HEAT TRANSFER BUTTONS Click here Clicking here will
HEAT TRANSFER BUTTONS Click here: Clicking here will move you the page. Clicking here will here bring reveal will reveal some back an information. to answer. a next this page. Clicking here on. Clicking will me will take will allow take you you totoayou to hear list SIback ofto multipliers some equations, information table. clicking Clicking on the here topic. here again Clicking again will here take return you again back you willto to stop your thethe previous sound. page. TOPICS Jump to… …temperature (one page). Jump to… …conduction (one page). Jump to… …convection (two pages). Jump to… …explanation (two pages - higher). Jump to… …radiation and summary (three pages). Jump to… …practice question (one page). Jump to… …exam questions (three pages).
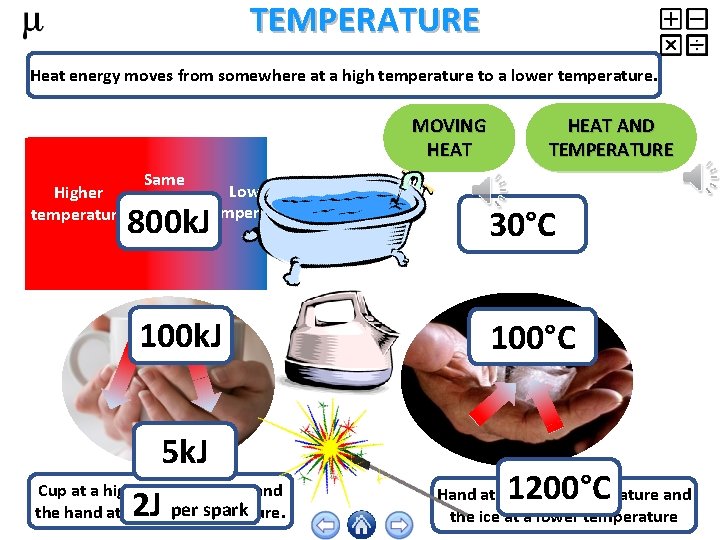
TEMPERATURE Heat energy moves from somewhere at a high temperature to a lower temperature. MOVING HEAT Same Higher temperature Lower temperaturethroughouttemperature 800 k. J 100 k. J 5 k. J Cup at a higher temperature and per spark the hand at a 2 J lower temperature. HEAT AND TEMPERATURE 30°C 100°C 1200°C Hand at a higher temperature and the ice at a lower temperature
