Hemostatic Balance Coagulation Fibrinolysis Inflammation InfectionTrauma Fetal DevelopmentPregnancy

Hemostatic Balance Coagulation & Fibrinolysis Inflammation Infection/Trauma Fetal Development/Pregnancy Loss

Overview Introduction Platelet Plug Formation Fibrin Clot Formation Fibrinolysis Antiplatelet & Anticoagulant Drugs Disorders of Hemostatic Balance Resources

Hemostatic Balance Coagulation Fibrinolysis Pro-coagulant Forces Anti-coagulant Forces
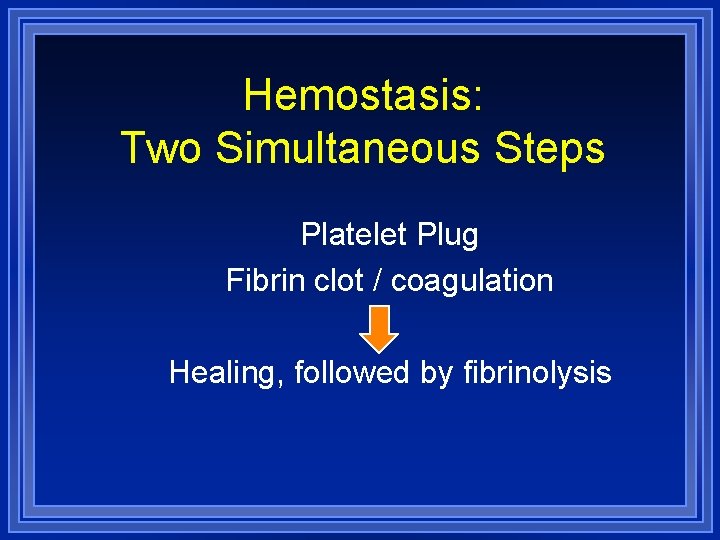
Hemostasis: Two Simultaneous Steps Platelet Plug Fibrin clot / coagulation Healing, followed by fibrinolysis
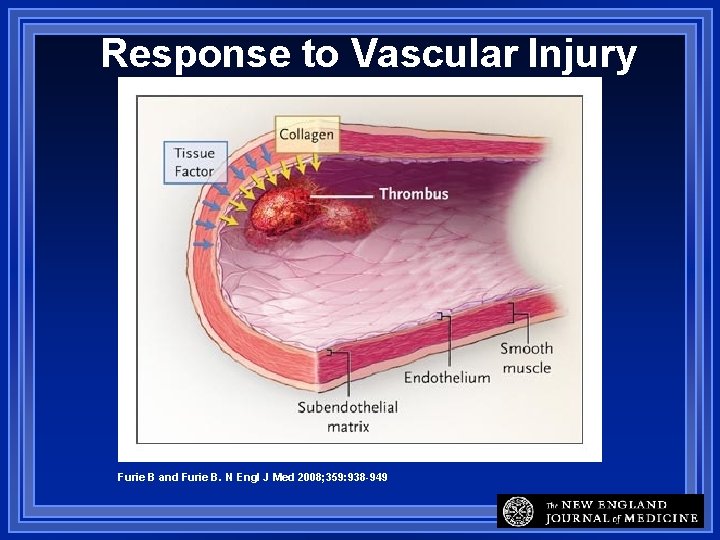
Response to Vascular Injury Furie B and Furie B. N Engl J Med 2008; 359: 938 -949

Coagulation & Fibrinolysis Formation of platelet plug Binding to collagen / v. WF Activation & secretion Aggregation Formation of fibrin clot – coagulation Tissue factor – key initiator Phases of coagulation Initiation, Propagation, Termination Fibrinolysis - elimination of clot
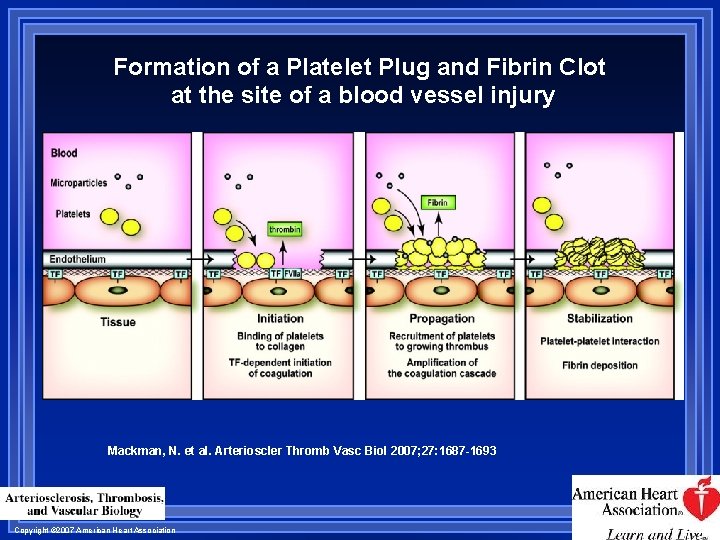
Formation of a Platelet Plug and Fibrin Clot at the site of a blood vessel injury Mackman, N. et al. Arterioscler Thromb Vasc Biol 2007; 27: 1687 -1693 Copyright © 2007 American Heart Association

Hemostatic System Components Platelets Plasma proteins Procoagulant Anticoagulant Fibrinolytic present as zymogens, serine proteases Vascular endothelial cells, leukocytes

