Hep B BD Guidance in RSA Hep B

Hep. B BD Guidance in RSA Hep. B BD Introduction in Africa Meeting 05 May 2021 Dr Kgomotso Vilakazi Nhlapo, National Department of Health, Pretoria, South Africa Dr Nishi Prabdial-Sing National Institute for Communicable Diseases (NICD), Johannesburg, South Africa
South Africa Population of South Africa approximately 59 million The average number of children born per year is approximately 1. 1 Million, with an estimated 90% hospital births Vaccine coverage: BCG dose 2020=86%(84. 4% in 2019). HBV 3 rd dose 2020=83, 6%(85% in 2019)

RSA Hepatitis B Epidemiology Risk of horizontal transmission- early childhood <5 years old • Lower prevalence of HBe. Ag positive mothers-pre-HIV era • Close household contracts, medical or traditional scarification procedures Peri-natal Transmission: Impact of HIV/HBV co-infection in pregnancy • Pregnant women 3 x more likely to test positive for HBV DNA, with higher HBV DNA • Twice as likely to test positive for HBe. Ag • Increased risk of HBV MTCT: 90% risk of chronicity Sexual Transmission • Adults born pre-1995 Percutaneous Transmission • Needle stick injuries (HCWs) Lancet infect Dis 2007; 7: 402
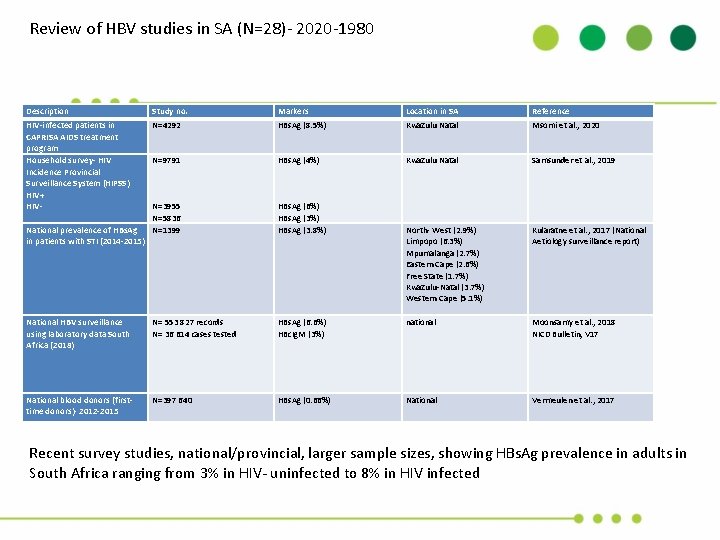
Review of HBV studies in SA (N=28)- 2020 -1980 Description Study no. Markers Location in SA Reference HIV-infected patients in CAPRISA AIDS treatment program Household survey- HIV Incidence Provincial Surveillance System (HIPSS) HIV+ HIV- N=4292 HBs. Ag (8. 5%) Kwa. Zulu Natal Msomi et al. , 2020 N=9791 HBs. Ag (4%) Kwa. Zulu Natal Samsunder et al. , 2019 N=3955 N=5836 N=1399 HBs. Ag (6%) HBs. Ag (3. 8%) North- West (2. 9%) Limpopo (6. 3%) Mpumalanga (2. 7%) Eastern Cape (2. 6%) Free State (1. 7%) Kwa. Zulu-Natal (3. 7%) Western Cape (5. 1%) Kularatne et al. , 2017 (National Aetiology surveillance report) National HBV surveillance using laboratory data South Africa (2018) N= 553 827 records N= 36 614 cases tested HBs. Ag (6. 6%) HBc. Ig. M (3%) national Moonsamy et al. , 2018 NICD Bulletin, V 17 National blood donors (firsttime donors)- 2012 -2015 N=397 640 HBs. Ag (0. 66%) National Vermeulen et al. , 2017 National prevalence of HBs. Ag in patients with STI (2014 -2015) Recent survey studies, national/provincial, larger sample sizes, showing HBs. Ag prevalence in adults in South Africa ranging from 3% in HIV- uninfected to 8% in HIV infected
