HeparinInduced Thrombocytopenia HIT Treatment with danaparoid Orgaran Management

Heparin-Induced Thrombocytopenia (HIT) Treatment with danaparoid (Orgaran )

Management of HIT – treatment When HIT is strongly-suspected: • Stop all heparin (both unfractionated and lowmolecular-weight heparin) • Initiate alternative non-heparin anticoagulant because of high risk of symptomatic thrombosis • Test for HIT antibodies • Duplex ultrasonography to exclude DVT

Management of HIT – treatment When the diagnosis of HIT is confirmed: • Therapeutic doses of alternative non-heparin anticoagulants are usually required • Postpone starting overlapping coumarin until the platelet count has recovered to at least 100 (and preferably) 150 x 109/L • If a sensitive test for HIT is negative, heparin therapy may be re-started with regular platelet count monitoring * 7 th. ACCP Conference 2004 Chest, 126, 311 S-337 S

Management of HIT – treatment Alternative non-heparin antithrombotic therapies include: Grade of recommendation* • Danaparoid 1 B • Direct thrombin inhibitors Ø Lepirudin 1 C+ Ø Argatroban 1 C Ø Bivalirudin 2 C *Grading as per 7 th American College of Chest Physicians Conference. Chest 2004, 126: 311 S-337 S

Heparin-Induced Thrombocytopenia (HIT) Rationale for initiating or continuing antithrombotic therapy after discontinuing heparin

Initiating or continuing antithrombotic therapy Rationale for initiating or continuing antithrombotic therapy after stopping heparin because of HIT • Patient typically has pre-existing indication for prophylactic or therapeutic anticoagulation • HIT greatly increases baseline risk of thrombosis (odds ratio, 20— 40)
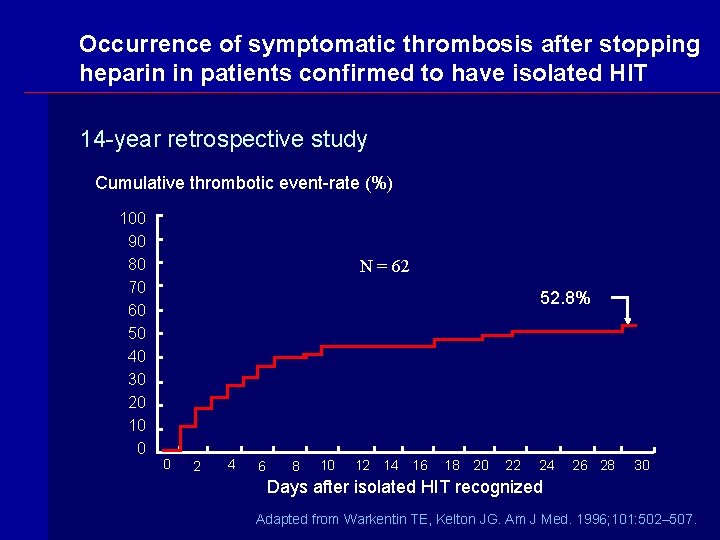
Occurrence of symptomatic thrombosis after stopping heparin in patients confirmed to have isolated HIT 14 -year retrospective study Cumulative thrombotic event-rate (%) 100 90 80 70 60 50 40 30 20 10 0 N = 62 52. 8% 0 2 4 6 8 10 12 14 16 18 20 22 24 26 28 30 Days after isolated HIT recognized Adapted from Warkentin TE, Kelton JG. Am J Med. 1996; 101: 502– 507.

Odds ratios for risk of thrombosis • • Prothrombin anomaly Lupus anticoagulant Factor V Leiden 6. 6 Protein S deficiency Dysfibrinogenemia Protein C deficiency Antithrombin deficiency • HIT 2. 0 5. 4 10. 9 11. 3 14. 4 24. 1 20 -40 Warkentin TE. Can J Cardiol 1995; 11(Suppl C): 29 C-34 C Warkentin TE. Thromb Res 2003; 110: 73 -82

Heparin-Induced Thrombocytopenia (HIT) Rationale for using danaparoid – Orgaran as the antithrombotic therapy of choice

