Heat Treatment of Metals HeatTreatment Heat treatment is















- Slides: 29

Heat Treatment of Metals

Heat-Treatment Heat treatment is a method used to alter the physical, and sometimes chemical properties of a material. The most common application is metallurgical It involves the use of heating or chilling, normally to extreme temperatures, to achieve a desired result such as hardening or softening of a material It applies only to processes where the heating and cooling are done for the specific purpose of altering properties intentionally Generally, heat treatment uses phase transformation during heating and cooling to change a microstructure in a solid state.


Types of Heat-Treatment (Steel) Annealing Tempering, and Quenching Precipitation hardening Case hardening

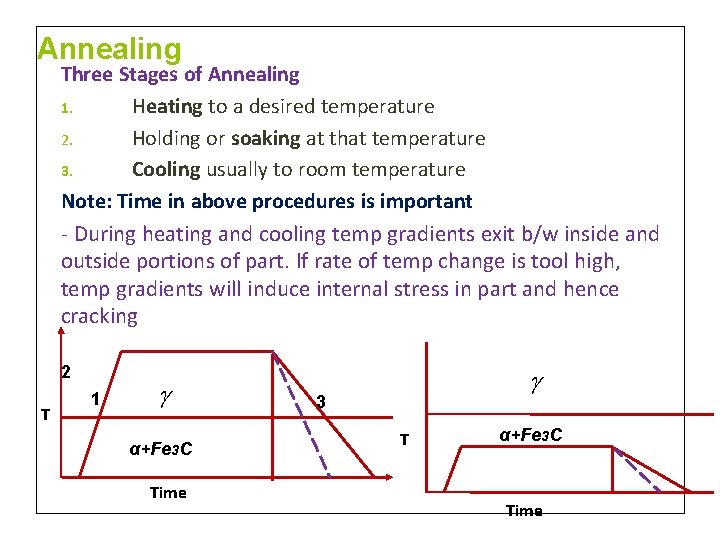
Annealing A heat treatment process in which a metal is exposed to an elevated temperature for an extended time period and then slowly cooled. Purpose: • Relieve stresses of cold working • Increase softness, ductility and toughness • Produce specific microstructure

Annealing Three Stages of Annealing 1. Heating to a desired temperature 2. Holding or soaking at that temperature 3. Cooling usually to room temperature Note: Time in above procedures is important - During heating and cooling temp gradients exit b/w inside and outside portions of part. If rate of temp change is tool high, temp gradients will induce internal stress in part and hence cracking 2 T 1 α+Fe 3 C Time 3 T α+Fe 3 C Time

Types of Annealing 1. 2. 3. 4. 5. Stress-Relief Annealing (or Stress-relieving) Normalizing Full Annealing Spheroidizing Annealing (or Spheroidizing ) Isothermal Annealing

