Hematopoietic stem cell transplantation Prof Dr Bar Kukonmaz

Hematopoietic stem cell transplantation Prof Dr Barış Kuşkonmaz HÜTF, Ped. Haematology

Objectives § Definition and classification § Indications of transplantation § Assessment of patient and donor before transplantation § Source of hematopoietic stem cells (HSC) § Hematopoietic stem cell transplantation process § Complications of transplantation: Acute graft versus host disease (GVHD) Infections Venoocclussive disease (VOD) The other early complications Chronic GVHD The other late complications

Definitions and classification

Characteristics of stem cells § Self proliferation (high capacity of proliferation) § Long term survival § Undifferentiated cells § Multi/pluri differentiation capacity § Clonality
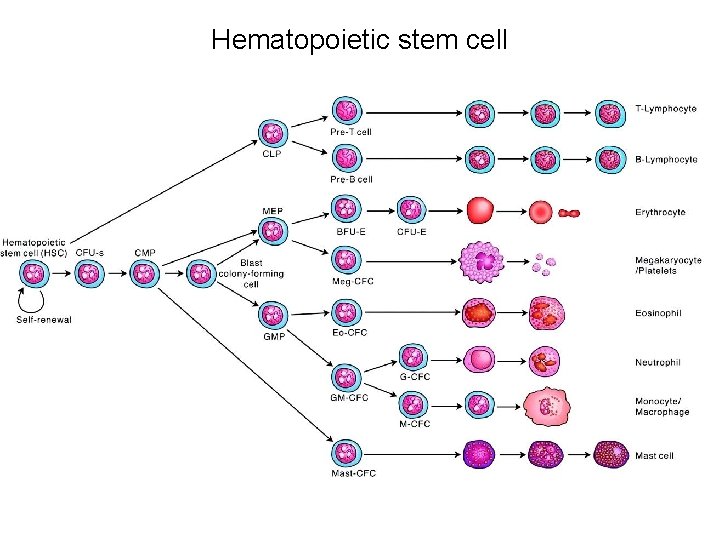
Hematopoietic stem cell
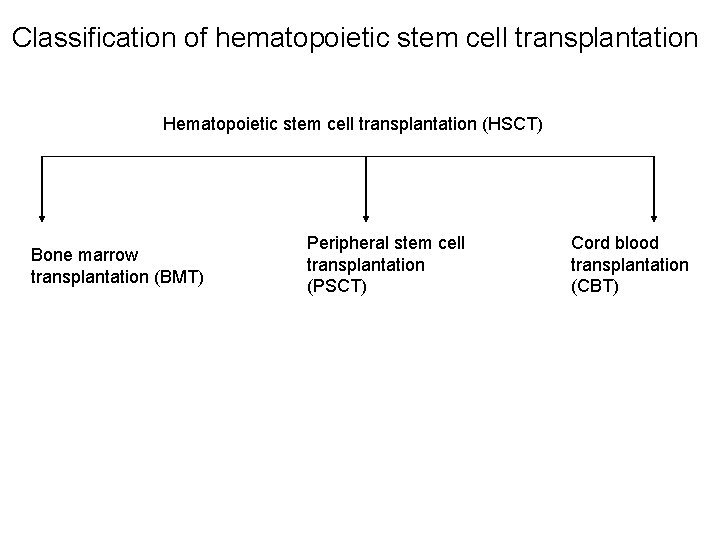
Classification of hematopoietic stem cell transplantation Hematopoietic stem cell transplantation (HSCT) Bone marrow transplantation (BMT) Peripheral stem cell transplantation (PSCT) Cord blood transplantation (CBT)
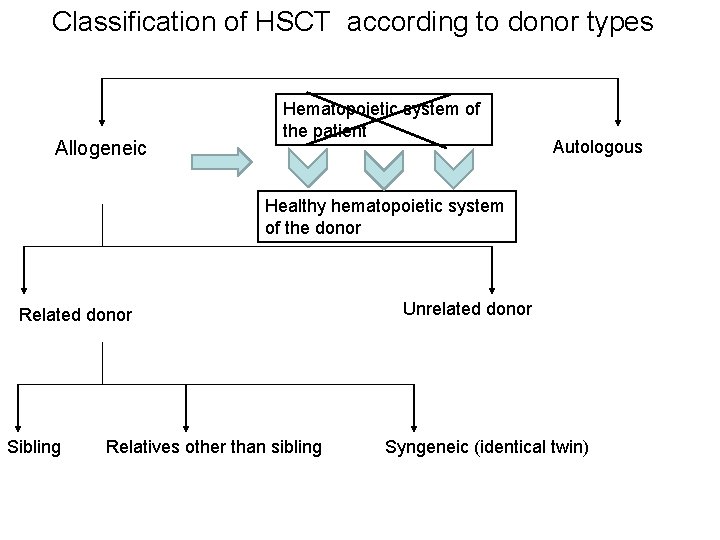
Classification of HSCT according to donor types Allogeneic Hematopoietic system of the patient Autologous Healthy hematopoietic system of the donor Related donor Sibling Relatives other than sibling Unrelated donor Syngeneic (identical twin)