Rationale for using Orgaran –danaparoid as the antithrombotic therapy of choice • Danaparoid is a nonheparin antithrombotic • It has been shown to be an effective antithrombotic with a high benefit-to-risk ratio in the treatment of HIT in an open-label randomized controlled trial and in studies using historical controls • In a minority (<5%) of HIT patients treated with danaparoid has clinically-evident cross-reactivity been implicated, most often because of platelet count fall
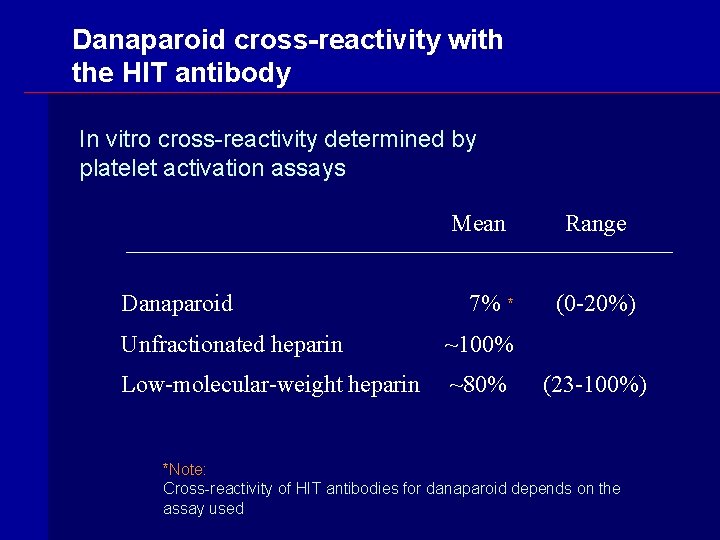
Danaparoid cross-reactivity with the HIT antibody In vitro cross-reactivity determined by platelet activation assays Mean Danaparoid 7% * Unfractionated heparin ~100% Low-molecular-weight heparin ~80% Range (0 -20%) (23 -100%) *Note: Cross-reactivity of HIT antibodies for danaparoid depends on the assay used
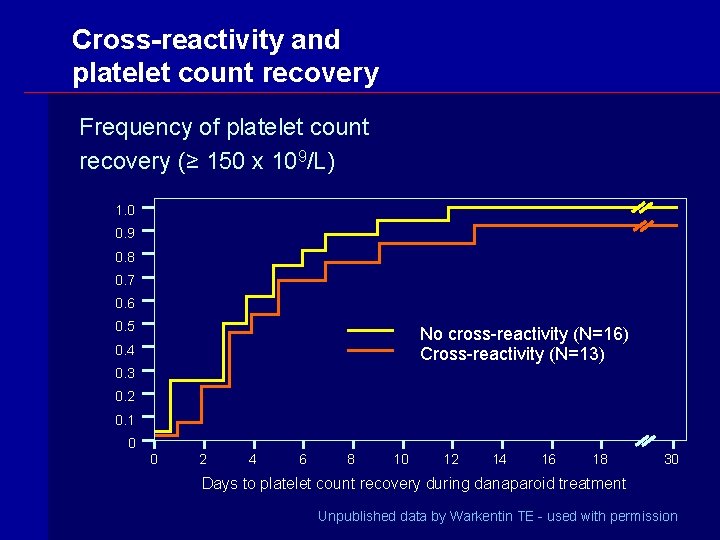
Cross-reactivity and platelet count recovery Frequency of platelet count recovery (≥ 150 x 109/L) 1. 0 0. 9 0. 8 0. 7 0. 6 0. 5 No cross-reactivity (N=16) Cross-reactivity (N=13) 0. 4 0. 3 0. 2 0. 1 0 0 2 4 6 8 10 12 14 16 18 30 Days to platelet count recovery during danaparoid treatment Unpublished data by Warkentin TE - used with permission

Danaparoid cross-reactivity with the HIT antibody “Potential in vivo cross-reactivity (rare) is not predictable by in vitro testing; thus, cross-reactivity testing is not recommended prior to use [of danaparoid]” 7 th. American College of Chest Physicians Conference Chest 2004, 126: 311 S-337 S