Platelet Plug Formation
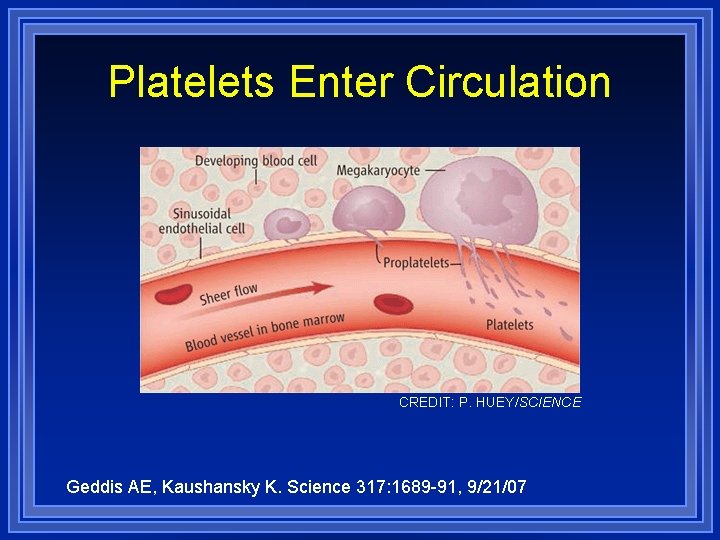
Platelets Enter Circulation CREDIT: P. HUEY/SCIENCE Geddis AE, Kaushansky K. Science 317: 1689 -91, 9/21/07

Platelets are important in: Hemostasis - primary focus today Inflammation Help neutrophils, monocytes & Th 1 cells attach to endothelium at injury sites - called “secondary capture”; also activates them Secrete cytokines & chemokines Wound healing Secrete growth factors such as PDGF, VEGF, b. FGF Zarblock A et al. Blood Reviews 21: 99, 2007

Platelets Circulate in resting, inactive form Activation secretion expression of surface proteins & receptors increased receptor affinity adhering to surfaces aggregate formation Provide surface for clotting cascade activity GPIIb/IIIa integrin receptor binds fibrinogen, von Willebrand factor & other ligands Aggrastat, Integrilin, Repro (abciximab)
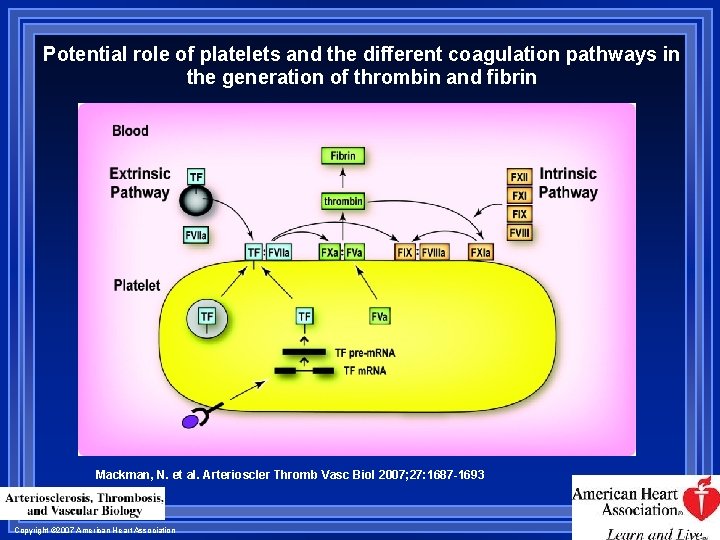
Potential role of platelets and the different coagulation pathways in the generation of thrombin and fibrin Mackman, N. et al. Arterioscler Thromb Vasc Biol 2007; 27: 1687 -1693 Copyright © 2007 American Heart Association

Formation of Platelet Plug Adhesion Weak binding to collagen, easily detached Binding to von Willebrand factor • Much stronger attachment Activation & secretion Platelets change shape, secrete bioactive molecules Express new surface receptors Aggregation via fibrinogen/receptors

von Willebrand Factor (v. WF) Functions: adhesion to damaged bv & other platelets Protects FVIII from proteolysis Structure: glycoprotein Storage in 2 locations α-granules of platelets Weibel-Palade bodies of endothelial cells Increased with stress, pregnancy, surgery
Platelet Pictures http: //www. platelet-research. org/1/function_hemo. htm#

Vascular Injury results in Vasoconstriction Formation of platelet plug Generation of fibrin clot Tissue Factor is exposed Deposition of WBCs in injury area Initiation of inflammation & repair Fibrinolysis

Vascular Endothelium Expresses adhesion molecules when stimulated, selectins & integrins t-PA = tissue plasminogen activator Thrombomodulin - when binds thrombin Protein C is activated Endothelial protein C receptor (EPCR) Does not usually express tissue factor Damage exposes sub-endothelial tissue that always has tissue factor