Definition Engraftment: achievement of donor hematopoiesis Neutrophil engraftment: first of three consecutive days with a neutrophil count above 500/mm 3 Platelet engraftment: platelet count higher than 20. 000/mm 3 with no transfusion requirements for at least 7 days Graft failure: the absence of hematopoietic recovery at day 60 and autologous hematopoietic reconstruction were considered engraftment failure Primary graft failure : Engraftment never occurs Secondary graft failure: Same criteria as primary graft failure but occurring after initial engraftment

Donor lymphocyte infusion Definition: infusion of donor’s lymphocytes The goals of the procedure • Enhancement of engraftment • Prevention of rejection • Prevention and treatment of disease relapse
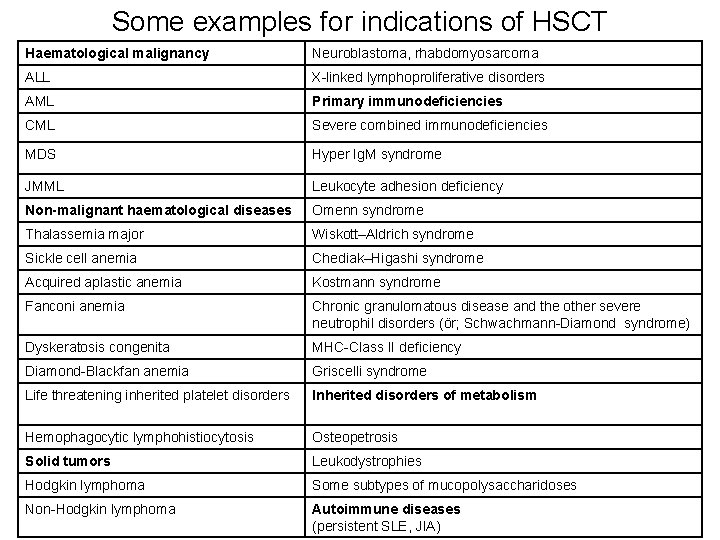
Some examples for indications of HSCT Haematological malignancy Neuroblastoma, rhabdomyosarcoma ALL X-linked lymphoproliferative disorders AML Primary immunodeficiencies CML Severe combined immunodeficiencies MDS Hyper Ig. M syndrome JMML Leukocyte adhesion deficiency Non-malignant haematological diseases Omenn syndrome Thalassemia major Wiskott–Aldrich syndrome Sickle cell anemia Chediak–Higashi syndrome Acquired aplastic anemia Kostmann syndrome Fanconi anemia Chronic granulomatous disease and the other severe neutrophil disorders (ör; Schwachmann-Diamond syndrome) Dyskeratosis congenita MHC-Class II deficiency Diamond-Blackfan anemia Griscelli syndrome Life threatening inherited platelet disorders Inherited disorders of metabolism Hemophagocytic lymphohistiocytosis Osteopetrosis Solid tumors Leukodystrophies Hodgkin lymphoma Some subtypes of mucopolysaccharidoses Non-Hodgkin lymphoma Autoimmune diseases (persistent SLE, JIA)
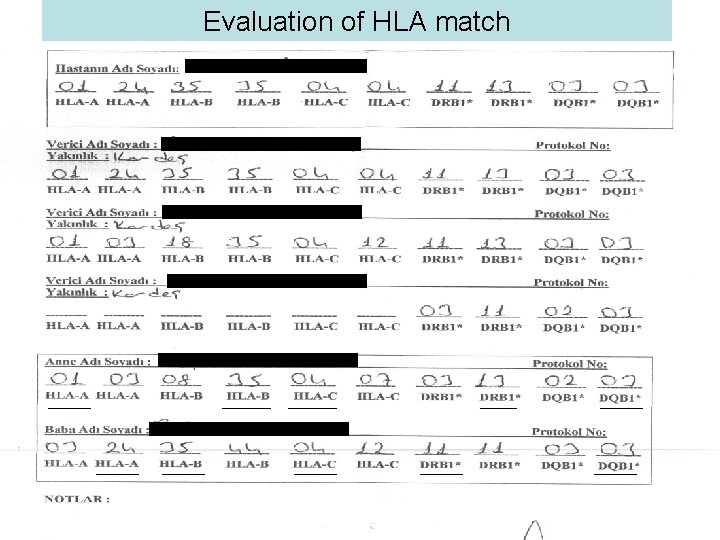
Evaluation of HLA match
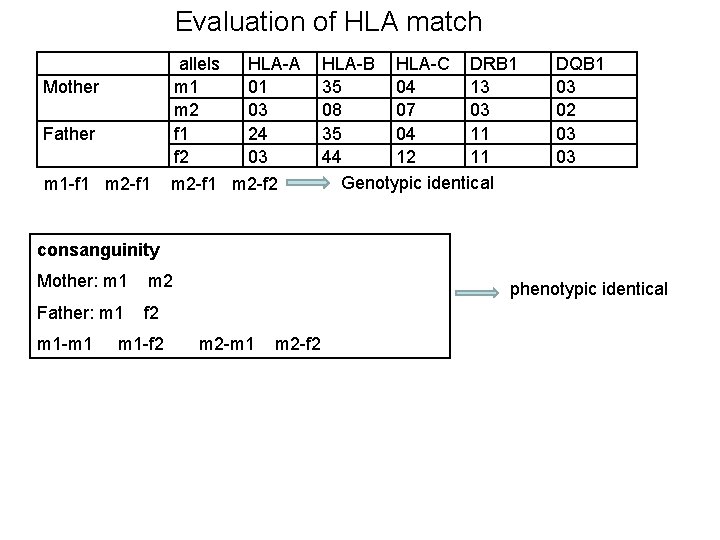
Evaluation of HLA match allels m 1 m 2 f 1 f 2 Mother Father m 1 -f 1 m 2 -f 1 HLA-A 01 03 24 03 m 2 -f 1 m 2 -f 2 HLA-B HLA-C DRB 1 35 04 13 08 07 03 35 04 11 44 12 11 Genotypic identical DQB 1 03 02 03 03 consanguinity Mother: m 1 m 2 Father: m 1 f 2 m 1 -m 1 m 1 -f 2 phenotypic identical m 2 -m 1 m 2 -f 2

