HEMOSTAZ Yard Do Dr Murat RMEN Hemostaz Bleeding

HEMOSTAZ Yard. Doç. Dr. Murat ÖRMEN
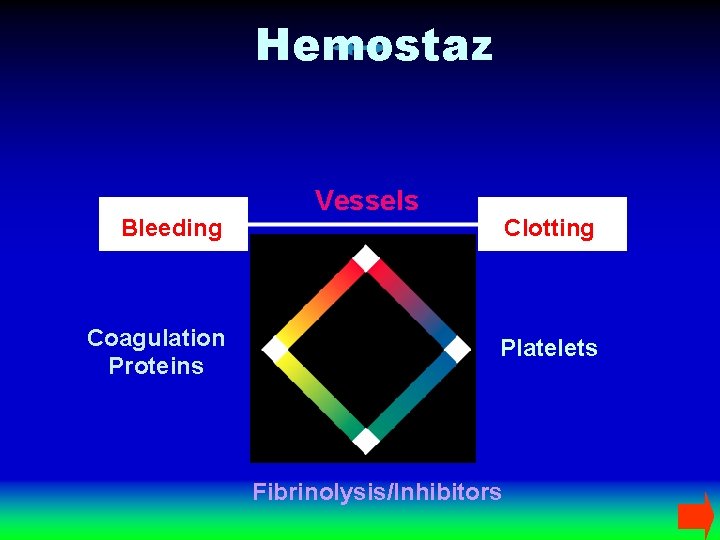
Hemostaz Bleeding Coagulation Proteins Vessels Clotting Platelets Fibrinolysis/Inhibitors
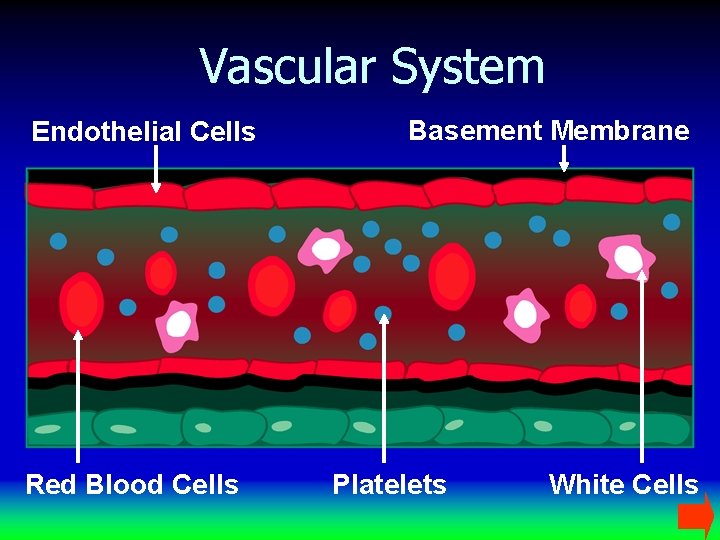
Vascular System Endothelial Cells Red Blood Cells Basement Membrane Platelets White Cells
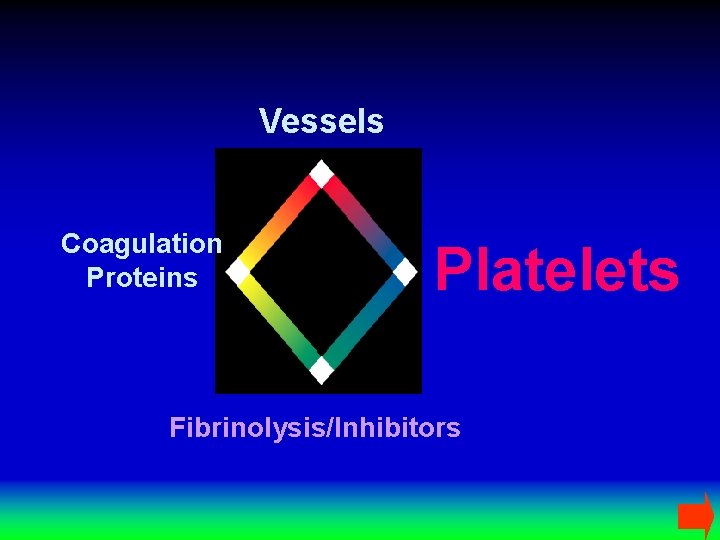
Vessels Coagulation Proteins Platelets Fibrinolysis/Inhibitors
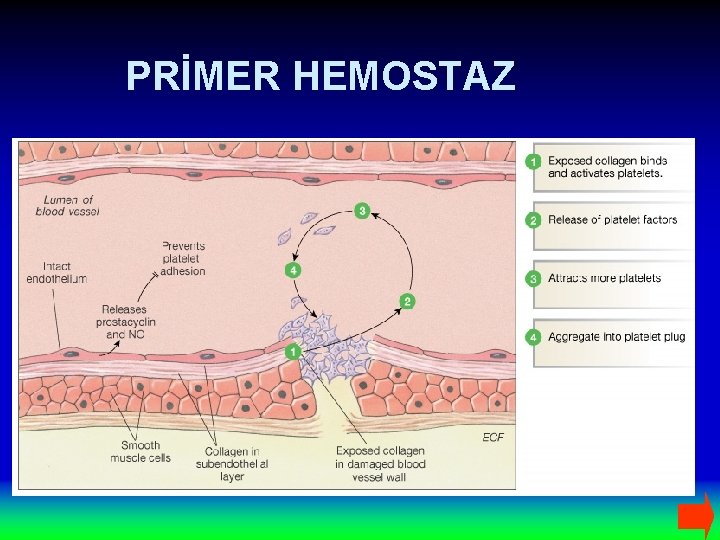
PRİMER HEMOSTAZ

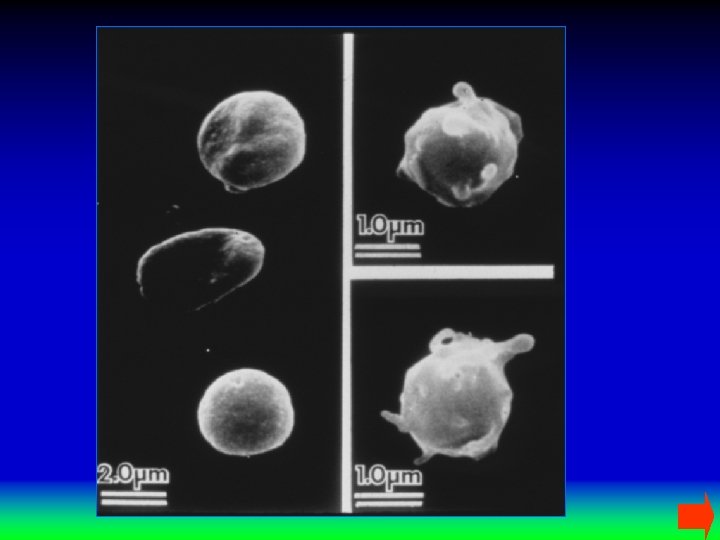

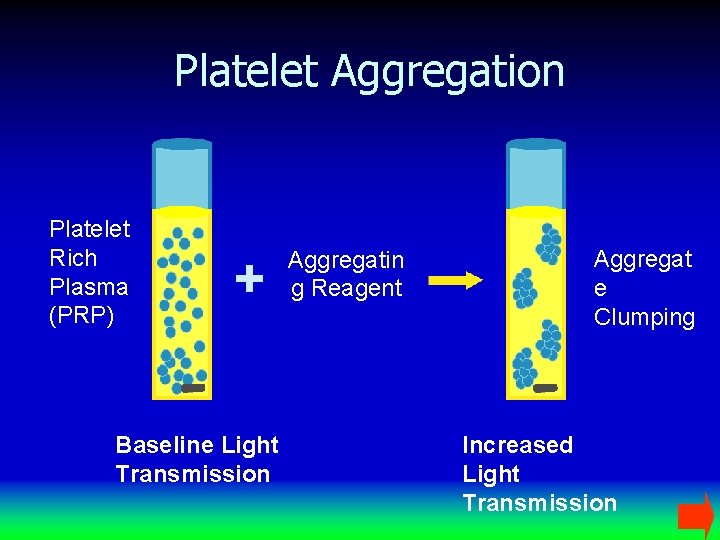
Platelet Aggregation Platelet Rich Plasma (PRP) + Baseline Light Transmission Aggregatin g Reagent Aggregat e Clumping Increased Light Transmission

Function HEMOSTASİS BLEEDİNG AGREGOMETRİ ANİMASYON
Vessels Coagulation Proteins Fibrinolysis/Inhibitors Platelets
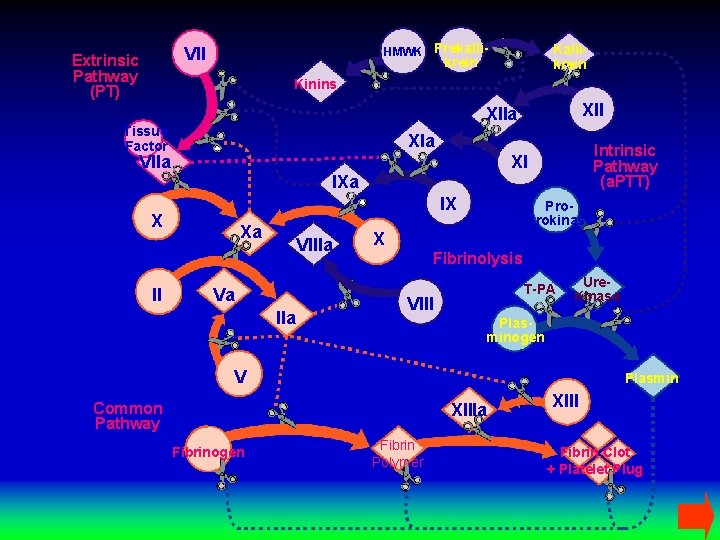
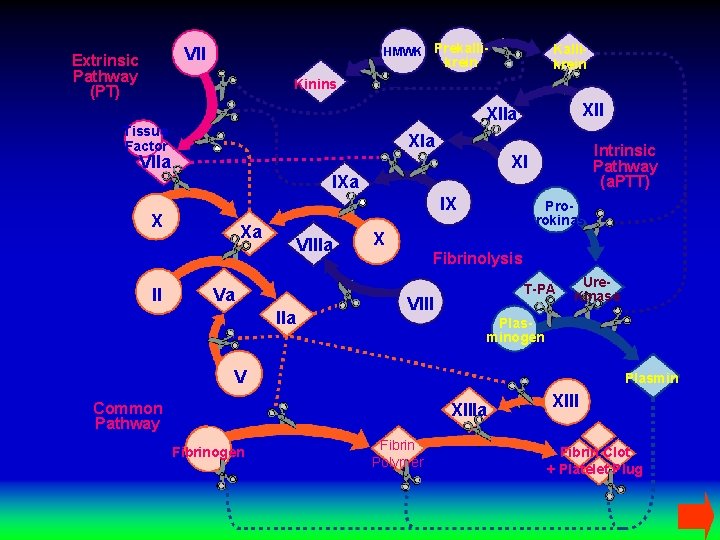
HMWK Prekalli- VII Extrinsic Pathway (PT) Kallikrein Kinins Tissue Factor XIa Intrinsic Pathway (a. PTT) XI VIIa IX X II XIIa Xa VIIIa Va IIa Pro. Urokinase X Fibrinolysis T-PA VIII Ure. Kinase Plasminogen V Plasmin Common Pathway XIIIa Fibrinogen Fibrin Polymer XIII Fibrin Clot + Platelet Plug
