Hemoglobin Biochemistry Free For All Hemoglobin and the

Hemoglobin Biochemistry Free For All

Hemoglobin and the Movement of Oxygen

Hemoglobin and the Movement of Oxygen • Introduction Animals Have Widely Varying Needs for Oxygen Demand for Oxygen Can Change in Seconds Basal Needs are Significant - Diffusion not Enough Exercise, Fight/Flight Add to the Need ATP Energy Produced Aerobically 15 Times More Efficiently Than Anaerobically Respiration versus Fermentation Efficient, Adaptable Oxygen Delivery is Necessary
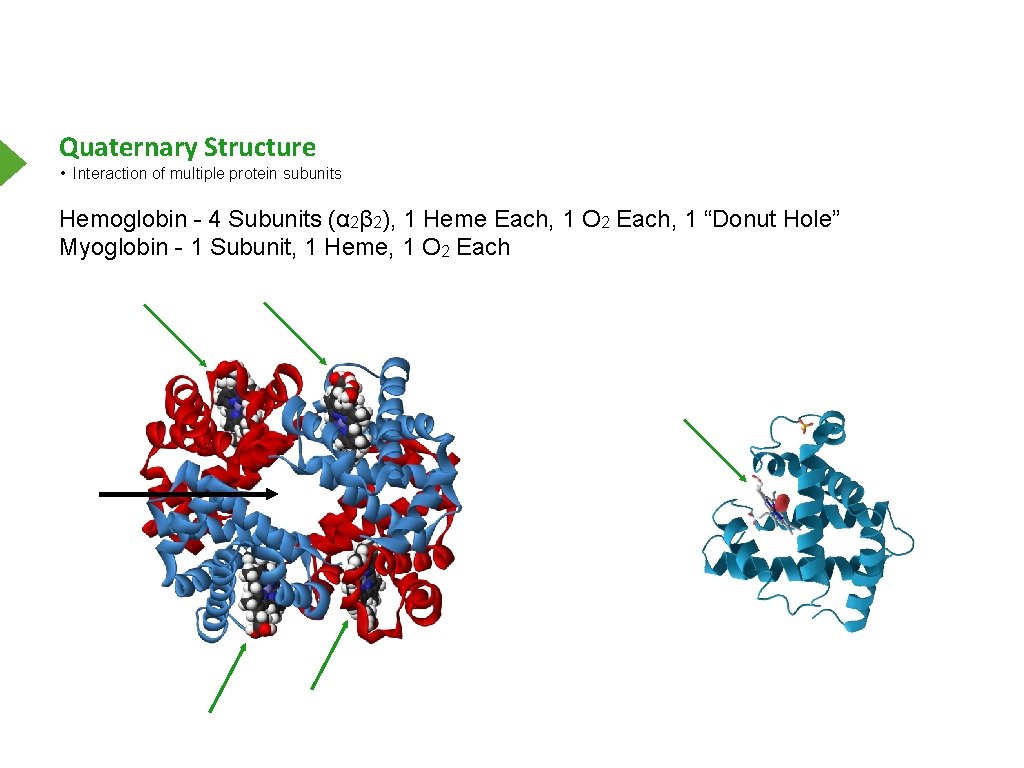
Quaternary Structure • Interaction of multiple protein subunits Hemoglobin - 4 Subunits (α 2β 2), 1 Heme Each, 1 O 2 Each, 1 “Donut Hole” Myoglobin - 1 Subunit, 1 Heme, 1 O 2 Each
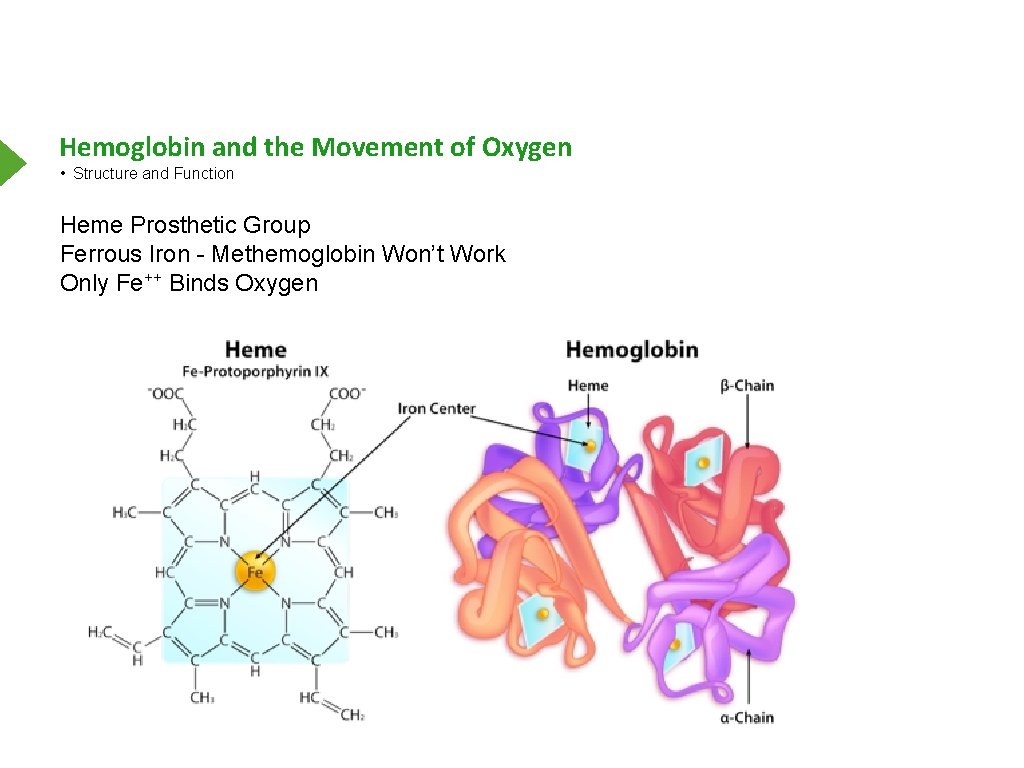