Laboratory evalaution prior to transplantation *CBC, liver and kidney function tests, blood glucose, blood group/anti-A, anti-B *Serological testing (HBV, HCV, CMV, EBV, HIV, VDRL HSVI, HSVII, toxoplasma, salmonella, brucella) *INR, a. PTT, fibrinogen *Chest X ray *PPD *Electrocardiography Echocardiography Pulmonary function test Ig. A, Ig. G, Ig. M levels, lymphocyte subsets Abdominal USG Evaluation of primary disase Medical history of donor and test for primary disease if indicated Required consultations *Required tests for donor
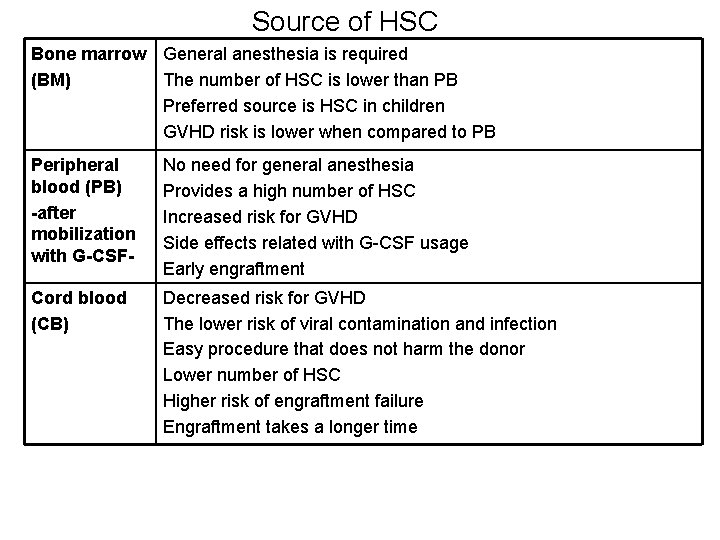
Source of HSC Bone marrow General anesthesia is required (BM) The number of HSC is lower than PB Preferred source is HSC in children GVHD risk is lower when compared to PB Peripheral blood (PB) -after mobilization with G-CSF- No need for general anesthesia Provides a high number of HSC Increased risk for GVHD Side effects related with G-CSF usage Early engraftment Cord blood (CB) Decreased risk for GVHD The lower risk of viral contamination and infection Easy procedure that does not harm the donor Lower number of HSC Higher risk of engraftment failure Engraftment takes a longer time

Conditioning regimen and GVHD prophylaxis 1) Myeloablation: Provide stem cell niches in the host BM for the new stem cell (Busulfan, treosulphan, melphalan, total body irradiation) 2) Immunosupression (IS): Provide sufficient IS to ensure engraftment and to prevent both rejection and GVHD (cyclophosphamide, fludaribine, thiotepa, ATG, alemtuzumab) 3) Disease eradication: Eradicate the hematologic malignancy in case of malignant indication for HSCT. Myeloablative conditioning: Intensive regimen, more side effects Non-myeloablative condtioning: Non-intensive regimen, less side effects -Disease driven (Fanconi anemia, acquired aplastic anemia) -In patients who cannot tolerate myeloablative conditioning Reduced intensity: The regimen that is not classified as myeloablative or nonmyeloablative GVHD prophylaxis: Cyclosporine A+methotrexate