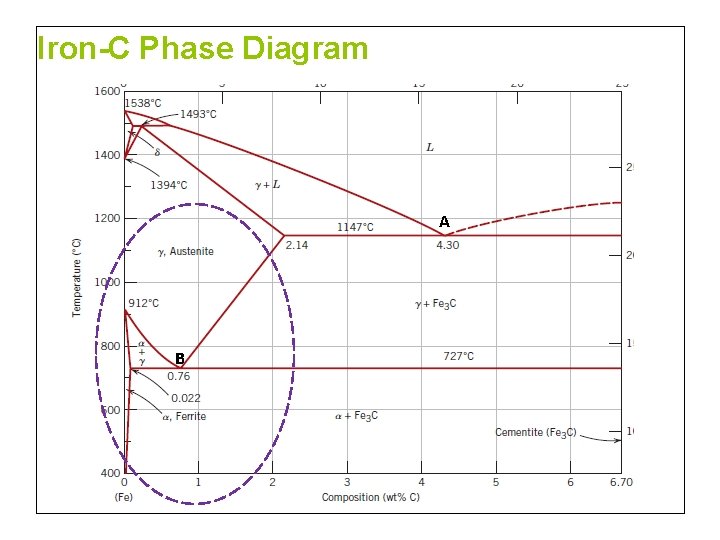
Iron-C Phase Diagram A B

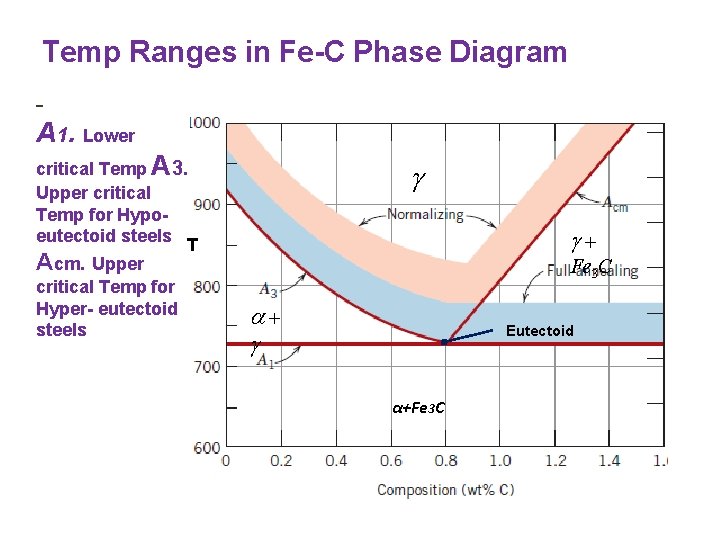
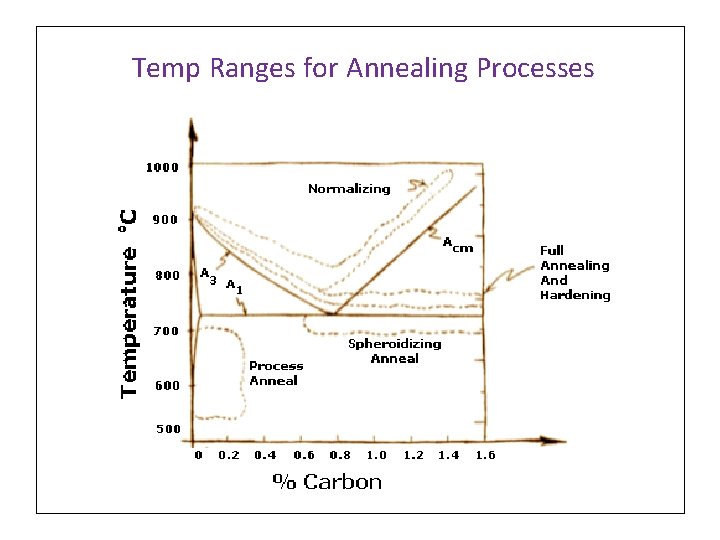
Temp Ranges in Fe-C Phase Diagram - A 1. Lower critical Temp A 3. Upper critical Temp for Hypoeutectoid steels T Acm. Upper critical Temp for Hyper- eutectoid steels Fe 3 C Eutectoid α+Fe 3 C

Temp Ranges for Annealing Processes

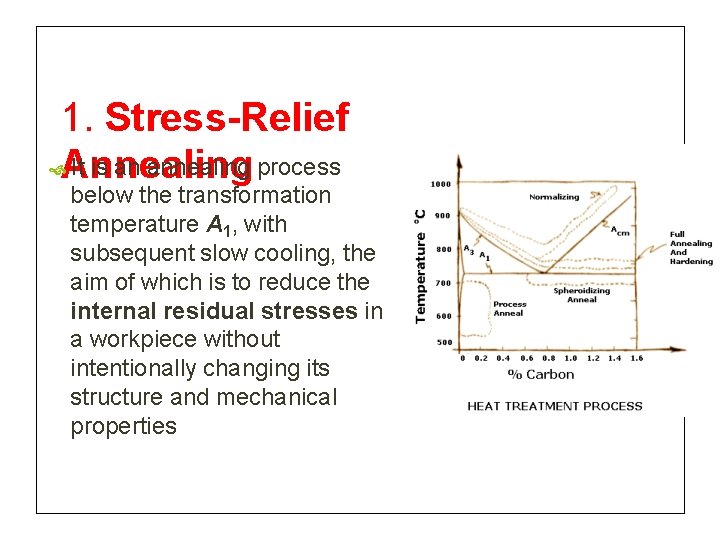
1. Stress-Relief Annealing It is an annealing process below the transformation temperature A 1, with subsequent slow cooling, the aim of which is to reduce the internal residual stresses in a workpiece without intentionally changing its structure and mechanical properties