Platelets – Vascular Integrity In post-capillary venules release trophogens that help endothelial cells stay attached to each other • VEGF-A = key trophogen Thrombocytopenia • 10, 000 - 20, 000 platlets/mm 3 • Post-capillary venules allow RBCs to leak out • Result = petechiae in skin, mucous membranes • Illustrated in next 3 slides
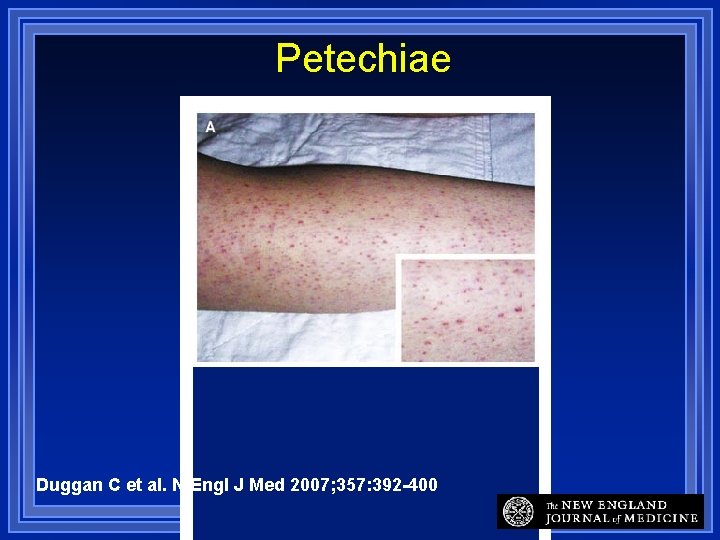
Petechiae Duggan C et al. N Engl J Med 2007; 357: 392 -400
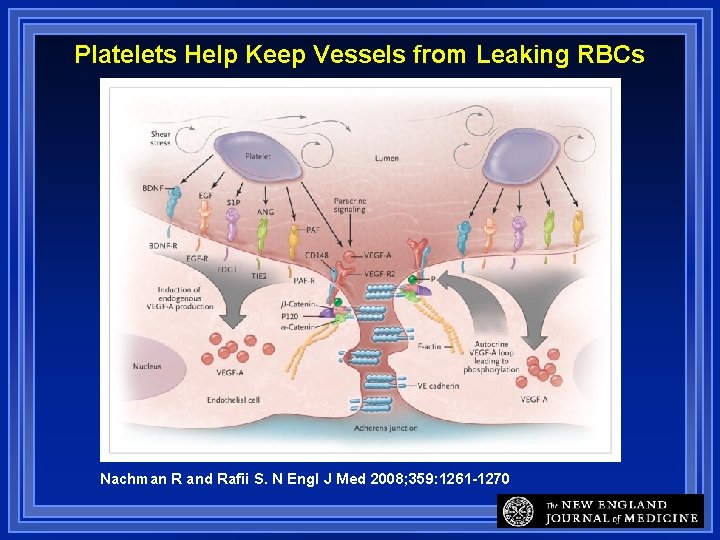
Platelets Help Keep Vessels from Leaking RBCs Nachman R and Rafii S. N Engl J Med 2008; 359: 1261 -1270
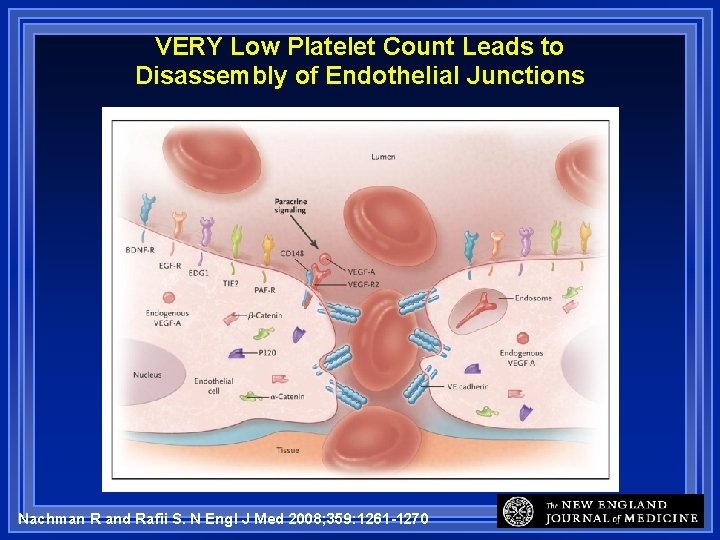
VERY Low Platelet Count Leads to Disassembly of Endothelial Junctions Nachman R and Rafii S. N Engl J Med 2008; 359: 1261 -1270

Formation of Fibrin Clot Coagulation
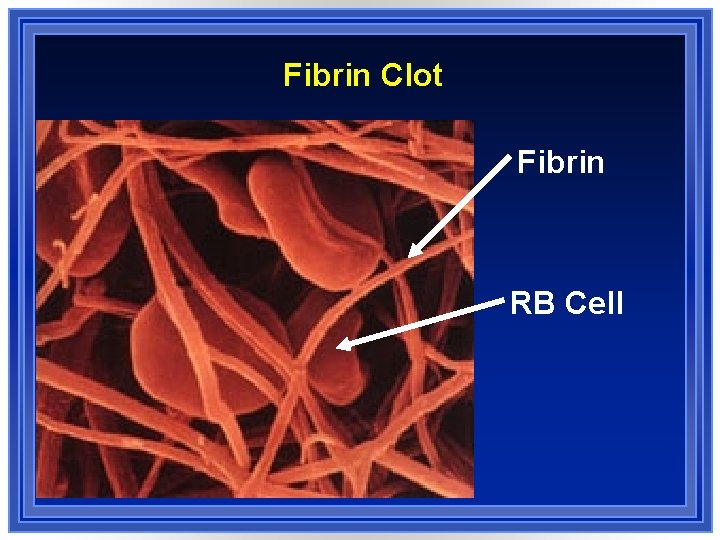
Fibrin Clot Fibrin RB Cell

Blood Clotting Proteins 3 categories Procoagulant Anti-coagulant Fibrinolytic Made in liver, some need Vit K for synthesis Proenzymes, procofactors or regulatory Require activation

Models of Clotting Cascades Cascade Model Suggests two alternate pathways for activation of clotting - extrinsic & intrinsic Still presented as THE clotting cascade in texts but this model is not supported by current data Cell-based Model Based on current data Clotting initiated through Tissue Factor pathway Intrinsic pathway may amplify initial response

Cascade Model of Coagulation 2 distinct pathways, intrinsic & extrinsic BUT Intrinsic occurs in test tube probably not in body Intrinsic - function measured by Activated partial thromboplastin time (a. PTT or PTT) • Normal is 25 - 40 seconds Extrinsic - Prothrombin time (PT) & International Normalized Ratio (INR) Cascade Model has some usefulness to understand clot formation in vitro for laboratory monitoring of anticoagulation differential diagnosis of coagulation disorders
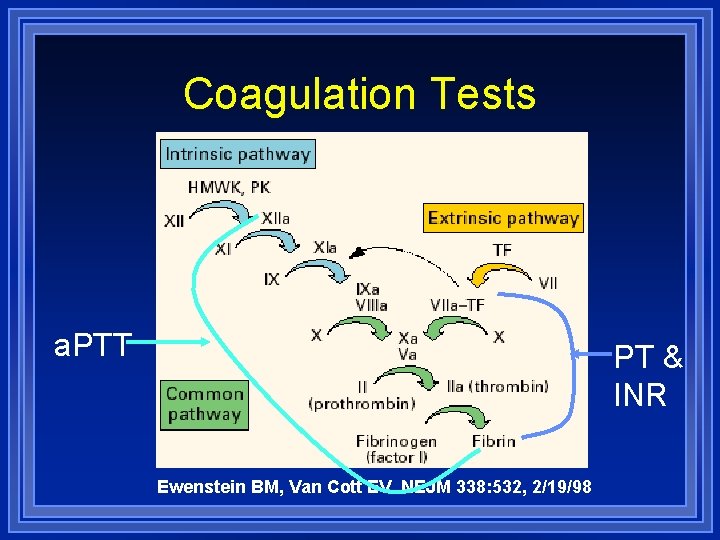
Coagulation Tests a. PTT PT & INR Ewenstein BM, Van Cott EV. NEJM 338: 532, 2/19/98