Hemoglobin and the Movement of Oxygen • Structure and Function Heme Prosthetic Group Ferrous Iron - Methemoglobin Won’t Work Only Fe++ Binds Oxygen
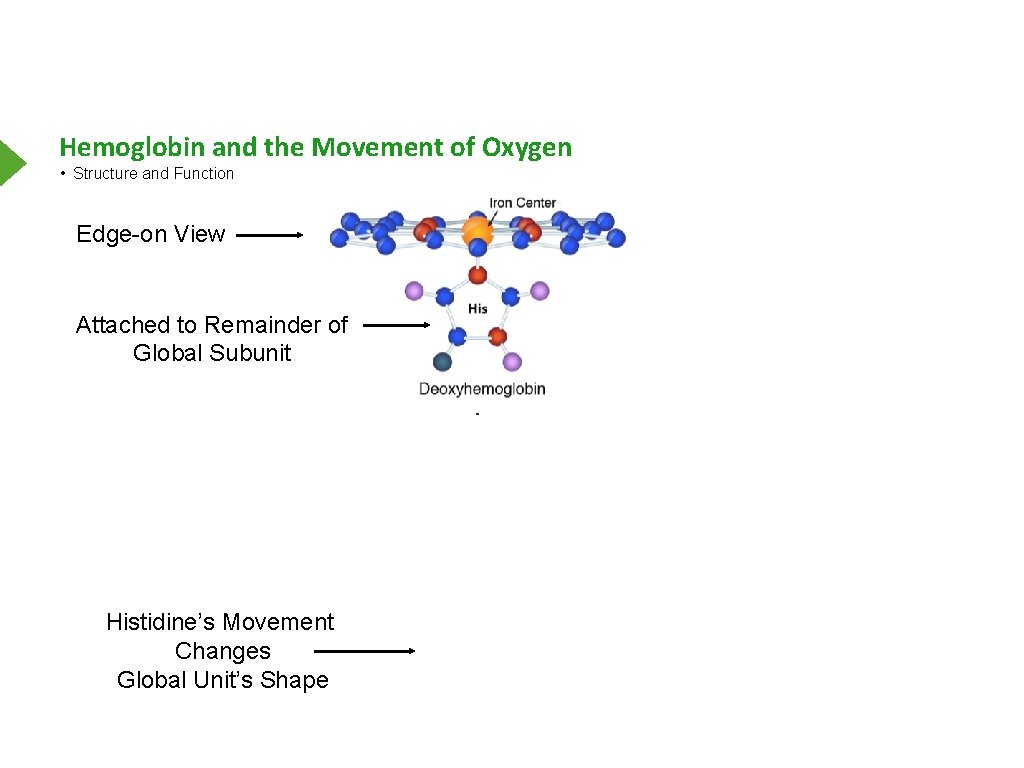
Hemoglobin and the Movement of Oxygen • Structure and Function Edge-on View Attached to Remainder of Global Subunit Histidine’s Movement Changes Global Unit’s Shape
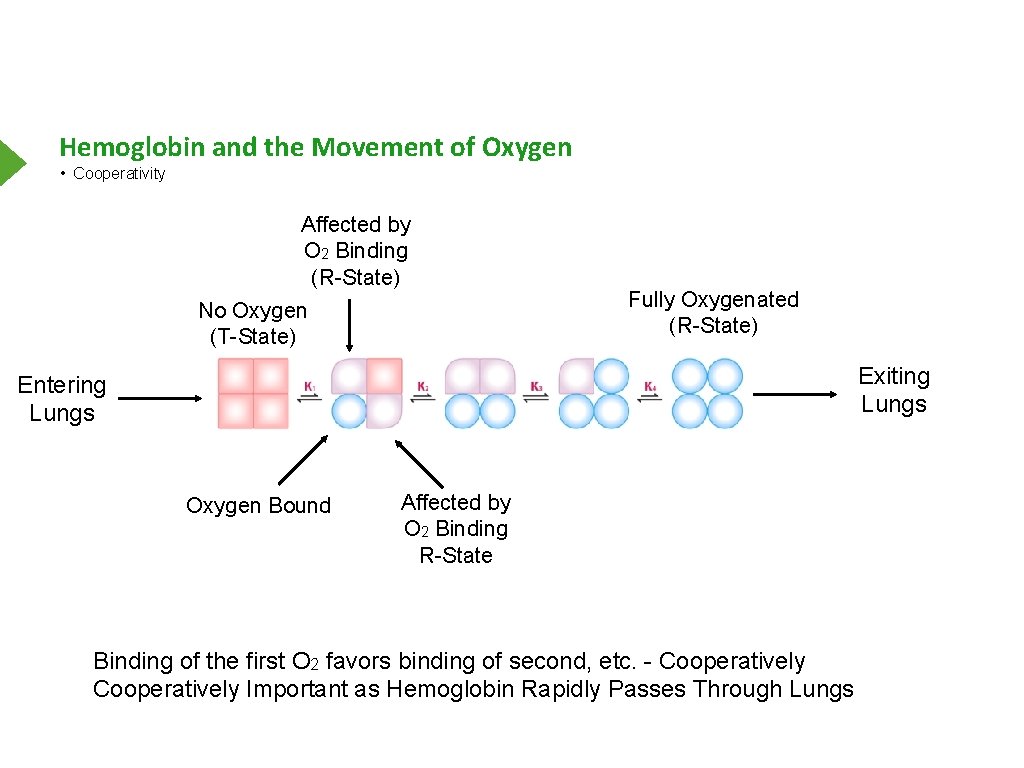
Hemoglobin and the Movement of Oxygen • Cooperativity Affected by O 2 Binding (R-State) No Oxygen (T-State) Fully Oxygenated (R-State) Exiting Lungs Entering Lungs Oxygen Bound Affected by O 2 Binding R-State Binding of the first O 2 favors binding of second, etc. - Cooperatively Important as Hemoglobin Rapidly Passes Through Lungs
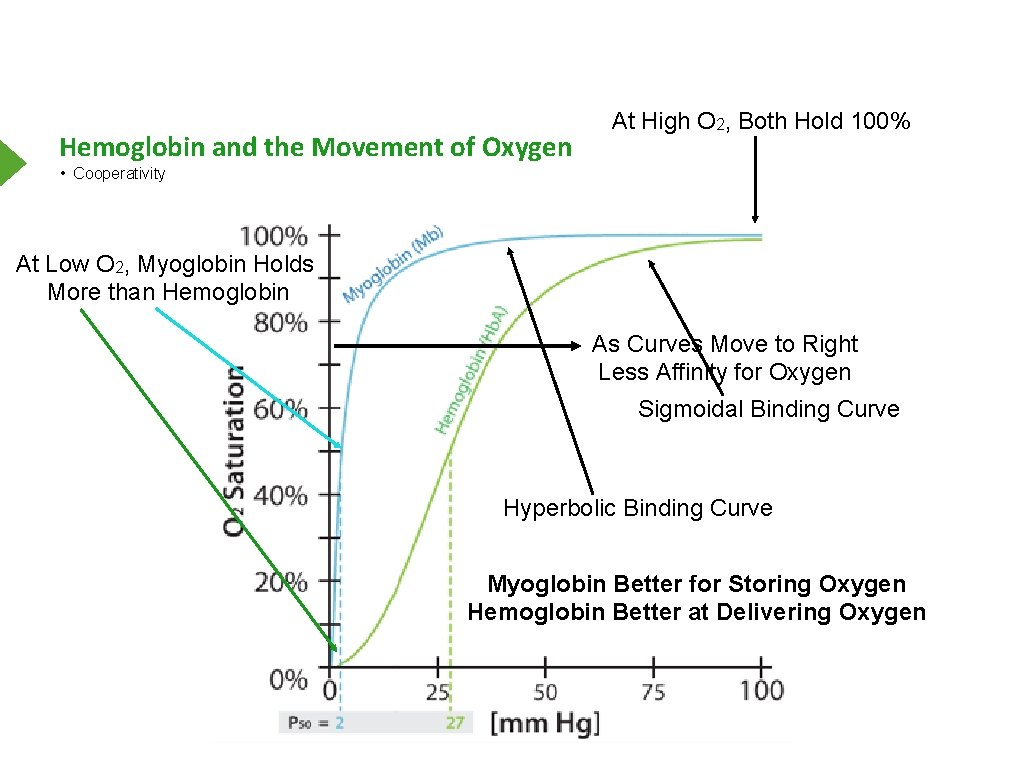
Hemoglobin and the Movement of Oxygen At High O 2, Both Hold 100% • Cooperativity At Low O 2, Myoglobin Holds More than Hemoglobin As Curves