Manipulation of graft in haploidentical transplantation Increased GVHD risk with haploidentical HSCT Stem cell selection/depletion -CD 34 selection -T cell depletion -T cell subgroup depletion (α, β) ±CD 19 depletion -Graft failure -Decreased graft versus leukemia effect/ increased risk of relapse -Reactivation of viral infection -Increased risk for posttransplant lymphoproliferative disorders
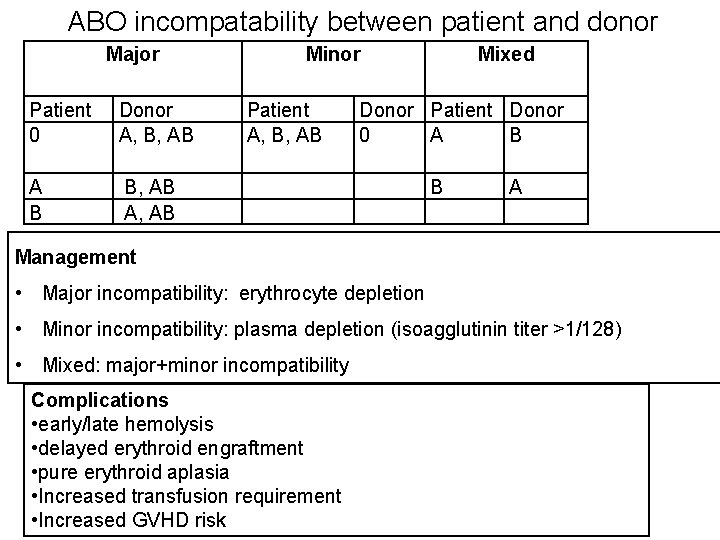
ABO incompatability between patient and donor Major Patient 0 Donor A, B, AB A B B, AB A, AB Minor Patient A, B, AB Mixed Donor Patient Donor 0 A B B A Management • Major incompatibility: erythrocyte depletion • Minor incompatibility: plasma depletion (isoagglutinin titer >1/128) • Mixed: major+minor incompatibility Complications • early/late hemolysis • delayed erythroid engraftment • pure erythroid aplasia • Increased transfusion requirement • Increased GVHD risk
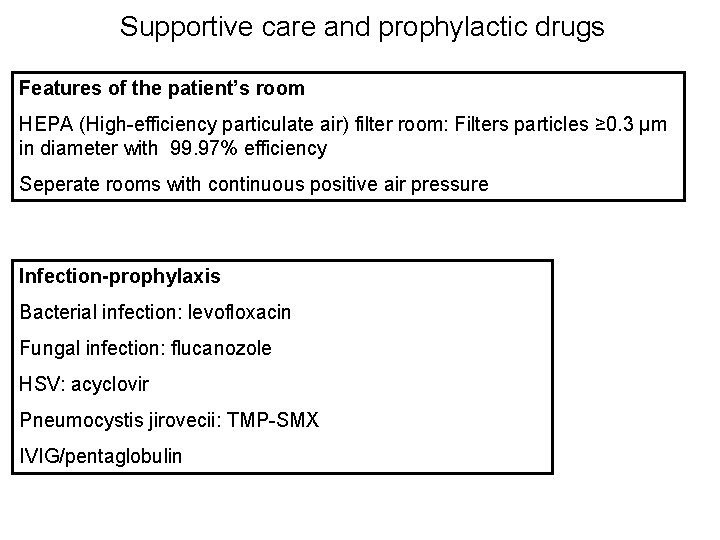
Supportive care and prophylactic drugs Features of the patient’s room HEPA (High-efficiency particulate air) filter room: Filters particles ≥ 0. 3 μm in diameter with 99. 97% efficiency Seperate rooms with continuous positive air pressure Infection-prophylaxis Bacterial infection: levofloxacin Fungal infection: flucanozole HSV: acyclovir Pneumocystis jirovecii: TMP-SMX IVIG/pentaglobulin