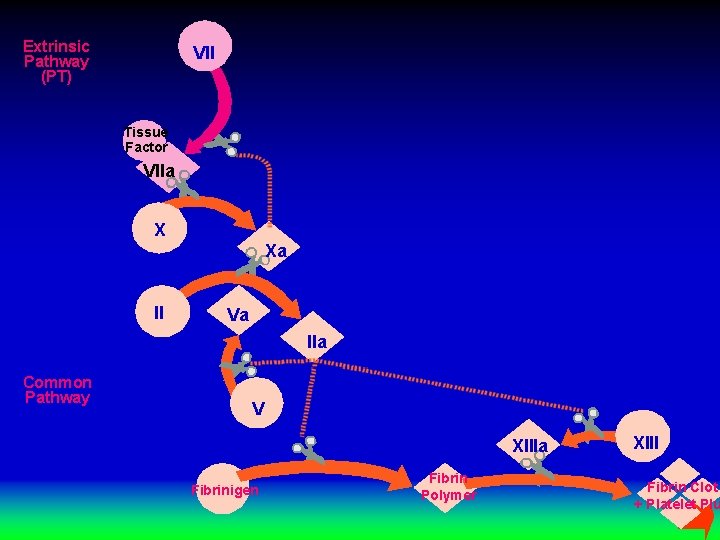
Extrinsic Pathway (PT) VII Tissue Factor VIIa X Xa II Va IIa Common Pathway V XIIIa Fibrinigen Fibrin Polymer XIII Fibrin Clot + Platelet Plu
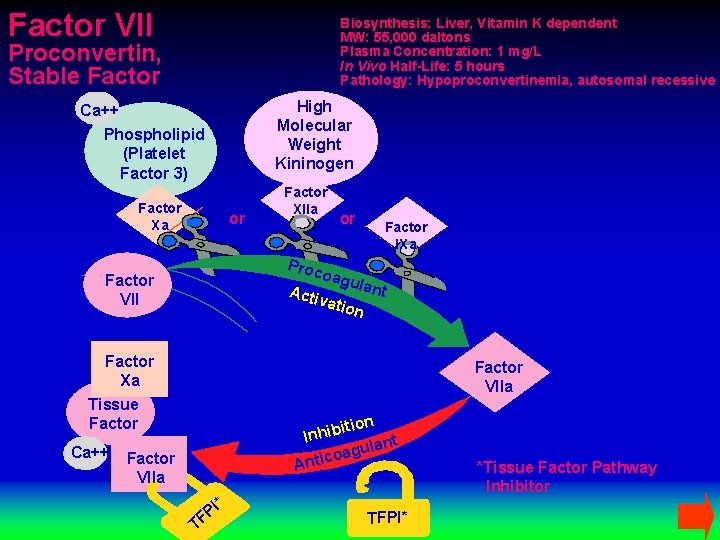
Factor VII Biosynthesis: Liver, Vitamin K dependent MW: 55, 000 daltons Plasma Concentration: 1 mg/L In Vivo Half-Life: 5 hours Pathology: Hypoproconvertinemia, autosomal recessive Proconvertin, Stable Factor High Molecular Weight Kininogen Ca++ Phospholipid (Platelet Factor 3) Factor Xa or Factor XIIa or Proc o Factor VII Acti Factor IXa agul vatio ant n Factor Xa Factor VIIa Tissue Factor Ca++ ition b i h In lant u g a o Antic Factor VIIa * I P TF TFPI* *Tissue Factor Pathway Inhibitor
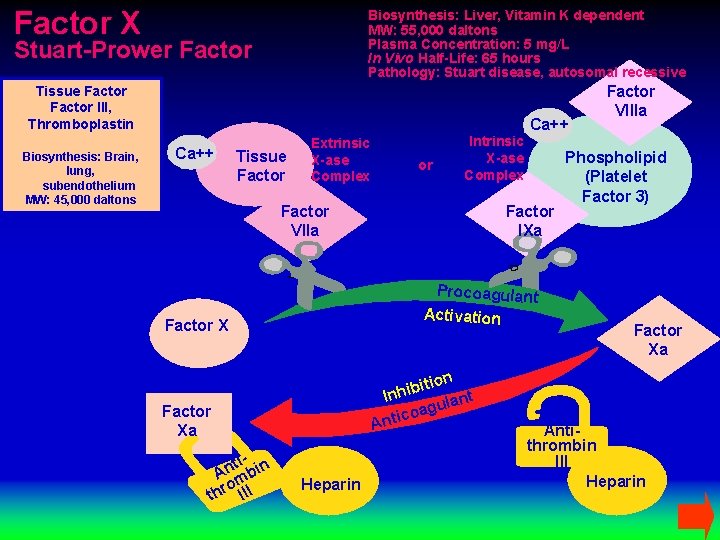
Factor X Biosynthesis: Liver, Vitamin K dependent MW: 55, 000 daltons Plasma Concentration: 5 mg/L In Vivo Half-Life: 65 hours Pathology: Stuart disease, autosomal recessive Stuart-Prower Factor Tissue Factor III, Thromboplastin Biosynthesis: Brain, lung, subendothelium MW: 45, 000 daltons Ca++ Tissue Factor Extrinsic X-ase Complex or Intrinsic X-ase Complex Factor VIIa Factor IXa Procoagulant Activation Factor Xa ti- n An mbi o thr III A Heparin ition b i h In ant l u g a ntico Factor VIIIa Phospholipid (Platelet Factor 3) Factor Xa Antithrombin III Heparin