Move to Right Less Affinity for Oxygen Sigmoidal Binding Curve Hyperbolic Binding Curve Myoglobin Better for Storing Oxygen Hemoglobin Better at Delivering Oxygen
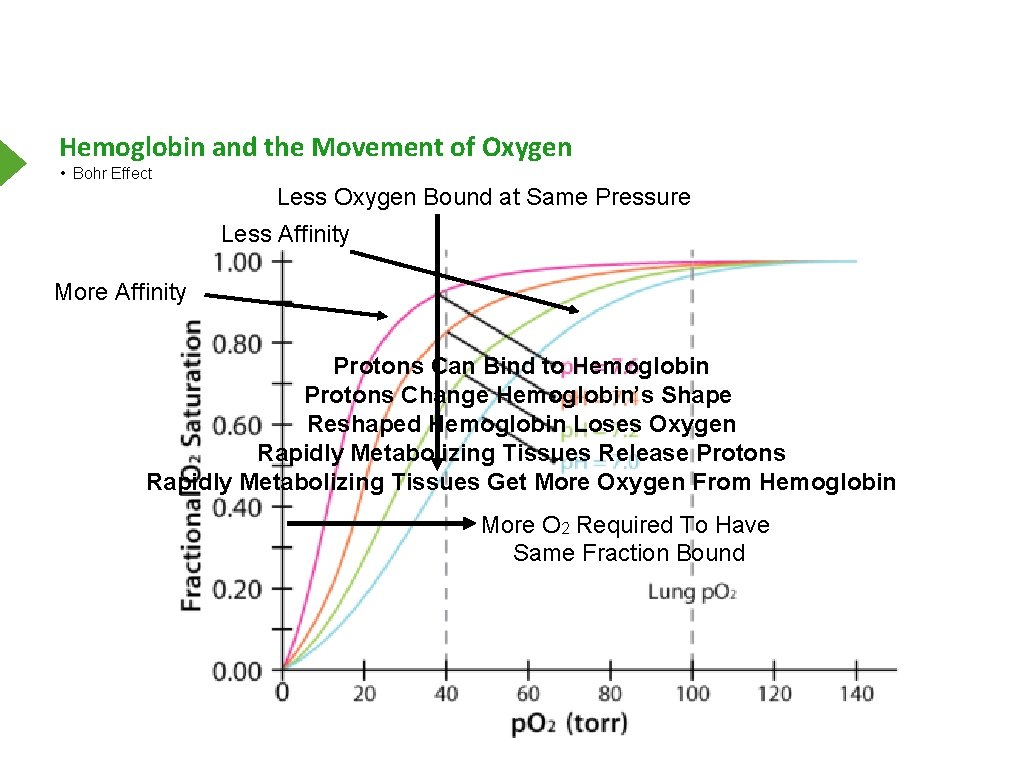
Hemoglobin and the Movement of Oxygen • Bohr Effect Less Oxygen Bound at Same Pressure Less Affinity More Affinity Protons Can Bind to Hemoglobin Protons Change Hemoglobin’s Shape Reshaped Hemoglobin Loses Oxygen Rapidly Metabolizing Tissues Release Protons Rapidly Metabolizing Tissues Get More Oxygen From Hemoglobin More O 2 Required To Have Same Fraction Bound
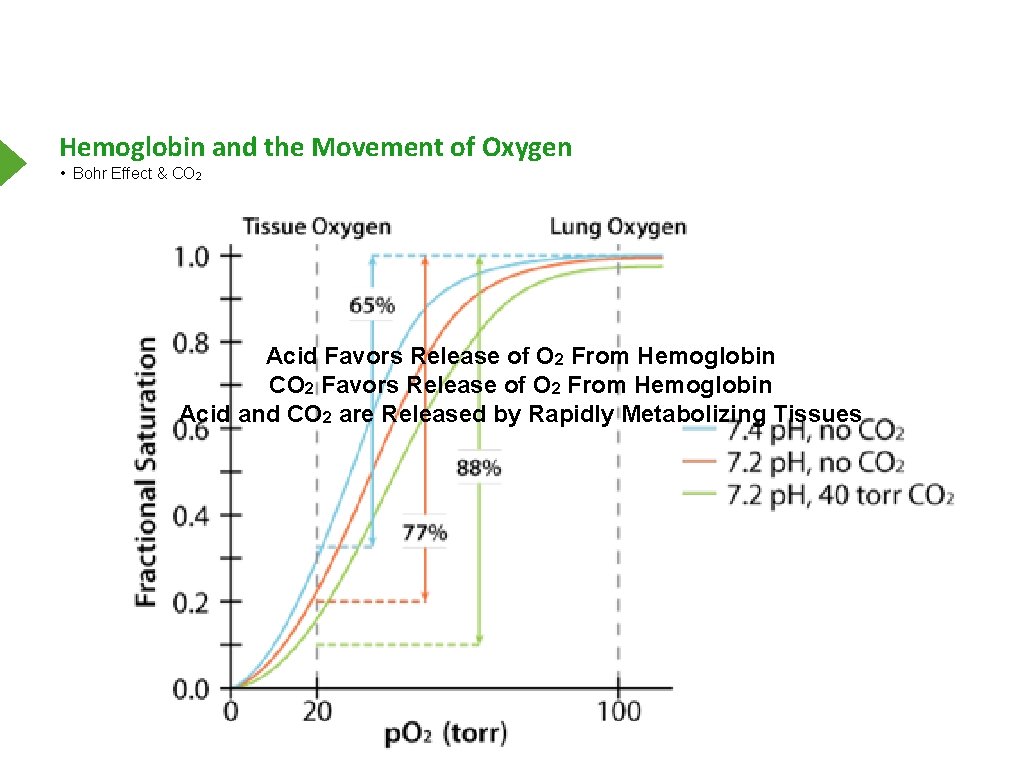
Hemoglobin and the Movement of Oxygen • Bohr Effect & CO 2 Acid Favors Release of O 2 From Hemoglobin CO 2 Favors Release of O 2 From Hemoglobin Acid and CO 2 are Released by Rapidly Metabolizing Tissues
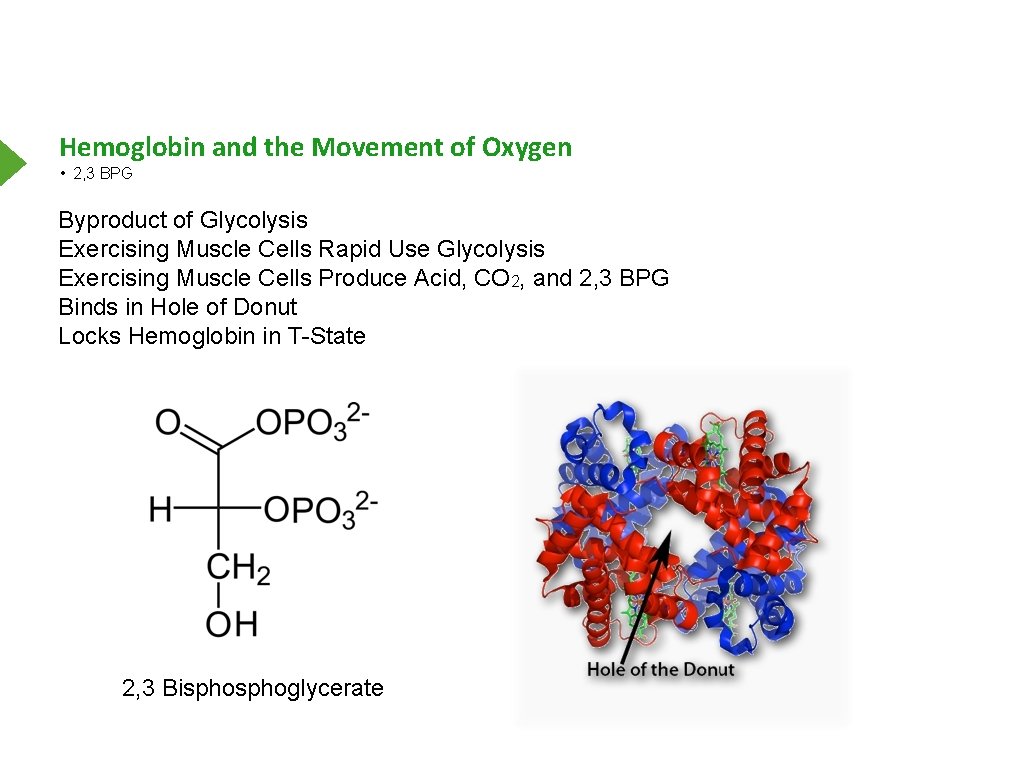
