Hemoglobin Hb is an ironcontaining metalloprotein that functions

Hemoglobin (Hb) is an iron-containing metalloprotein that functions to transport oxygen in the red blood cells of vertebrates. The protein, which accounts for 97% of the RBC’s dry content is responsible for delivering oxygen from the lungs to all other tissues in the body where it is taken up for cell use. Deepa John Harini Chandra
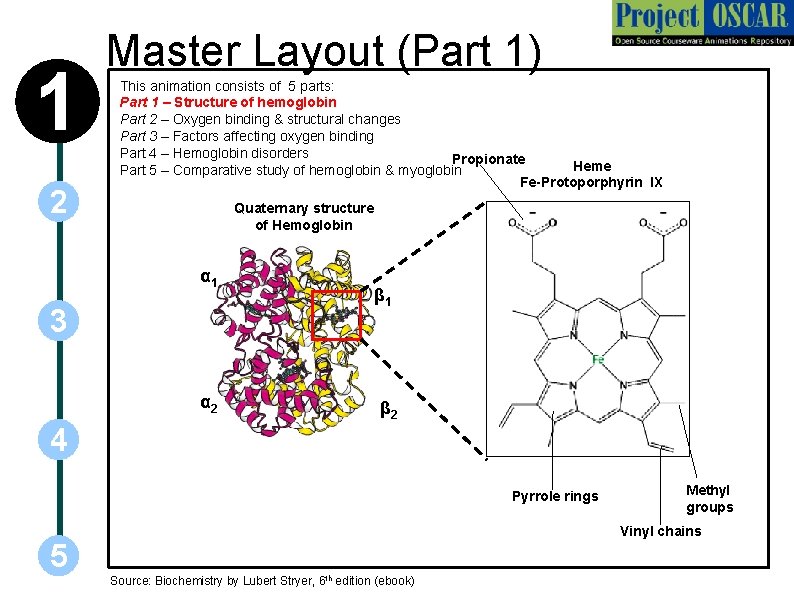
1 2 Master Layout (Part 1) This animation consists of 5 parts: Part 1 – Structure of hemoglobin Part 2 – Oxygen binding & structural changes Part 3 – Factors affecting oxygen binding Part 4 – Hemoglobin disorders Propionate Heme Part 5 – Comparative study of hemoglobin & myoglobin Fe-Protoporphyrin IX Quaternary structure of Hemoglobin α 1 3 α 2 β 1 β 2 4 Pyrrole rings 5 Methyl groups Vinyl chains Source: Biochemistry by Lubert Stryer, 6 th edition (ebook)

1 2 Definitions of the components: Part 1 – Structure of hemoglobin 1. Hemoglobin: Hemoglobin is the predominat metalloprotein in the red blood cells that carries oxygen. Each hemoglobin molecule is made up of four heme groups surrounding a globin group. 2. a, b subunits: Subunits that occur in the functional organization of macromolecules, usually proteins. 3. Heme: Heme is a prosthetic group containing carbon, nitrogen and hydrogen atoms, with a single Fe 2+ ion at the center. 3 4. Prosthetic group: A tightly bound non – protein chemical compound that is required for the activity of an enzyme. 5. Protoporphyrin IX: A metal-free porphyrin that combines with iron to form the heme of iron-containing proteins. It is made up of four pyrrole rings linked by methene bridges to form a tetrapyrrole ring. Four methyl groups, two vinyl groups, and two propionate side chains are attached. 4 5
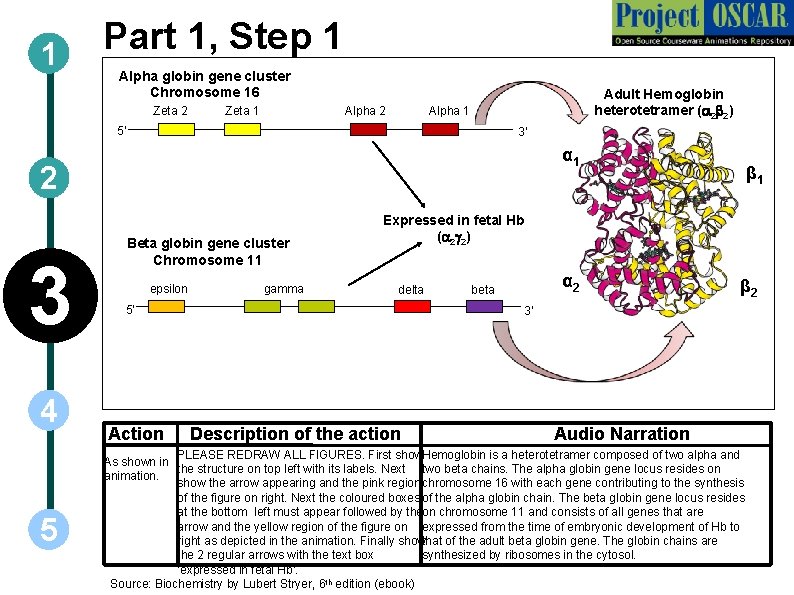
1 Part 1, Step 1 Alpha globin gene cluster Chromosome 16 Zeta 2 Zeta 1 Alpha 2 Adult Hemoglobin heterotetramer (a 2 b 2) Alpha 1 5' 3' α 1 2 3 4 5 Beta globin gene cluster Chromosome 11 epsilon gamma Expressed in fetal Hb (a 2 g 2) delta 5' Action β 1 α 2 beta β 2 3' Description of the action Audio Narration PLEASE REDRAW ALL FIGURES. First show Hemoglobin is a heterotetramer composed of two alpha and As shown in the structure on top left with its labels. Next two beta chains. The alpha globin gene locus resides on animation. show the arrow appearing and the pink region chromosome 16 with each gene contributing to the synthesis of the figure on right. Next the coloured boxes of the alpha globin chain. The beta globin gene locus resides at the bottom left must appear followed by the on chromosome 11 and consists of all genes that are arrow and the yellow region of the figure on expressed from the time of embryonic development of Hb to right as depicted in the animation. Finally show that of the adult beta globin gene. The globin chains are the 2 regular arrows with the text box synthesized by ribosomes in the cytosol. ‘expressed in fetal Hb’. Source: Biochemistry by Lubert Stryer, 6 th edition (ebook)
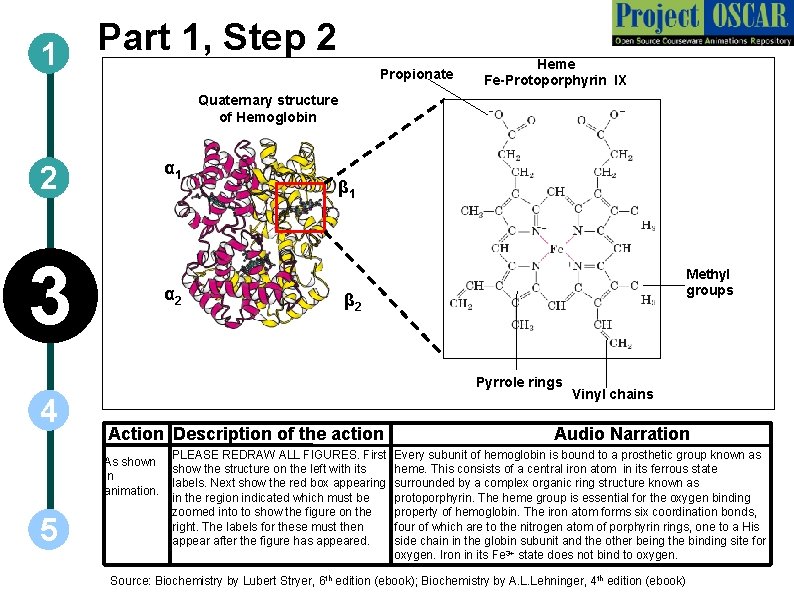
1 Part 1, Step 2 Propionate Heme Fe-Protoporphyrin IX Quaternary structure of Hemoglobin α 1 2 3 α 2 β 1 Methyl groups β 2 Pyrrole rings 4 Action Description of the action As shown in animation. 5 PLEASE REDRAW ALL FIGURES. First show the structure on the left with its labels. Next show the red box appearing in the region indicated which must be zoomed into to show the figure on the right. The labels for these must then appear after the figure has appeared. Vinyl chains Audio Narration Every subunit of hemoglobin is bound to a prosthetic group known as heme. This consists of a central iron atom in its ferrous state surrounded by a complex organic ring structure known as protoporphyrin. The heme group is essential for the oxygen binding property of hemoglobin. The iron atom forms six coordination bonds, four of which are to the nitrogen atom of porphyrin rings, one to a His side chain in the globin subunit and the other being the binding site for oxygen. Iron in its Fe 3+ state does not bind to oxygen. Source: Biochemistry by Lubert Stryer, 6 th edition (ebook); Biochemistry by A. L. Lehninger, 4 th edition (ebook)