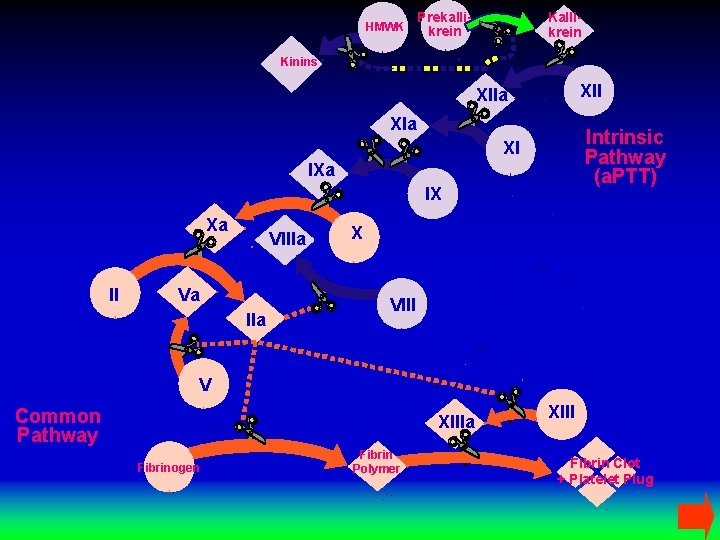
HMWK Kallikrein Prekallikrein Kinins XIIa XIa Intrinsic Pathway (a. PTT) XI IXa IX Xa II VIIIa Va IIa X VIII V Common Pathway XIIIa Fibrinogen Fibrin Polymer XIII Fibrin Clot + Platelet Plug
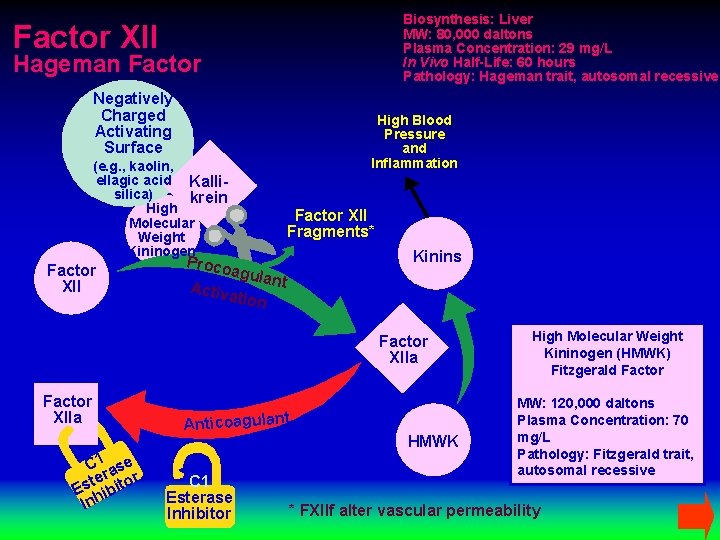
Biosynthesis: Liver MW: 80, 000 daltons Plasma Concentration: 29 mg/L In Vivo Half-Life: 60 hours Pathology: Hageman trait, autosomal recessive Factor XII Hageman Factor Negatively Charged Activating Surface (e. g. , kaolin, ellagic acid Kallisilica) krein High Molecular Weight Kininogen Factor XII High Blood Pressure and Inflammation Factor XII Fragments* Kinins Proco agula nt Activ ation Factor XIIa C 1 ase r ter Es hibito In Inhibition Anticoagulant C 1 Esterase Inhibitor HMWK High Molecular Weight Kininogen (HMWK) Fitzgerald Factor MW: 120, 000 daltons Plasma Concentration: 70 mg/L Pathology: Fitzgerald trait, autosomal recessive * FXIIf alter vascular permeability
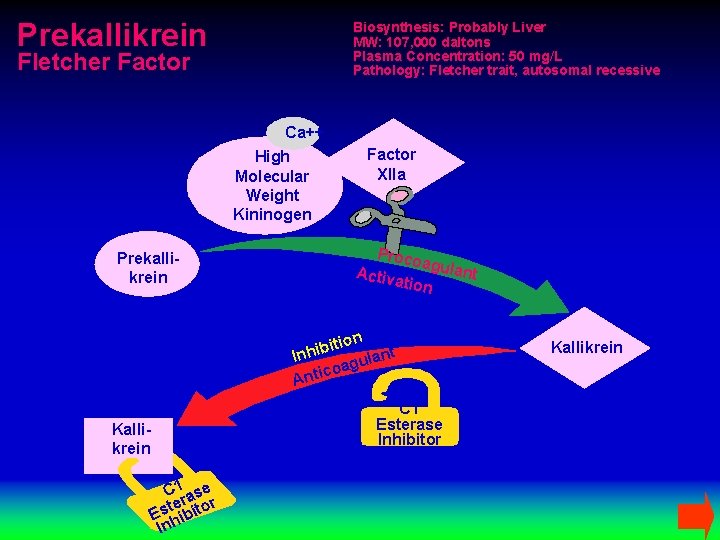