Hemoglobin and the Movement of Oxygen • 2, 3 BPG Byproduct of Glycolysis Exercising Muscle Cells Rapid Use Glycolysis Exercising Muscle Cells Produce Acid, CO 2, and 2, 3 BPG Binds in Hole of Donut Locks Hemoglobin in T-State 2, 3 Bisphoglycerate
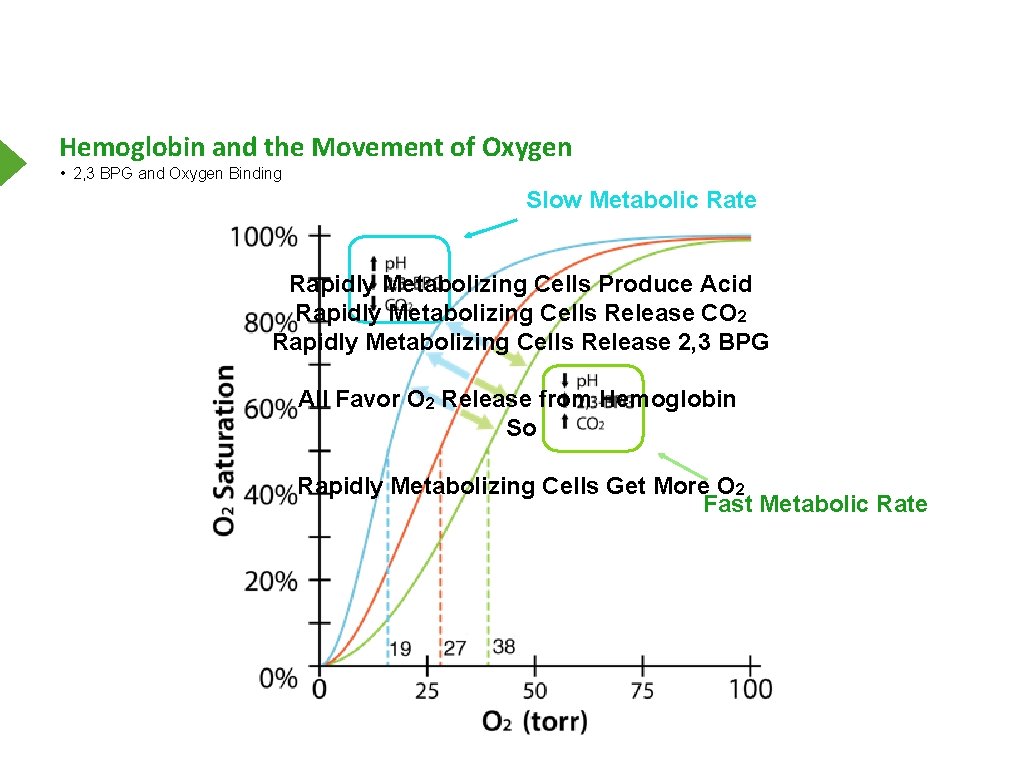
Hemoglobin and the Movement of Oxygen • 2, 3 BPG and Oxygen Binding Slow Metabolic Rate Rapidly Metabolizing Cells Produce Acid Rapidly Metabolizing Cells Release CO 2 Rapidly Metabolizing Cells Release 2, 3 BPG All Favor O 2 Release from Hemoglobin So Rapidly Metabolizing Cells Get More O 2 Fast Metabolic Rate

Hemoglobin and the Movement of Oxygen • 2, 3 BPG and Smoking 2, 3 BPG Big Concern for Smokers Blood of Smokers has High Levels of 2, 3 BPG Hemoglobin Gets Locked in T-state in Passage Through Lungs Oxygen Carrying Capacity of Blood Reduced Carbon Monoxide Levels Also Higher in Smokers 2, 3 Bisphoglycerate