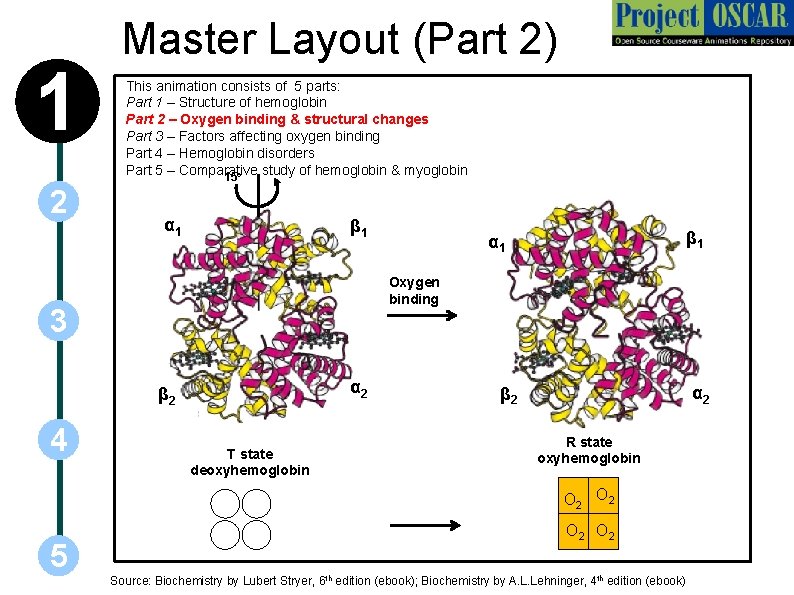
1 2 Master Layout (Part 2) This animation consists of 5 parts: Part 1 – Structure of hemoglobin Part 2 – Oxygen binding & structural changes Part 3 – Factors affecting oxygen binding Part 4 – Hemoglobin disorders Part 5 – Comparative study of hemoglobin & myoglobin 15 o α 1 β 1 Oxygen binding 3 α 2 β 2 4 β 1 α 1 T state deoxyhemoglobin α 2 β 2 R state oxyhemoglobin O 2 5 O 2 Source: Biochemistry by Lubert Stryer, 6 th edition (ebook); Biochemistry by A. L. Lehninger, 4 th edition (ebook)

1 2 3 4 Definitions of the components: Part 2 – Oxygen binding & structural changes 1. Cooperative binding: It is a form of allosteric binding in which ligand binding to macromolecules having more than one binding site is carried out in a cooperative manner such that binding of ligand at one site increases the affinity of another site for the ligand. In tetrameric hemoglobin, binding of first oxygen molecule to one subunit brings about structural changes which in turn positively influences the binding of the remaining subunits to oxygen. 2. Models for oxygen binding to Hb: Two models have been proposed by different groups of scientists to explain the binding of oxygen to hemoglobin. These are the concerted and sequential models: a) Concerted model: The first model proposed by Monod, Wyman and Changeux, also known as the MWC model, assumes that each subunit can exist in at least two conformational states and hypothesizes that all subunits make the transition from one state to the other simultaneously. According to this model, each subunit of a cooperatively binding protein is functionally identical. Binding of ligand can occur with either conformation but with varying degrees of affinity for each. Binding of ligand to a low affinity state makes the transition to the high affinity state more likely. b) Sequential model: The second model, proposed by Daniel Koshland in 1966, postulates that binding of a ligand to one subunit can induce conformational changes in the other subunits. Unlike the concerted model, it does not state that all subunits must exhibit transition from one state to the other simultaneously, thereby making a larger number of intermediate states more likely. 5

1 2 Definitions of the components: Part 2 – Oxygen binding & structural changes 3. Oxyhemoglobin: When all subunits of hemoglobin are bound to oxygen, it is known as oxyhemoglobin and it transports oxygen to the various tissues of the body. 4. Deoxyhemoglobin: Hemoglobin in oxygen unloaded form is called deoxyhemoglobin. 5. T state: T stands for the tense state. It is one of the two quaternary forms of hemoglobin that predominates in absence of oxygen. It has a lower affinity for substrates and, hence, lower catalytic activity. 3 6. R state: R stands for relaxed state. One of the two quaternary forms of hemoglobin that predominates when oxygen is bound. . It has a higher affinity for substrates and, hence, higher catalytic activity. 7. Distal histidine: Distal histidine in vertebrate hemoglobins plays an important role in oxygen binding and has been strongly conserved during evolution. It is considered to be important in fine-tuning the ligand affinities of hemoglobin. 4 5 8. Proximal histidine: The heme in hemoglobin is held in the cleft both by hydrophobic interactions and by a covalent bond between the iron and a nitrogen atom of a nearby histidine side chain. This histidine is referred to as the proximal histidine. 9. Sigmoidal plot: The binding of oxygen to hemoglobin displays marked sigmoidal behaviour, which is indicative of cooperation between subunits. The binding of first oxygen causes a conformational change in the other binding sites that makes it easier for oxygen to bind there. This explains the initial up-swing in the sigmoidal curve. The "leveling out" at the top of the curve is caused by saturation of the hemoglobin-oxygen binding sites.
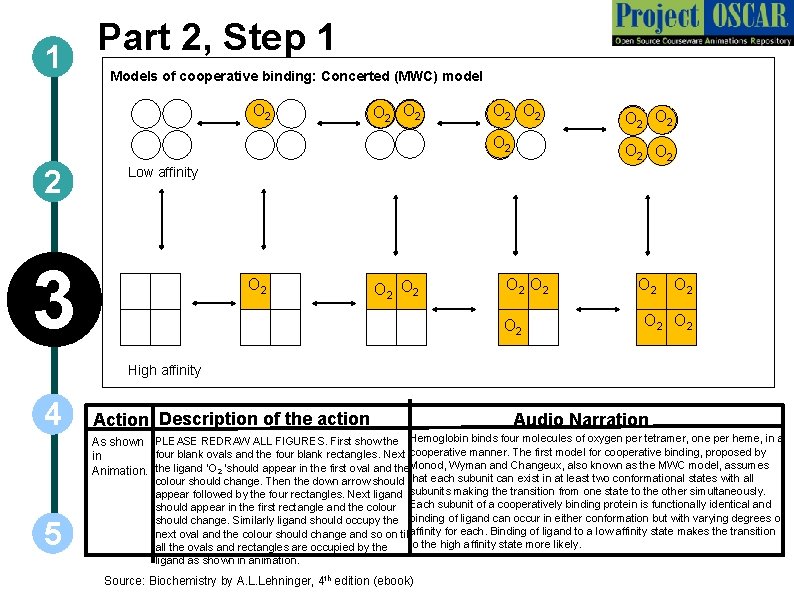