Supportive care and prophylactic drugs VOD prophylaxis Defibrotide Ursodeoxycholic acid Enoxaparine Vitamin E • Protection of GIS mucosa • Anticonvulsive prophylaxis (busulfan) • Diuresis • Analgesia • Antiemetics • Vitamin/mineral (multivitamins, Vitamin K, folic acid, Mg) • Oral care • Nutrition: TPN in case of decreased oral intake • Growth factor (G-CSF)
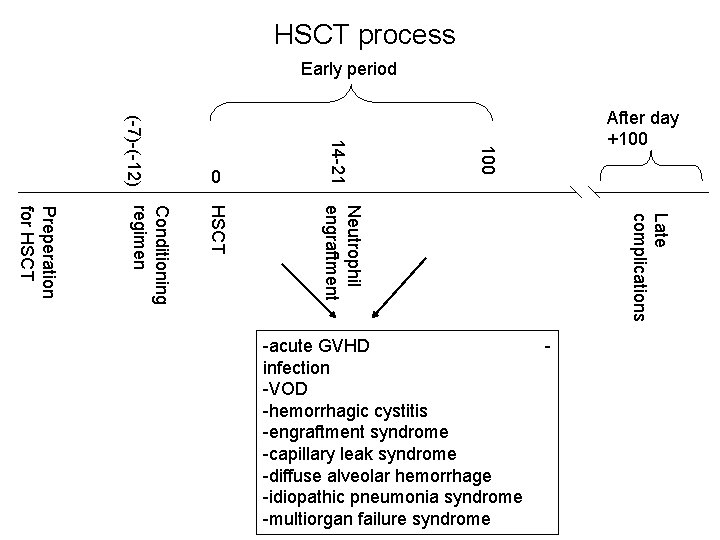
HSCT process Early period 100 14 -21 (-7)-(-12) 0 After day +100 Late complications Neutrophil engraftment HSCT Conditioning regimen Preperation for HSCT -acute GVHD infection -VOD -hemorrhagic cystitis -engraftment syndrome -capillary leak syndrome -diffuse alveolar hemorrhage -idiopathic pneumonia syndrome -multiorgan failure syndrome -
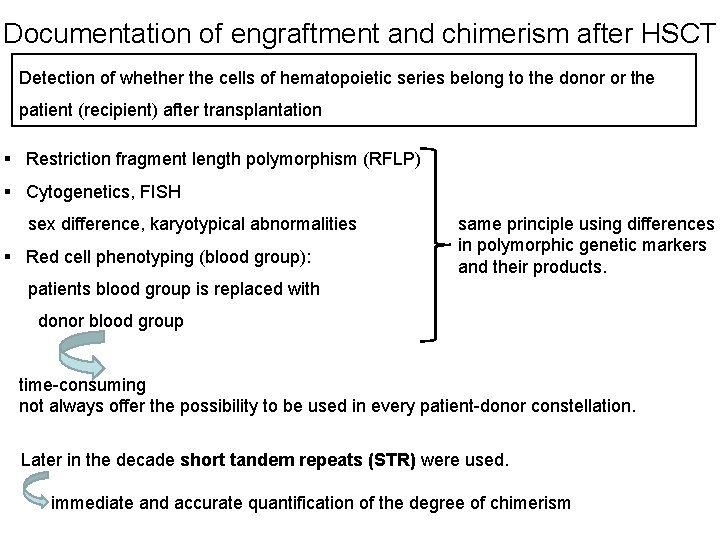
Documentation of engraftment and chimerism after HSCT Detection of whether the cells of hematopoietic series belong to the donor or the patient (recipient) after transplantation § Restriction fragment length polymorphism (RFLP) § Cytogenetics, FISH sex difference, karyotypical abnormalities § Red cell phenotyping (blood group): same principle using differences in polymorphic genetic markers and their products. patients blood group is replaced with donor blood group time-consuming not always offer the possibility to be used in every patient-donor constellation. Later in the decade short tandem repeats (STR) were used. immediate and accurate quantification of the degree of chimerism

Complications of HSCT
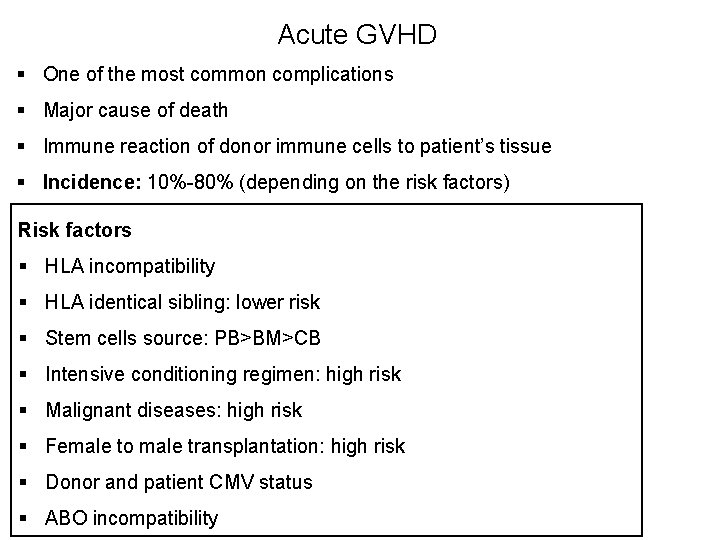
Acute GVHD § One of the most common complications § Major cause of death § Immune reaction of donor immune cells to patient’s tissue § Incidence: 10%-80% (depending on the risk factors) Risk factors § HLA incompatibility § HLA identical sibling: lower risk § Stem cells source: PB>BM>CB § Intensive conditioning regimen: high risk § Malignant diseases: high risk § Female to male transplantation: high risk § Donor and patient CMV status § ABO incompatibility