Prekallikrein Biosynthesis: Probably Liver MW: 107, 000 daltons Plasma Concentration: 50 mg/L Pathology: Fletcher trait, autosomal recessive Fletcher Factor Ca++ High Molecular Weight Kininogen Prekallikrein Factor XIIa Proco agula nt Activ ation ition b i h In ant l u g coa Anti C 1 Esterase Inhibitor Kallikrein C 1 ase ter itor s E hib In Kallikrein
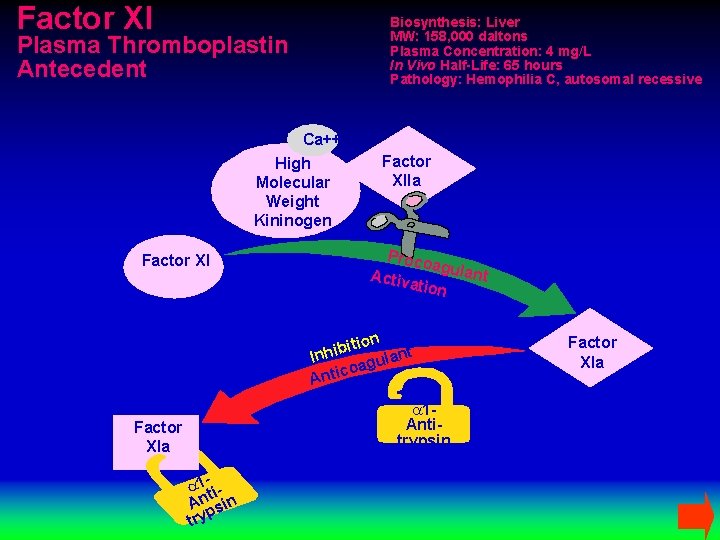
Factor XI Biosynthesis: Liver MW: 158, 000 daltons Plasma Concentration: 4 mg/L In Vivo Half-Life: 65 hours Pathology: Hemophilia C, autosomal recessive Plasma Thromboplastin Antecedent Ca++ High Molecular Weight Kininogen Factor XIIa Proco agula nt Activ ation ition b i h In lant u g a co Anti a 1 Antitrypsin Factor XIa a 1 ti. An sin p try Factor XIa
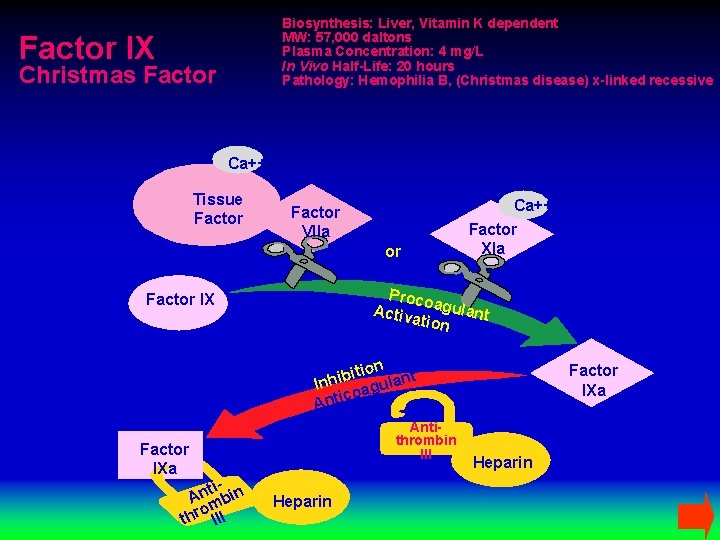
Biosynthesis: Liver, Vitamin K dependent MW: 57, 000 daltons Plasma Concentration: 4 mg/L In Vivo Half-Life: 20 hours Pathology: Hemophilia B, (Christmas disease) x-linked recessive Factor IX Christmas Factor Ca++ Tissue Factor VIIa or Ca++ Factor XIa Proco Activ agulant ation Factor IX tion nt i b i Inh agula co Antithrombin III Factor IXa ti. An mbin o thr III Heparin Factor IXa Heparin
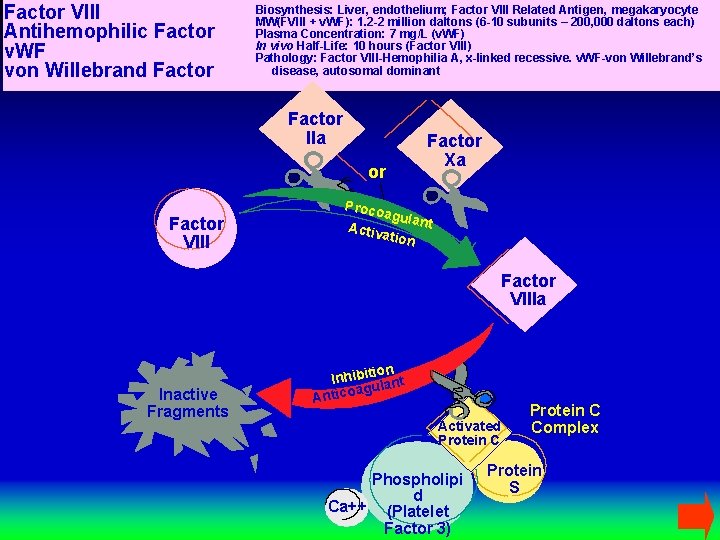