INR International Normalized Ratio Standardizes PT ratio worldwide • Normal PT value is 11. 5 - 14 seconds • Needed - variability in test reagents Normal value = 1 - 1. 4 Clinical use - titrate warfarin (Coumadin) For example, goals of Rx • INR = 2 - 3 venous thrombosis etc • INR = 2. 5 - 3. 5 mechanical heart valve
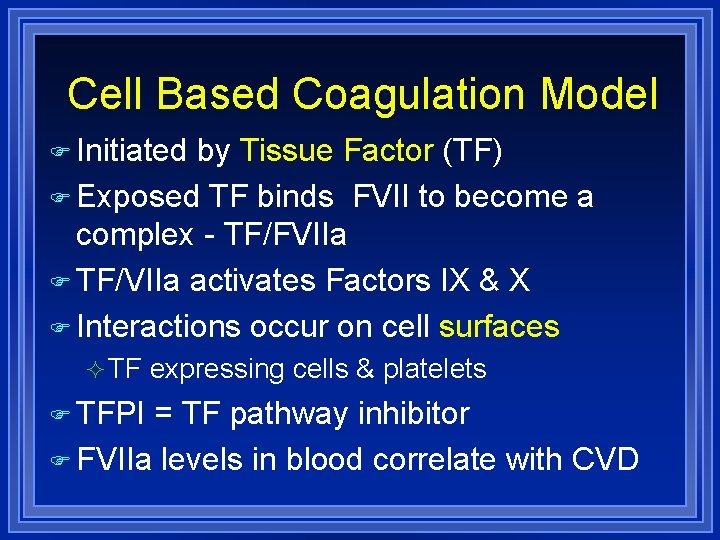
Cell Based Coagulation Model Initiated by Tissue Factor (TF) Exposed TF binds FVII to become a complex - TF/FVIIa TF/VIIa activates Factors IX & X Interactions occur on cell surfaces TF expressing cells & platelets TFPI = TF pathway inhibitor FVIIa levels in blood correlate with CVD
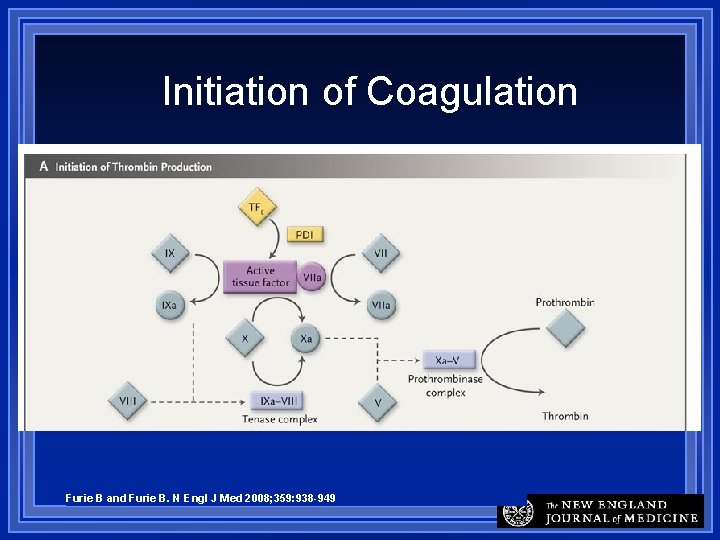
Initiation of Coagulation Furie B and Furie B. N Engl J Med 2008; 359: 938 -949
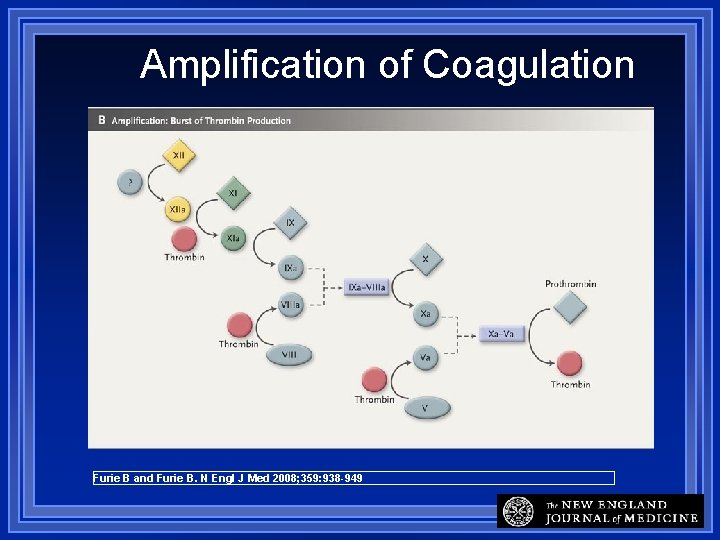
Amplification of Coagulation Furie B and Furie B. N Engl J Med 2008; 359: 938 -949
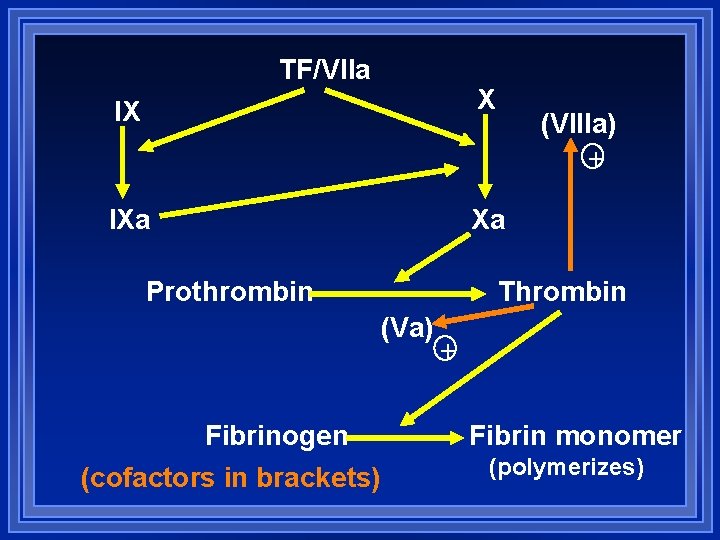
TF/VIIa X IX (VIIIa) IXa Xa Prothrombin Thrombin (Va) Fibrinogen (cofactors in brackets) Fibrin monomer (polymerizes)