Definition of acute GVHD Classical acute GVHD: before day +100 Definition (in the absence of chronic GVHD) Persistent, recurrent or late acute GVHD: After day +100 In general, after cessation of immunsuprresive treatment or donor lymphocyte infusion
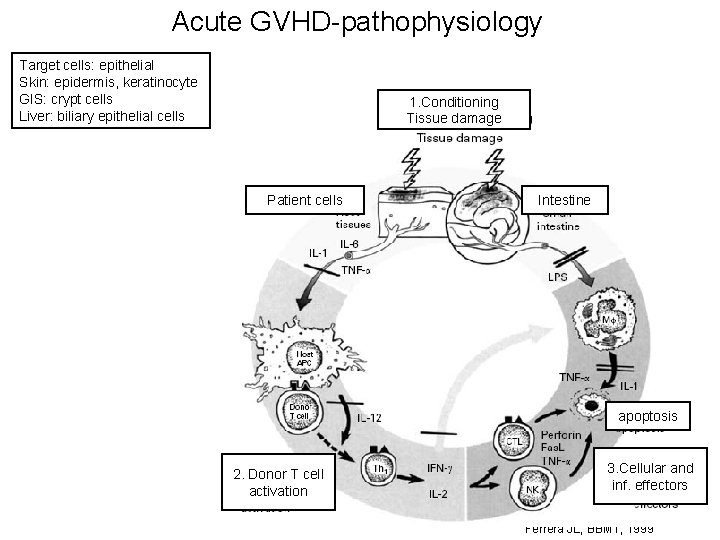
Acute GVHD-pathophysiology Target cells: epithelial Skin: epidermis, keratinocyte GIS: crypt cells Liver: biliary epithelial cells 1. Conditioning Tissue damage Patient cells Intestine apoptosis 2. Donor T cell activation 3. Cellular and inf. effectors Ferrera JL, BBMT, 1999
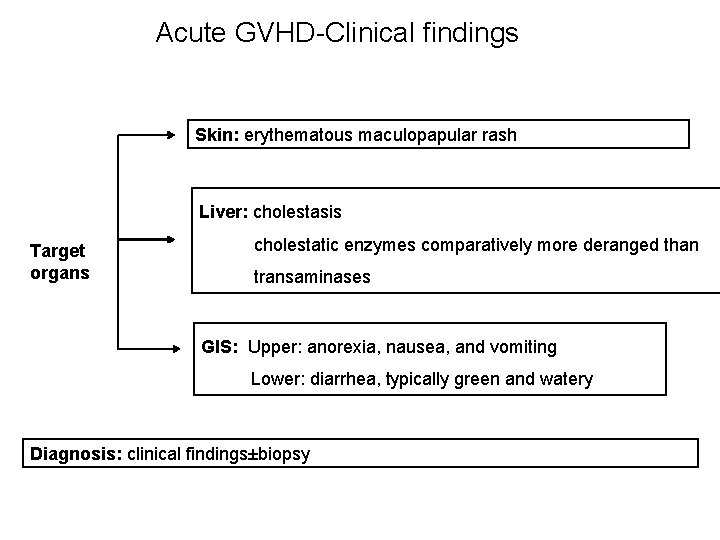
Acute GVHD-Clinical findings Skin: erythematous maculopapular rash Liver: cholestasis Target organs cholestatic enzymes comparatively more deranged than transaminases GIS: Upper: anorexia, nausea, and vomiting Lower: diarrhea, typically green and watery Diagnosis: clinical findings±biopsy
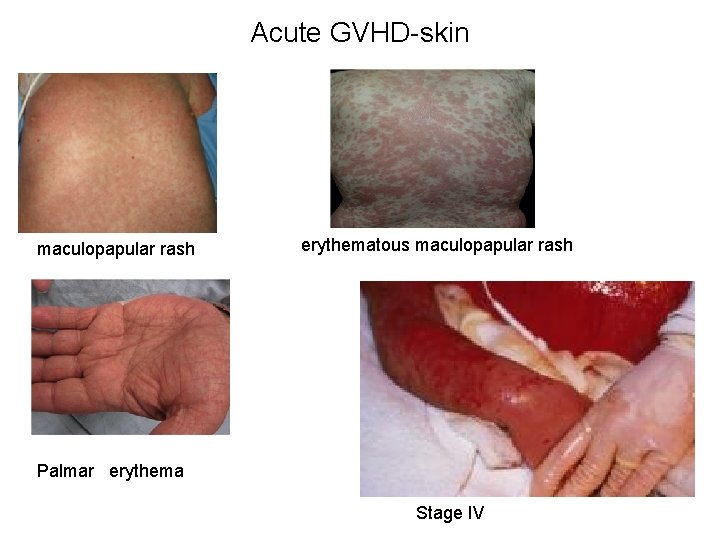
Acute GVHD-skin maculopapular rash erythematous maculopapular rash Palmar erythema Stage IV
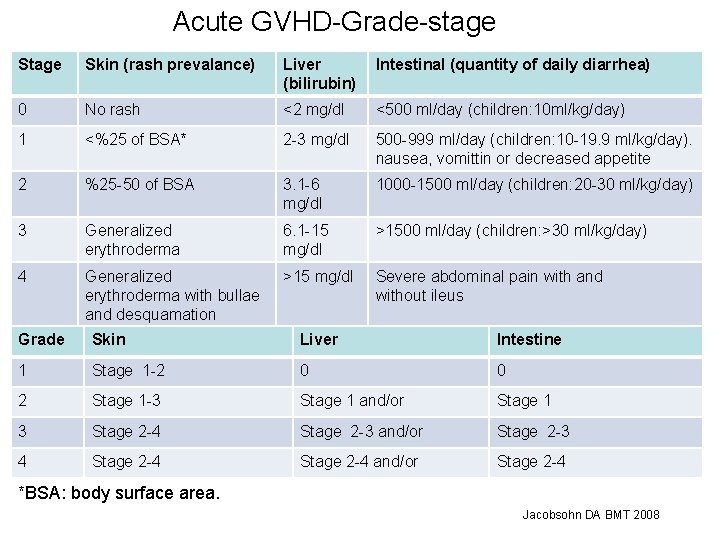
Acute GVHD-Grade-stage Skin (rash prevalance) Liver (bilirubin) Intestinal (quantity of daily diarrhea) 0 No rash <2 mg/dl <500 ml/day (children: 10 ml/kg/day) 1 <%25 of BSA* 2 -3 mg/dl 500 -999 ml/day (children: 10 -19. 9 ml/kg/day). nausea, vomittin or decreased appetite 2 %25 -50 of BSA 3. 1 -6 mg/dl 1000 -1500 ml/day (children: 20 -30 ml/kg/day) 3 Generalized erythroderma 6. 1 -15 mg/dl >1500 ml/day (children: >30 ml/kg/day) 4 Generalized erythroderma with bullae and desquamation >15 mg/dl Severe abdominal pain with and without ileus Grade Skin Liver Intestine 1 Stage 1 -2 0 0 2 Stage 1 -3 Stage 1 and/or Stage 1 3 Stage 2 -4 Stage 2 -3 and/or Stage 2 -3 4 Stage 2 -4 and/or Stage 2 -4 *BSA: body surface area. Jacobsohn DA BMT 2008