Factor VIII Antihemophilic Factor v. WF von Willebrand Factor Biosynthesis: Liver, endothelium; Factor VIII Related Antigen, megakaryocyte MW(FVIII + v. WF): 1. 2 -2 million daltons (6 -10 subunits – 200, 000 daltons each) Plasma Concentration: 7 mg/L (v. WF) In vivo Half-Life: 10 hours (Factor VIII) Pathology: Factor VIII-Hemophilia A, x-linked recessive. v. WF-von Willebrand’s disease, autosomal dominant Factor IIa or Factor Xa Factor VIII Proco Activ agula ation nt Factor VIIIa Inactive Fragments ion Inhibit lant agu Antico Activated Protein C Phospholipi d Ca++ (Platelet Factor 3) Protein C Complex Protein S
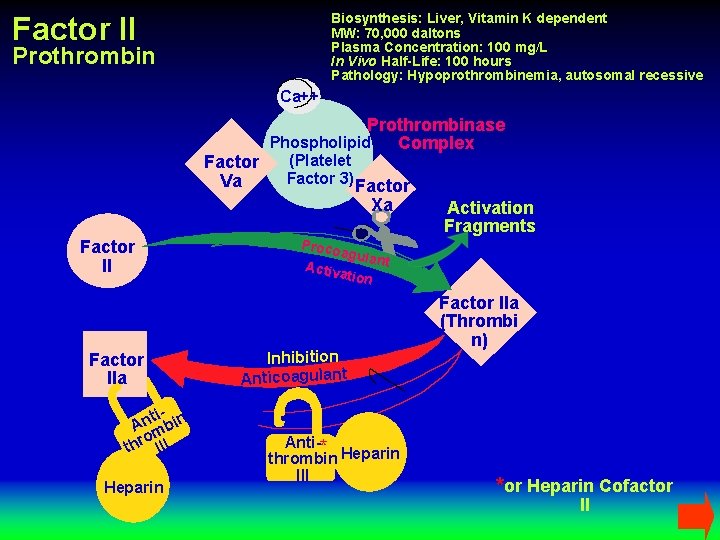
Biosynthesis: Liver, Vitamin K dependent MW: 70, 000 daltons Plasma Concentration: 100 mg/L In Vivo Half-Life: 100 hours Pathology: Hypoprothrombinemia, autosomal recessive Factor II Prothrombin Ca++ Factor Va Factor IIa ti- in n A mb o thr III Heparin Prothrombinase Phospholipid Complex (Platelet Factor 3) Factor Xa Activation Fragments Proco agula nt Activ ation Inhibition Anticoagulant Anti-* thrombin Heparin III Factor IIa (Thrombi n) *or Heparin Cofactor II
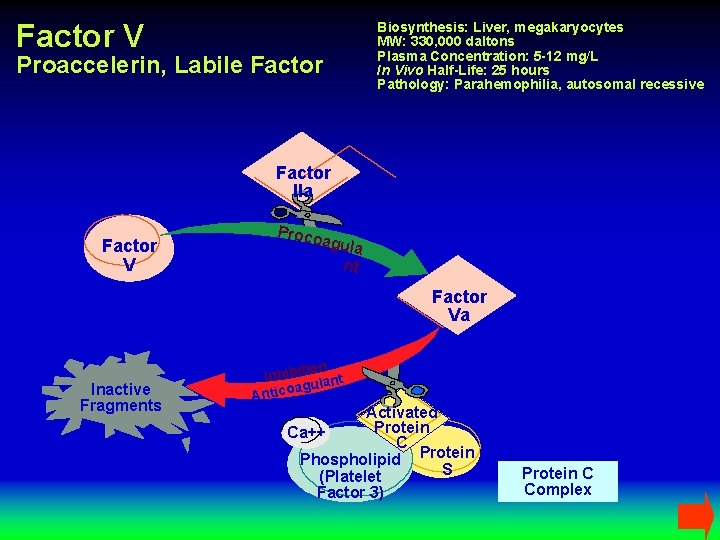
