HEPATITIS 2014 BC CHRONIC HEPATITIS B THE PEOPLE

HEPATITIS 2014 BC

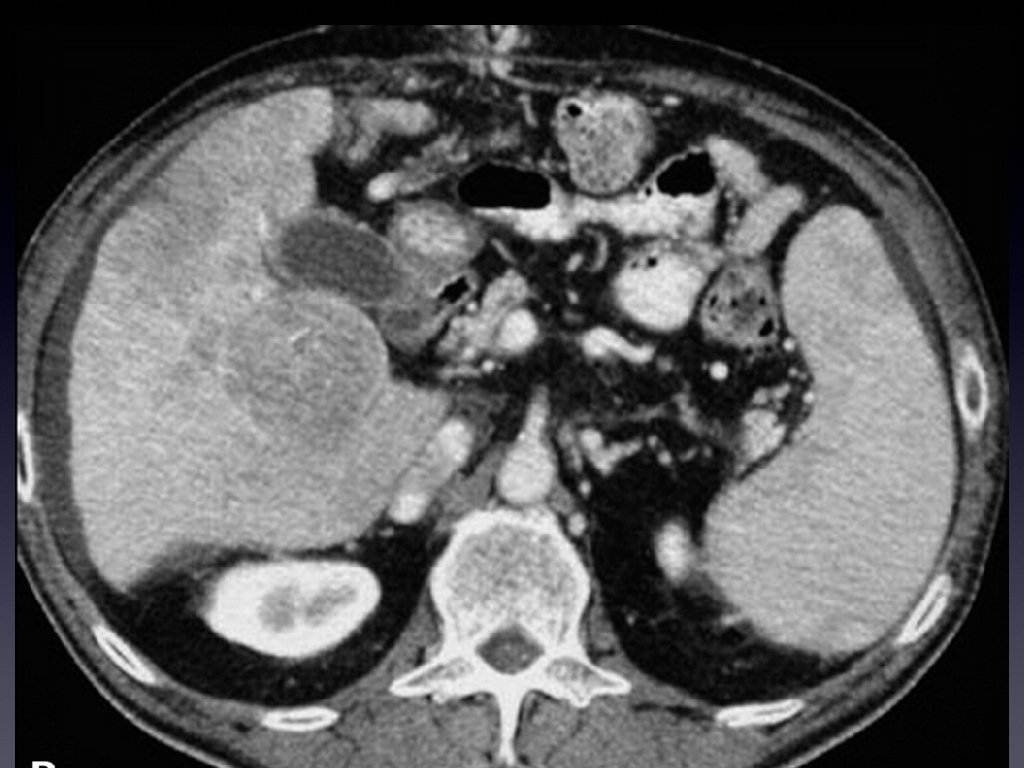
CHRONIC HEPATITIS B THE PEOPLE PROBLEM • 350, 000 HAVE IT • • IT IS TRANSMITTED MOTHER TO CHILD WHERE IT IS ENDEMIC IT CAN BE TRANSMITTED VIA BODY FLUIDS IT OFTEN PROGRESSES TO CIRRHOSIS IT CAUSES HEPATOCELLULAR CARCINOMA
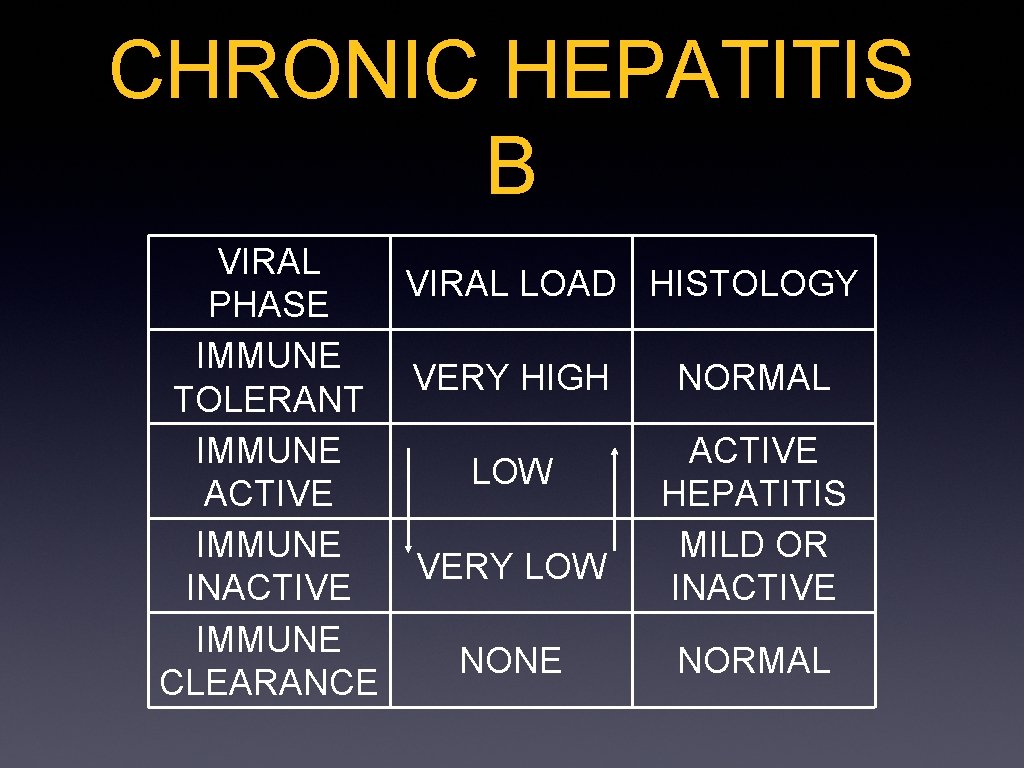
CHRONIC HEPATITIS B VIRAL LOAD HISTOLOGY PHASE IMMUNE VERY HIGH NORMAL TOLERANT IMMUNE ACTIVE LOW ACTIVE HEPATITIS IMMUNE MILD OR VERY LOW INACTIVE IMMUNE NORMAL CLEARANCE