Acute GVHD- treatment Primary treatment : Corticosteroid+cyclosporine A Steroid resistance Secondary treatment: No standard management § mycophenolate mophetil, TNF α blockers, sirolimus, extracorporeal photopheresis (ECP), ATG, monoclonal antibody, …. . § As the grade of the acute GVHD increases the response rate decreases § Steroid resistance is associated with poor prognosis
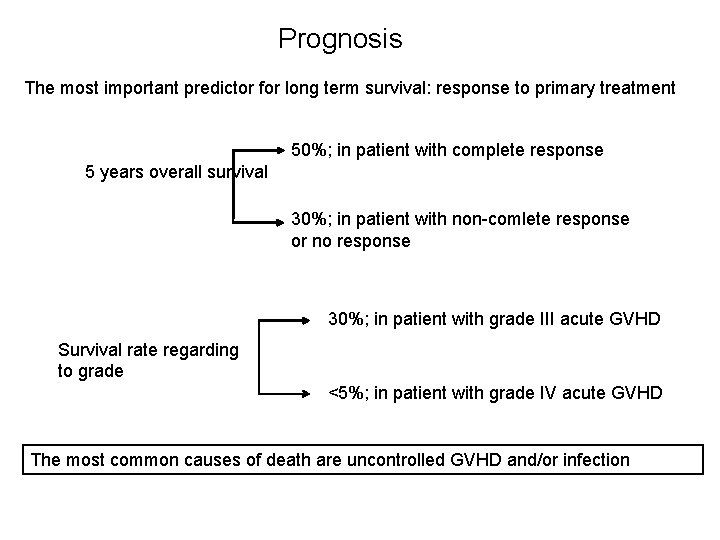
Prognosis The most important predictor for long term survival: response to primary treatment 50%; in patient with complete response 5 years overall survival 30%; in patient with non-comlete response or no response 30%; in patient with grade III acute GVHD Survival rate regarding to grade <5%; in patient with grade IV acute GVHD The most common causes of death are uncontrolled GVHD and/or infection

Infections
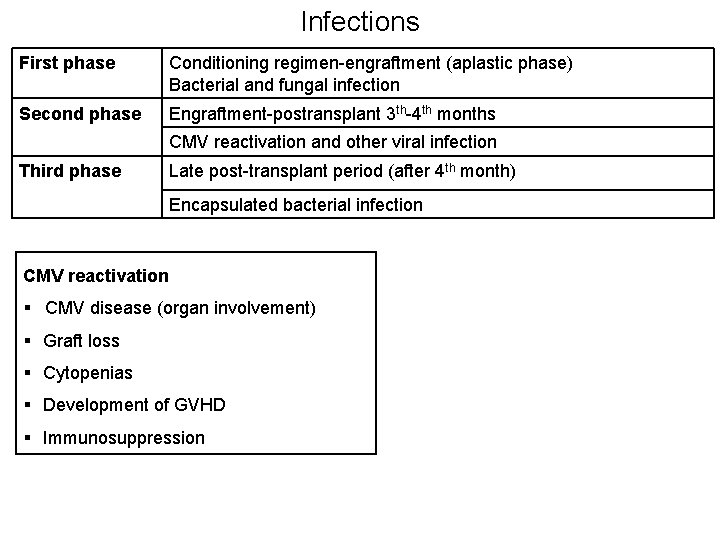
Infections First phase Conditioning regimen-engraftment (aplastic phase) Bacterial and fungal infection Second phase Engraftment-postransplant 3 th-4 th months CMV reactivation and other viral infection Third phase Late post-transplant period (after 4 th month) Encapsulated bacterial infection CMV reactivation § CMV disease (organ involvement) § Graft loss § Cytopenias § Development of GVHD § Immunosuppression

VOD

VOD § Conditioning regimen-related hepatic toxicity. § Incidence (pediatric patients): %27 -40 Baltimore Criteria* In the first 21 days after HSCT Bilirubin ≥ 2 mg/d. L and ≥ 2 of the following – Painful hepatomegaly – Weight gain >5% – Ascites *These symptoms/signs should not be attributable to other causes How to Confirm the Diagnosis? Transjugular hemodynamic study Transvenous liver biopsies Ultrasonography/Doppler USG Composite biomarkers: hyaluronic acid, vascular cell adhesion molecule-1, suppressor of tumorigenicity-2, angiopoietin-2, L-Ficolin

VOD-Prophylaxis and treatment § Defibrotide: The only agent that has established efficiency anti-thrombotic, anti-ischemic, anti-inflammatory, protection of endothelial cells, decreasing platelet activation and fibrinolytic effects. § Ursodeoxycholic acid, vitamin E, prostaglandin E 1, Heparin, antithrombin III, . .