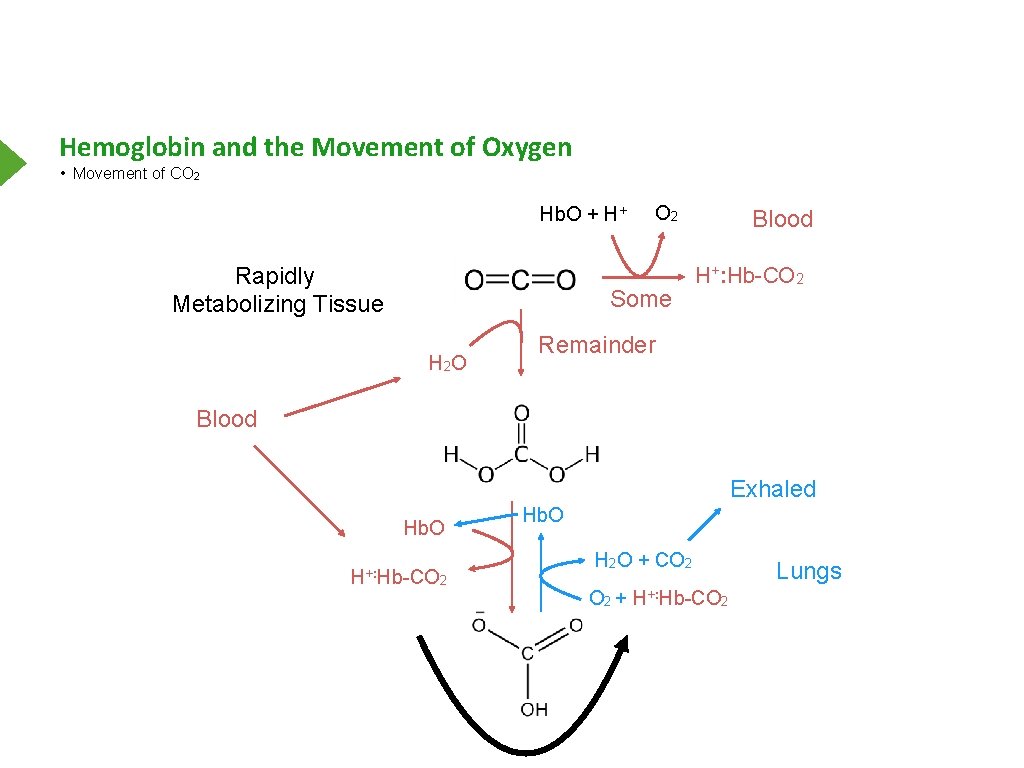
Hemoglobin and the Movement of Oxygen • Movement of CO 2 Hb. O + H+ Rapidly Metabolizing Tissue O 2 Some H 2 O Blood H+: Hb-CO 2 Remainder Blood Exhaled Hb. O H+: Hb-CO Hb. O H 2 O + CO 2 2 O 2 + H+: Hb-CO 2 Lungs
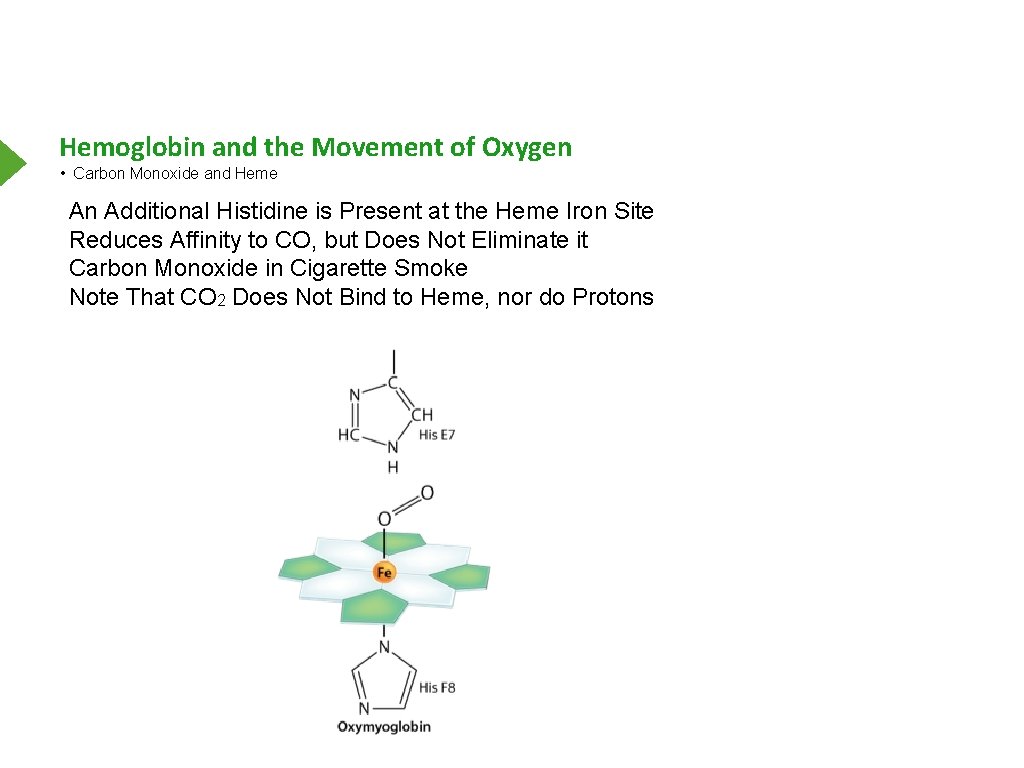
Hemoglobin and the Movement of Oxygen • Carbon Monoxide and Heme An Additional Histidine is Present at the Heme Iron Site Reduces Affinity to CO, but Does Not Eliminate it Carbon Monoxide in Cigarette Smoke Note That CO 2 Does Not Bind to Heme, nor do Protons
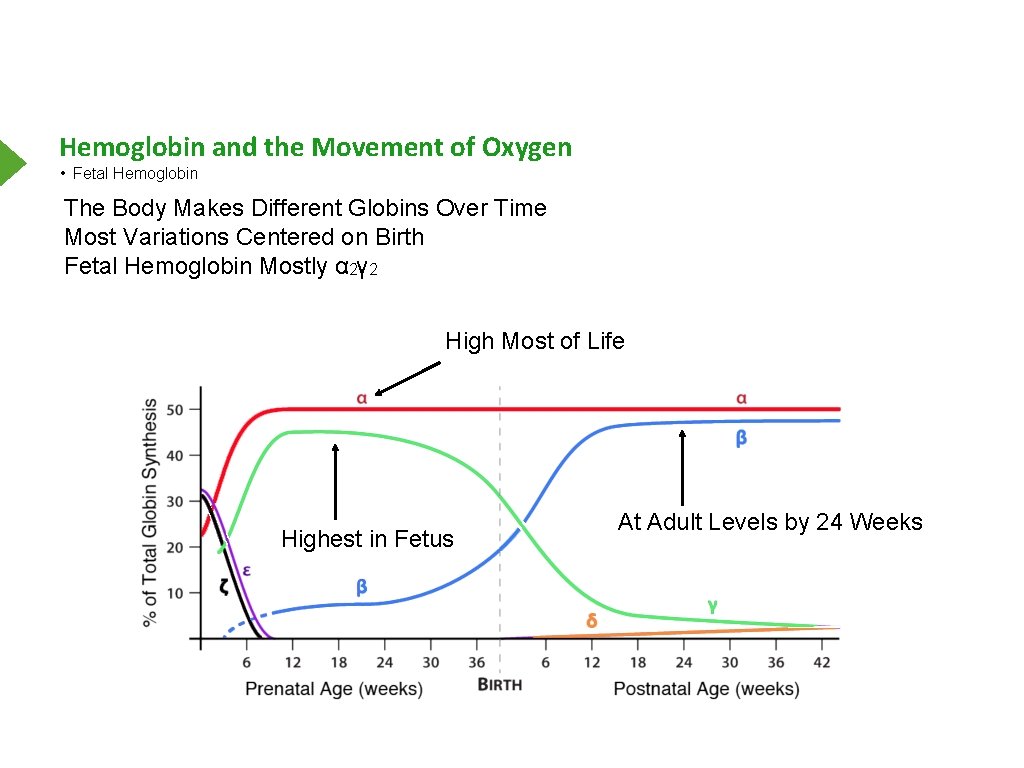
Hemoglobin and the Movement of Oxygen • Fetal Hemoglobin The Body Makes Different Globins Over Time Most Variations Centered on Birth Fetal Hemoglobin Mostly α 2γ 2 High Most of Life Highest in Fetus At Adult Levels by 24 Weeks