Part 2, Step 1 1 Models of cooperative binding: Concerted (MWC) model O 2 2 O 2 O 2 O 2 Low affinity 3 O 2 O 2 O 2 High affinity 4 Action Description of the action Audio Narration As shown PLEASE REDRAW ALL FIGURES. First show the Hemoglobin binds four molecules of oxygen per tetramer, one per heme, in a four blank ovals and the four blank rectangles. Next cooperative manner. The first model for cooperative binding, proposed by in Animation. the ligand ‘O 2 ‘should appear in the first oval and the Monod, Wyman and Changeux, also known as the MWC model, assumes 5 colour should change. Then the down arrow should that each subunit can exist in at least two conformational states with all appear followed by the four rectangles. Next ligand subunits making the transition from one state to the other simultaneously. should appear in the first rectangle and the colour Each subunit of a cooperatively binding protein is functionally identical and should change. Similarly ligand should occupy the binding of ligand can occur in either conformation but with varying degrees of next oval and the colour should change and so on till affinity for each. Binding of ligand to a low affinity state makes the transition to the high affinity state more likely. all the ovals and rectangles are occupied by the ligand as shown in animation. Source: Biochemistry by A. L. Lehninger, 4 th edition (ebook)
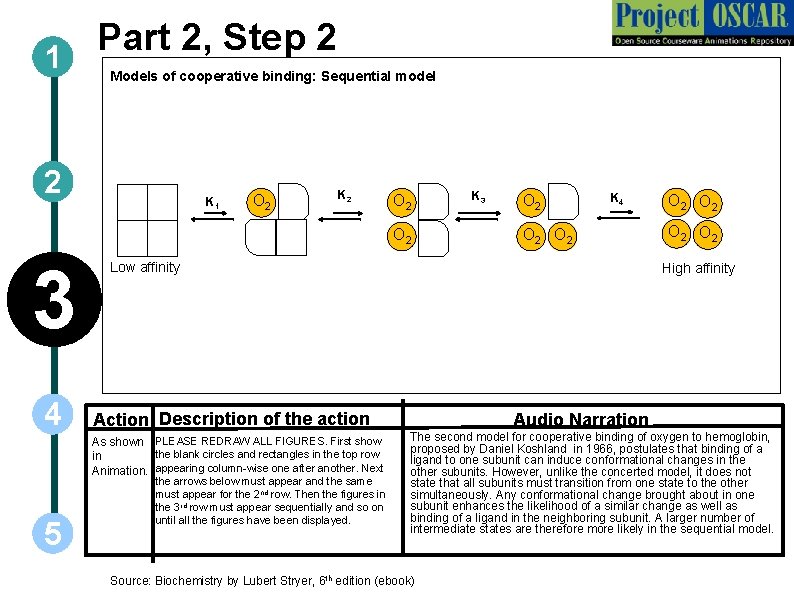
Part 2, Step 2 1 Models of cooperative binding: Sequential model 2 K 1 O 2 K 2 O 2 3 4 O 2 K 4 O 2 Low affinity the arrows below must appear and the same must appear for the 2 nd row. Then the figures in the 3 rd row must appear sequentially and so on until all the figures have been displayed. O 2 O 2 High affinity Action Description of the action As shown PLEASE REDRAW ALL FIGURES. First show the blank circles and rectangles in the top row in Animation. appearing column-wise one after another. Next 5 K 3 Audio Narration The second model for cooperative binding of oxygen to hemoglobin, proposed by Daniel Koshland in 1966, postulates that binding of a ligand to one subunit can induce conformational changes in the other subunits. However, unlike the concerted model, it does not state that all subunits must transition from one state to the other simultaneously. Any conformational change brought about in one subunit enhances the likelihood of a similar change as well as binding of a ligand in the neighboring subunit. A larger number of intermediate states are therefore more likely in the sequential model. Source: Biochemistry by Lubert Stryer, 6 th edition (ebook)
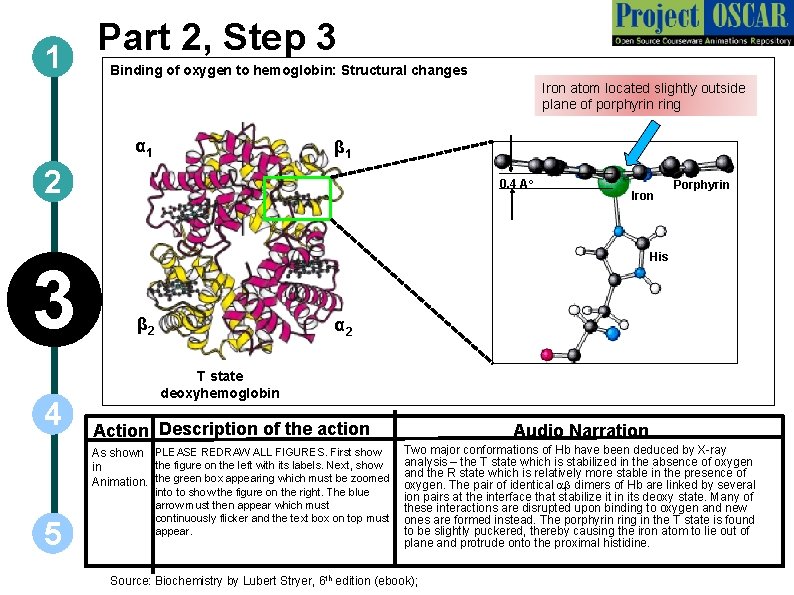
Part 2, Step 3 1 Binding of oxygen to hemoglobin: Structural changes Iron atom located slightly outside plane of porphyrin ring α 1 β 1 2 3 4 0. 4 Ao Porphyrin His β 2 α 2 T state deoxyhemoglobin Action Description of the action Audio Narration Two major conformations of Hb have been deduced by X-ray analysis – the T state which is stabilized in the absence of oxygen and the R state which is relatively more stable in the presence of oxygen. The pair of identical ab dimers of Hb are linked by several into to show the figure on the right. The blue ion pairs at the interface that stabilize it in its deoxy state. Many of arrow must then appear which must these interactions are disrupted upon binding to oxygen and new continuously flicker and the text box on top must ones are formed instead. The porphyrin ring in the T state is found to be slightly puckered, thereby causing the iron atom to lie out of appear. plane and protrude onto the proximal histidine. As shown PLEASE REDRAW ALL FIGURES. First show the figure on the left with its labels. Next, show in Animation. the green box appearing which must be zoomed 5 Iron Source: Biochemistry by Lubert Stryer, 6 th edition (ebook);
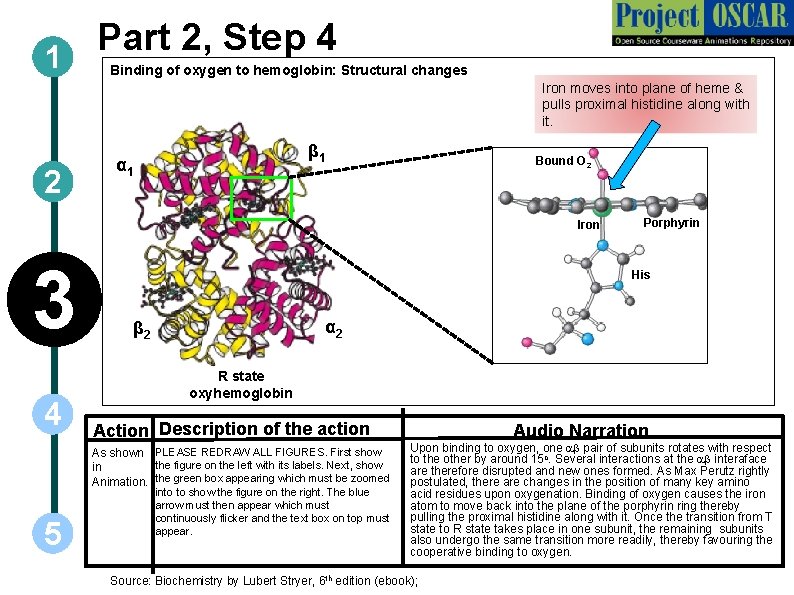