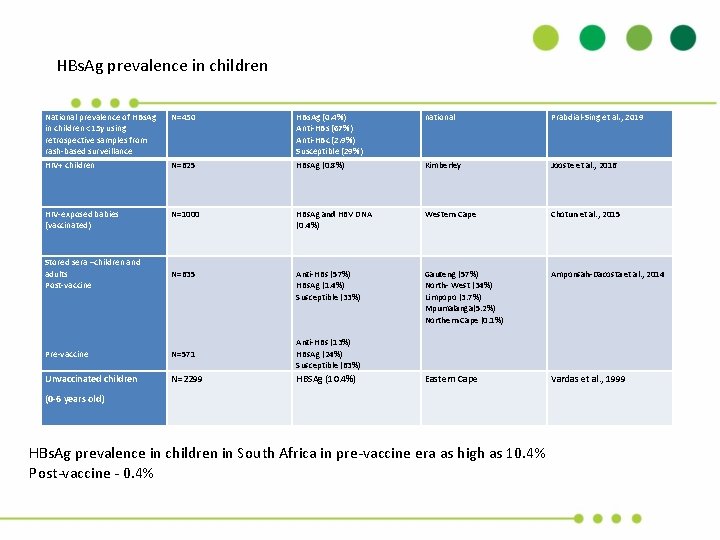
HBs. Ag prevalence in children National prevalence of HBs. Ag in children <15 y using retrospective samples from rash-based surveillance N=450 HBs. Ag (0. 4%) Anti-HBs (67%) Anti-HBc (2. 9%) Susceptible (29%) national Prabdial-Sing et al. , 2019 HIV+ children N=625 HBs. Ag (0. 8%) Kimberley Jooste et al. , 2016 HIV-exposed babies (vaccinated) N=1000 HBs. Ag and HBV DNA (0. 4%) Western Cape Chotun et al. , 2015 Stored sera –children and adults Post-vaccine N=635 Anti-HBs (57%) HBs. Ag (1. 4%) Susceptible (33%) Gauteng (57%) North- West (34%) Limpopo (3. 7%) Mpumalanga(5. 2%) Northern Cape (0. 1%) Amponsah-Dacosta et al. , 2014 Pre-vaccine N=571 Anti-HBs (13%) HBs. Ag (24%) Susceptible (63%) Unvaccinated children N=2299 HBSAg (10. 4%) Eastern Cape Vardas et al. , 1999 (0 -6 years old) HBs. Ag prevalence in children in South Africa in pre-vaccine era as high as 10. 4% Post-vaccine - 0. 4%
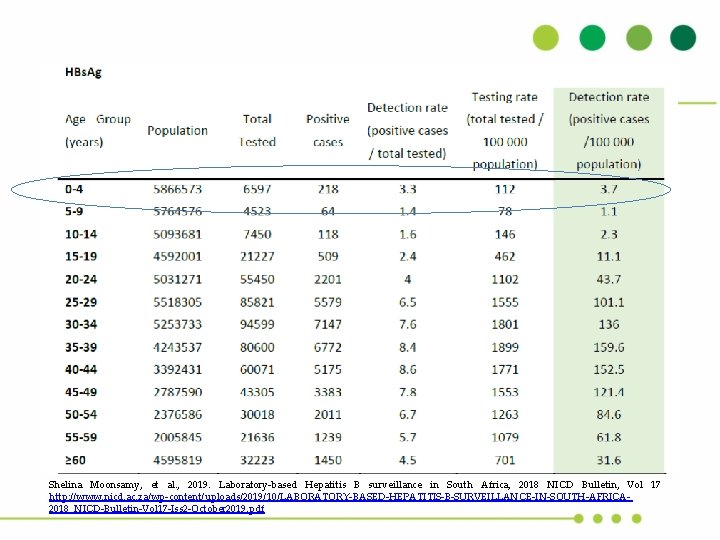
Shelina Moonsamy, et al. , 2019. Laboratory-based Hepatitis B surveillance in South Africa, 2018 NICD Bulletin, Vol 17 http: //www. nicd. ac. za/wp-content/uploads/2019/10/LABORATORY-BASED-HEPATITIS-B-SURVEILLANCE-IN-SOUTH-AFRICA 2018_NICD-Bulletin-Vol 17 -Iss 2 -October 2019. pdf
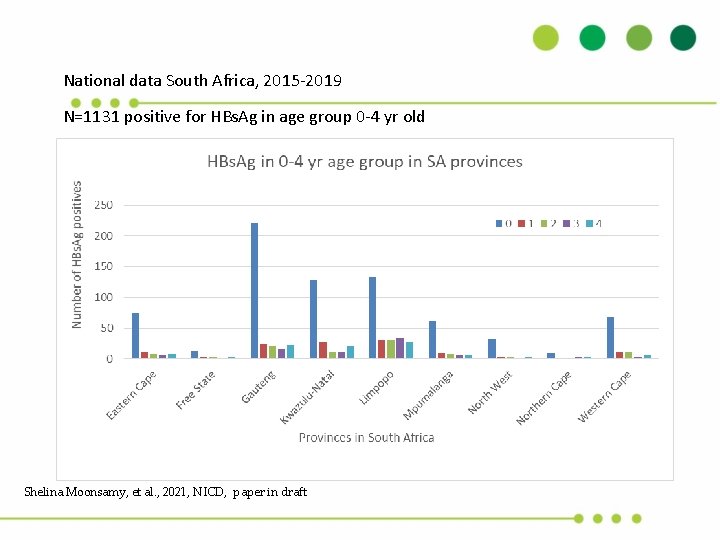
National data South Africa, 2015 -2019 N=1131 positive for HBs. Ag in age group 0 -4 yr old Shelina Moonsamy, et al. , 2021, NICD, paper in draft

CONCLUSION Summarized data from as early as 1999 to present There is known evidence presented on the gains of HBV vaccination in South Africa, comparing data pre-1995 and post. Data provided that there are still cases of HBV infection in the country in the age group that should be protected - Poor vaccine coverage - MTCT is taking place and concerning data shows this in 0 -1 age group - Vaccine hesitancy Roll-out programs - HB BD - HBV screening antenatal - Follow-up