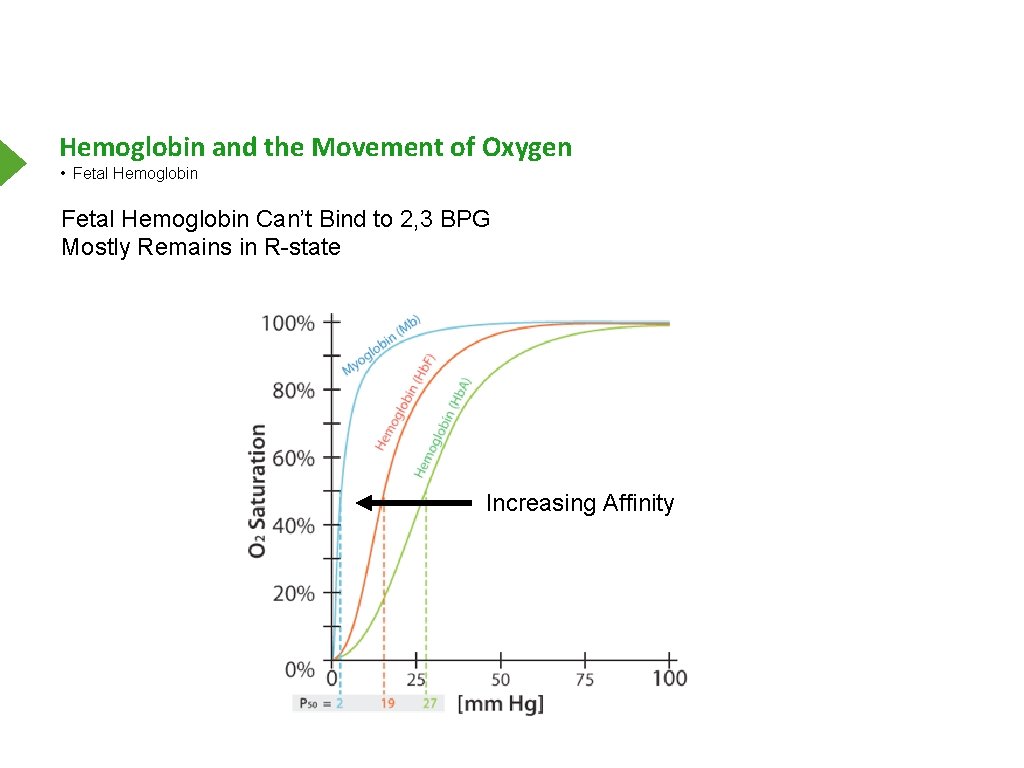
Hemoglobin and the Movement of Oxygen • Fetal Hemoglobin Can’t Bind to 2, 3 BPG Mostly Remains in R-state Increasing Affinity
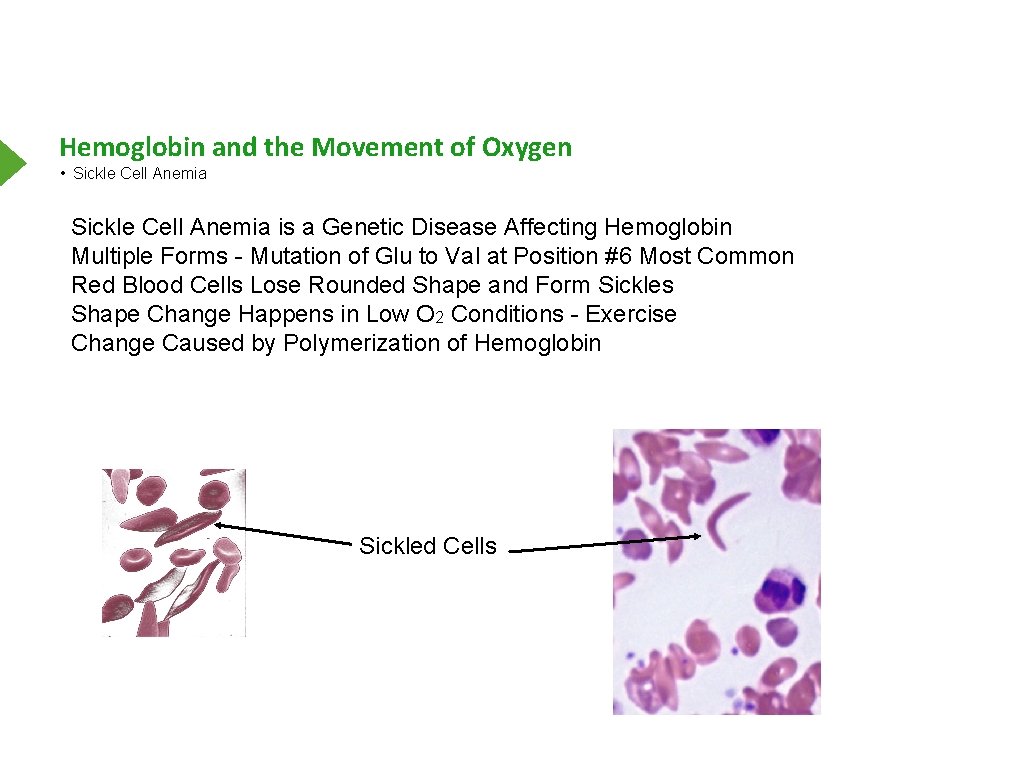
Hemoglobin and the Movement of Oxygen • Sickle Cell Anemia is a Genetic Disease Affecting Hemoglobin Multiple Forms - Mutation of Glu to Val at Position #6 Most Common Red Blood Cells Lose Rounded Shape and Form Sickles Shape Change Happens in Low O 2 Conditions - Exercise Change Caused by Polymerization of Hemoglobin Sickled Cells
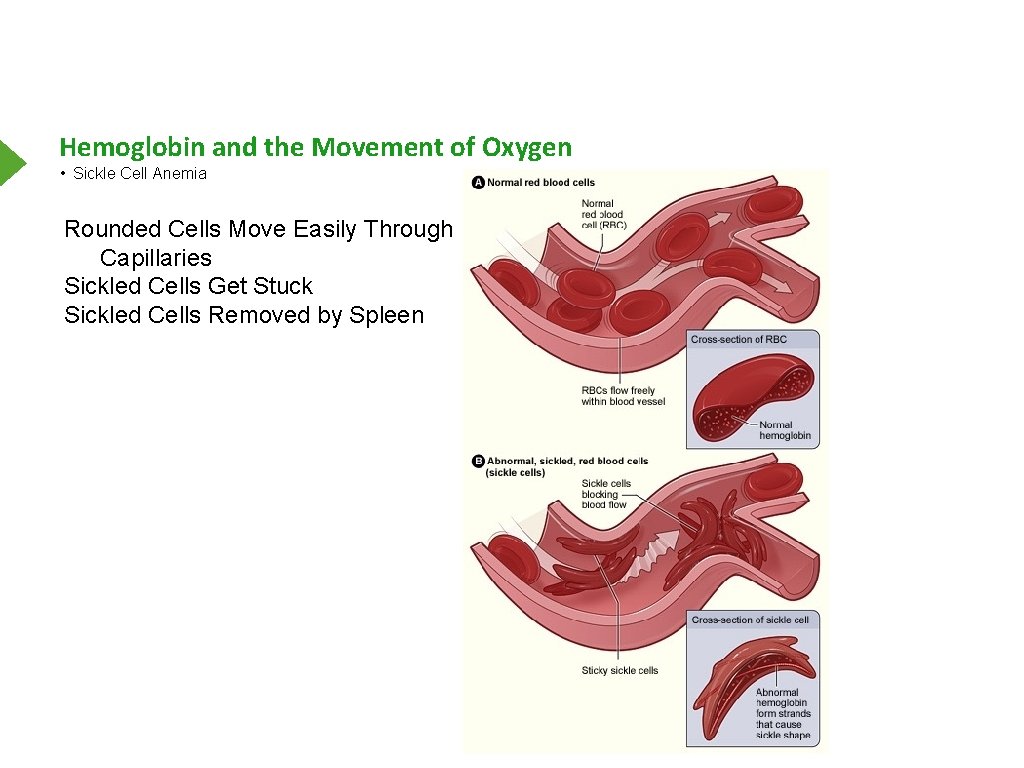
Hemoglobin and the Movement of Oxygen • Sickle Cell Anemia Rounded Cells Move Easily Through Capillaries Sickled Cells Get Stuck Sickled Cells Removed by Spleen
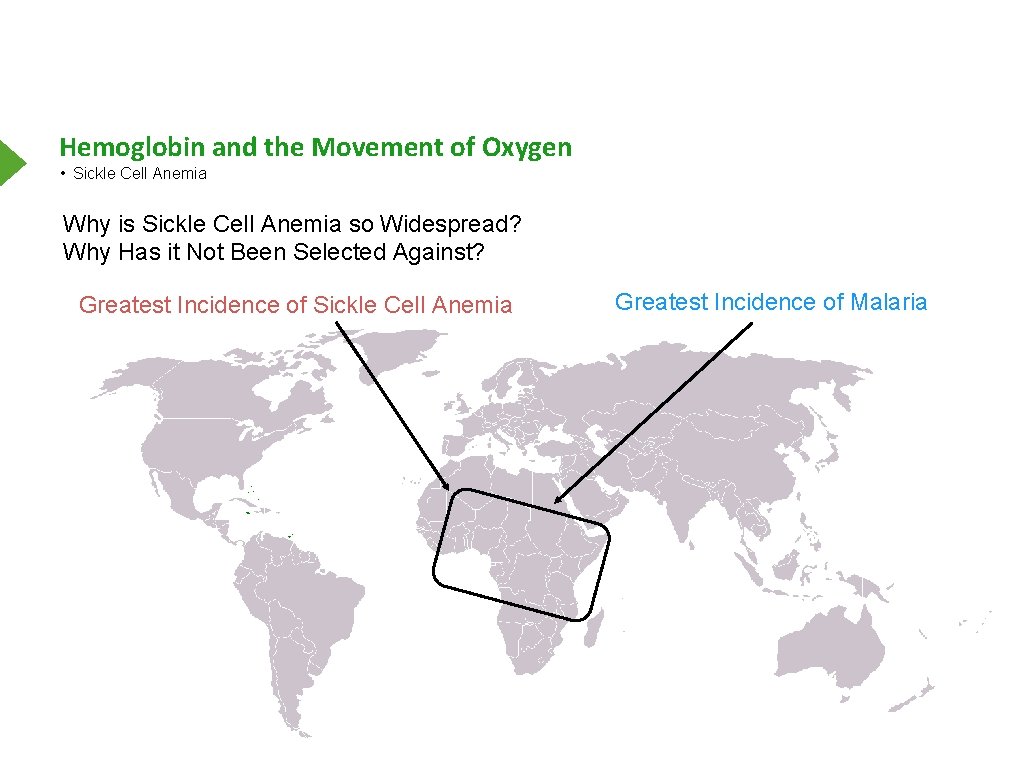