Clinical Experience with Danaparoid in the Management of HIT
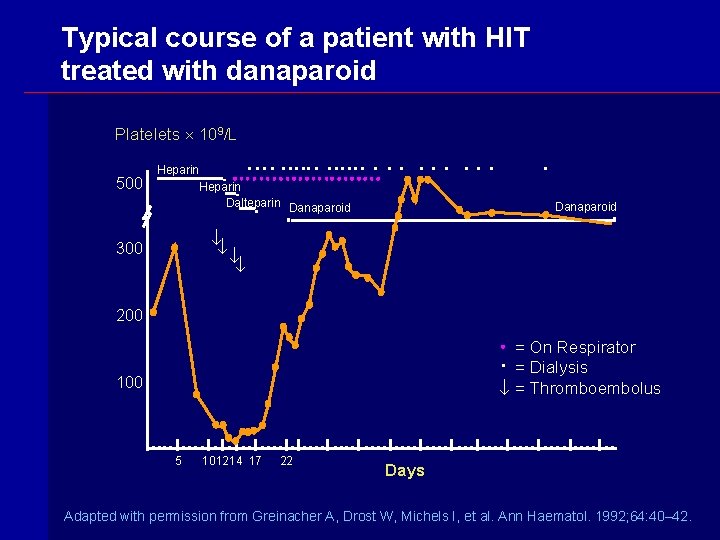
Typical course of a patient with HIT treated with danaparoid Platelets 109/L 500 Heparin Dalteparin Danaparoid 300 200 = On Respirator = Dialysis = Thromboembolus 100 5 101214 17 22 Days Adapted with permission from Greinacher A, Drost W, Michels I, et al. Ann Haematol. 1992; 64: 40– 42.

Clinical Experience with Danaparoid in the Management of HIT Comparative Clinical Studies

Danaparoid vs. Dextran Randomized, open-label study* Chong BH et al. Thromb Haemost 2001; 81: 1170 -1175. Inclusion Criteria: • All patients with strong clinical evidence of HIT - platelet count < 100 X 109/L while on heparin with no other obvious cause for thrombocytopenia • All patients were tested for HIT antibodies by platelet activation assay but negative patients were not excluded if there was strong clinical suspicion of HIT • All had thrombosis: in 50% of patients in each treatment group, thrombosis was severe and progressive * This represents the only randomized controlled trial performed on patients with HIT

Danaparoid vs. Dextran Randomized, open-label study Chong BH et al. Thrombos Haemost 2001; 81: 1170 -1175. Exclusion Criteria: • Alternative explanation for platelet count • Initiation of VKA therapy and in the target therapeutic range (INR >2. 0) prior to consideration for inclusion • Patients with renal failure, heart failure, pregnancy or requiring surgery were excluded from the study * This represents the only randomized controlled trial performed on patients with HIT

Danaparoid vs. Dextran Treatment regimens: • Comparison Therapies Ø Danaparoid i. v. bolus + infusion for 5 days Ø Control: Dextran 1, 000 ml on Day 1 followed by 500 ml/day for 5 days. • All received oral anticoagulant (VKA) therapy from Day 1 (Target INR >2)
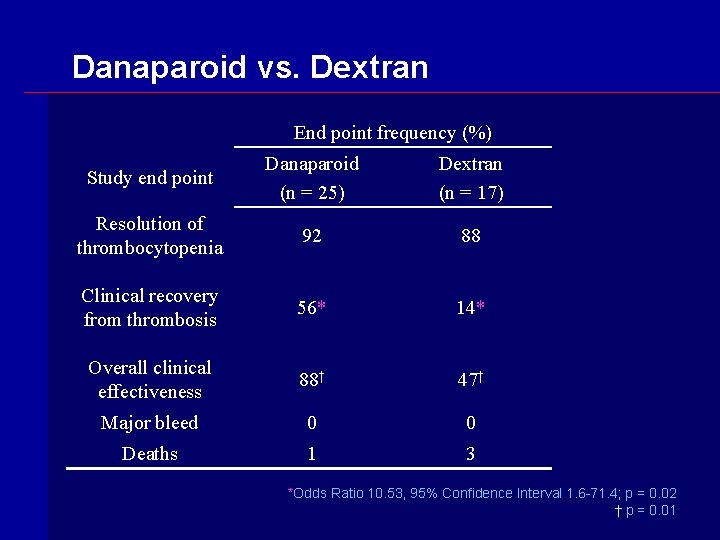
Danaparoid vs. Dextran End point frequency (%) Study end point Danaparoid (n = 25) Dextran (n = 17) Resolution of thrombocytopenia 92 88 Clinical recovery from thrombosis 56* 14* Overall clinical effectiveness 88† 47† Major bleed 0 0 Deaths 1 3 *Odds Ratio 10. 53, 95% Confidence Interval 1. 6 -71. 4; p = 0. 02 † p = 0. 01