Factor V Biosynthesis: Liver, megakaryocytes MW: 330, 000 daltons Plasma Concentration: 5 -12 mg/L In Vivo Half-Life: 25 hours Pathology: Parahemophilia, autosomal recessive Proaccelerin, Labile Factor IIa Factor V Proco Activ agula ation n t Factor Va Inactive Fragments ion Inhibit lant agu Antico Activated Protein Ca++ C Phospholipid Protein S (Platelet Factor 3) Protein C Complex
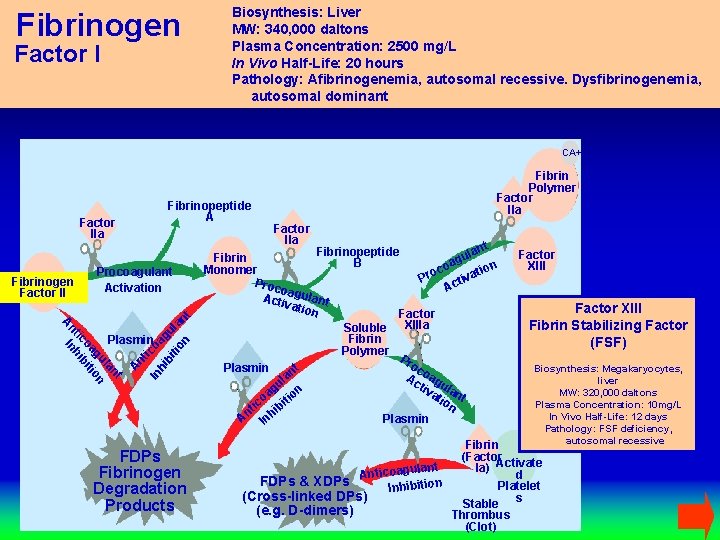
Fibrinogen Factor I Biosynthesis: Liver MW: 340, 000 daltons Plasma Concentration: 2500 mg/L In Vivo Half-Life: 20 hours Pathology: Afibrinogenemia, autosomal recessive. Dysfibrinogenemia, autosomal dominant CA+ Fibrinopeptide A Factor IIa Procoagulant Activation nt la gu on oa iti ic ib nt Inh A ic oa hi bi gu tio la n n t Fibrinogen Factor II In A nt Plasmin FDPs Fibrinogen Degradation Products Fibrin Polymer Factor IIa t Fibrinopeptide lan u Fibrin B ag on co Monomer o ati r v P i t Proc Ac oagu la Acti vati nt on Factor Soluble XIIIa Fibrin Polymer Pr oc t Plasmin n a Ac oag l tiv ula gu n a ati nt o i o on ic ibit t n h A In Plasmin Factor XIII Fibrin Stabilizing Factor (FSF) Biosynthesis: Megakaryocytes, liver MW: 320, 000 daltons Plasma Concentration: 10 mg/L In Vivo Half-Life: 12 days Pathology: FSF deficiency, autosomal recessive Fibrin (Factor Ia) Activate lant d Anticoagu FDPs & XDPs Platelet Inhibition (Cross-linked DPs) s Stable (e. g. D-dimers) Thrombus (Clot)