Part 2, Step 4 1 Binding of oxygen to hemoglobin: Structural changes Iron moves into plane of heme & pulls proximal histidine along with it. 2 β 1 α 1 Bound O 2 Iron 3 4 His α 2 β 2 R state oxyhemoglobin Action Description of the action As shown PLEASE REDRAW ALL FIGURES. First show the figure on the left with its labels. Next, show in Animation. the green box appearing which must be zoomed 5 Porphyrin into to show the figure on the right. The blue arrow must then appear which must continuously flicker and the text box on top must appear. Audio Narration Upon binding to oxygen, one ab pair of subunits rotates with respect to the other by around 15 o. Several interactions at the ab interaface are therefore disrupted and new ones formed. As Max Perutz rightly postulated, there are changes in the position of many key amino acid residues upon oxygenation. Binding of oxygen causes the iron atom to move back into the plane of the porphyrin ring thereby pulling the proximal histidine along with it. Once the transition from T state to R state takes place in one subunit, the remaining subunits also undergo the same transition more readily, thereby favouring the cooperative binding to oxygen. Source: Biochemistry by Lubert Stryer, 6 th edition (ebook);
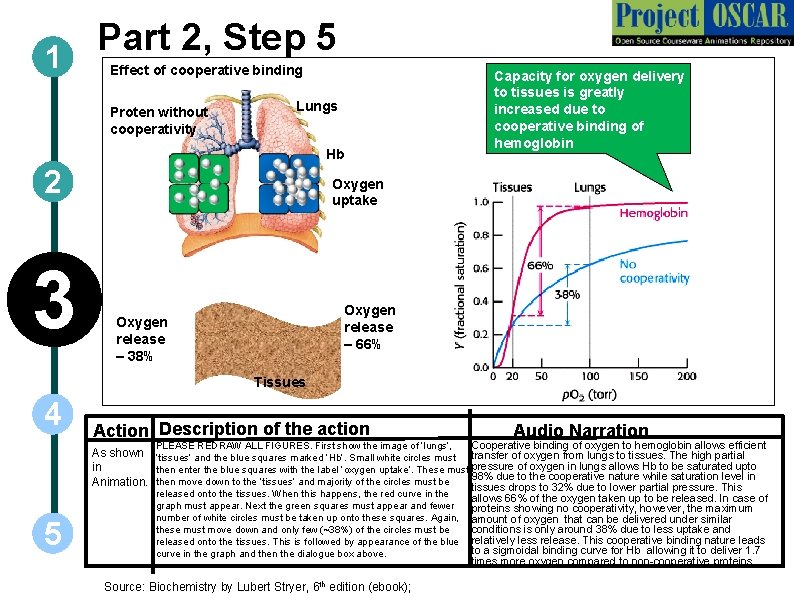
Part 2, Step 5 1 Effect of cooperative binding Proten without cooperativity Lungs Hb 2 3 Capacity for oxygen delivery to tissues is greatly increased due to cooperative binding of hemoglobin Oxygen uptake Oxygen release – 66% Oxygen release – 38% Tissues 4 Action Description of the action As shown in Animation. 5 Audio Narration Cooperative binding of oxygen to hemoglobin allows efficient PLEASE REDRAW ALL FIGURES. First show the image of ‘lungs’, ‘tissues’ and the blue squares marked ‘Hb’. Small white circles must transfer of oxygen from lungs to tissues. The high partial then enter the blue squares with the label ‘oxygen uptake’. These must pressure of oxygen in lungs allows Hb to be saturated upto 98% due to the cooperative nature while saturation level in then move down to the ‘tissues’ and majority of the circles must be tissues drops to 32% due to lower partial pressure. This released onto the tissues. When this happens, the red curve in the allows 66% of the oxygen taken up to be released. In case of graph must appear. Next the green squares must appear and fewer proteins showing no cooperativity, however, the maximum number of white circles must be taken up onto these squares. Again, amount of oxygen that can be delivered under similar conditions is only around 38% due to less uptake and these must move down and only few (~38%) of the circles must be released onto the tissues. This is followed by appearance of the blue relatively less release. This cooperative binding nature leads to a sigmoidal binding curve for Hb allowing it to deliver 1. 7 curve in the graph and then the dialogue box above. times more oxygen compared to non-cooperative proteins. Source: Biochemistry by Lubert Stryer, 6 th edition (ebook);
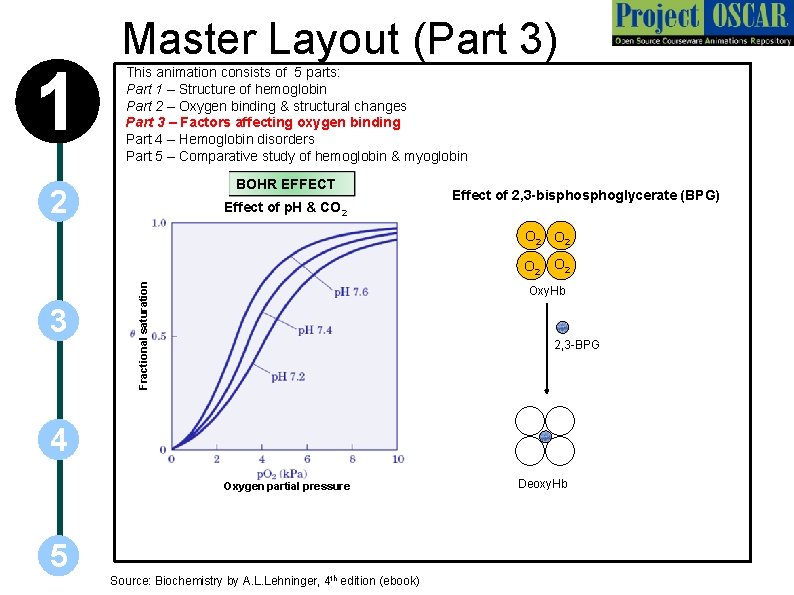
1 Master Layout (Part 3) This animation consists of 5 parts: Part 1 – Structure of hemoglobin Part 2 – Oxygen binding & structural changes Part 3 – Factors affecting oxygen binding Part 4 – Hemoglobin disorders Part 5 – Comparative study of hemoglobin & myoglobin BOHR EFFECT 2 Effect of p. H & CO 2 Effect of 2, 3 -bisphoglycerate (BPG) O 2 3 Fractional saturation O 2 Oxy. Hb 2, 3 -BPG 4 Oxygen partial pressure 5 Source: Biochemistry by A. L. Lehninger, 4 th edition (ebook) Deoxy. Hb

1 2 3 4 5 Definitions of the components: Part 3 – Factors affecting oxygen binding 1. Bohr Effect: The effect of p. H and CO 2 concentration on the binding and release of oxygen by hemoglobin is called the Bohr effect. Lowering the p. H and raising the partial pressure of carbon dioxide results in the release of O 2 from oxyhemoglobin. 2. Effect of 2, 3 -bisphoglycerate: 2, 3 -bisphoglycerate is a highly anionic compound that is present in RBCs at around the same concentration as Hb. 2, 3 -BPG binds to a central pocket of the T form of Hb tetramer and stabilizes it by interacting with three positively charged amino acids on each β-chain. 3. Fractional saturation: It is the fraction of active sites that are bound to the substrate and is directly proportional to reaction velocity. 4. Oxygen partial pressure: It is the partial pressure of oxygen in the gas phase above the solution.