1. Stress-Relief For plain carbon and low-alloy steels the Annealing temperature to which the specimen is heated is usually between 450 and 650˚C, whereas for hotworking tool steels and high-speed steels it is between 600 and 750˚C This treatment will not cause any phase changes, but recovery & recrystallization may take place. Machining allowance sufficient to compensate for any warping/distrotion resulting from stress relieving should be provided

Causes of Residual Stresses 1. Mechanical factors (e. g. , cold-working during metal forming/machining) 2. Thermal factors (e. g. , thermal stresses caused by temperature gradients within the work-piece during heating or cooling) 3. Metallurgical factors (e. g. , phase transformation upon cooling wherein parent and product phases have different densities 4. - In the heat treatment of metals, quenching or rapid cooling is the cause of the greatest residual stresses

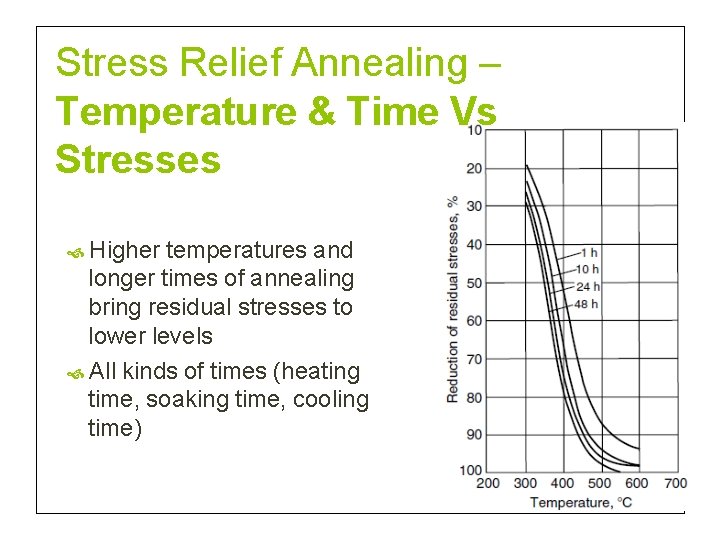
Stress Relief Annealing – Temperature & Time Vs Stresses Higher temperatures and longer times of annealing bring residual stresses to lower levels All kinds of times (heating time, soaking time, cooling time)

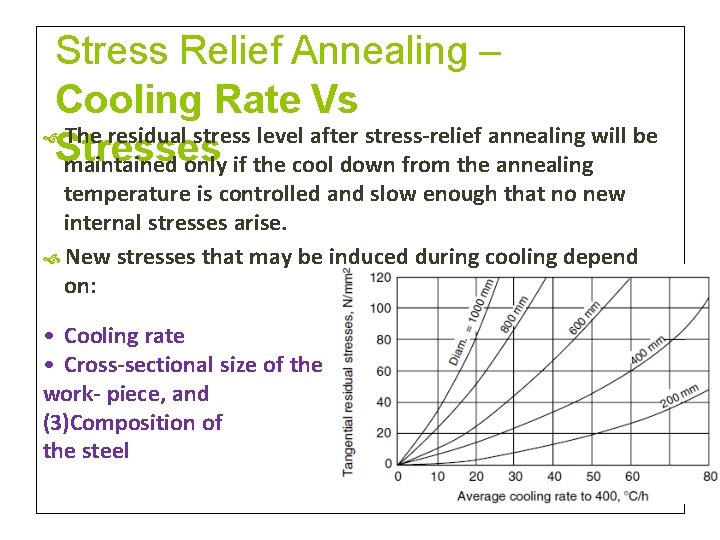
Stress Relief Annealing – Cooling Rate Vs The residual stress level after stress-relief annealing will be Stresses maintained only if the cool down from the annealing temperature is controlled and slow enough that no new internal stresses arise. New stresses that may be induced during cooling depend on: • Cooling rate • Cross-sectional size of the work- piece, and (3)Composition of the steel

