Hench Wilson Surface active biomaterials Two classes of

Hench & Wilson : Surface active biomaterials Two classes of endosseous implants 1. Bone-bonding : calcium phosphate coated implant 2. Nonbonding : metals such as titanium Science 1984, 226: 630 -636

Why plasma sprayed hydroxyapatite coating method is declined ?
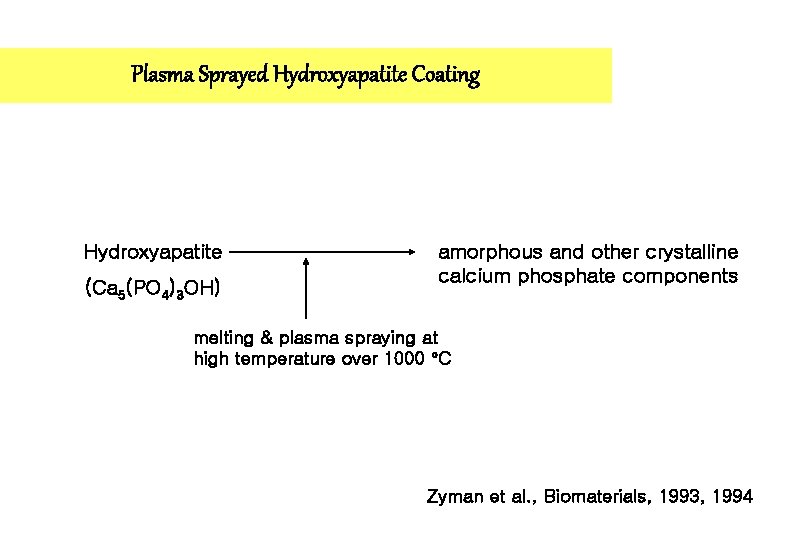
Plasma Sprayed Hydroxyapatite Coating Hydroxyapatite (Ca 5(PO 4)3 OH) amorphous and other crystalline calcium phosphate components melting & plasma spraying at high temperature over 1000 C Zyman et al. , Biomaterials, 1993, 1994
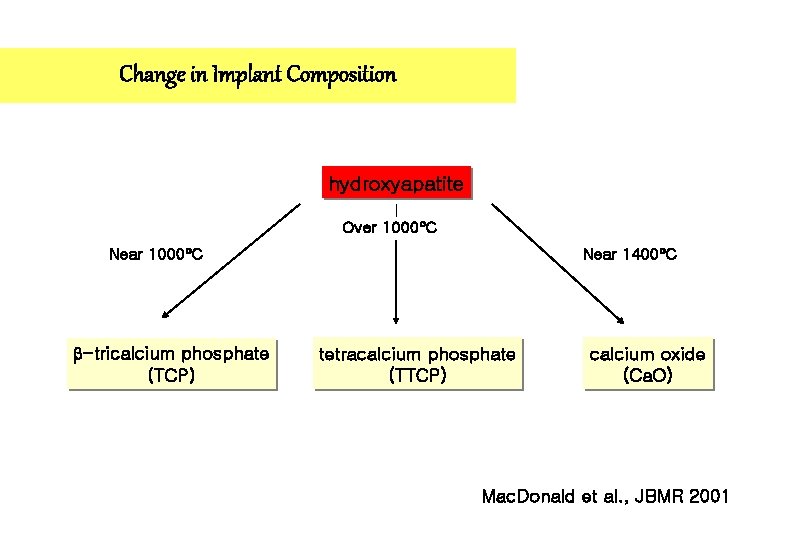
Change in Implant Composition hydroxyapatite Over 1000 C Near 1000 C -tricalcium phosphate (TCP) Near 1400 C tetracalcium phosphate (TTCP) calcium oxide (Ca. O) Mac. Donald et al. , JBMR 2001

Change in Implant Composition Therefore, HA coating after plasma spraying procedure is not longer HA, but contains non-HA constituents(TTCP, ACP and Ca. O). Mac. Donald et al. , JBMR 2001

Less Crystallinity and Coating Dissolution Constituents of less crystallinity (ACP, TTCP, TCP and Ca. O) within th e c o a tin g w h ic h d e m o n s tra te a g re a te r p re d is p o s itio n to w a rd d is s o lu tio n w ill p a s s in to th e b io lo g ic a l e n v iro n m e n t to a g re a te r degree than more highly crystalline phases, most notably HA. Mac. Donald et al. , JBMR 2001

Factors that impede crystalline phase formation 1. Melting of the HA powder during plasma spraying 2. Rapid cooling of molten material (Heat dissipation to the metallic substrate is sufficiently rapid to constitute amorphous phase formation) 3. Dehydroxylation of the surface HA powder Mac. Donald et al. , JBMR 2001

Results of non-HA constituents solubilization 1. Resultant microvoids develop between the HA splats 2. Loss of these soluble Ca. P phases may potentially compromise the integrity of the remaining coating 3. Solubilized Ca. P inorganics potentially reprecipitating, 4. The coating interacting with bone and proteins facilitating the formation and entry of bone into these microvoids. Mac. Donald et al. , JBMR 2001

Results of non-HA constituents solubilization 5. As a result of coating disintegration and fragmentation within the biological environment, these HA particles may incorporate into the surrounding bone or potentially stimulate an inflammatory reaction with localized bone loss. Mac. Donald et al. , JBMR 2001

Thickness of Plasma Sprayed Coating 1. As applied by manufacturers, varies between 50 and 200 m. 2. It should be noted that a coating thickness over 100 m can introduce fatigue under tensile loadings.

Thickness of Plasma Sprayed Coating 3. Excessive thickness can lead to coating delamination and fragmentation, which in time can result in implant mobility. 4. Søballe(1993) reported a thin coating 50 m gave a stronger fixation than a thick coating of 200 m

Future perspectives of Ca-P-coated implants 1. Sol-gel coating 2. Electrophoretic deposition 3. Pulsed laser deposition 4. Biomimetic deposition 5. Sputter deposition

Other than living cells, the main constituent of bones and teeth is a calcium phosphate called hydroxyapatite (Ca 5(PO 4)3 OH)

Why not bulk calcium-phosphate implant ? 1. Bulk calcium phosphates are weak and brittle, making them unsuitable for replacing parts of the body that experience large stresses, such as teeth. 2. To eliminate of this problems, combine the mechanical strength of titanium with the biological properties of calcium phosphate.