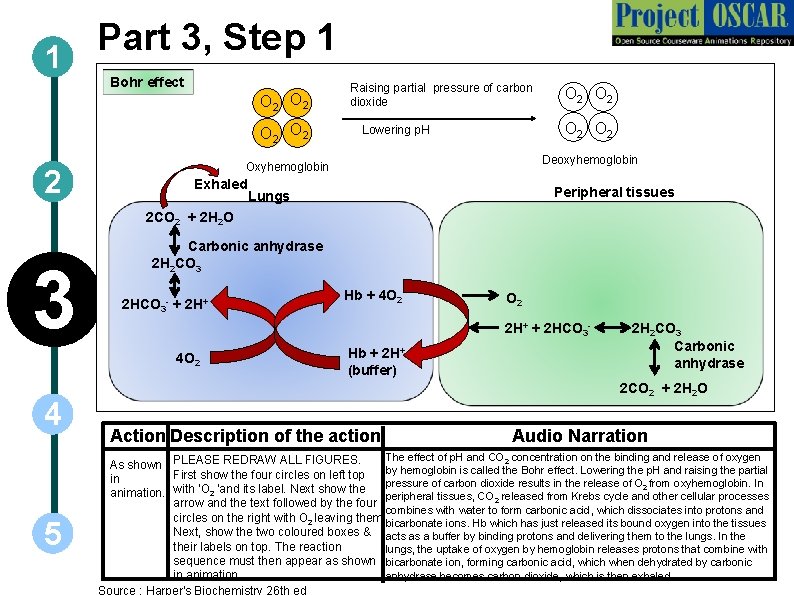
Part 3, Step 1 1 Bohr effect O 2 O 2 2 Raising partial pressure of carbon dioxide O 2 O 2 Lowering p. H Deoxyhemoglobin Oxyhemoglobin Exhaled Lungs Peripheral tissues 2 CO 2 + 2 H 2 O 3 Carbonic anhydrase 2 H 2 CO 3 2 HCO 3 - + 2 H+ 5 O 2 2 H+ + 2 HCO 3 - 4 O 2 4 Hb + 4 O 2 Hb + 2 H+ (buffer) 2 H 2 CO 3 Carbonic anhydrase 2 CO 2 + 2 H 2 O Action Description of the action Audio Narration The effect of p. H and CO 2 concentration on the binding and release of oxygen As shown PLEASE REDRAW ALL FIGURES. First show the four circles on left top by hemoglobin is called the Bohr effect. Lowering the p. H and raising the partial in pressure of carbon dioxide results in the release of O 2 from oxyhemoglobin. In animation. with ‘O 2 ‘and its label. Next show the peripheral tissues, CO 2 released from Krebs cycle and other cellular processes arrow and the text followed by the four combines with water to form carbonic acid, which dissociates into protons and circles on the right with O 2 leaving them. bicarbonate ions. Hb which has just released its bound oxygen into the tissues Next, show the two coloured boxes & acts as a buffer by binding protons and delivering them to the lungs. In their labels on top. The reaction lungs, the uptake of oxygen by hemoglobin releases protons that combine with sequence must then appear as shown bicarbonate ion, forming carbonic acid, which when dehydrated by carbonic in animation. anhydrase becomes carbon dioxide, which is then exhaled. Source : Harper's Biochemistry 26 th ed
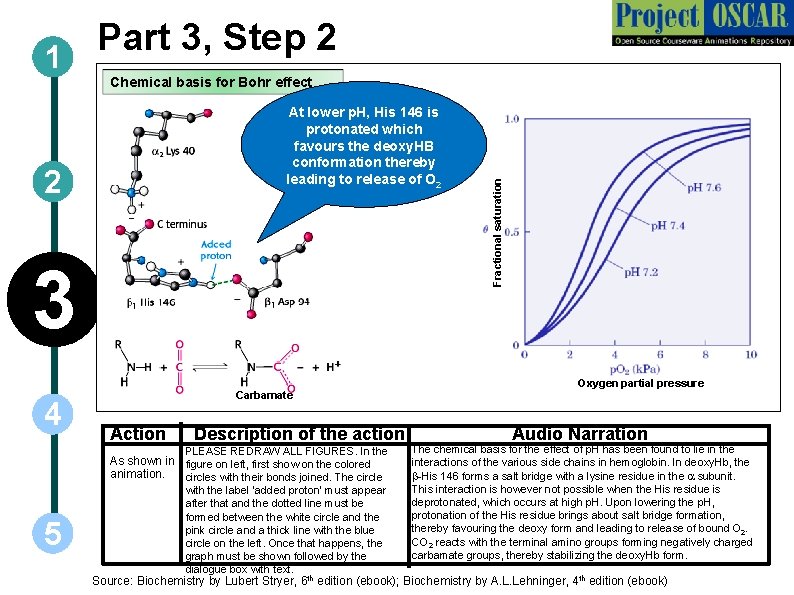
Part 3, Step 2 1 At lower p. H, His 146 is protonated which favours the deoxy. HB conformation thereby leading to release of O 2 2 3 4 5 Carbamate Action Description of the action PLEASE REDRAW ALL FIGURES. In the As shown in figure on left, first show on the colored animation. circles with their bonds joined. The circle with the label ‘added proton’ must appear after that and the dotted line must be formed between the white circle and the pink circle and a thick line with the blue circle on the left. Once that happens, the graph must be shown followed by the dialogue box with text. Fractional saturation Chemical basis for Bohr effect Oxygen partial pressure Audio Narration The chemical basis for the effect of p. H has been found to lie in the interactions of the various side chains in hemoglobin. In deoxy. Hb, the b-His 146 forms a salt bridge with a lysine residue in the a subunit. This interaction is however not possible when the His residue is deprotonated, which occurs at high p. H. Upon lowering the p. H, protonation of the His residue brings about salt bridge formation, thereby favouring the deoxy form and leading to release of bound O 2. CO 2 reacts with the terminal amino groups forming negatively charged carbamate groups, thereby stabilizing the deoxy. Hb form. Source: Biochemistry by Lubert Stryer, 6 th edition (ebook); Biochemistry by A. L. Lehninger, 4 th edition (ebook)
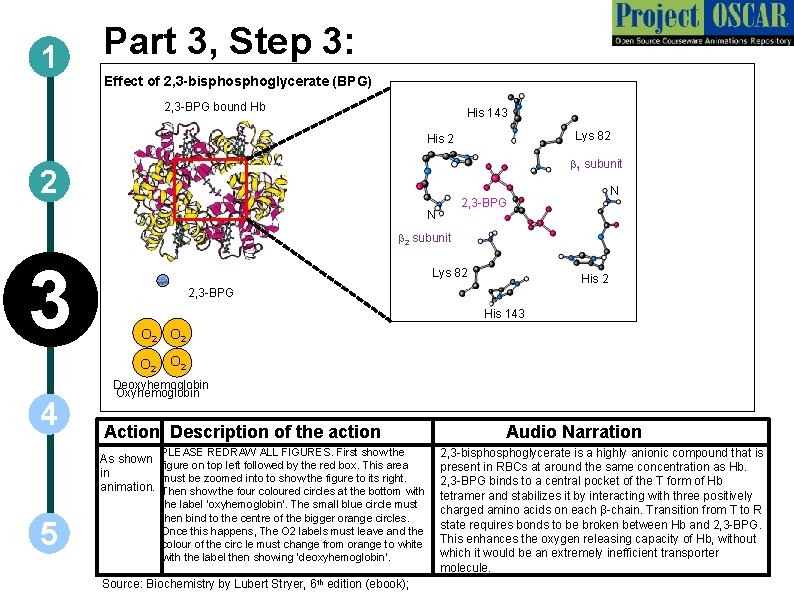
1 Part 3, Step 3: Effect of 2, 3 -bisphoglycerate (BPG) 2, 3 -BPG bound Hb His 143 Lys 82 His 2 b 1 subunit 2 N N 2, 3 -BPG b 2 subunit 3 Lys 82 His 2 2, 3 -BPG His 143 O 2 O 2 4 5 Deoxyhemoglobin Oxyhemoglobin Action Description of the action Audio Narration PLEASE REDRAW ALL FIGURES. First show the 2, 3 -bisphoglycerate is a highly anionic compound that is As shown figure on top left followed by the red box. This area present in RBCs at around the same concentration as Hb. in must be zoomed into to show the figure to its right. animation. Then show the four coloured circles at the bottom with 2, 3 -BPG binds to a central pocket of the T form of Hb tetramer and stabilizes it by interacting with three positively the label ‘oxyhemoglobin’. The small blue circle must charged amino acids on each β-chain. Transition from T to R then bind to the centre of the bigger orange circles. state requires bonds to be broken between Hb and 2, 3 -BPG. Once this happens, The O 2 labels must leave and the colour of the circ le must change from orange to white This enhances the oxygen releasing capacity of Hb, without which it would be an extremely inefficient transporter with the label then showing ‘deoxyhemoglobin’. molecule. th Source: Biochemistry by Lubert Stryer, 6 edition (ebook);
1 Master Layout (Part 4) This animation consists of 5 parts: Part 1 – Structure of hemoglobin Part 2 – Oxygen binding & structural changes Part 3 – Factors affecting oxygen binding Part 4 – Hemoglobin disorders Part 5 – Comparative study of hemoglobin & myoglobin 2 SICKLE CELL ANEMIA Thalassemia 3 Normal red blood cell 4 5 Sickled red blood cell α thalassemia β thalassemia