Hemoglobin and the Movement of Oxygen • Sickle Cell Anemia Why is Sickle Cell Anemia so Widespread? Why Has it Not Been Selected Against? Greatest Incidence of Sickle Cell Anemia Greatest Incidence of Malaria
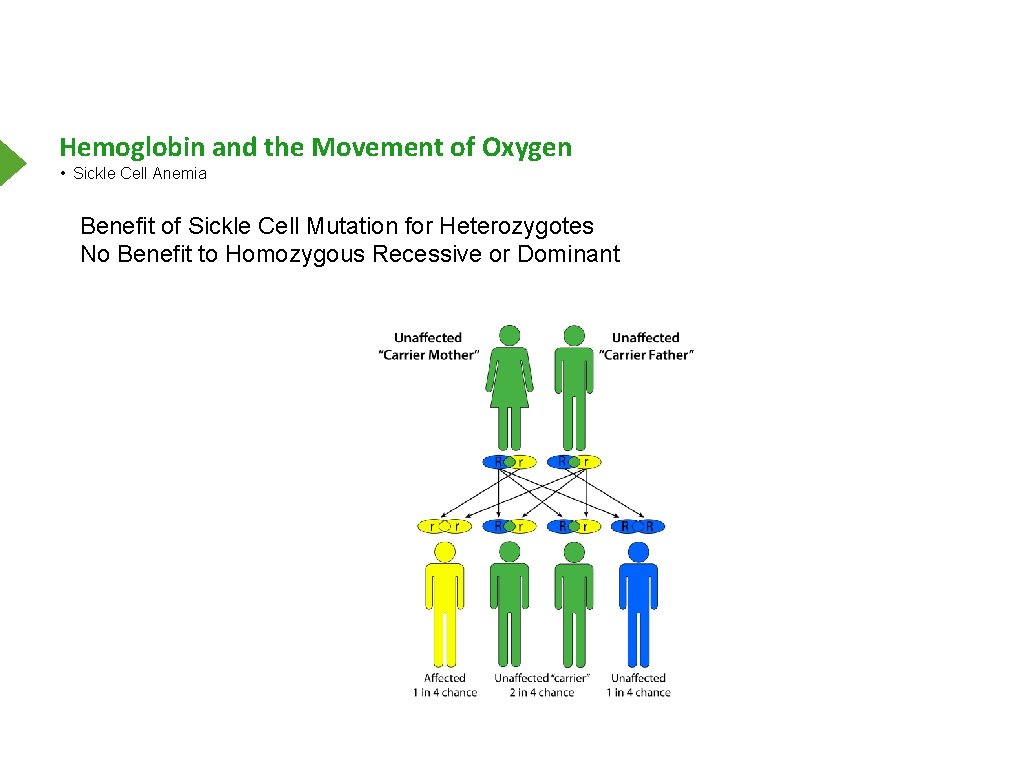
Hemoglobin and the Movement of Oxygen • Sickle Cell Anemia Benefit of Sickle Cell Mutation for Heterozygotes No Benefit to Homozygous Recessive or Dominant

Hemoglobin and the Movement of Oxygen • Summary Animals Have Widely Varying O 2 Needs ATP Generated Much More Efficiently in Presence of O 2 Hemoglobin and Myoglobin are Related, but Have Different Functions Hemoglobin has Four Subunits and Hemes. Myoglobin has One of Each Bind of O 2 by Heme’s Iron Pulls up on a Histidine and Change’s Hemoglobin’s Shape Changing Hemoglobin’s Shape Converts Hemoglobin from T-state to R-state Binds Oxygen Better. T-state