The Quality of Plasma Spray-deposited Coating 1. Plasma temperature 2. Nature of plasma gas 3. Particle size of the powder 4. Chemical nature of the ceramic powder Wolke et al. J. Thermal Spray Technology 1992

Quality Analysis of HA-coated Implants 1. 2. 3. 4. 5. 6. 7. Chemical composition Ca/P ratio Crystallinity Density Tensile strength Thickness and Uniformity Trace element analysis But these control tests will be no guarantee for the final biologic performance of the coated implants

First HA coated Dental Implant • In 1984, were sold under the name Integral • 75 m thick layer of hydroxyapatite • Clinically apparently satisfactory results

Histologic Studies of HA-coated Implants 1. Piatelli & Trisi (1993), Piatelli et al. (1998) Oguchi (1994) 2. Very intimate bone-HA coating contact 3. The bonding of HA coating to the metal was strong enough to resist loading forces.

Crystallinity and Biologic Behavior of Plasma Sprayed Ca-P Coatings 1. The more crystalline a Ca-P coating is, the lower its dissolution rate 2. The combination of stress and dissolution had a dramatic influence on the integrity of the amorphous or glassy phase of the HA coating Klein et al. Biomaterials, 1994

Biodegradation of Ca-P coatings 1. 2. 3. 4. 5. 6. 7. 8. Crystal structure Microporosity Crystallinity Chemical composition Ca/P ratio Lattice defects Particle size Purity of starting powder

Biodegradation of Ca-P coatings 1. The degradation of plasma-spray Ca-P coatings occurs by the rapid dissolution of the relatively soluble amorphous phase, followed by loosening of crystalline particles. 2. This can cause a foreign body response (Bloebaum and Dupont, J. Arthroplasty 1993)

Amorphous or Glassy phase after Plasma-Spraying 1. This state of Ca-P ceramic is unstable and more liable to biodegradation : physicochemical dissolution (Buser et al. J. Biomed. Mater. Res. 1991) 2. Degradation can also occur by cell mediated processes (Frayssinet et al. Cells and Materials, 1995, )

Future Perspectives of Ca-P Coated Implants Merit Dental Clinic Hur, Yin-Shik, Periodontist

1. Sol-gel coating 2. Electrophoretic deposition 3. Pulsed laser deposition 4. Biomimetic deposition 5. Sputter deposition

Sol-gel Process for Ca-P coating Merit Dental Clinic Yin-Shik Hur, Periodontist

Sol-gel Process 1. Inorganic polymerization procedure based on the hydrolysis and condensation reactions of metal alkoxides. 2. Conventional sol-gel precursor solutions often contain metal alkoxides diluted in ethanol, water and acid. Vititala et al. , Thin Solid Films 2000

Sol-gel Process 3. Titanium alkoxides are known to react readily with water. 4. Solution composition and preparation procedures play a key role in the sol-gel manufacturing of titania thin films. Vititala et al. , Thin Solid Films 2000

Various Sol-gel Processes 1. Dip-coating method 2. Spin-coating method 3. Aerosol-gel method Vititala et al. , Thin Solid Films 2000

Dip-Coating Methods 1. Based on linear spreading of the sol on the substrate 2. The film thickness is essentially controlled by the solution viscosity and the linear speed of the substrate. Vititala et al. , Thin Solid Films 2000

Sol-gel coating by Dipping 1. HA sols are prepared and pure titanium rods are dipped into HA sols. 2. Extremely dense, thickness of about 10 m. 3. Superior bone behavior compared to uncoated implants (Li et al. 1996, J. Materials Science : Material in Medicine)

Spin-Coating Methods 1. Based on centrifugal spreading of the sol on the substrate 2. The film thickness is essentially controlled by the solution viscosity and the centrifugal speed of the substrate. Vititala et al. , Thin Solid Films 2000

Aerosol-gel Techniques 1. Based on the sol-gel polymerization of a liquid film deposited from an ultrasonically generated aerosol. 2. This process displays several advantages compared to conventional sol-gel deposition techniques. Vititala et al. , Thin Solid Films 2000
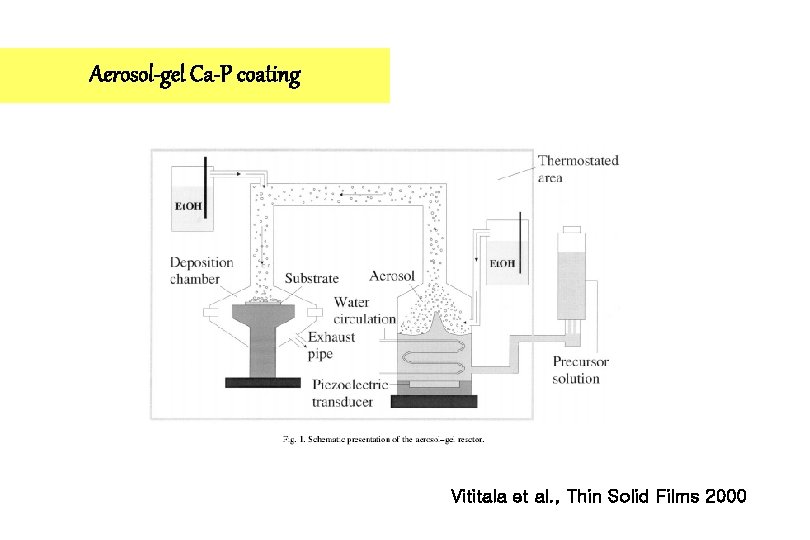
Aerosol-gel Ca-P coating Vititala et al. , Thin Solid Films 2000

Advantages of Aerosol-gel Techniques 1. Due to aerosol it is possible to coat substrates with complex shapes 2. The layer thickness can be accurately controlled by variation of several experimental parameters, including dilution of the precursor, applied ultrasonic excitation power, deposition duration and carrier gas flow rate. Vititala et al. , Thin Solid Films 2000

Advantages of Aerosol-gel Techniques 3. A suitable adjustment of the substrate temperature enables the manufacturing of films with different morphologies, ranging from smooth optical quality layers to rough or non-continuous coatings. 4. From an industrial point of view, the aerosol-gel process is fully compatible with inline technologies for highly efficient production. Vititala et al. , Thin Solid Films 2000

Electrophoretic Deposition 1. Charged Ca-P ceramic particles suspended in solution are uniformly deposited onto the metal substrate by electrophoresis. 2. The coating is densified and bonded to the metal by sintering. 3. Low adherence to the metal surface 4. Only a short-term advantage (Gottlander et al. 1997 b. Biomaterials)

Pulsed Laser Deposition 1. Ablation of an HA target using a pulsed Kr. F excimer laser beam ( =248 nm) in Torr/H 2 O atmosphere and deposition of ejected HA material on heated substrate (400800ºC) 2. Thin HA coating (0. 05 -5 m) 3. The adhesion strength decreases for deposited films produced with high substrate temperature and appears to be related to the existence of an oxide at the interface between the film and the substrate.

