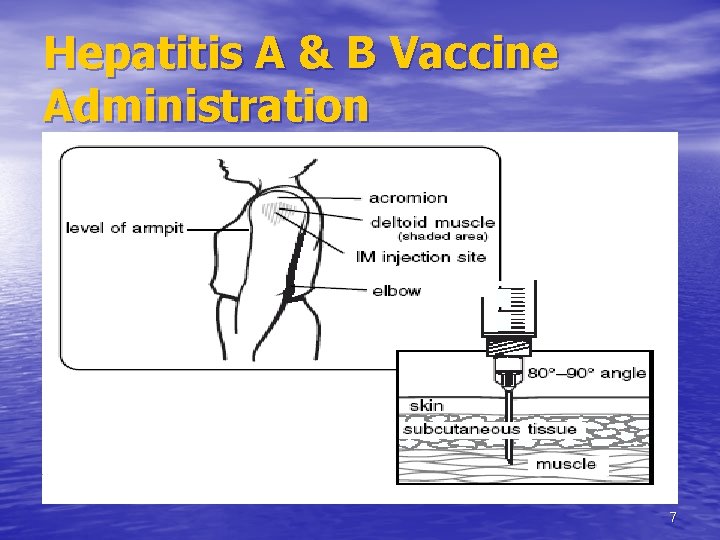
Hepatitis A & B Vaccine Administration 7

Hepatitis A Vaccination • Who should be vaccinated – Adults: – – – International travelers, Men who have sex with men, Users of either injectable or non-injectable drugs, Persons with HIV/AIDS, Persons with chronic liver disease, Persons with a diagnosis of clotting factor disorder, and – Persons with occupational risks.

Hepatitis B Vaccination • Who should be vaccinated – Adults: – Household contacts and sex partners of persons infected with HBV, – Persons with > 1 sex partner in 6 months, – Men who have sex with men, – Persons seeking evaluation or treatment for an STD, – Users of injectable drugs, – Persons with HIV/AIDS, – Persons with chronic liver disease, – Persons with end-stage kidney disease, and – Persons with jobs that expose them to human blood.

Risk Assessment & Pre-Vaccination Screening Three questions that should be asked prior to hepatitis vaccination: 1. Are you sick today? 2. Do you have any allergies to medications, food or any vaccine? 3. Have you ever had a serious reaction after receiving a vaccination?

Contraindications and Precautions • Severe allergic reaction to a vaccine component or following a prior dose, or • Moderate or severe acute illness.

Types of Vaccine Reactions in Adults • • Local Reaction, Systemic Reaction, Allergic Reaction (anaphylactic), and Post-Vaccination Syncope.

Local Reactions • Symptoms include pain, swelling, and redness at the injection site. • These reactions occur about 50% of the time. • These reactions occur within a few hours after • • • vaccination and are mild and self-limited. If patient has soreness, redness, itching or swelling, apply a cold compress and give analgesics if necessary. If patient has slight bleeding, apply adhesive over the injection site. If patient continues bleeding, apply gauze pads and firm pressure; raise bleeding site above level of heart.

Systemic Reaction • Fever, malaise, myalgias, headache, loss of appetite, etc. • Rarely occur when inactivated vaccines are used, • • as in the case with Hepatitis A and B vaccines. Typically mild and self-limited. Treat fever, myalgia and/or headache with analgesics, if necessary.

Allergic Reaction (anaphylactic) • Extremely rare, life-threatening reactions. • Itching, redness, hives, swelling of lips, face or throat, wheezing, shortness of breath, shock, cramping or cardiovascular collapse. • If itching & swelling confined to injection site, observe patient for other symptoms. • Call 911 and seek emergency help if symptoms are generalized.

Post-Vaccination Syncope • Vaccine providers should strongly consider observing patients for 15 minutes after they are vaccinated. – Having patients sit or lie down for 15 minutes following vaccination could prevent most syncopal episodes and secondary injuries. • If syncope develops, patients should be observed until symptoms resolve.

Reporting Adverse Reactions for Hepatitis A & B Vaccination Vaccine Adverse Events Reporting System (VAERS) • Adverse events following vaccination may be reported by providers or the vaccinee. • For more information and to download report forms: http: //www. vaers. hhs. gov/.

Vaccination Documentation • Vaccine Information Statements – It’s a Federal Law! • Must be provided with each dose of vaccine. • Available in many languages. – www. cdc. gov/vaccines – www. immunize. org/vis • Explains benefits and risks of a vaccine.

Vaccination Documentation Providers must document: • Which VIS was given, • Date of publication of VIS, • Date VIS was given, • Name and title of person administering vaccine, • Date of administration, • Site of administration, and • Vaccine manufacturer and lot number.

Vaccine Storage and Handling Hepatitis A and B Vaccines • When vaccine is received, immediately store between 2° - 8°C (35° - 46°F). – Frozen vaccine loses potency and effectiveness. • Refrigerators for vaccine should be combination household or stand-alone commercial style, not dormitory style.

Vaccine Storage and Handling • Food and drink should NOT be stored in refrigerator. • Storage should be in middle of refrigerator and not in the door. Ø

Vaccine Storage and Handling • Assign a staff member and backup to be in • charge of vaccine storage and handling. Keep a thermometer in the refrigerator. – Check and record the temperature twice a day. • Keep extra containers of water in the refrigerator to stabilize temperatures. Please do not leave my door open. Thank you!

Vaccine Storage and Handling • Rotate vaccine stock using shortest expiration date first. • Have a DO NOT UNPLUG sign posted near refrigerator’s outlet. • Have a back up plan in event of power outage.

Don’t Miss An Opportunity to Vaccinate !!!!!

We can prevent this!!!!! Severe ascites (abdominal swelling) in a woman with chronic Hepatitis B infection Photo courtesy of Patricia Walker, MD, Ramsey Clinic Associates, St. Paul, MN

Additional References NYSDOH Hepatitis Vaccine Information: http: //www. nyhealth. gov/diseases/communicable/hepatitis /vaccine. htm CDC: www. cdc. gov/hepatitis ACIP Recommendations for Hepatitis A Vaccination: http: //www. cdc. gov/vaccines/pubs/ACIP-list. htm#hepa ACIP Recommendations for Hepatitis B Vaccination: http: //www. cdc. gov/vaccines/pubs/ACIP-list. htm#hepb

Additional References (con’t) NYSDOH Immunization Page: http: //www. nyhealth. gov/prevention/immunization/ CDC Vaccine Page: http: //www. cdc. gov/vaccines/ Immunization Action Coalition: http: //www. immunize. org/ NYSDOH Bureau of Immunization e-mail: immunize@health. state. ny. us NYSDOH Bureau of Immunization phone: (518) 473 -4437
- Slides: 27