More on Cell-Based Model Once formed thrombin activates platelets accelerates clot formation converts fibrinogen to fibrin network or mesh activates anticoagulation influences inflammation (more later) Fibrinogen fibrin monomer Fibrin monomers polymerize into fibrin strands (requires FXIIIa to stabilize)

Fibrinogen Synthesized in liver, 1. 7 - 5 gm/day Can increase production 20 x prn In inflammation - involved in signaling necessary for wound healing

Fibrinogen Fibrin Conversion Soluble single molecule converts into an insoluble polymer Conversion is a 3 -step process fibrin cleaved by thrombin into fibrin monomers assembled in fibers & then into bundles stabilization by cross-linking (Factor XIIIa) Factor XIII activated by thrombin Fibrin binds thrombin, plasminogen, t-PA

Fibrin Polymerization Factor XIIIa Lorand L. ATVB 2000 20: 2 -9

Factor XIII Pleiotropic Functions in addition to stabilizing fibrin clot include: Support wound healing • Cross-link extracellular matrix proteins • Promote angiogenesis Prevent pregnancy loss Reduce vascular permeability Muszbek et al. Factor XIII. Physiol Rev 91: 931 -72, 2011

Fibrinolysis Failure of fibrinolysis can lead to excess clotting and risk of thrombosis
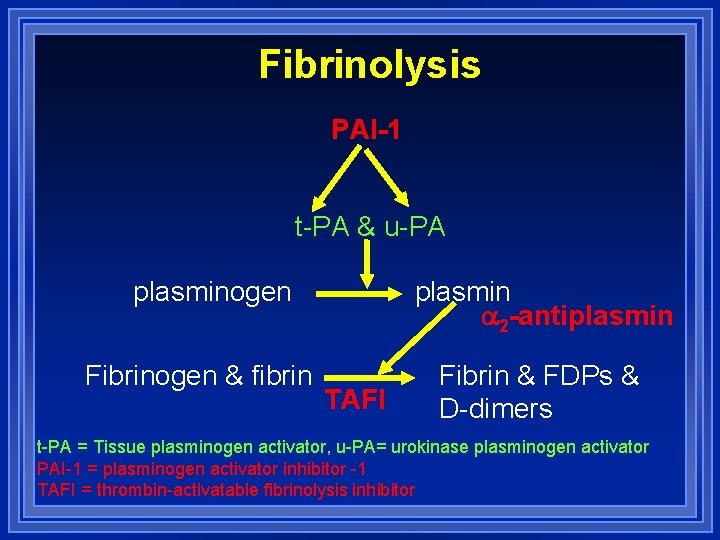
Fibrinolysis PAI-1 t-PA & u-PA plasminogen Fibrinogen & fibrin plasmin 2 -antiplasmin TAFI Fibrin & FDPs & D-dimers t-PA = Tissue plasminogen activator, u-PA= urokinase plasminogen activator PAI-1 = plasminogen activator inhibitor -1 TAFI = thrombin-activatable fibrinolysis inhibitor

Plasmin Activated by t-PA = tissue-type plasminogen activator t-PA is activated by binding to fibrin u-PA = urokinase-type plasminogen activator Plasminogen activation -inhibited by PAI-1 Plasminogen activator inhibitor 1 Plasmin inhibited by 2 -antiplasmin

D-dimers (1) Formed when fibrinolytic system breaks down fibrin Composed of 2 cross-linked fibrin molecules 1/2 life about 8 hours Levels increased when fibrin is being degraded by plasmin Indicator of coagulation activation ACCP guidelines recommend along with ultrasound for DVT testing

D-dimers (2) Levels increase ≈ 8 x with VTE Levels also increase with other thrombotic events Levels usually down to near normal by 15 20 days Levels correlate best with clot surface area Increased with infections, cancer, renal or cardiac failure, acute coronary syndromes, some strokes, pregnancy, sickle cell crisis Kelly J et al. Arch Intern Med 162: 747 -56, 2002 (Apr 8)

Natural Anticoagulant Mechanisms Tissue Factor Pathway Inhibitor Antithrombin III Thrombomodulin / Protein C / Protein S These 3 form a complex on endothelium Inhibition - Plasmin Activation - PAI 1, TAFI “Every pro-coagulant step in coagulation is balanced by 1 or more natural anticoagulants” Aird - WHY?

