Heating Cooling Curves D Crowley 2008 Wednesday December

Heating & Cooling Curves D. Crowley, 2008

Wednesday, December 22, 2021 Heating & Cooling Curves n To understand heating and cooling curves

Temperature n The differences between solids, liquids and gases can be explained by the particle model: ¨ All substances are made up of particles (atoms, ions or molecules) ¨ These particles are attracted to each other, some strongly and others weakly ¨ These particles move around (i. e. have kinetic energy) ¨ The kinetic energy of particles increases with temperature
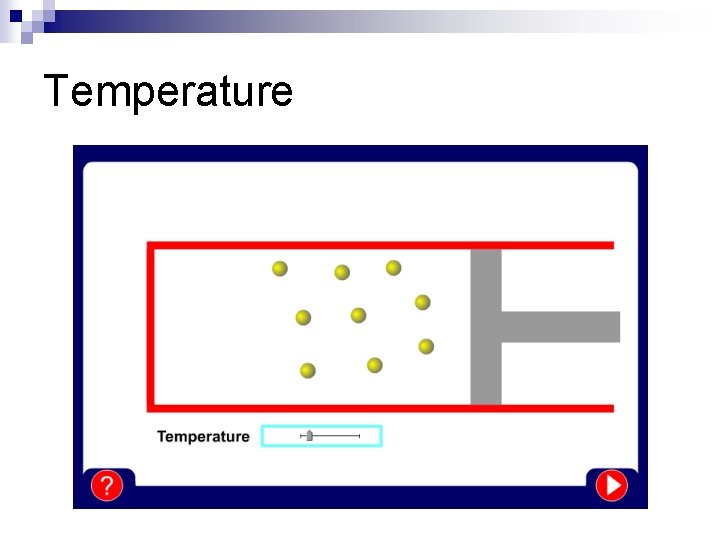
Temperature

Chemical & Physical n Watch the demo of chemical and physical changes… Chemical Changes Physical Changes New substances are formed No new substances are formed Changes are usually permanent (irreversible) Changes are usually not permanent (reversible)

States Of Matter n Ice melting into water is an example of a physical change n No new substances are formed during physical changes n At a cold enough temperature, even substances that are normally gases will become solid n At higher temperatures, solids change to become liquids or gases – as long as they don’t catch fire or decompose first