Biomimetic Deposition 1. Biologically active bone-like apatite layer is formed on a substrate surface by immersion in a Hank’s Balanced Salt Solution or Simulated Body Fluid at 37ºC for several days. 2. Coating thickness : 1 -5 m (Hanawa & Ota, Applied Surface Science , 1992)

Sputter Deposition 1. In a vacuum chamber, atoms or molecules of a material are ejected by bombardment of high-energy ions. The dislodged particles deposit on a substrate, also placed in to the vacuum chamber. 2. Coating thickness : 0. 5 -10 m 3. Ca/P ratio : 1. 5 -2. 6

Sputter Deposition 4. The in vitro dissolution appeared to be determined by the degree of crystallinity of the coating. 5. Strong adhesion of the coatings to titanium substrates 6. Magnetron sputtering appears to be the most promising method forming biocompatible Ca-P coatings.

Advantages of Magnetron Sputtering 1. Thin coating thickness 2. High deposition rate 3. Excellent adhesiveness 4. Ability to coat implants with difficult surface geometry But, the clinical efficacy of these coatings has to be proven in human clinical trials

New Trends for Ca-P Coating • Towards very thin (< 10 m) coatings • Well-controlled crystalline Ca-P phases on titanium

New Trends for Ca-P Coating 1. Magnetron sputtering 2. Pulsed laser deposition 3. Ion beam assisted deposition 4. Electrophoresis 5. Surface-induced mineralization 6. Biomimetic deposition 7. Sol-gel processing

Evaluating mechanical adhesion of sol-gel titanium dioxide coatings containing calcium phosphate for metal implant application Piveteau L. -D. , Gasser B. Schlapbach L Biomaterials , 2000

Advantages of Ti. O 2 -Ca. P sol-gel coating 1. The composition of coating can be constant over its entire thickness. 2. The composition of coating can present a concentration gradient (from a pure Ti. O 2 at the substrate surface to a pure HA at the implant/body interface) Piveteau et al. , Biomaterials 2000

Objective of Ti. O 2 -Ca. P sol-gel coating 1. To add a chemical component to the binding between the titanium substrate (covered with natural Ti. O 2) and the coating (containing Ti. O 2) to increase the adhesion strength 2. In vivo, the presence of Ti. O 2 -Ti. O 2 binding should reduce the risk of loosening through dissolution of the coating at the interface with the substrate. Piveteau et al. , Biomaterials 2000

Procedures of Ti. O 2 -Ca. P sol-gel coating 1. A sol of Ti. O 2 , obtained by hydrolyzing a 1 M solution of tetrabutyl ortho-titanate {Ti. O(CH 2)3 CH 3)4} in absolute ethanol denatured with 5% of methanol with acidified water 2. A solution of calcium nitrate {Ca(NO 3)2 } / phosphoric acid esters{PO(OH)3 -x(OEt)x: x = 1, 2; 50/50% } were mixed together and deposited at room temperature by dip- or spincoating Piveteau et al. , Biomaterials 2000

Procedures of Ti. O 2 -Ca. P sol-gel coating 3. In order to change thickness of the layer, precursors with various viscosities and varied coating speeds were used. 4. After deposition of three layers, the coatings were crystallized for 10 min at 850ºC in air. Piveteau et al. , Biomaterials 2000

Procedures of Pure Ca. P sol-gel coating 1. Pure Ca. P sol-gel coating were deposited in a similar way 2. A mixture of calcium nitrate and phosphoric acid esters in ethanol was coated onto Ti. Cp and crystallized for 10 min at 850ºC in air. Piveteau et al. , Biomaterials 2000

Sol-gel Preparation by Dip-coating 1. Viscosity of precursor : 1. 95 10 -2 Pa s, measured at 26ºC. 2. Withdrawal speed : Thin (1 m) coating: 0. 57± 0. 05 mm/s Thick(5 m) coating: 4. 26± 0. 05 mm/s Piveteau et al. , Biomaterials 2000

Sol-gel Preparation by Spin-coating 1. Single angular velocity : 2800 r. p. m. 2. Viscosity of precursor : measured at 26ºC. Thin (1 m) coating: 3. 52 10 -3 Pa s, Thick(5 m) coating: 1. 95 10 -2 Pa s, Piveteau et al. , Biomaterials 2000

Adhesion, Coating thickness, Roughness 1. For all surface pre-treatment, the tensile strength was reduced by 20 -40% for a thick coating. 2. Alumina-blasted samples showed a 30 -50% higher adhesion strength. Piveteau et al. , Biomaterials 2000

Failure Patterns of Coating 1. For thick coatings, failure always occurs at the interface between the coating and the substrate. 2. With thin layered samples having a smooth substrate, failure mostly occurs between the coating and the substrate and in some locations between the coating and the glue. 3. For the alumina-blasted surface, failure occurs at coating/glue and glue/stud interfaces. Piveteau et al. , Biomaterials 2000

Conclusion of Piveteau et al. (2000) 1. The adhesion increases for a decreasing thickness and an increasing roughness at a micrometer scale. 2. The difference observed between a pure Ca. P coating and a Ti. O 2 –Ca. P coating suggests also the involvement of a chemical component in the binding between the substrate and the coating. Piveteau et al. , Biomaterials 2000

Combination of Alkali Treatment and Heat Treatment for Ca-P Deposition Merit Dental Clinic Yin-Shik Hur, Periodontist

The effect of heat treatment on bone-forming ability of alkali-treated titanium Shigeru Nishiguchi, Takashi Nakamura, Masahiko Kobayashi, Hyun-Min Kim, Fumiaki Miyaji, Todashi Kokubo Biomaterials 1999
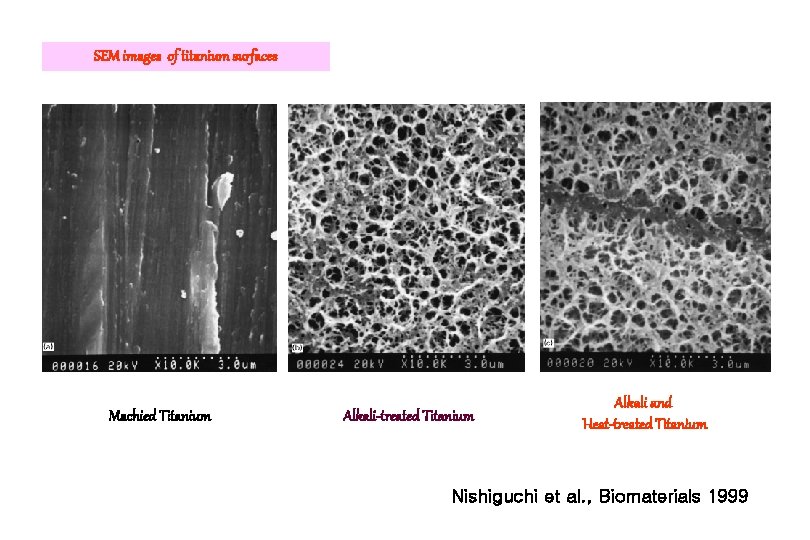
SEM images of titanium surfaces Machied Titanium Alkali-treated Titanium Alkali and Heat-treated Titanium Nishiguchi et al. , Biomaterials 1999