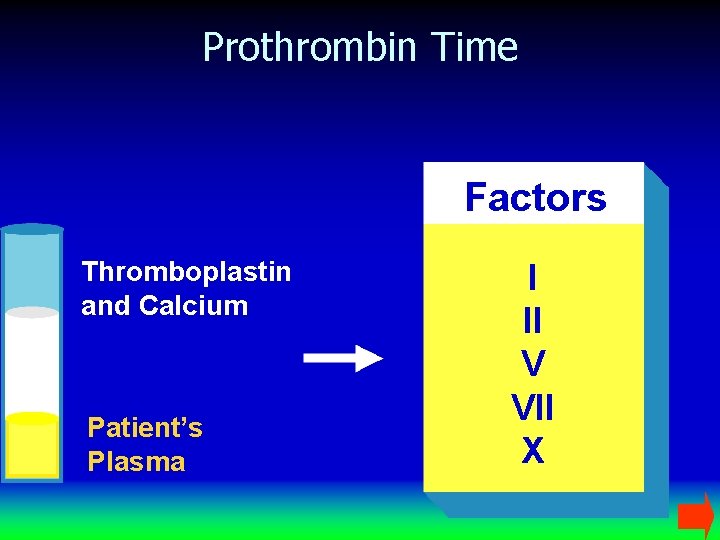
Prothrombin Time Factors Thromboplastin and Calcium Patient’s Plasma I II V VII X
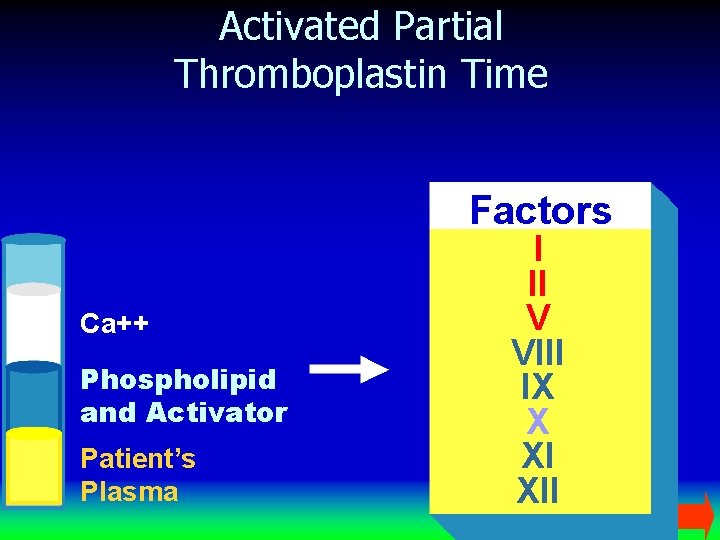
Activated Partial Thromboplastin Time Factors Ca++ Phospholipid and Activator Patient’s Plasma I II V VIII IX X XI XII
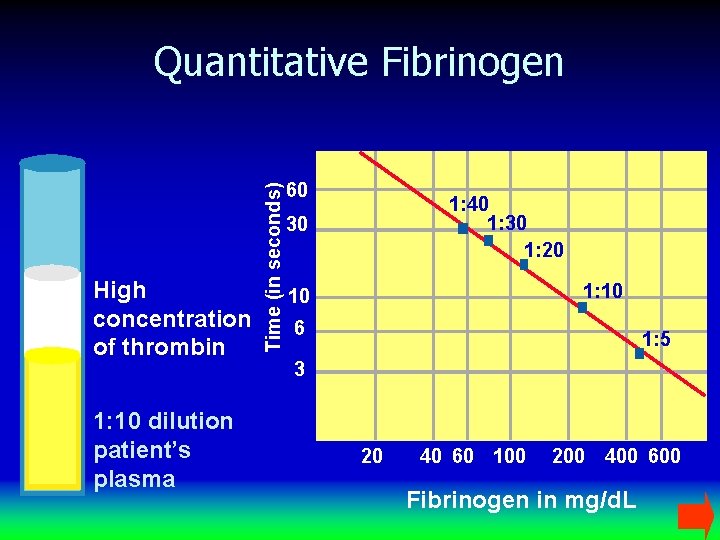
High concentration of thrombin 1: 10 dilution patient’s plasma Time (in seconds) Quantitative Fibrinogen 60 1: 40 1: 30 1: 20 30 1: 10 10 6 1: 5 3 20 40 60 100 200 400 600 Fibrinogen in mg/d. L
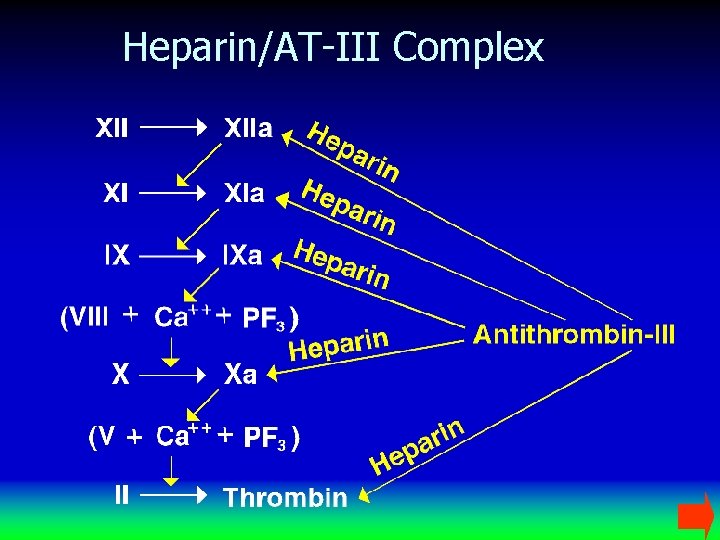
Heparin/AT-III Complex
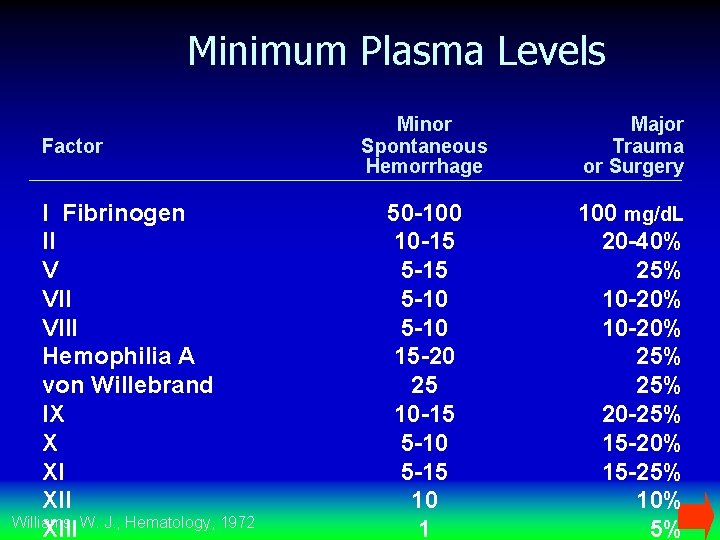
Minimum Plasma Levels Factor I Fibrinogen II V VIII Hemophilia A von Willebrand IX X XI XII Williams, W. J. , Hematology, 1972 XIII Minor Spontaneous Hemorrhage Major Trauma or Surgery 50 -100 10 -15 5 -10 15 -20 25 10 -15 5 -10 5 -15 10 1 100 mg/d. L 20 -40% 25% 10 -20% 25% 20 -25% 15 -20% 15 -25% 10% 5%
Vessels Coagulation Proteins Platelets Fibrinolysis/Inhibitors
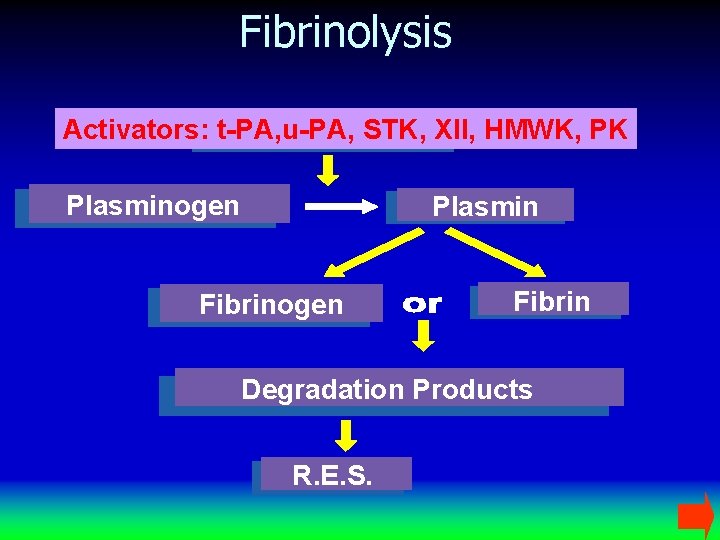
Fibrinolysis Activators: t-PA, u-PA, STK, XII, HMWK, PK Plasminogen Plasmin Fibrinogen Fibrin Degradation Products R. E. S.