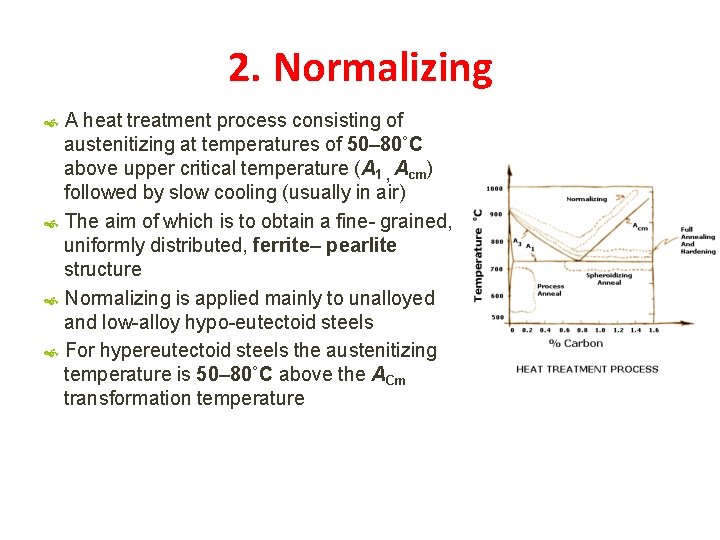
2. Normalizing A heat treatment process consisting of austenitizing at temperatures of 50– 80˚C above upper critical temperature (A 1 , Acm) followed by slow cooling (usually in air) The aim of which is to obtain a fine- grained, uniformly distributed, ferrite– pearlite structure Normalizing is applied mainly to unalloyed and low-alloy hypo-eutectoid steels For hypereutectoid steels the austenitizing temperature is 50– 80˚C above the ACm transformation temperature

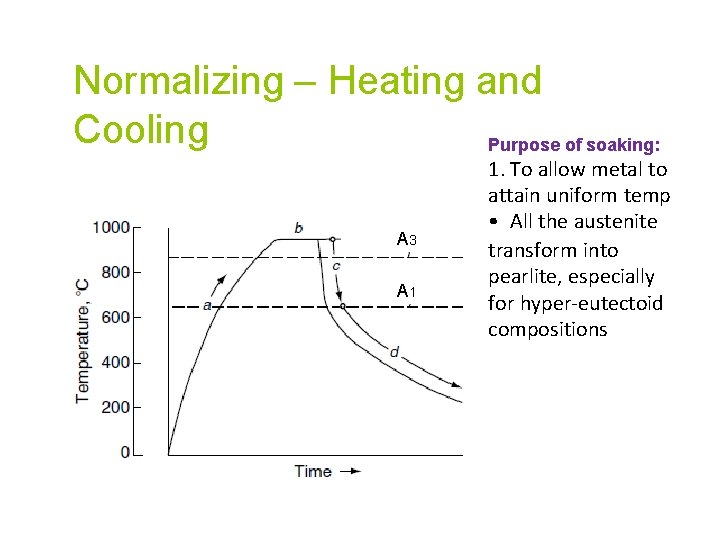
Normalizing – Heating and Cooling Purpose of soaking: A 3 A 1 1. To allow metal to attain uniform temp • All the austenite transform into pearlite, especially for hyper-eutectoid compositions