Surface structural and morphologic feature of the surface of Ti metal subjected to alkali and heat treatments and the subsequent soaking in SBF for 4 weeks. (SEM image) Yan et al. , J. Biomed. Mater. Res. 1997

Procedure for Alkali and Heat Treatment 1. Pure titanium plates were soaked in 5 mol Na. OH aqueous solution at 60ºC for 24 h. 2. The plates were washed gently with acetone and distilled water, and dried at 40ºC for 24 h in an air atmosphere. 3. The plates were heated to 600 ºC at a rate of 5 ºC min-1 in an electric furnace, kept at a given temperature for 1 h. Nishiguchi et al. , Biomaterials 1999
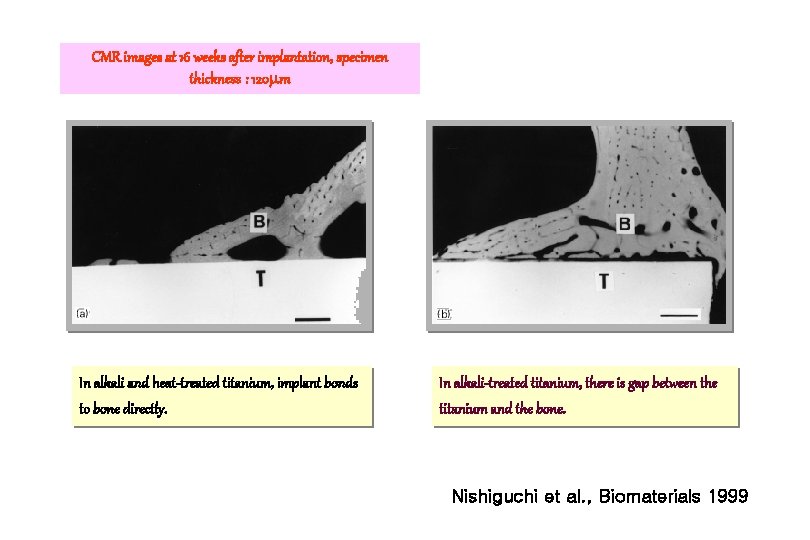
CMR images at 16 weeks after implantation, specimen thickness : 120 m In alkali and heat-treated titanium, implant bonds to bone directly. In alkali-treated titanium, there is gap between the titanium and the bone. Nishiguchi et al. , Biomaterials 1999
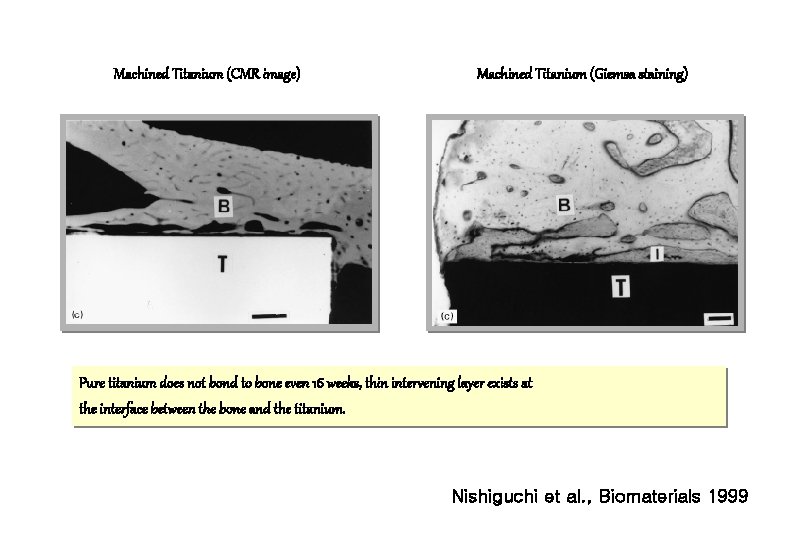
Machined Titanium (CMR image) Machined Titanium (Giemsa staining) Pure titanium does not bond to bone even 16 weeks, thin intervening layer exists at the interface between the bone and the titanium. Nishiguchi et al. , Biomaterials 1999
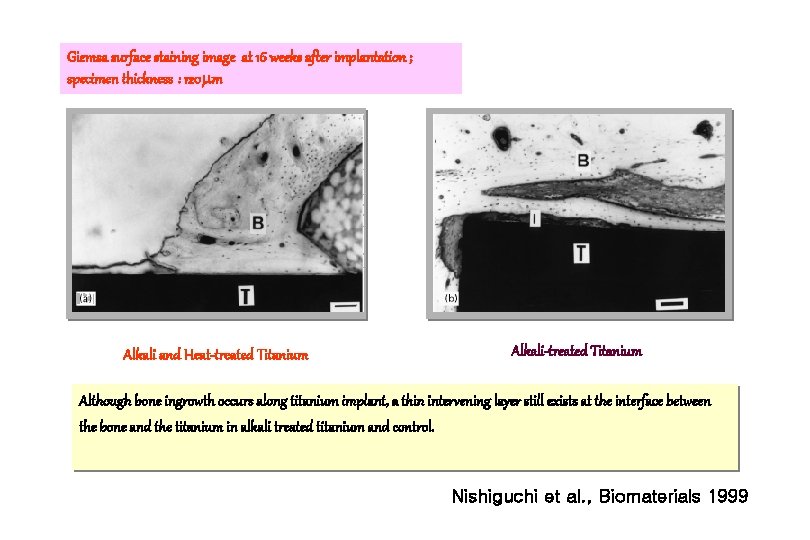
Giemsa surface staining image at 16 weeks after implantation ; specimen thickness : 120 m Alkali and Heat-treated Titanium Alkali-treated Titanium Although bone ingrowth occurs along titanium implant, a thin intervening layer still exists at the interface between the bone and the titanium in alkali treated titanium and control. Nishiguchi et al. , Biomaterials 1999