TF Pathway Inhibitor Often referred to as (TFPI) Inhibits Also TF inhibits activated Factors VII & X (VIIa & Xa)

Antithrombin (III) Inhibits coagulation by enzyme inactivation - thrombin & Xa Inhibits all procoagulant serine proteases Heparin potentiates activity of AT Key factor in keeping coagulation localized AT deficiency can be congenital or acquired What will be key symptom of AT deficiency?
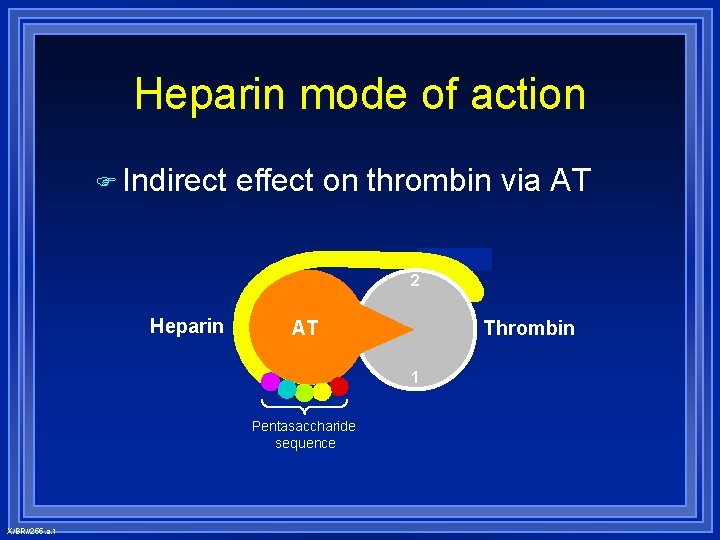
Heparin mode of action Indirect effect on thrombin via AT 2 Heparin AT Thrombin 1 Pentasaccharide sequence X/BR//255. a. 1

Thrombomodulin/Prot C/Prot S Thrombomodulin bound to endothelial cells is constitutively present on cells Thrombin activates Protein C Protein S is a cofactor Activated Prot C plus factor S inactivate factors VIII and V Low levels of Proteins C or S increase risk of thrombosis (fairly rare)
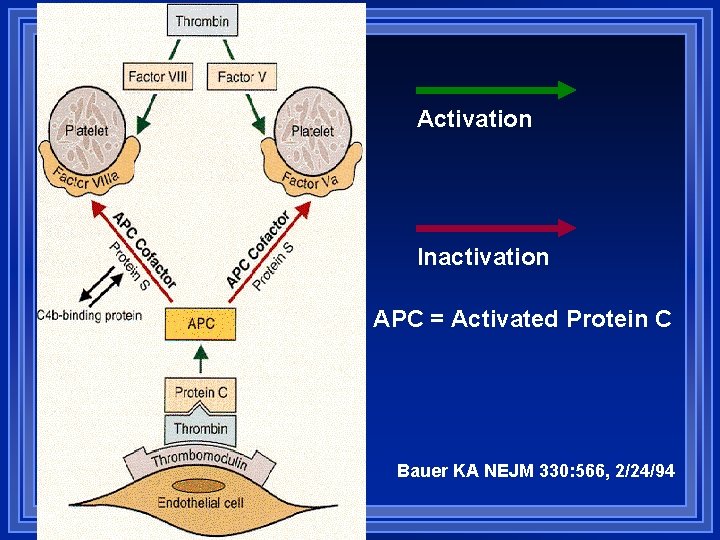
Activation Inactivation APC = Activated Protein C Bauer KA NEJM 330: 566, 2/24/94

Inhibition of Plasmin Activation PAI 1 - plasmin activator inhibitor 1 TAFI - thrombin activatable fibrinolysis inhibitor Alpha 2 -antiplasmin All work to inhibit fibrinolysis

Anti-platelet & Anticoagulant Medications
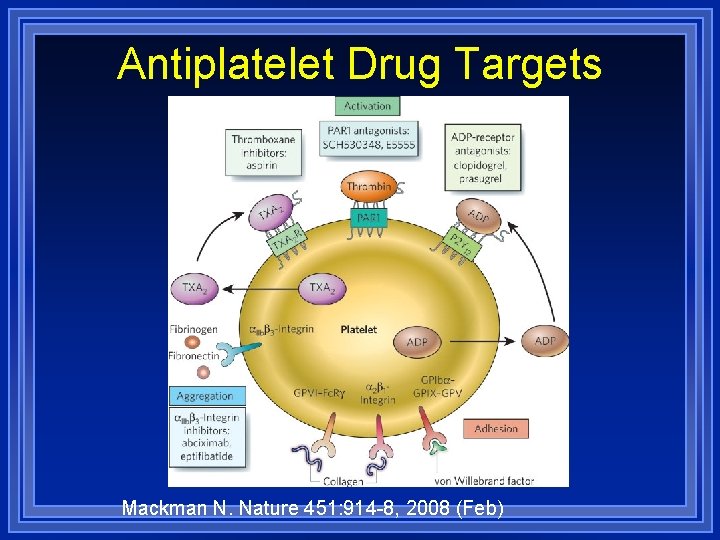
Antiplatelet Drug Targets Mackman N. Nature 451: 914 -8, 2008 (Feb)
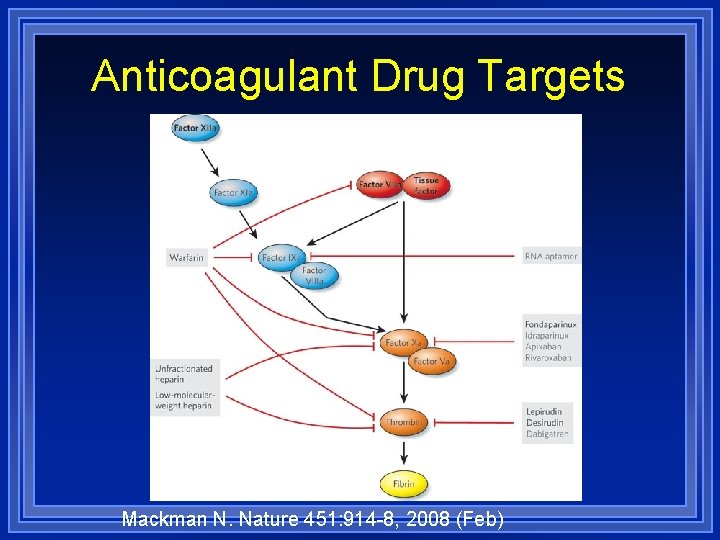
Anticoagulant Drug Targets Mackman N. Nature 451: 914 -8, 2008 (Feb)
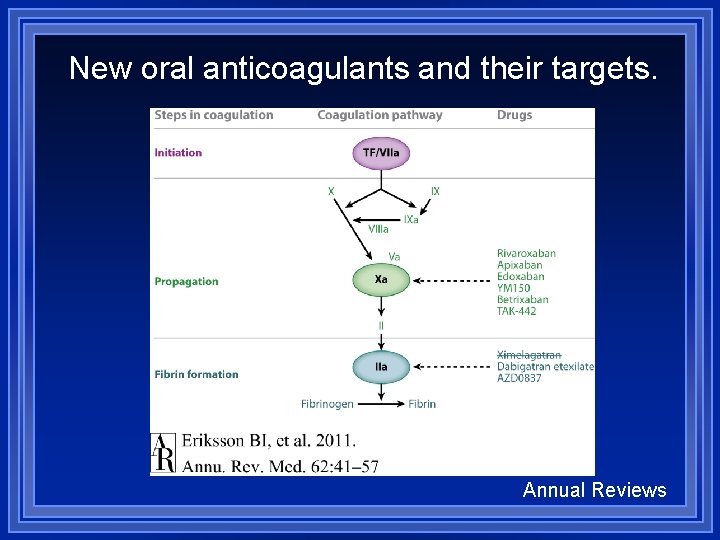
New oral anticoagulants and their targets. Annual Reviews