Danaparoid vs. Lepirudin A retrospective cohort (danaparoid) versus a prospective cohort (lepirudin) study Farner B et al. Thromb Haemost 2001; 85: 950 -957. Lepirudin Patients satisfying the study inclusion/exclusion criteria were treated with a. PTT-adjusted lepirudin i. v. either at therapeutic anticoagulation dose +/- thrombolysis or at thrombosis prophylaxis dose and followed prospectively Danaparoid HIT patients who otherwise fulfilled the same inclusion and exclusion criteria as in the prospective lepirudin study but who instead were treated with danaparoid (either in therapeutic or prophylactic doses i. v. or s. c. ) were evaluated retrospectively and compared with lepirudin-treated patients

Danaparoid vs. Lepirudin Inclusion Criteria Active HIT • Clinical criteria • Platelet Count 50% or <100 x 109/L and/or thromboembolism during i. v. or s. c heparin treatment • Skin inflammation at the heparin injection site • Laboratory criteria • Positive heparin-induced platelet aggregation (HIPA) test Exclusion Criteria • Renal impairment • Pregnancy • Overt or enhanced bleeding risk • Need for cardiopulmonary bypass surgery

Danaparoid vs. Lepirudin Study characteristics Danaparoid (n = 53) Lepirudin (n = 114) Mean age (yrs) 63 57 Treatment duration Days (median) 7 (1 -115) 10 (Unknown) Treatment schedule High dose 2250 U i. v. bolus 400 U/h – 4 hrs 300 U/h – 4 hrs 200 U/h PCR† at entry >95% Low dose High dose* Low dose 750 U s. c. b. i. d or t. i. d. 0. 4 mg/kg i. v. bolus 0. 15 mg/kg/hr 0. 10 mg/kg/hr i. v. >95% † PCR = platelet count reduction *Dose reduced in patients given thrombolytic therapy

Danaparoid vs. Lepirudin End point frequency (%) Study end point Danaparoid (n = 53) Lepirudin (n = 114) New thrombus 9. 4* 7. 9* Major bleed 2. 5† 10. 4† Deaths 6. 6† 6. 9† * Patients on full anticoagulant dosage schedule (p = 0. 913) † Included patients on low dose schedules
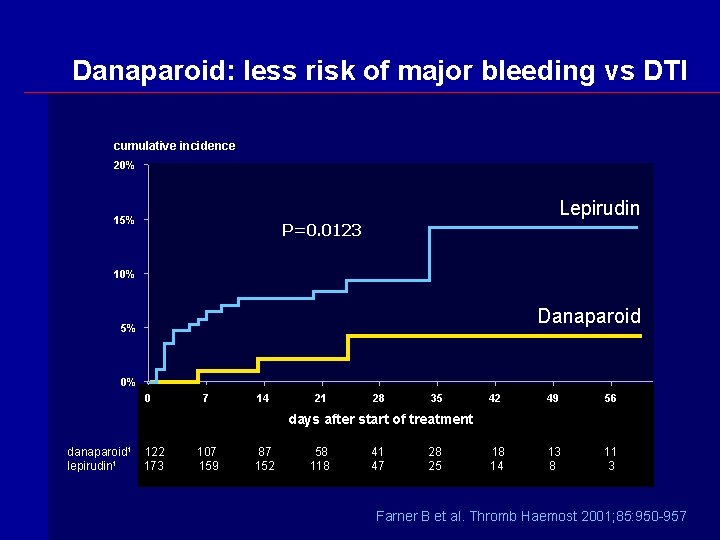
Danaparoid: less risk of major bleeding vs DTI cumulative incidence 20% 15% Lepirudin P=0. 0123 10% Danaparoid 5% 0% 0 7 14 21 28 35 42 49 56 18 14 13 8 11 3 days after start of treatment danaparoid 1 lepirudin 1 122 173 107 159 87 152 58 118 41 47 28 25 Farner B et al. Thromb Haemost 2001; 85: 950 -957