Histological Exam. of Alkali and Heat Treated Samples CMR reveals that all samples contacted the bone directly, without any intervening fibrous tissue at 8 weeks, and at 16 weeks, the amount of bone in direct contact with the plate increased. Nishiguchi et al. , Biomaterials 1999

Histological Exam. of Alkali Treated Samples and Control There was a fibrous tissue layer between the bone and the implant in control titanium group at 8 weeks. At 16 weeks new bone formation increased around the plates, but intervening layers, although thinner, still remained. Nishiguchi et al. , Biomaterials 1999

Alkali Titanate Hydrogel Layer 1. Titanium forms an alkali titanate hydrogel layer on its surface when soaked in an alkali solution. 2. This layer is unstable as to lose its apatite-forming ability during preservation or implantation. 3. Kim et al. (1997) recommended that heat treatment should be added to form a more stable alkali titanate layer. Nishiguchi et al. , Biomaterials 1999
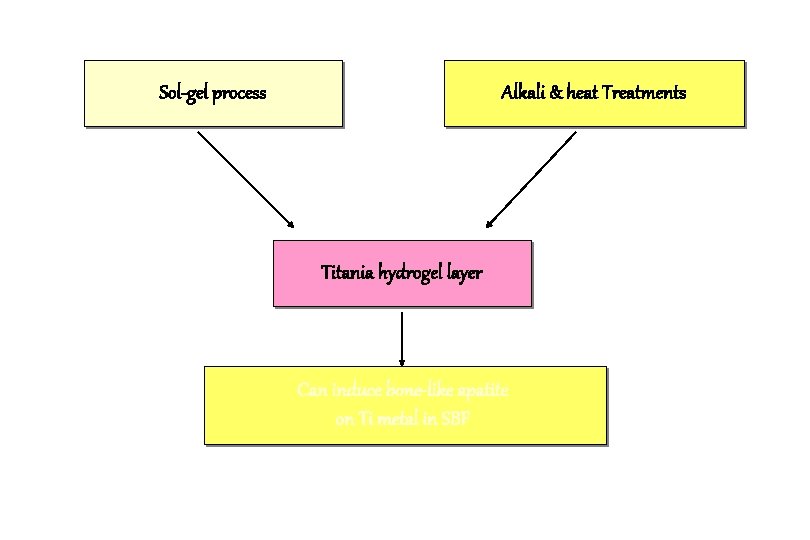
Sol-gel process Alkali & heat Treatments Titania hydrogel layer Can induce bone-like apatite on Ti metal in SBF
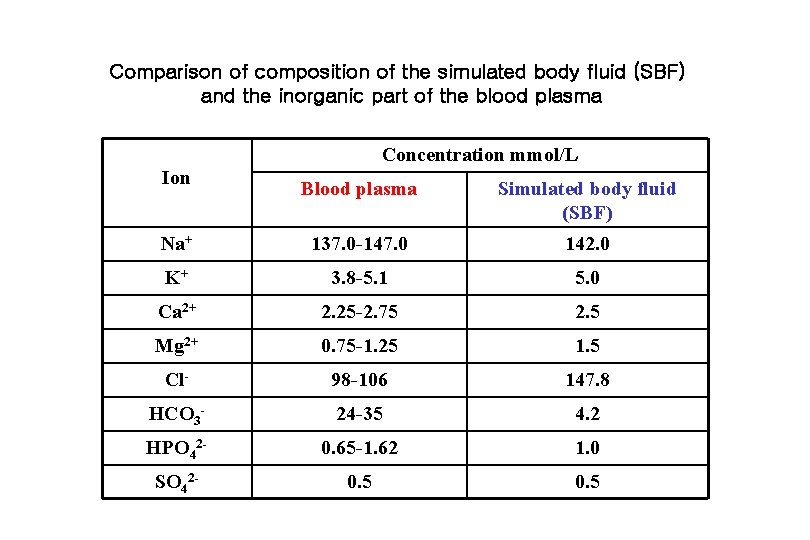
Comparison of composition of the simulated body fluid (SBF) and the inorganic part of the blood plasma Concentration mmol/L Ion Blood plasma Simulated body fluid (SBF) Na+ 137. 0 -147. 0 142. 0 K+ 3. 8 -5. 1 5. 0 Ca 2+ 2. 25 -2. 75 2. 5 Mg 2+ 0. 75 -1. 25 1. 5 Cl- 98 -106 147. 8 HCO 3 - 24 -35 4. 2 HPO 42 - 0. 65 -1. 62 1. 0 SO 42 - 0. 5
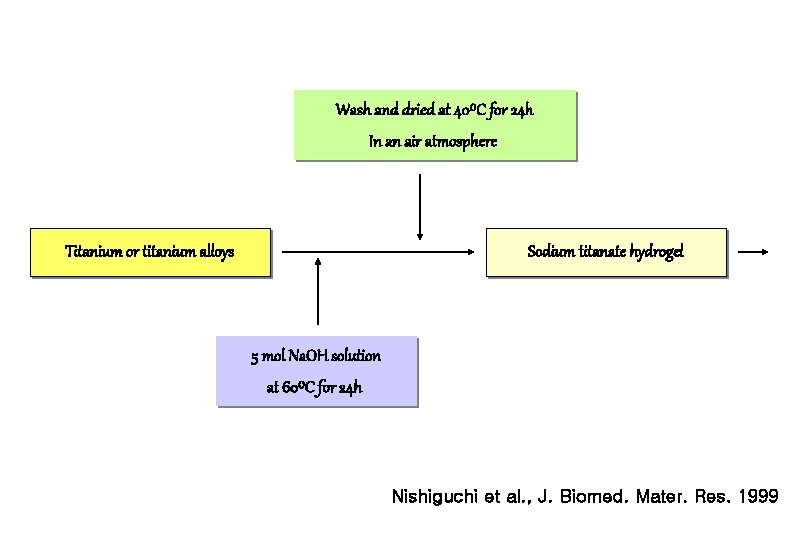
Wash and dried at 40⁰C for 24 h In an air atmosphere Titanium or titanium alloys Sodium titanate hydrogel 5 mol Na. OH solution at 60⁰C for 24 h Nishiguchi et al. , J. Biomed. Mater. Res. 1999
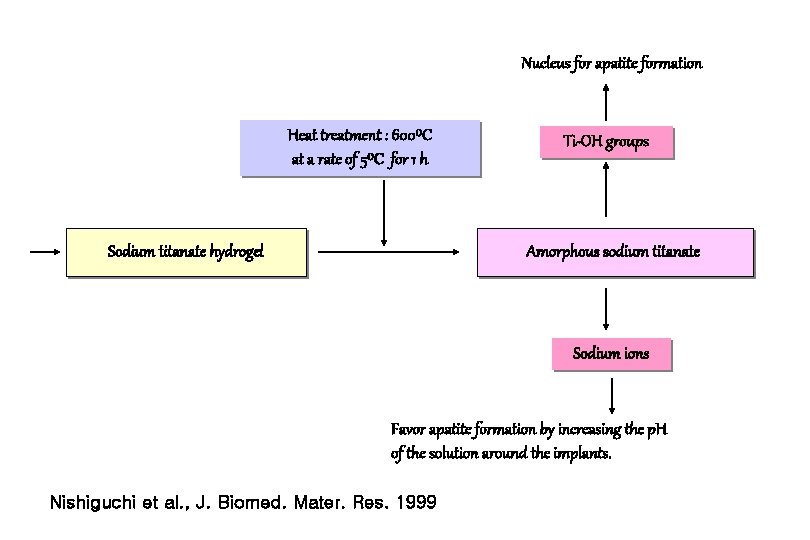
Nucleus for apatite formation Heat treatment : 600⁰C at a rate of 5⁰C for 1 h Sodium titanate hydrogel Ti-OH groups Amorphous sodium titanate Sodium ions Favor apatite formation by increasing the p. H of the solution around the implants. Nishiguchi et al. , J. Biomed. Mater. Res. 1999
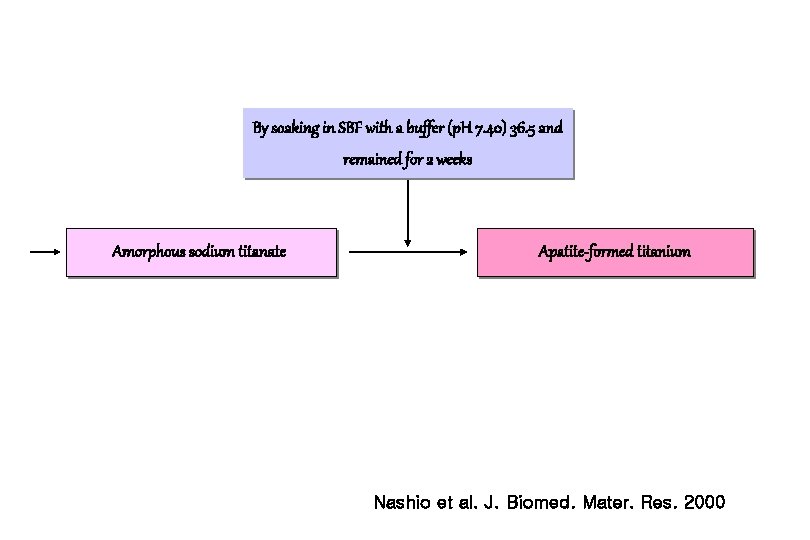
By soaking in SBF with a buffer (p. H 7. 40) 36. 5 and remained for 2 weeks Amorphous sodium titanate Apatite-formed titanium Nashio et al. J. Biomed. Mater. Res. 2000