Releases O 2 Better In the Bohr Effect, Binding of CO 2 and H+ Favors O 2 Release The Bohr Effect Explains How Oxygen and CO 2 Exchanged in Lungs 2, 3 BPG is Produced by Rapidly Metabolizing Cells. It too Favors O 2 Release Fetal Hemoglobin Can’t Bind 2, 3 BPG and has Greater O 2 Affinity Than Adult Hemoglobin Sickle Cell Anemia (SCA) is a Genetic Disease of Hemoglobin In Low O 2 Concentration, Red Blood Cells of SCA Sufferers Form Sickle Shapes Sickled Cells Stick in Capillaries and Can be Fatal People Heterozygous for the Mutated Gene Survive Malaria Better Than Others
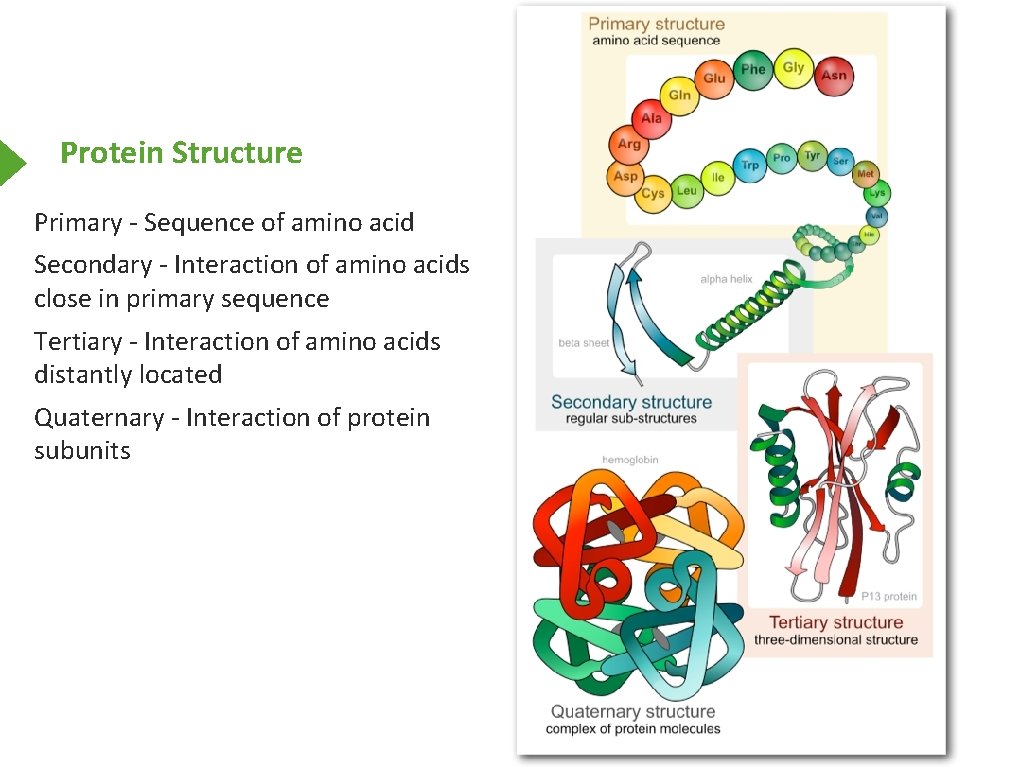
Protein Structure Primary - Sequence of amino acid Secondary - Interaction of amino acids close in primary sequence Tertiary - Interaction of amino acids distantly located Quaternary - Interaction of protein subunits
- Slides: 23