Drugs & Coagulation Direct thrombin inhibitors 4 are ? FDA approved All given IV lepirudin (Refludan) & argatroban • Indicated for HIT (heparin-induced thrombocytopenia) bivalirudin (Angiomax) - PCI Desirudin - VTE prev in hip replacement

Disorders of Hemostatic Balance Bleeding Risk Clotting – thrombophilic risk

Hemophilias Hemophilia A Most common, factor VIII deficiency Hemophilia B Factor IX deficiency, less severe Hemophilia C Factor XI deficiency von Willibrand Disease

Sepsis, Coagulation, Fibrinolysis Sepsis associated with changes in inflammation coagulation, & fibrinolysis Increases in coagulation & inflammation Decrease in fibrinolysis If acidosis – impaired thrombin formation Result is increase in probability of bleeding Can have coagulation abnormalities without DIC
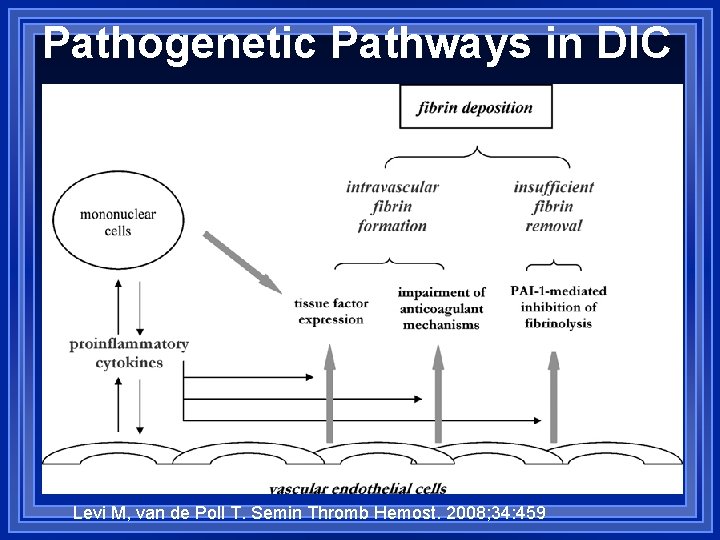
Pathogenetic Pathways in DIC Levi M, van de Poll T. Semin Thromb Hemost. 2008; 34: 459
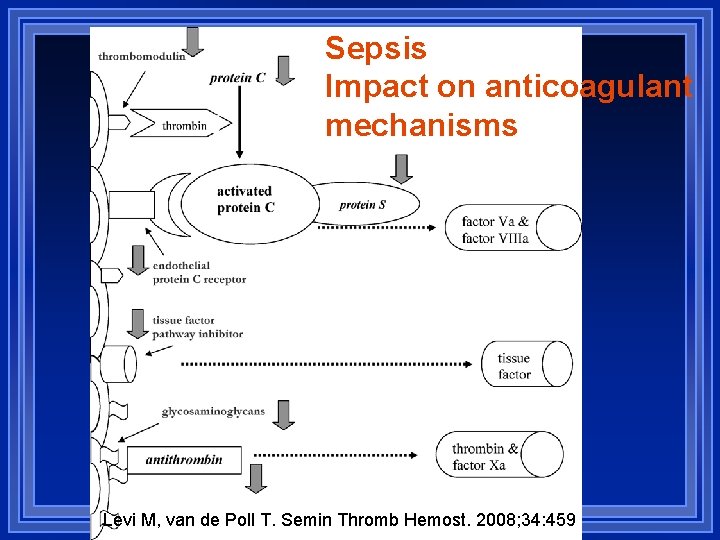
Sepsis Impact on anticoagulant mechanisms Levi M, van de Poll T. Semin Thromb Hemost. 2008; 34: 459

Coagulopathy of Trauma Impaired coagulation seen in 1 of 4 trauma cases Trauma coagulopathy – 4 x in mortality Associated factors include: Acidosis Hypocalcemia (low ionized calcium) Anemia Hypothermia Management – ? ? , treat underlying causes Hess JR et al. J Trauma 2008; 65: 748; Lier H et al. J Trauma 2008; 65: 951.
Inflammation & Coagulation Processes Related Conditions: Sepsis, Pregnancy Loss, ASCVD

Coagulation Inflammation Thrombin enhances inflammation by adhesion molecules on endothelium, neutophils & monocytes acts as a chemotactic agent for WBCs Thrombin enhances cell proliferation stimulates fibroblast cell mitosis PDGF release from platelets, endothelium stimulates PDGF formation in endothelium BUT thrombin & TM = anti-inflammatory Esmon et al. Haematologica 84: 254 -9, 1999.