CHRONIC HEPATITIS B THE PROBLEM • IT IS QUICKLY TRANSMITTED TO THE INFANT OF AN INFECTED MOTHER • IT CAN BE REACTIVATED EVEN WHEN APPARENTLY “CLEARED” BY CHEMOTX OR PERHAPS TNF INHIBITORS

CHRONIC HEP B WHO TO TX? • ALL PATIENTS WITH CIRRHOSIS AND DETECTABLE VIRUS • • • ALL PATIENTS WITH IMMUNE ACTIVE PHASE DISEASE ASYMPTOMATIC PREGNANT WOMEN WHO HAVE > 1, 000 COPIES/ML BEGINNING IN THE THIRD TRIMESTER PATIENTS WITH DETECTABLE HBs. Ag ABOUT TO UNDERGO IMMUNOSUPPRESSION

CHRONIC HEPATITIS B WHAT TO USE? • ENTECAVIR • TENOFOVIR • TELBIVUDINE • LAMIVUDINE

HEPATITIS C NOW A DISEASE FOR PRIMARY CARE THERAPY

THE SHORT VERSION ONE OR TWO SIDE-EFFECTFREE PILLS DAILY FOR 3 TO 6 MONTHS CURES >90% OF PEOPLE WITH CHRONIC HEPATITIS C
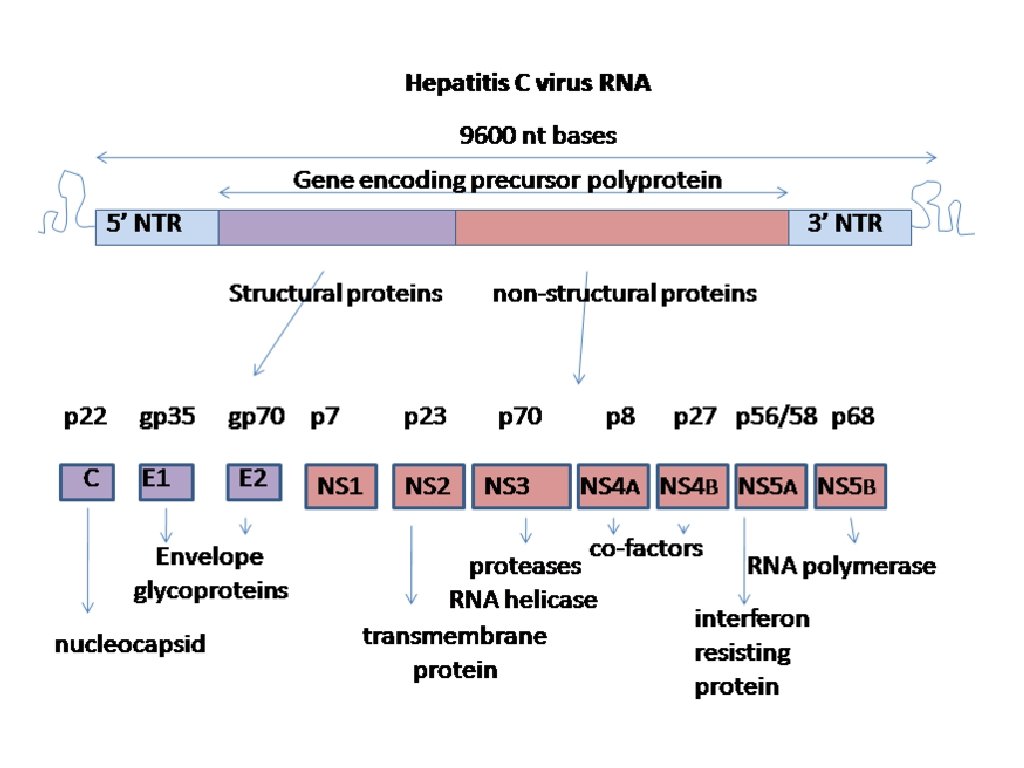