1 Definitions of the components: Part 4 – Hemoglobin disorders 2 1. Sickle cell anemia: Sickle-cell anemia is a genetic disease in which an individual has inherited the allele for sickle-cell hemoglobin from both parents characterized by abnormal, rigid, sickle shape (Hb. S) as compared to the normal flexible biconcave disk shaped red blood cells (Hb. A). It results from a single amino acid substitution, a Val instead of a Glu residue at position 6 in the two β chains. As a result of this change, deoxyhemoglobin S has a hydrophobic patch on its surface, which causes the molecules to aggregate into strands that align to form insoluble fibres. Sickle-cell disease may lead to various acute and chronic complications, several of which are potentially lethal. 3 2. Thalassemia: This is another inherited genetic disorder due to deletions or mutations in the globin genes which results in abnormalities and deficiencies in α-globin synthesis. Signs and symptoms of thalassemias are due to lack of oxygen in the bloodstream. This occurs because the body doesn't make enough healthy red blood cells and hemoglobin. The severity of symptoms depends on the severity of the disorder. 3. Point mutation: A type of mutation resulting from substitution of a single nucleotide base with another. 4 5
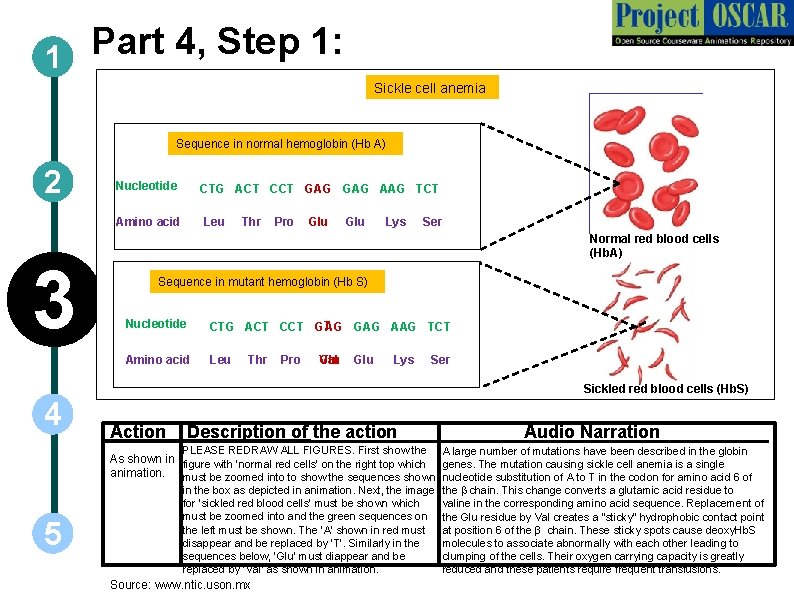
Part 4, Step 1: 1 Sickle cell anemia Sequence in normal hemoglobin (Hb A) 2 3 Nucleotide CTG ACT CCT GAG AAG TCT Amino acid Leu Thr Pro Glu Lys Ser Normal red blood cells (Hb. A) Sequence in mutant hemoglobin (Hb S) Nucleotide Amino acid T CTG ACT CCT G G GAG AAG TCT A Leu Thr Pro Glu Lys Ser Val Glu Sickled red blood cells (Hb. S) 4 Action Description of the action Audio Narration PLEASE REDRAW ALL FIGURES. First show the A large number of mutations have been described in the globin in the box as depicted in animation. Next, the image for ‘sickled red blood cells’ must be shown which must be zoomed into and the green sequences on the left must be shown. The ‘A’ shown in red must disappear and be replaced by ‘T’. Similarly in the sequences below, ‘Glu’ must diappear and be replaced by ‘Val’ as shown in animation. the b chain. This change converts a glutamic acid residue to valine in the corresponding amino acid sequence. Replacement of the Glu residue by Val creates a “sticky” hydrophobic contact point at position 6 of the β chain. These sticky spots cause deoxy. Hb. S molecules to associate abnormally with each other leading to clumping of the cells. Their oxygen carrying capacity is greatly reduced and these patients require frequent transfusions. As shown in figure with ‘normal red cells’ on the right top which genes. The mutation causing sickle cell anemia is a single animation. must be zoomed into to show the sequences shown nucleotide substitution of A to T in the codon for amino acid 6 of 5 Source: www. ntic. uson. mx
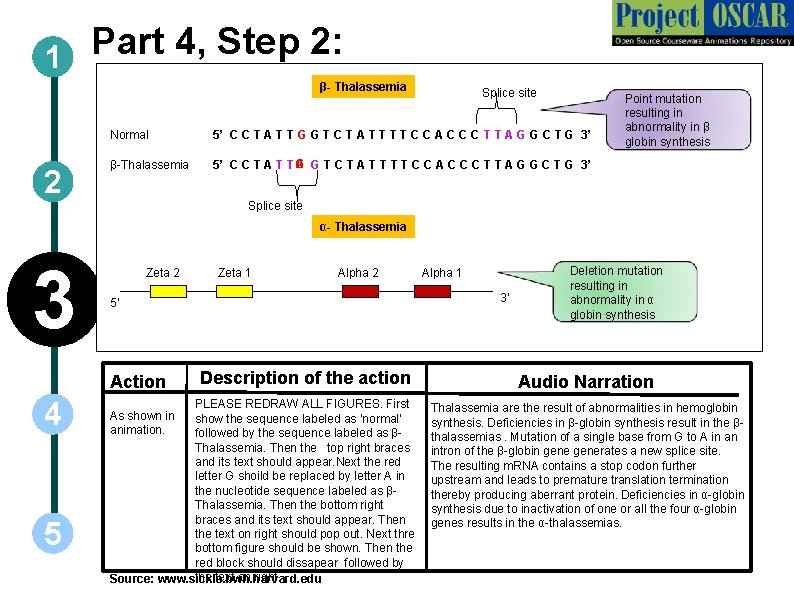
Part 4, Step 2: 1 β- Thalassemia 2 Splice site Normal 5’ C C T A T T G G T C T A T T C C A C C C T T A G G C T G 3’ β-Thalassemia A G 5’ C C T A T T G T C T A T T C C A C C C T T A G G C T G 3’ Point mutation resulting in abnormality in β globin synthesis Splice site α- Thalassemia 3 Zeta 2 5 Alpha 2 Description of the action PLEASE REDRAW ALL FIGURES. First show the sequence labeled as ‘normal’ followed by the sequence labeled as βThalassemia. Then the top right braces and its text should appear. Next the red letter G shoild be replaced by letter A in the nucleotide sequence labeled as βThalassemia. Then the bottom right braces and its text should appear. Then the text on right should pop out. Next thre bottom figure should be shown. Then the red block should dissapear followed by the text on right. Source: www. sickle. bwh. harvard. edu As shown in animation. Alpha 1 3' 5' Action 4 Zeta 1 Deletion mutation resulting in abnormality in α globin synthesis Audio Narration Thalassemia are the result of abnormalities in hemoglobin synthesis. Deficiencies in β-globin synthesis result in the βthalassemias. Mutation of a single base from G to A in an intron of the β-globin generates a new splice site. The resulting m. RNA contains a stop codon further upstream and leads to premature translation termination thereby producing aberrant protein. Deficiencies in α-globin synthesis due to inactivation of one or all the four α-globin genes results in the α-thalassemias.
1 2 Master Layout (Part 5) This animation consists of 5 parts: Part 1 – Structure of hemoglobin Part 2 – Oxygen binding & structural changes Part 3 – Factors affecting oxygen binding Part 4 – Hemoglobin disorders Part 5 – Comparative study of hemoglobin & myoglobin Heme group Iron atom α 1 β 1 3 α 2 4 Myoglobin 5 Source: Biochemistry by Lubert Stryer, 6 th edition (ebook); β 2 Hemoglobin

1 2 3 4 5 Definitions of the components: Part 5 – Comparative study of hemoglobin & myoglobin 1. Myoglobin (Mb): Myoglobin is a globular protein having a single polypeptide chain consisting of eight alpha helices linked by short polypeptide segments, with a total of 153 amino acid residues. It is found in muscle tissues of most mammals and plays a role in oxygen binding. Myoglobin resembles the alpha subunit of hemoglobin and like hemoglobin, it consists of a central heme group which enables it to bind oxygen. Myoglobin was the first protein to have its X-ray crystallography structure determined in 1959 by John Kendrew.