Conclusion of Nishiguchi et al. (1999) 1. Although alkali-treated titanium has the ability to form apatite in SBF, it dose not serve as a bone-bonding material in practical use. 2. Both alkali and heat treatments are essential for inducing bioactivity in titanium. Nishiguchi et al. , Biomaterials 1999

Conclusion of Nishiguchi et al. (1999) 3. In our study we measured the tensile strength, which is less affected by surface roughness. Also, alkali-treated samples had almost the same surface topography. 4. Thus it is supposed that chemical bonding between titanium and bone via an apatite layer plays a major role in bone-bonding behavoir Nishiguchi et al. , Biomaterials 1999

Conclusion of Nishiguchi et al. (1999) 1. Alkali-and heat treated Ti 6 Al 4 V may be more useful as a bioactive metal than alkaliand heat treated pure titanium, due to its enhanced mechanical properties. 2. T i 15 Mo 5 Zr 3 Al may be more useful rather than Ti 6 Al 4 V because of low elastic modulus and lack of vanadium. Nishiguchi et al. , J. Biomed. Mater. Res. 1999

Advantages of Alkali and Heat Treatment over Plasma-Spray Coating 1. Can be applied uniformly on complicated surface texture. 2. Since reactive layer is as thin as 1 m, can be applied without changing existing implant design. 3. Simple and econnomical compared with the H plasma-spray technique. Nishiguchi et al. , Biomaterials 1999

TPS control AW-glass ceramic TPS AH-TPS Nishiguchi et al. , J. Biomed. Mater. Res. 2001

TPS control and AW-GC coated TPS implant : bone ingrowth into pores was observed, but intervening fibrous tissue usually existed at the interface between the bone and the titanium. Alkali and heat treated TPS implant : direct bonding between the bone and the titanium. Nishiguchi et al. , J. Biomed. Mater. Res. 2001

Combination of Alkali and Heat Treatment on the TPS surface 1. Alkali and heat treatments on porous titanium (TPS) implants may provide earlier stabilization and better clinical results of porous titanium (TPS) implants. 2. Alkali and heat treatments do not reduce the pore space available for bone ingrowth, because they cause little change in the surface morphology of the implant, affecting only the top 1 m of the surface. Nishiguchi et al. , J. Biomed. Mater. Res. 2001

Combination of Alkali and Heat Treatment on the TPS surface 3. Direct bonding between bone and the titanium implant obtained by alkali and heat treatments may prevent the invasion of fibrous tissue, metal particles, subsequent osteolysis. 4. Their effects extend all over the irregular surface of the implant, because the implants are immersed into Na. OH solution. Nishiguchi et al. , J. Biomed. Mater. Res. 2001

The effect of alkali- and heat-treated titanium and apatite-formed titanium on osteoblastic differentiation of bone marrow cells (in vitro study) Ken Nashio, Masashi Neo, Haruhiko Akiyama, Shigeru Nishiguchi, Hyun-Min Kim, Tadashi Kokubo, Takashi Nakamura J. Biomed. Mater. Res. , 2000

Apatite formation and Bioactivity Formation of a surface layer of apatite is a prerequisite for the bioactivity of materials, not only because apatite is a main component of bone crystal, but also because the apatite layer preferentially adsorbs acellular proteins that serve as growth factors. Hench et al. , The bone-biomaterial interface, 1991

Bioactive glass and Local factors 1. Covering a bioactive glass surface with carbonated HA in Tris buffer enhanced the expression of ALP activity and the production of mineralized extracellular matrix of bone cells. 2. They proposed that this enhancement was due to the adsorption of a high quantity of fibronectin from the serum onto the reacted HA. El-Ghannam and Ducheyne, et al. , Biomaterials, 1997