The National Clinical guidelines and Hepatitis Action Plan • The national clinical guidelines and Action plan for Viral hepatitis were approved in 2018. • Guidelines were used to extract a set of activities required to implement guidelines in practice(making guidelines practical) Hepatitis Action Plan Priority Activities 1. Raise awareness of Hepatitis Viral infection amongst the health care workers and the general population (Knowledge and information on VH is very limited in these 2 populations) 2. Strengthen knowledge of hepatitis burden of disease (Research and surveillance-no existing system in the country, most data is from studies performed in different parts of the country) 3. Prevent transmission of viral hepatitis (education, vaccination, diagnosis, treatment ) 4. Improve diagnosis and treatment of chronic hepatitis 5. Management, coordination and evidence-based policy 9

Priority interventions in the Action Plan include: • HBV birth dose vaccination –Current EPI is at 6, 10&14 weeks, with a booster at 18 months. HBVBD vaccine has a significant impact at low cost in further reducing the burden of HBV and producing an HBV free generation. • Awareness, diagnosis and treatment for HBV to be strengthened – notably as part of antenatal care, include PMTCT screening and treatment of HBs. Ag +ve /HBV VL>200 000 in 3 rd trimester. TDF can be stopped 12 weeks post delivery, if only used for HBV MTCT prevention in HIV-negative pregnant women. HIV/TB/STIs PMTCT guidelines and Maternal Care Guidelines include Hep. B-BD • A one-time campaign to screen all healthcare workers for HBV and HCV and vaccinate for HAV and HBV, and add integration of screening and vaccination at entry into clinical service. 10

Cost of Birth dose implementation • • • Vaccine and consumables Additional cold chain and dry storage needs Expansion of vaccine distribution sites (i. e. delivery facilities) Distribution costs New delivery strategies (postnatal wards, outreach for home births) Training of health care workers Waste management costs Development of communication strategy and roll-out of information Education and communication (IEC) media and materials Revision, printing home-based records, registries, forms AEFI monitoring and response

EPI schedule implications • To prevent mother-to-child (perinatal) HBV transmission , Hepatitis B vaccine preferably administered within 24 hours • This should be followed by at least two subsequent doses – Hexaxim (4) • In interest of access and public health – complete roll-out • Evidence from observational studies shows that birth dose vaccination can still be effective against perinatal transmission, even if given later than 24 hours after birth, less effective over time -Catch-up will be provided up to 2 weeks after birth • Access infants born at home – Discussions ongoing

EPI schedule implications • Inclusion into the private EPI schedule – should be discussed • Survey of the range of documents used by obstetric, maternal and new-born care staff should be conducted • Need to revise guidance on care at childbirth or in the early newborn period-Maternal Care Guidelines revised • Implementation activities: • Situation analysis (EPI programme ) • National policies support implementation of Hep. B-BD vaccination • Health-facility policies and procedures require and authorize appropriate staff to give Hep. B-BD • Private sector provides Hep. B-BD and has access to vaccine supply

EPI schedule implications • Hep B-BD vaccination integrated with new-born care for both facility births and home births • Monovalent Hep B vaccine integrated fully into vaccine supply chain • Health facility, cold chain and strategic placement of vaccine in postnatal care ward • Train all appropriate staff working in immunizations and maternal and newborn health • Supervise and monitor at national, subnational and facility level in all settings where births take place (public-health facility, private facility, community)

Action • Amend RSA registration with SAHPRA of Hep B paediatric 10 dose, to include birth dose. • Submission to DG for change of the EPI schedule, for inclusion of Hep B BD in EML • Addition of Hep. B-BD indicators in the next NIDS Programme management & coordination • Will be coordinated from NDOH. Stakeholder mapping to be done from other programmes, including HIV/TB, Maternal Health, PHC and District health services. These stakeholders should be identified, including private sector, community forums or interest groups which could assist with the implementation of the Hep. B-BD. Planning & preparation • Project plan for implementation, done after stakeholder analysis. • Identified Lead persons for implementation of Hep BD for each activity according to expertise Post Implementation Evaluation – 1 year post implementation

Conclusion • The WHO 2030 targets challenges us to strengthen & up-scale Hepatitis focal Programmes : • • • Reduce HBV and HCV deaths by 65% Reduce incidence of HBV and HCV by 90% Reduce mother-to-child transmission of HBV by 90% Diagnose 90% of HBV and HCV cases Treat 80% of eligible HBV and HCV patients • While the guidelines and action plan create a roadmap towards realizing the WHO 2030 targets • Our ability to reach the targets is dependent on consistent commitment from all stakeholders and the availability of resources

Acknowledgement • • • Prof W. Spearman Prof M. Sonderup Dr N. Prabdial-Singh World Health Organization PHAROS Team National Viral Hepatitis Technical Working Group

• • • Ndi a livhuwa Thank you Ke a leboga Dankie Ha khensa Ndiyabulela
- Slides: 18