Danaparoid vs. Lepirudin Characteristics Danaparoid Lepirudin Mode of action Anti-Xa >> anti-IIa Anti-IIa 25 hr (anti-Xa) >1. 3 hr. Route of administration i. v. or s. c. i. v. Dose adjustment (bolus) <60 or >75 kg body wt mg/kg body wt or body wt; renal failure Routine Activated protein C generated No Yes Antibody development 7% HIT cross-reactive (clinical significance? ) 40% anti-lepirudin antibodies No Yes <10% 40% Half-life Monitoring recommended Anaphylaxis Major bleeding

Danaparoid HIT Dosing Regimen The following dosing regimen is recommended for patients with HIT (with or without associated thromboembosis): • Bolus: • 2, 250 u* • Adjustment phase: • 400 u/hr for 4 hrs • 300 u/hr for 4 hrs • Maintenance: • 150 -200 u/hr † * for body weight of 60 -75 kg (if <60 kg, give 1500 U bolus; if 75 -90 kg, give 3000 U bolus; if >90 kg, give 3, 750 U bolus) † Adjust by anti-Xa assay levels, if available 7 th. American College of Chest Physicians Conference Chest 2004, 126: 311 S-337 S

Danaparoid HIT Monitoring Recommendations The anti-Xa levels (U/ml) achieved should be: • Post-bolus: • Adjustment phase: • Maintenance: 0. 5 -0. 7 U/ml 1. 0 U/ml 0. 5 -0. 8 U/ml

Danaparoid HIT Monitoring Recommendations • Platelet counts should be determined daily for 1 week, then on alternate days for 2 weeks, then weekly to monthly thereafter (while on danaparoid) • In vitro cross-reactivity testing should be performed if: • Recovery in platelet count does not occur • An existing thrombus extends or a new thromboembolic event occurs

Use of danaparoid in cardiopulmonary bypass (CPB) Danaparoid is: • Not generally recommended for anticoagulation during CPB • Is an option for • Post-CPB anticoagulation • “Off-pump” cardiac surgery

Heparin-Induced Thrombocytopenia: Recognition, Treatment & Prevention Theodore E. Warkentin & Andreas Greinacher ‘Certain of the pharmacokinetic features of danaparoid, such as its long half-life, lack of effect on the INR, and its potential for SC administration make it an appropriate choice for an otherwise uncomplicated patient with venous thromboembolism in whom eventual overlap with oral anticoagulants is required. Danaparoid does not cross the placenta, and thus should be safe for management of pregnant patients with HIT. ’ 7 th. American College of Chest Physicians Conference Chest 2004, 126: 311 S-337 S Danaparoid is not secreted into the breast milk and can used in nursing mothers

The use of Danaparoid in the management of HIT Summary & Conclusions 1. Danaparoid has been used in at least 100, 000 treatment episodes in patients with HIT 2. Clinical studies in HIT suggest a 94% success rate (investigator-reported) 3. It can be given by both i. v. & s. c routes with 100% bioavailability

The use of Danaparoid in the management of HIT Summary & Conclusions 4. Unlike the DTIs (especially argatroban), danaparoid does not prolong the INR, thus simplifying overlapping VKA therapy 5. It demonstrates a favorable anti-thrombotic efficacy: safety ratio 6. Cross-reactivity of danaparoid with HIT antibodies is uncommon and of doubtful clinical significance

The use of Danaparoid in the management of HIT Summary & Conclusions 7. Apart from evidence of prior in vivo cross-reactivity, there are no known contraindications for its use in HIT patients 8. Danaparoid-induced HIT has not been reported 9. Similar efficacy as lepirudin but has better safety profile with regard to: • Major bleeding • Accumulation during renal failure • Immunization and allergy/anaphylaxis

Heparin-Induced Thrombocytopenia (HIT) Treatment with danaparoid (Orgaran )
- Slides: 35