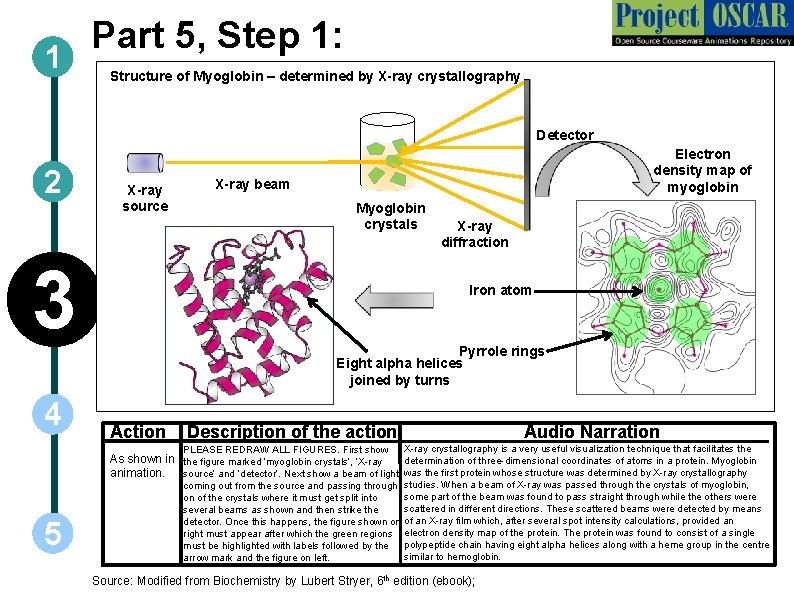
1 Part 5, Step 1: Structure of Myoglobin – determined by X-ray crystallography Detector 2 X-ray source 3 4 Electron density map of myoglobin X-ray beam Myoglobin crystals X-ray diffraction Iron atom Pyrrole rings Eight alpha helices joined by turns Action Description of the action PLEASE REDRAW ALL FIGURES. First show Audio Narration X-ray crystallography is a very useful visualization technique that facilitates the As shown in the figure marked ‘myoglobin crystals’, ‘X-ray determination of three-dimensional coordinates of atoms in a protein. Myoglobin animation. source’ and ‘detector’. Next show a beam of light was the first protein whose structure was determined by X-ray crystallography 5 coming out from the source and passing through studies. When a beam of X-ray was passed through the crystals of myoglobin, some part of the beam was found to pass straight through while the others were on of the crystals where it must get split into scattered in different directions. These scattered beams were detected by means several beams as shown and then strike the detector. Once this happens, the figure shown on of an X-ray film which, after several spot intensity calculations, provided an right must appear after which the green regions electron density map of the protein. The protein was found to consist of a single must be highlighted with labels followed by the polypeptide chain having eight alpha helices along with a heme group in the centre similar to hemoglobin. arrow mark and the figure on left. Source: Modified from Biochemistry by Lubert Stryer, 6 th edition (ebook);
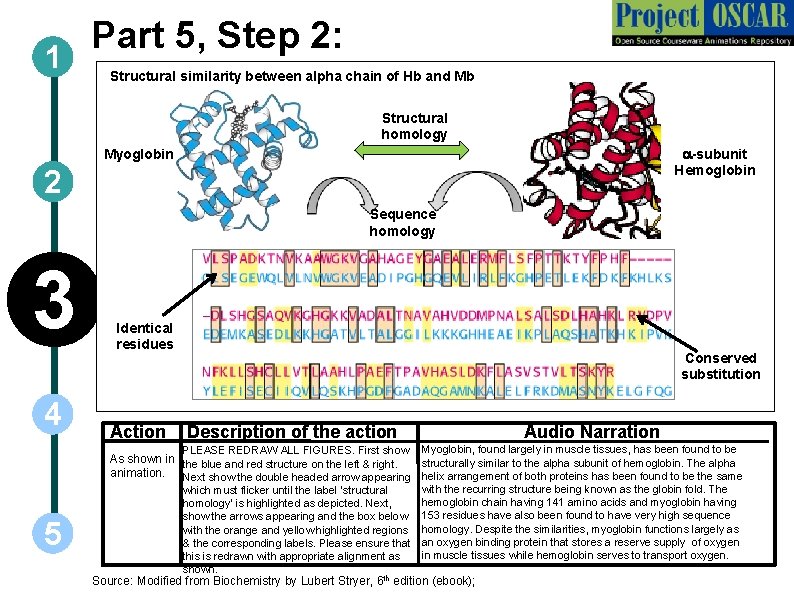
1 Part 5, Step 2: Structural similarity between alpha chain of Hb and Mb Structural homology a-subunit Hemoglobin Myoglobin 2 Sequence homology 3 4 Identical residues Action Conserved substitution Description of the action Audio Narration PLEASE REDRAW ALL FIGURES. First show Myoglobin, found largely in muscle tissues, has been found to be As shown in the blue and red structure on the left & right. structurally similar to the alpha subunit of hemoglobin. The alpha animation. Next show the double headed arrow appearing helix arrangement of both proteins has been found to be the same 5 which must flicker until the label ‘structural homology’ is highlighted as depicted. Next, show the arrows appearing and the box below with the orange and yellow highlighted regions & the corresponding labels. Please ensure that this is redrawn with appropriate alignment as shown. with the recurring structure being known as the globin fold. The hemoglobin chain having 141 amino acids and myoglobin having 153 residues have also been found to have very high sequence homology. Despite the similarities, myoglobin functions largely as an oxygen binding protein that stores a reserve supply of oxygen in muscle tissues while hemoglobin serves to transport oxygen. Source: Modified from Biochemistry by Lubert Stryer, 6 th edition (ebook);
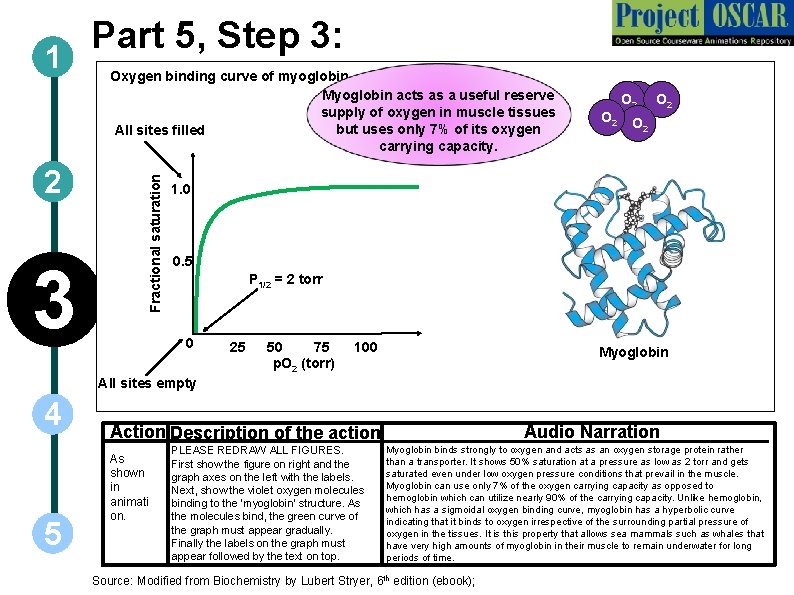
2 3 Oxygen binding curve of myoglobin Myoglobin acts as a useful reserve supply of oxygen in muscle tissues but uses only 7% of its oxygen All sites filled carrying capacity. Fractional saturation 1 Part 5, Step 3: O 2 O 2 O 2 1. 0 0. 5 P 1/2 = 2 torr 0 25 50 75 p. O 2 (torr) 100 Myoglobin All sites empty 4 5 Action Description of the action As shown in animati on. PLEASE REDRAW ALL FIGURES. First show the figure on right and the graph axes on the left with the labels. Next, show the violet oxygen molecules binding to the ‘myoglobin’ structure. As the molecules bind, the green curve of the graph must appear gradually. Finally the labels on the graph must appear followed by the text on top. Audio Narration Myoglobin binds strongly to oxygen and acts as an oxygen storage protein rather than a transporter. It shows 50% saturation at a pressure as low as 2 torr and gets saturated even under low oxygen pressure conditions that prevail in the muscle. Myoglobin can use only 7% of the oxygen carrying capacity as opposed to hemoglobin which can utilize nearly 90% of the carrying capacity. Unlike hemoglobin, which has a sigmoidal oxygen binding curve, myoglobin has a hyperbolic curve indicating that it binds to oxygen irrespective of the surrounding partial pressure of oxygen in the tissues. It is this property that allows sea mammals such as whales that have very high amounts of myoglobin in their muscle to remain underwater for long periods of time. Source: Modified from Biochemistry by Lubert Stryer, 6 th edition (ebook);