HEPATITIS C-THE PROBLEM 3. 8 MILLION AMERICANS HAVE IT 170 MILLION PEOPLE WORLDWIDE HAVE IT IT PROGRESSES TO CIRRHOSIS IN AT LEAST 25% IT CAUSES HEPATOCELLULAR CARCINOMA WE CAN’T PREDICT WHO WILL PROGRESS

HEPATITIS C-THE PROBLEMEXTRAHEPATIC MANIFESTATIONS ARTHRITIS VASCULITIS CRYOIMMUNOGLOBULINEMIA RENAL DISEASE INSULIN RESISTANCE AND TYPE II DIABETES MELLITUS LYMPHOMA THYROIDITIS LICHEN PLANUS ETC


HEPATITIS C-THE PAST TREATMENT WAS DIFFICULT AND OFTEN INEFFECTIVE IT REQUIRED CLOSE MONITORING AND A CLEAR WORKING KNOWLEDGE OF THE NUANCES OF THE MEDICATIONS USED SIDE EFFECTS WERE UNIVERSAL AND OFTEN TERRIBLE PATIENTS HAD FREQUENT PHYSICIAN VISITS OVER 6 TO 12 MONTHS MOST PATIENTS UNDERGOING TREATMENT HAD TO MISS SOME WORK AND OFTEN COULD NOT WORK AT ALL WE COULDN’T TREAT THE SICKEST PATIENTS

HEPATITIS C-THE PAST WE EMPLOYED VERY SELECTIVE CRITERIA FOR TREATMENT: ILLNESS FROM THE INFECTION--RARE UNTIL CIRRHOSIS ENSUED, WHEN TREATMENTS WERE LESS EFFECTIVE HISTOLOGIC EVIDENCE SUGGESTING PROGRESSIVE FIBROSIS NOT TOO SICK TO TREAT

HEPATITIS C THE PAST THE BEST RECENT TREATMENT WAS EFFECTIVE IN ABOUT: 1/2 OF GENOTYPE 1 b 2/3 OF GENOTYPE 1 a 3/4 OF GENOTYPE 2 AND 3 EXCEPT IF YOU WERE OF MAINLY SUBSAHARAN AFRICAN ORIGIN THEN IT WAS < 1/2 THE ABOVE

HEPATITIS C THE PAST THE TREATMENT WAS EXPENSIVE $25, 000 -$30, 000 WITH PEGI/RIBA >$80, OOO IF YOU ADDED TELAPREVIR OR BOCEPREVIR YOU LOST WORK USUALLY TOO

HEPATITIS C THE FUTURE • NUMEROUS MEDS ARE COMING • THEY WILL ATTACK THE VIRUS AT DIFFERENT SITES • THEY HAVE MINIMAL SIDE EFFECTS • TREATMENT WILL BE 12 -24 WEEKS FOR MOST PATIENTS

HEPATITIS C THE FUTURE • THE MEDS WILL BE GIVEN IN TWO DRUG COMBINATIONS FOR MOST • NO RIBAVIRIN WILL BE NEEDED • WHEN AND IN WHAT COMBINATIONS WILL BE DETERMINED BY THE FDA