Engraftment syndrome § Within 96 hours after neutrophil engraftment Major criteria: § Non-infectious fever § Skin rash (>25 of BSA) § Pulmonary infiltration (in the absence of fluid retention, emboli, CHF) Minor criteria: § Hepatic dysfunction, renal insufficiency, weight gain, encephalopathy Diagnosis: 3 major or 2 major 1 minor criteria Treatment: methylprednisolone

Chronic GVHD

Chronic GVHD §most relevant cause of late non-relapse morbidity and mortality §incidence of c. GVHD is 20– 40% in children §Chronic GVHD usually begins between 3 months and 2 years after HSCT, but earlier onset is possible § 40% of the children have the history of acute GVHD §Chronic GVHD can affect a number of organs and sytems §Chronic GVHD can imitate almost any autoimmune disease

Chronic GVHD-common organ involvement Skin (most frequently involved organ) § Diagnostic features: poikiloderma, lichen planus-like, lichen sclerosus-like, morphealike, and deep sclerotic eruptions § The other findings: Depigmentation and papulosquamous lesions or ichthyosis, keratosis pilaris, pigmental changes, loss of skin appendages, and sweat impairment Lung § Diagnostic features: Bronchiolitis obliterans diagnosed with lung biopsy § lymphocytic alveolitis, resulting in interstitial fibrosis or bronchiolitis obliterans organizing pneumonia Oral Mucosa § Diagnostic features: lichenoid changes, lichen-type features, restriction of mouth opening from sclerosis, erythema or ulcers and mucoceles § The other findings: Sicca symptoms, gingivitis, periodontitis, increased tooth decay, and tooth loss

Chronic GVHD-common organ involvement Eyes § atrophy of the lacrimal gland with sicca syndrome, severe blepharitis, chronic persistent inflammation with visible erythema of the conjunctiva Liver § Cholestasis, hepatitic forms with high transaminases Gastrointestinal Tract § Diagnostic features: Esophageal web, Strictures or stenosis in the upper to mid third of the esophagus § The other findings: Dysphagia, nausea and vomiting, chronic, diarrhea and malabsorption syndrome, immune-mediated pancreatitis Joints, Fasciae, muscle § Diagnostic features: Fasciitis Joint stiffness or contractures secondary to sclerosis, myositis, muscle weakness, cramping, edema, and pain

Chronic GVHD-diagnosis, grading and treatmet § Chronic GVHD is diagnosed on the basis of the symptoms of eight organs, laboratory values (for hepatic manifestations), and PFTs. § Each organ is graded between 0 and 3. § The overall severity of c. GVHD is classified as mild, moderate, or severe Firts line treatment: steroids±calcineurin inhibitors
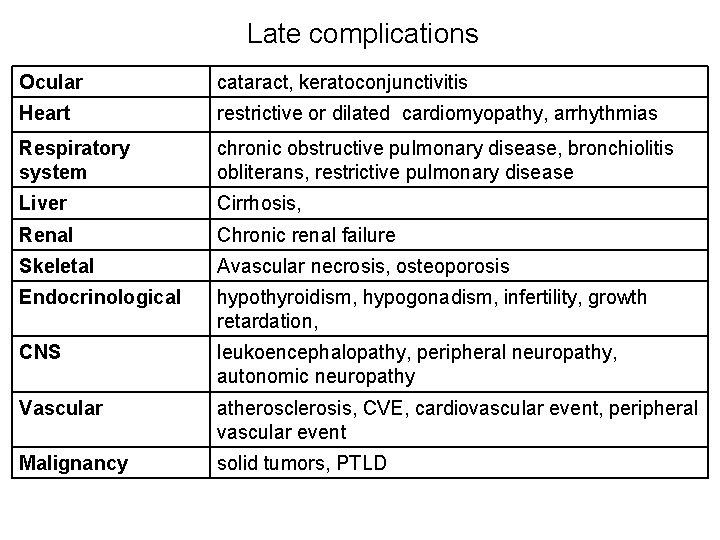
Late complications Ocular cataract, keratoconjunctivitis Heart restrictive or dilated cardiomyopathy, arrhythmias Respiratory system chronic obstructive pulmonary disease, bronchiolitis obliterans, restrictive pulmonary disease Liver Cirrhosis, Renal Chronic renal failure Skeletal Avascular necrosis, osteoporosis Endocrinological hypothyroidism, hypogonadism, infertility, growth retardation, CNS leukoencephalopathy, peripheral neuropathy, autonomic neuropathy Vascular atherosclerosis, CVE, cardiovascular event, peripheral vascular event Malignancy solid tumors, PTLD
- Slides: 42