ALP activity on HA surface The cellular DNA levels of rat bone marrow cells cultured on HA or pure titanium were not significantly different, but ALP activity of the cells cultured on HA was higher than on pure titanium. Ozawa et al. , Biomaterails 1996

Bioactive surface and Osteoblast differentiation The promotion of osteoblastic differentiation of bone marrow cells on bioactive glass ceramics could be further enhanced by the formation of a biological apatite layer on ceramic surfaces. Ohgushi et al. , J. Biomed. Mater. Res. 1996

ECM production and Ca-P coating 1. The bone marrow cells formed more mineralized extracellular matrices on magnetron -sputtered Ca-P coating titanium than pure titanium. 2. The proliferation of the cells was significantly higher on the pure titanium than the coated titanium Hulshoff et al. , J. Biomed. Mater. Res. 1998

Cp titanium AH titanium Ap titanium Nashio et al. , J. Biomed. Mater. Res. 2000

Material and Methods 1. Commercially pure titanium (Cp Ti) disks 2. Alkali- and heat- treated titanium (AH Ti) disks 3. Apatite-formed titanium (Ap Ti) disks : produced by soaking the AH Ti disks in SBF. Nashio et al. , J. Biomed. Mater. Res. 2000

Results of Nashio et al. (2000) 1. The ALP activity of cells cultured on Ap Ti was significantly higher at both day 7, 14, than on other types of Ti. 2. The expression of osteocalcin m. RNA of cells on Ap Ti was highest, followed by on AH Ti, lastly on Cp Ti. 3. The expression of 1(I) collagen m. RNA of cells on Ap Ti was significantly higher at day 14 than on the other types of Ti. Nashio et al. , J. Biomed. Mater. Res. 2000

Conclusions of Nashio et al. (2000) 1. Apatite formed on titanium probably promotes adsorption of proteins from the serum, which are then subsequently utilized by the osteoblasts to form a mineralized extracellular matrix. 2. The difference in structure, composition, and quantity of calcium phosphate may affect the growth and differentiation of osteoblastic cells. Nashio et al. , J. Biomed. Mater. Res. 2000

Conclusions of Nashio et al. (2000) 3. Bonelike apatite-formed titanium after alkali and heat treatment provided the most favorable conditions for bone marrow cell differentiation. 4. At a later stage, alkali and heat treated titanium also provided favorable conditions, perhaps because of the calcium phosphate layer that formed on its surface. Nashio et al. , J. Biomed. Mater. Res. 2000

Bonding of alkali- and heat-treated tantalum implants to bone Hirofumi Kato, Takashi Nakamura, Shigeru Nishiguchi, Yoshitaka Matsusue, Masahiko Kobayashi, Toshiki Miyazaki, Hyun-Min Kim, Todashi Kokubo J. Biomed. Mater. Res. 2000
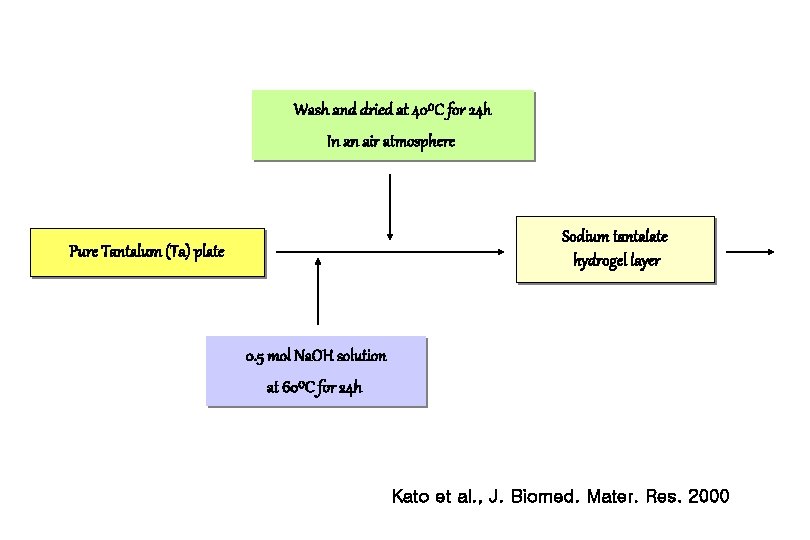
Wash and dried at 40⁰C for 24 h In an air atmosphere Sodium tantalate hydrogel layer Pure Tantalum (Ta) plate 0. 5 mol Na. OH solution at 60⁰C for 24 h Kato et al. , J. Biomed. Mater. Res. 2000
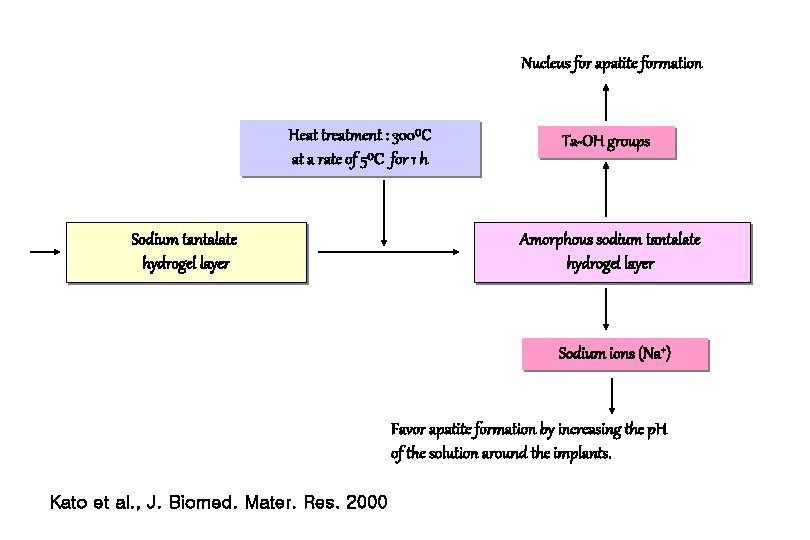
Nucleus for apatite formation Heat treatment : 300⁰C at a rate of 5⁰C for 1 h Sodium tantalate hydrogel layer Ta-OH groups Amorphous sodium tantalate hydrogel layer Sodium ions (Na+) Favor apatite formation by increasing the p. H of the solution around the implants. Kato et al. , J. Biomed. Mater. Res. 2000

3 possible reasons for the lower detaching failure load of AH tantalum 1. AH treated titanium is thought to give much higher apatite layer bonding strength than AH treated tantalum. 2. With titanium, no multinucleated macrophages were observed in the interface, while could occasionally be recognized in the bone-tantalum interface. 3. Difference in the concentrations of Na. OH solution : AH titanium : 5 mol Na. OH, AH tantalum : 0. 5 mol Na. OH Kato et al. , J. Biomed. Mater. Res. 2000