State Changes

Temperature Change n What happens to the temperature of a block of ice when a medium constant heat is applied to it? n Your task is to measure the temperatures of the ice as it is heated every 30 seconds (as well as noting the state of the water (solid, liquid, gas)): Time (seconds) 0 30 60 90 120 Etc… Temperature (o. C) State (solid / liquid / gas)
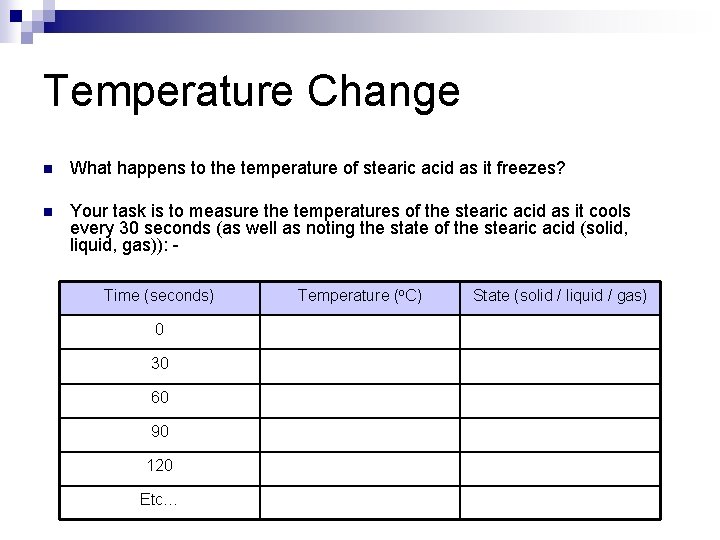
Temperature Change n What happens to the temperature of stearic acid as it freezes? n Your task is to measure the temperatures of the stearic acid as it cools every 30 seconds (as well as noting the state of the stearic acid (solid, liquid, gas)): Time (seconds) 0 30 60 90 120 Etc… Temperature (o. C) State (solid / liquid / gas)
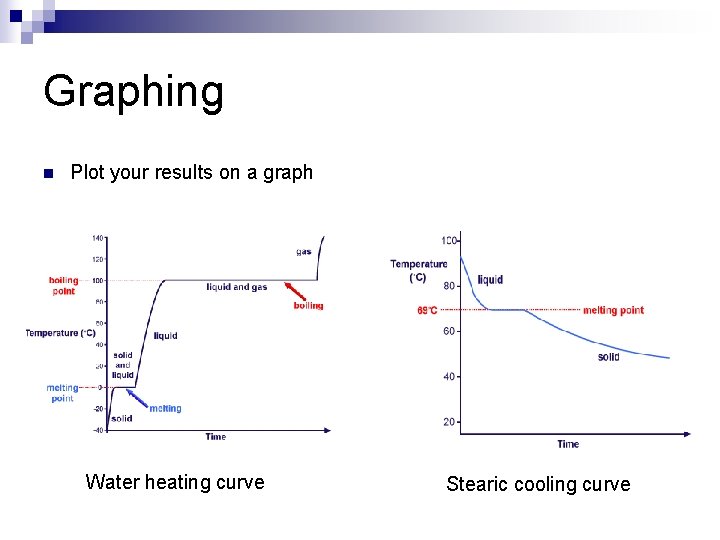
Graphing n Plot your results on a graph Water heating curve Stearic cooling curve
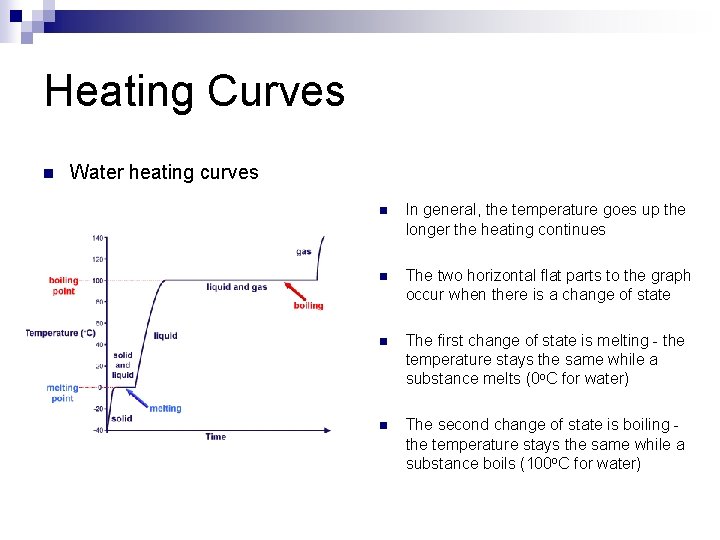
Heating Curves n Water heating curves n In general, the temperature goes up the longer the heating continues n The two horizontal flat parts to the graph occur when there is a change of state n The first change of state is melting - the temperature stays the same while a substance melts (0 o. C for water) n The second change of state is boiling the temperature stays the same while a substance boils (100 o. C for water)
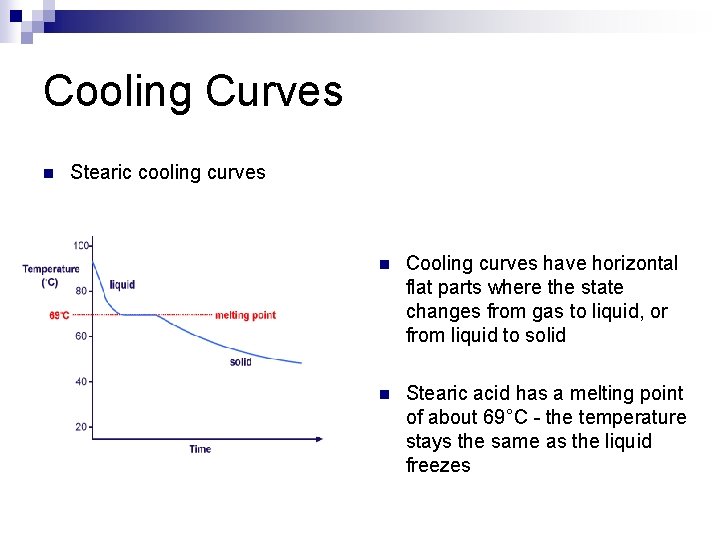
Cooling Curves n Stearic cooling curves n Cooling curves have horizontal flat parts where the state changes from gas to liquid, or from liquid to solid n Stearic acid has a melting point of about 69°C - the temperature stays the same as the liquid freezes
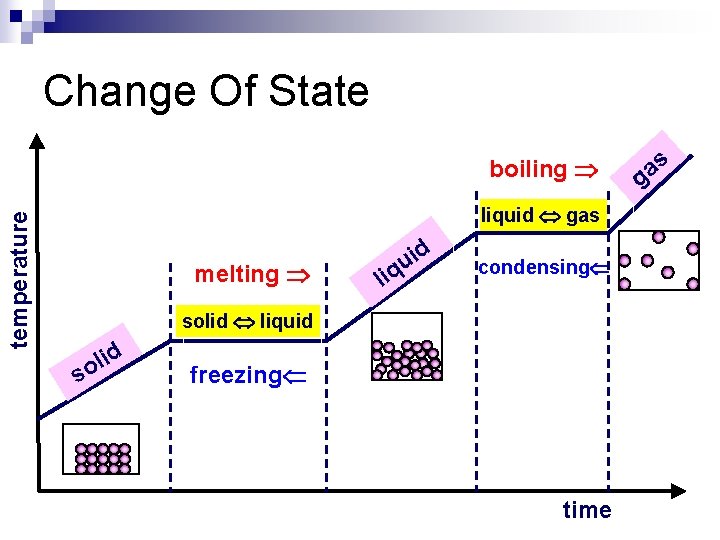
Change Of State temperature boiling liquid gas melting li id u q condensing solid liquid d i l so freezing time s a g
- Slides: 13