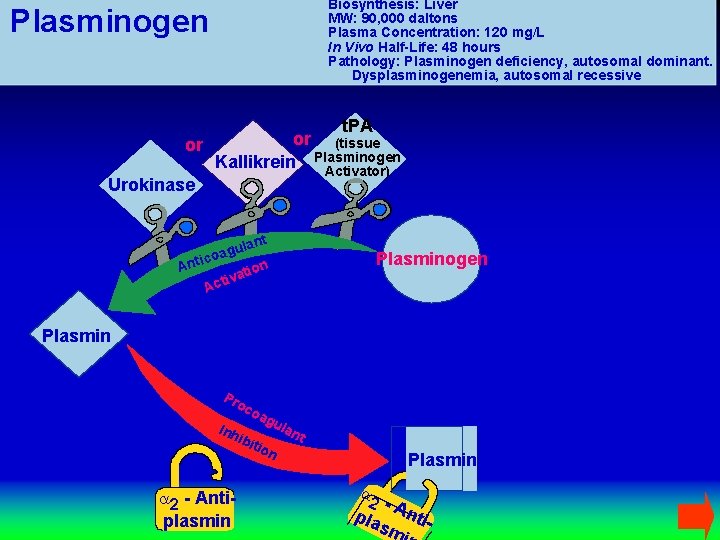
Biosynthesis: Liver MW: 90, 000 daltons Plasma Concentration: 120 mg/L In Vivo Half-Life: 48 hours Pathology: Plasminogen deficiency, autosomal dominant. Dysplasminogenemia, autosomal recessive Plasminogen t. PA or (tissue or Kallikrein Plasminogen Activator) Urokinase t ulan g a ico Ant tion a v i Act Plasminogen Plasmin Pro c oa gu lan Inh t ibi tio n a 2 - Antiplasmin Plasmin a 2 -A pla ntism
HMWK Prekalli- VII Extrinsic Pathway (PT) Kallikrein Kinins Tissue Factor XIa Intrinsic Pathway (a. PTT) XI VIIa IX X II XIIa Xa VIIIa Va IIa Pro. Urokinase X Fibrinolysis T-PA VIII Ure. Kinase Plasminogen V Plasmin Common Pathway XIIIa Fibrinogen Fibrin Polymer XIII Fibrin Clot + Platelet Plug
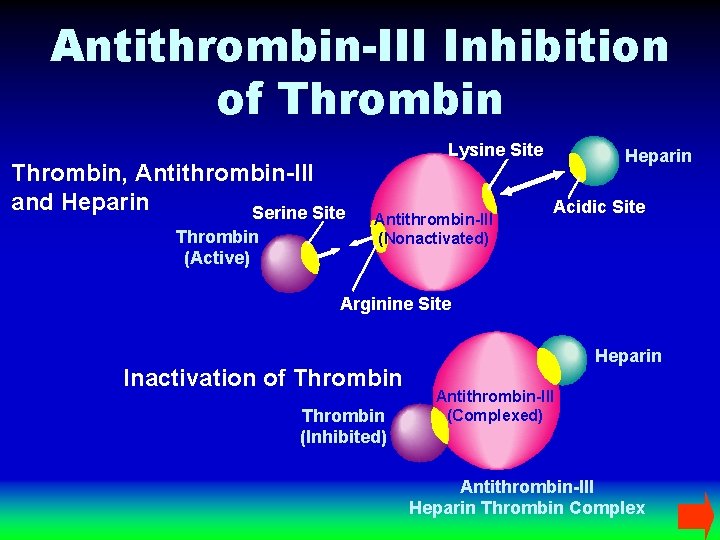
Antithrombin-III Inhibition of Thrombin, Antithrombin-III and Heparin Serine Site Thrombin (Active) Lysine Site Antithrombin-III (Nonactivated) Heparin Acidic Site Arginine Site Inactivation of Thrombin (Inhibited) Heparin Antithrombin-III (Complexed) Antithrombin-III Heparin Thrombin Complex

Antithrombin-III Decreased Levels 1. Congenital 2. Acquired – decreased synthesis 3. Acquired – increased utilization 4. Drug-induced

Protein C • Vitamin K-dependent plasma protein • Inactivates Factors V and VIII • Stimulates fibrinolysis
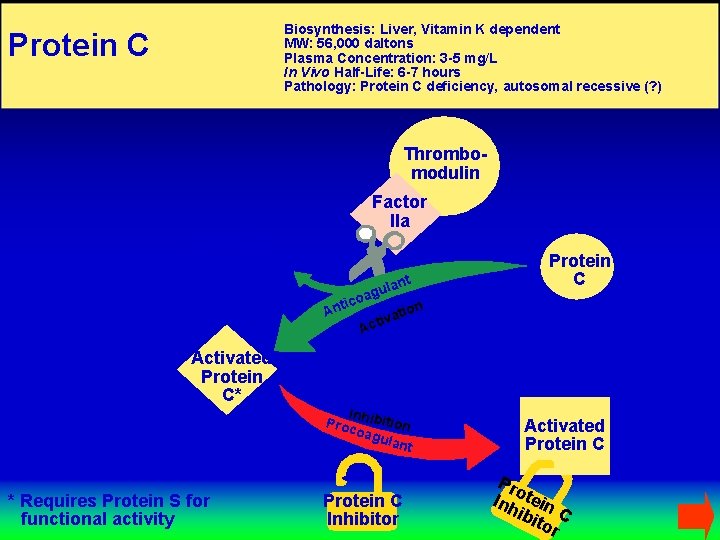
Biosynthesis: Liver, Vitamin K dependent MW: 56, 000 daltons Plasma Concentration: 3 -5 mg/L In Vivo Half-Life: 6 -7 hours Pathology: Protein C deficiency, autosomal recessive (? ) Protein C Thrombomodulin Factor IIa Protein C Activation Peptide ant ic ul oag Ant Protein C tion a v i Activated Protein C* In Proc hibition oag ulan t * Requires Protein S for functional activity Protein C Inhibitor Activated Protein C Pro Inh tein ibi C tor

Deficiencies of Protein C I. Congenital Hereditary autosomal dominant II. Acquired A. DIC B. Liver disease C. During post-operative period D. Anticoagulant therapy

Clinical Manifestations • Superficial thrombophlebitis • Venous thromboses in adolescents or young adults • Arterial thromboses rarely observed • Skin necrosis during onset of oral anticoagulant therapy

Protein S • Cofactor for Protein C • Vitamin K-dependent protein • Enhances binding of Protein C to phospholipid surfaces


- Slides: 42