SO HOW GOOD ARE THE NEW TREATMENTS? • VERY, VERY GOOD
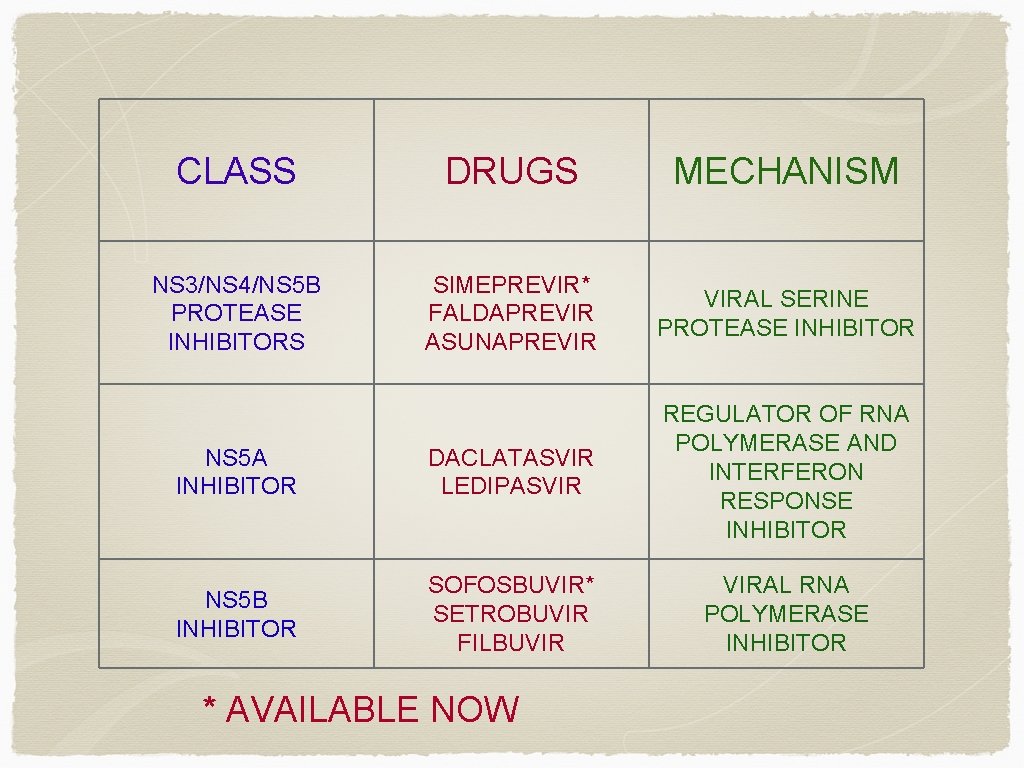
CLASS DRUGS MECHANISM NS 3/NS 4/NS 5 B PROTEASE INHIBITORS SIMEPREVIR* FALDAPREVIR ASUNAPREVIR VIRAL SERINE PROTEASE INHIBITOR NS 5 A INHIBITOR DACLATASVIR LEDIPASVIR REGULATOR OF RNA POLYMERASE AND INTERFERON RESPONSE INHIBITOR NS 5 B INHIBITOR SOFOSBUVIR* SETROBUVIR FILBUVIR VIRAL RNA POLYMERASE INHIBITOR * AVAILABLE NOW

SOFOSBUVIR AND DACLATASVIR FOR 24 WEEKS GENOTYPE 1 a/1 b TX NAIVE SVR 100% GENOTYPE 1 a/1 b TX FAILURE SVR 100% GENOTYPE 2/3 TX NAIVE SVR 100% THESE WERE ALL NON CIRRHOTIC •

SOFOSBUVIR, LEDIPASVIR, RIBAVIRIN IN GENOTYPE 1 a AND 1 b • TREATMENT SVR RESULTS NAIVE S+L X 8 WKS 95% NAIVE S+L+R X 8 WKS 100% NAIVE S+L X 12 WKS 95% TX FAILURE S+L X 12 WKS 95%* TX FAILURE S+L+R X 12 WKS 100%* *40% HAD CIRRHOSIS

HEPATITIS C THE FUTURE • SOME RESULTS WILL VARY • GENOTYPE 3 WILL BE SLOWEST TO TX • CIRRHOTICS WILL LIKELY HAVE SLOWER RESPONSES BUT LIKELY JUST NEED LONGER TX • VIRAL RESISTANCE CAN OCCUR • WE KNOW LITTLE ABOUT NON 1, 2, 3 GENOTYPES

HEPATITIS C THE FUTURE • COST: $160, 000! • TIME INVOLVED IN GETTING THE DRUG IS 2 HOURS • BRIGHT • GETTING BRIGHTER

HEPATITIS C THE FUTURE • CURRENTLY THE DRUG COMPANIES ARE VERY, VERY HELPFUL AT GETTING A GOOD DEAL FOR THE PATIENTS

WHO SHOULD THE NON HEPATOLOGIST/GASTROENTEROLOGIST REFER? n CIRRHOTICS n ANYONE YOU ARE NOT COMFORTABLE TREATING n TREATMENT FAILURES n ANYONE NEEDING TREATMENT UNTIL THESE NEW AGENTS AND COMBINATIONS ARE READY n ANYONE WHO NEEDS A COLONOSCOPY--JUST KIDDING

HEPATITIS C THE FUTURE • WHEN WILL THE DRUGS BE HERE • THEY’RE HERE NOW--BUT ARE NOT APPROVED BY THE FDA IN COMBINATION • SO INSURANCE WON’T BUY THEM • MORE WILL COME LATER THIS YEAR AND FOR THE NEXT FEW YEARS

HEPATITIS C SUMMARY IT IS A COMMON DISEASE, WATCH FOR IT IT IS A CAUSE OF REMARKABLE MORBIDITY AND MORTALITY IT CAN BE CURED YOU CAN CURE IT IF WE CAN PAY FOR IT IF YOU HAVE THE TIME TO PRESCRIBE THE MEDS
- Slides: 29