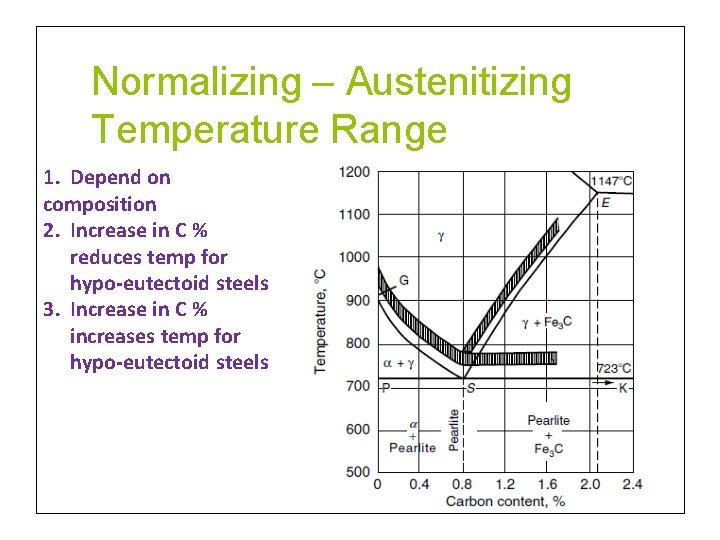
Normalizing – Austenitizing Temperature Range 1. Depend on composition 2. Increase in C % reduces temp for hypo-eutectoid steels 3. Increase in C % increases temp for hypo-eutectoid steels

Effect of Normalizing on Grain Size Normalizing refines (reduces) the grains of a steel that have become coarse (long and irregular) as a result of heavy deformations as in forging or in rolling The fine grains have higher toughness than coarse grains, Steel with 0. 5% C

Normalizing after Rolling After hot rolling, the structure of steel is usually oriented in the rolling direction To remove the oriented structure and obtain the uniform mechanical properties in all directions, a normalizing annealing has to be performed

Normalizing after Forging After forging at high temperatures, especially with work-pieces that vary widely in cross sectional size, because of the different rates of cooling from the forging temperature, a heterogeneous structure is obtained that can be made uniform by normalizing • Normalizing is also done to improve • machinability of low-c steels •

Normalizing – Holding Time Holding time at austenitizing temperature may be calculated using the empirical formula: t = 60 + D where t is the holding time (min) and D is the maximum diameter of the workpiece (mm).

3. Full Annealing For compositions less than eutectoid, the metal is heated above A 3 line to form austenite - For compositions larger than eutectoid, the metal is heated above A 1 line to form austenite and Fe 3 C - Cooled slowly in a furnace instead in air as in Normalizing. Furnace is switched off, both metal and furnace cool at the same rate Usually applied for low -Microstructure outcome: Coarse and medium C steel Pearlite. In Normalizing, structure? -Structure is relatively softer than that in Normalizing -Full annealing is normally used when material needs to be deformed further. -

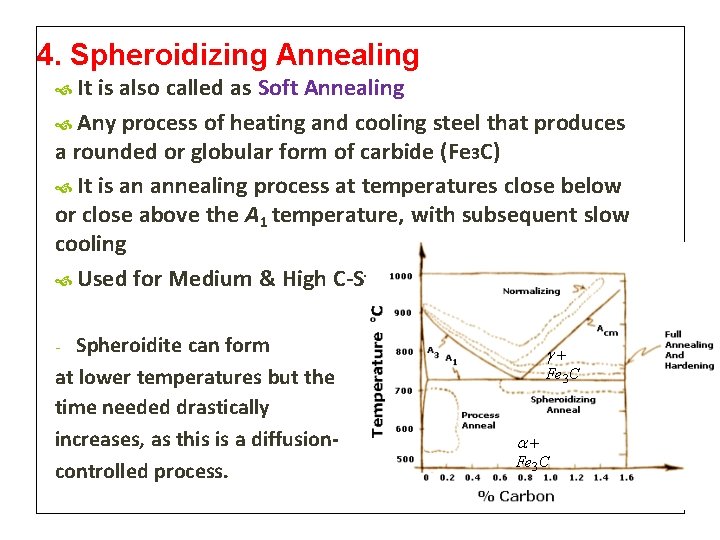
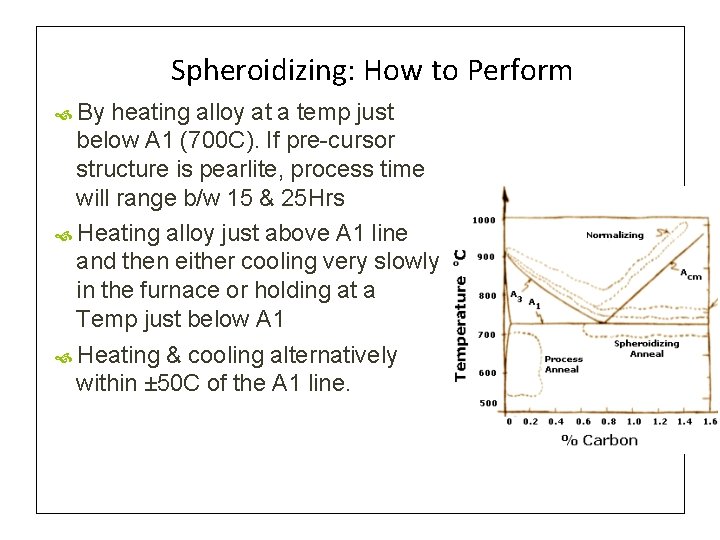
4. Spheroidizing Annealing It is also called as Soft Annealing Any process of heating and cooling steel that produces a rounded or globular form of carbide (Fe 3 C) It is an annealing process at temperatures close below or close above the A 1 temperature, with subsequent slow cooling Used for Medium & High C-Steels Spheroidite can form at lower temperatures but the time needed drastically increases, as this is a diffusioncontrolled process. - Fe 3 C