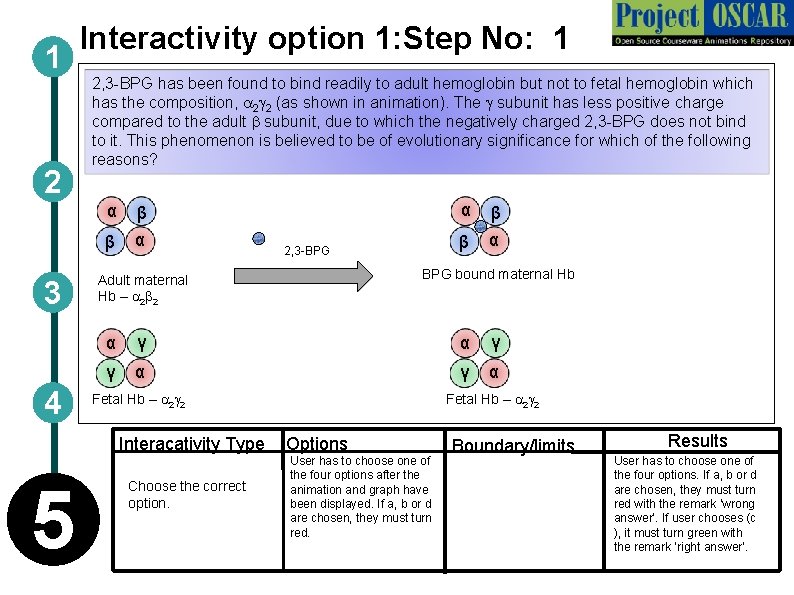
1 2 3 4 Interactivity option 1: Step No: 1 2, 3 -BPG has been found to bind readily to adult hemoglobin but not to fetal hemoglobin which has the composition, a 2 g 2 (as shown in animation). The g subunit has less positive charge compared to the adult b subunit, due to which the negatively charged 2, 3 -BPG does not bind to it. This phenomenon is believed to be of evolutionary significance for which of the following reasons? α β β α 2, 3 -BPG β β α BPG bound maternal Hb Adult maternal Hb – a 2 b 2 α γ γ α Fetal Hb – a 2 g 2 Interacativity Type Options 5 α Choose the correct option. User has to choose one of the four options after the animation and graph have been displayed. If a, b or d are chosen, they must turn red. Boundary/limits Results User has to choose one of the four options. If a, b or d are chosen, they must turn red with the remark ‘wrong answer’. If user chooses (c ), it must turn green with the remark ‘right answer’.
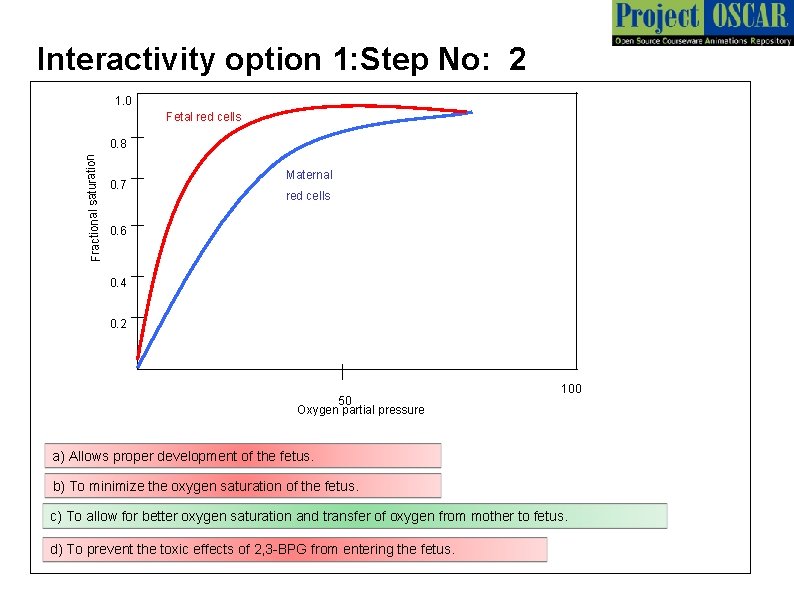
Interactivity option 1: Step No: 2 1. 0 Fetal red cells Fractional saturation 0. 8 0. 7 Maternal red cells 0. 6 0. 4 0. 2 50 Oxygen partial pressure 100 a) Allows proper development of the fetus. b) To minimize the oxygen saturation of the fetus. c) To allow for better oxygen saturation and transfer of oxygen from mother to fetus. d) To prevent the toxic effects of 2, 3 -BPG from entering the fetus.
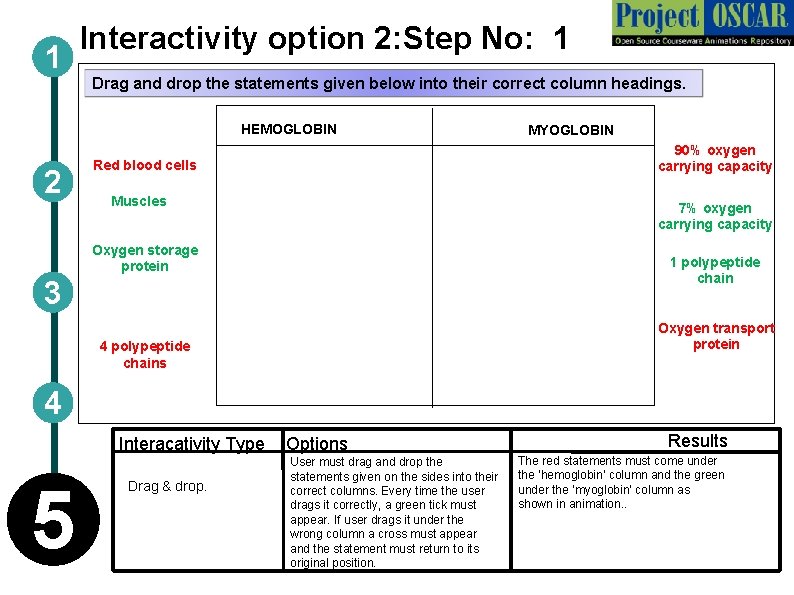
1 Interactivity option 2: Step No: 1 Drag and drop the statements given below into their correct column headings. HEMOGLOBIN 2 MYOGLOBIN 90% oxygen carrying capacity Red blood cells Muscles 7% oxygen carrying capacity Oxygen storage protein 1 polypeptide chain 3 Oxygen transport protein 4 polypeptide chains 4 Interacativity Type Options 5 Drag & drop. User must drag and drop the statements given on the sides into their correct columns. Every time the user drags it correctly, a green tick must appear. If user drags it under the wrong column a cross must appear and the statement must return to its original position. Results The red statements must come under the ‘hemoglobin’ column and the green under the ‘myoglobin’ column as shown in animation. .

1 Questionnaire 1. Which of the following is the major form of hemoglobin produced in red blood cells during normal adult life? Answers: a) a 2 b 2 b) a 2 g 2 c) z 2 e 2 d) a 2 e 2 2 2. All of the following factors influence hemoglobin dissociation curve except ? Answers: a) p. H b) CO 2 tension c) Temperature d) 2, 3 BPG levels 3 4 3. The cause of sicke cell anemia is : Answers: a) a deletion of the beta globin gene promoter b) a missense mutation in the coding region of the beta globin gene c) a nonsense mutation in the coding region of the beta globin gene d) the increased production of the alpha globin gene due to a duplication 4. Bohr effect explains the effect of following on the binding and release of oxygen by hemoglobin: Answers: a) CO 2 b) p. H c) 2, 3, BPG d) both a and b 5. What is the heme group? 5 Answers: a) lipid b) tetrapyrrole c) protein d) steroid

Links for further reading Books: Biochemistry by Lubert Stryer et al. , 6 th edition (ebook) Biochemistry by A. L. Lehninger et al. , 4 th edition Biochemistry by Harper, 26 th edition (ebook) Research papers: The crystal structure of a tetrameric hemoglobin in a partial hemichrome state. Antonio Riccio by Luigi Vitagliano, Guido di Prisco, Adriana Zagari and Lelio Mazzarella
- Slides: 32