Bioactive tantalum metal prepared by Na. OH treatment Toshiki Miyazaki, Hyun-Min Kim, Fumiaki Miyaji, Todashi Kokubo, Hirofumi Kato, Takashi Nakamura J. Biomed. Mater. Res. 2000
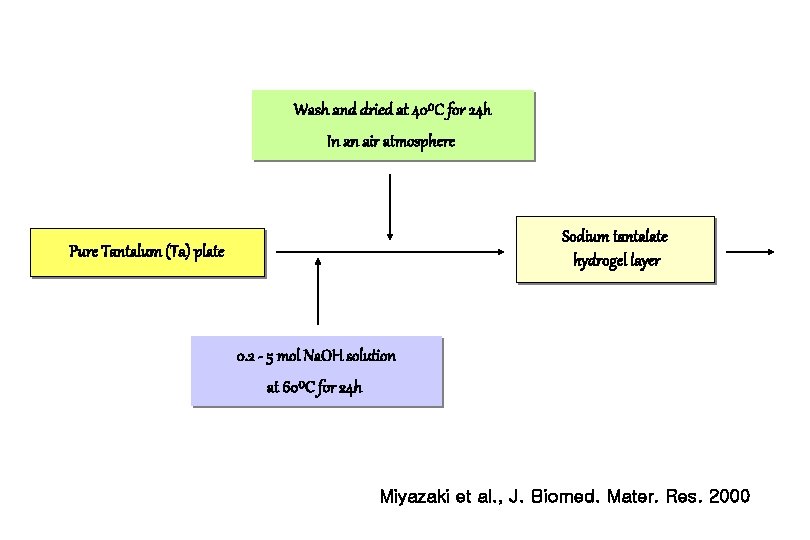
Wash and dried at 40⁰C for 24 h In an air atmosphere Sodium tantalate hydrogel layer Pure Tantalum (Ta) plate 0. 2 - 5 mol Na. OH solution at 60⁰C for 24 h Miyazaki et al. , J. Biomed. Mater. Res. 2000
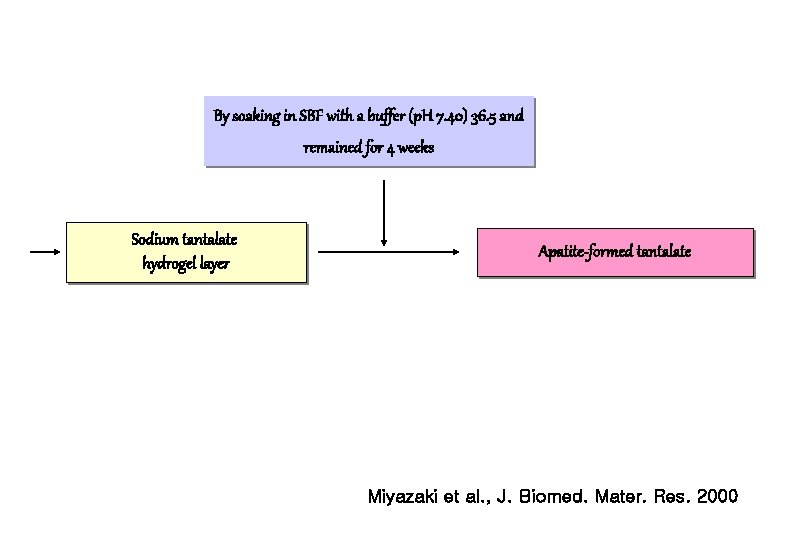
By soaking in SBF with a buffer (p. H 7. 40) 36. 5 and remained for 4 weeks Sodium tantalate hydrogel layer Apatite-formed tantalate Miyazaki et al. , J. Biomed. Mater. Res. 2000

Materials, Methods, Results 1. Untreated tantalum : covered with a thin tantalum oxide layer of less than 0. 1 m in thickness. 2. Treated with 0. 2 and 0. 5 M Na. OH solutions : Amorphous sodium tantalate layer about 1 m thick was formed. 3. Treated with 5. 0 M Na. OH solutions : Crystalline sodium tantalate layer was formed. Miyazaki et al. , J. Biomed. Mater. Res. 2000

Na. OH concentrations and Results 1. The tantalum metal treated with 5. 0 M Na. OH solutions does not form apatite on its surface in SBF. 2. The induction periods for apatite formation on the surfaces of the tantalum metals treated with 0. 2 -0. 5 M Na. OH solution were about 1 week. 3. For obtaining practically useful bioactive tantalum metal, hydrogel layer must be stabilized by a heat treatment. Miyazaki et al. , J. Biomed. Mater. Res. 2000
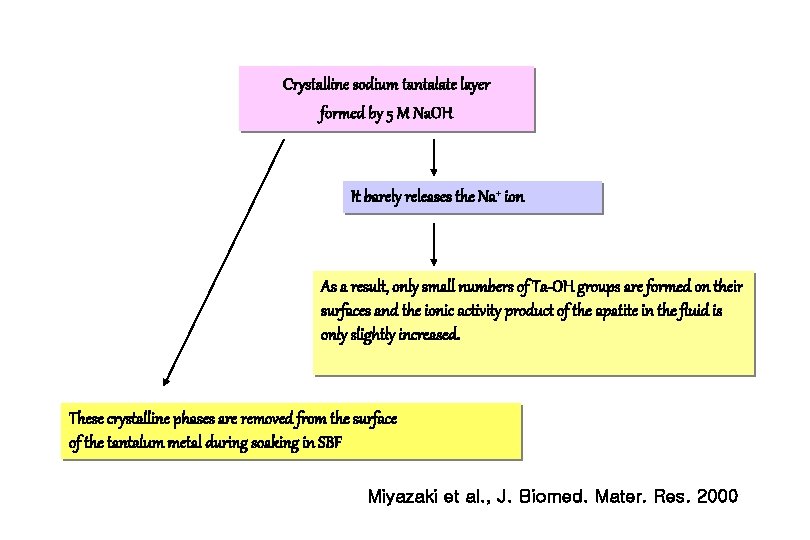
Crystalline sodium tantalate layer formed by 5 M Na. OH It barely releases the Na+ ion As a result, only small numbers of Ta-OH groups are formed on their surfaces and the ionic activity product of the apatite in the fluid is only slightly increased. These crystalline phases are removed from the surface of the tantalum metal during soaking in SBF Miyazaki et al. , J. Biomed. Mater. Res. 2000
- Slides: 99