Inflammation Coagulation Inhibits anticoagulants, ie. thrombomodulin Stimulates coagulation, ie. tissue factor Inhibits fibrinolysis, ie. PAI-1 Chemokines (some) activate platelets Esmon et al. Haematologica 84: 254 -9, 1999. Lambert MP et al. Thromb Haemost. 97: 722, 2007

Thrombosis A major healthcare challenge

Thrombophilic Risk Strongest risk of VTE Antithrombin III deficiency Protein C or S deficiency homozygous mutations in FV Leiden, Prothrombin B 20210 A Milder Risk of VTE heterozygous mutations in FV Leiden, Prothrombin B 20210 A Increased levels of Factor VIII

Pregnancy & Thrombosis Developed World thrombotic problems = most common cause of maternal death Risk of mortality by 4 -5 times VTE risk incidence ≈ 2/1000 deliveries Contraceptive use also an issue

Thrombophilic Mutations & Adverse Pregnancy Outcomes Common thrombophilic mutations Factor V Leiden - resists inactivation by protein C • Heterozygotes have 8 x risk thrombosis • Homozygotes - 30 x risk thrombosis Prothrombin gene mutations (1 - 4% US popn) • Increased risk venous thrombosis, MI, CVA Women with mutations may have adverse pregnancy outcomes – debated

Thrombosis & CVD Acute coronary syndromes Stroke Peripheral vascular disease Pulmonary embolism
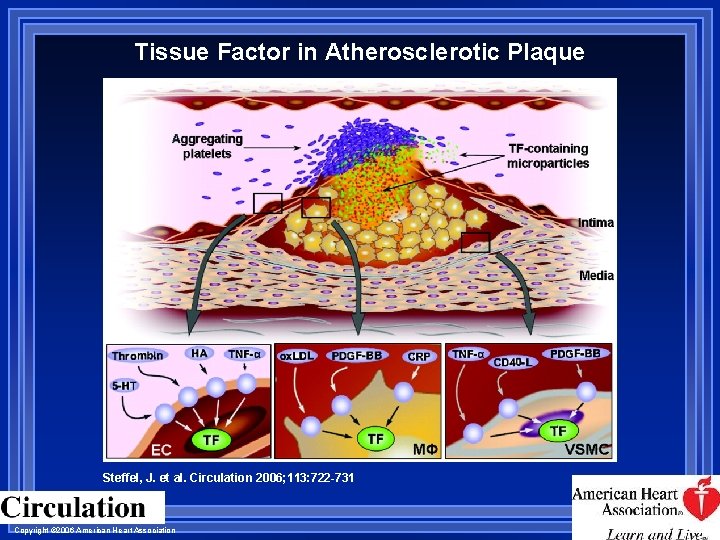
Tissue Factor in Atherosclerotic Plaque Steffel, J. et al. Circulation 2006; 113: 722 -731 Copyright © 2006 American Heart Association
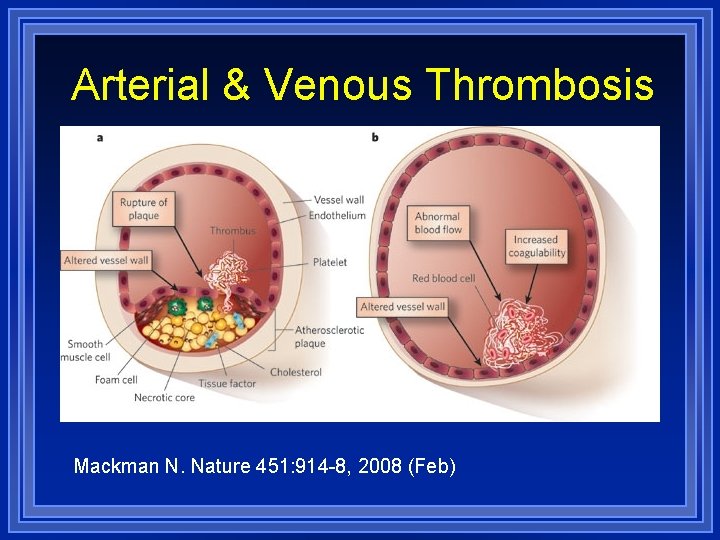
Arterial & Venous Thrombosis Mackman N. Nature 451: 914 -8, 2008 (Feb)
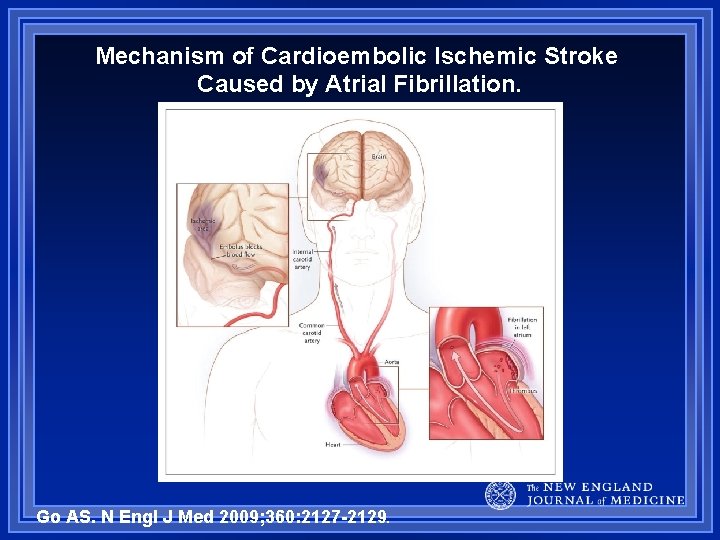
Mechanism of Cardioembolic Ischemic Stroke Caused by Atrial Fibrillation. Go AS. N Engl J Med 2009; 360: 2127 -2129.

Self Test For each of the situations on the next slide indicate which of the following apply. a. There is an increased risk of bleeding / hemorrhage b. There is an increased risk of thrombosis c. No change in risk d. Risk of hemorrhage / bleeding and of thromboses are both increased.

Self-Test Situations _____ _____ 1. Presence of Factor V Leiden 2. Low levels of PAI 1 3. Liver disease 4. Unable to absorb fat 5. Lack of functional factor VIII 6. Lack of von Willibrand’s factor 7. Increased plasma levels of fibrinogen 8. Septicemia
- Slides: 74