Spheroidizing: How to Perform By heating alloy at a temp just below A 1 (700 C). If pre-cursor structure is pearlite, process time will range b/w 15 & 25 Hrs Heating alloy just above A 1 line and then either cooling very slowly in the furnace or holding at a Temp just below A 1 Heating & cooling alternatively within ± 50 C of the A 1 line.

Spheroidizing - Purpose The aim is to produce a soft structure by changing all hard micro-constituents like pearlite, bainite, and martensite (especially in steels with carbon contents above 0. 5% and in tool steels) into a structure of spheroidized carbides in a ferritic matrix (a) (b) a medium-carbon low-alloy steel after soft annealing at 720 C; a high-speed steel soft annealed at 820 C.

Spheroidizing - Uses Such a soft structure is required for good machinability of steels having more than 0. 6%C and for all cold-working processes that include plastic deformation. Spheroidite steel is the softest and most ductile form of steel

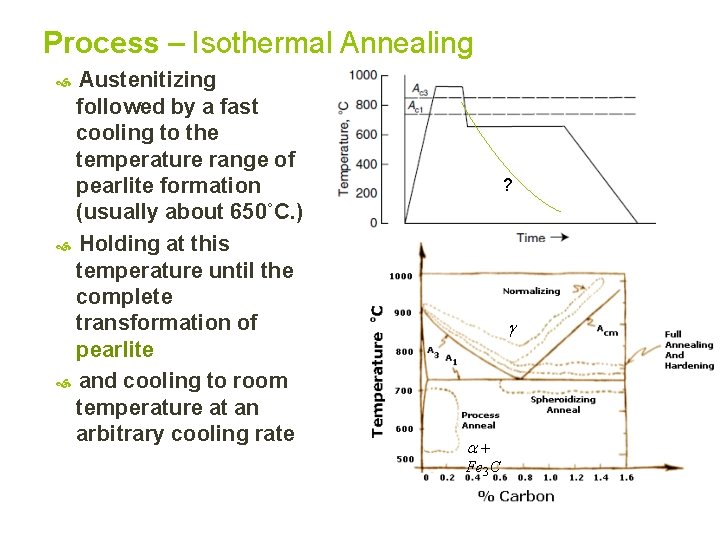
5. Isothermal Annealing Spheroidizing is more useful for improving machinability of high C steel than that of low and medium C steels. In fact, spherodized low and medium C steels become over soft for machining and give long shavings which accumulate on tool cutting edge and produce poor surface. Hypoeutectoid low-carbon steels as well as medium-carbon structural steels are often isothermally annealed, for best machinability An isothermally annealed structure should have the following characteristics: • High proportion of ferrite • Uniformly distributed pearlite grains • Fine lamellar pearlite grains

Process – Isothermal Annealing Austenitizing followed by a fast cooling to the temperature range of pearlite formation (usually about 650˚C. ) Holding at this temperature until the complete transformation of pearlite and cooling to room temperature at an arbitrary cooling rate ? Fe 3 C