CONDUCTION Heat moves through a solid by Conduction. The Wire Gauze Different Metals Copper and Plastic Summary
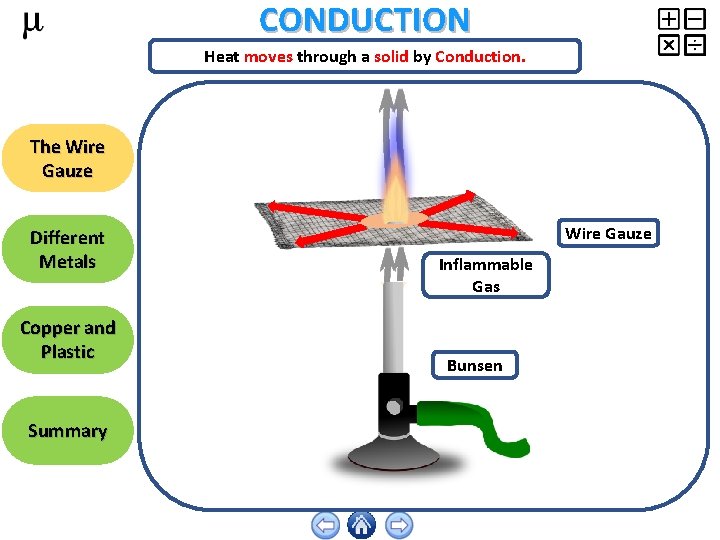
CONDUCTION Heat moves through a solid by Conduction. The Wire Gauze Different Metals Copper and Plastic Summary Wire Gauze Inflammable Gas Bunsen
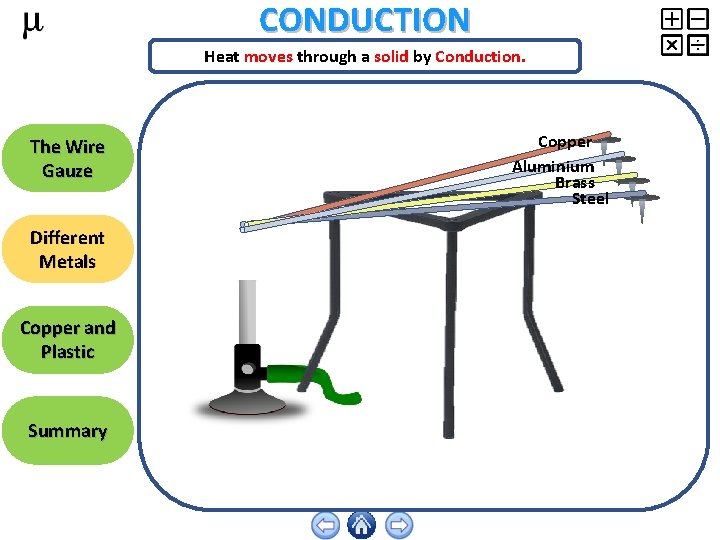
CONDUCTION Heat moves through a solid by Conduction. The Wire Gauze Different Metals Copper and Plastic Summary Copper Aluminium Brass Steel
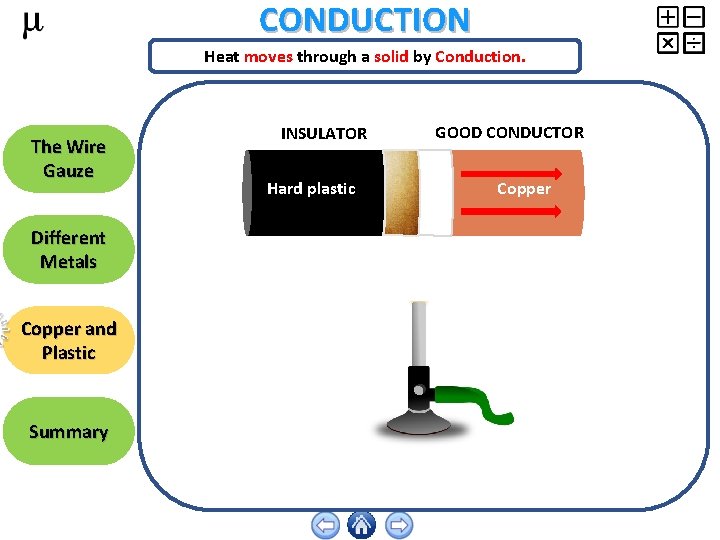
CONDUCTION Heat moves through a solid by Conduction. The Wire Gauze Different Metals Copper and Plastic Summary INSULATOR Hard plastic GOOD CONDUCTOR Copper
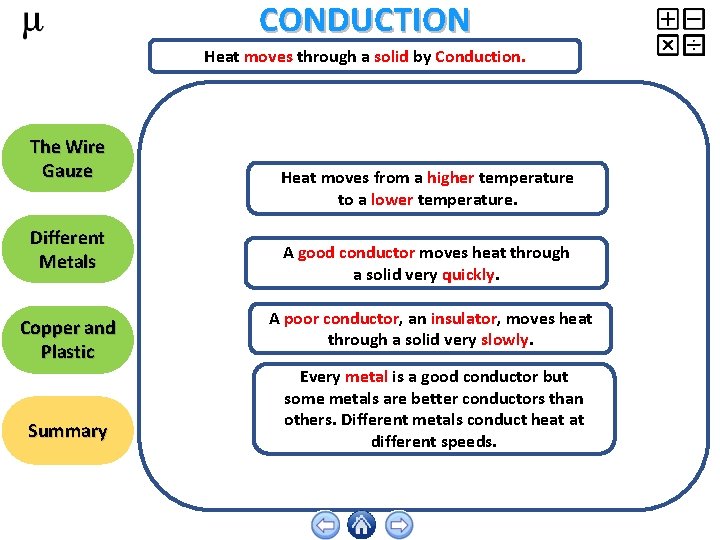
CONDUCTION Heat moves through a solid by Conduction. The Wire Gauze Different Metals Copper and Plastic Summary Heat moves from a higher temperature to a lower temperature. A good conductor moves heat through a solid very quickly. A poor conductor, an insulator, moves heat through a solid very slowly. Every metal is a good conductor but some metals are better conductors than others. Different metals conduct heat at different speeds.
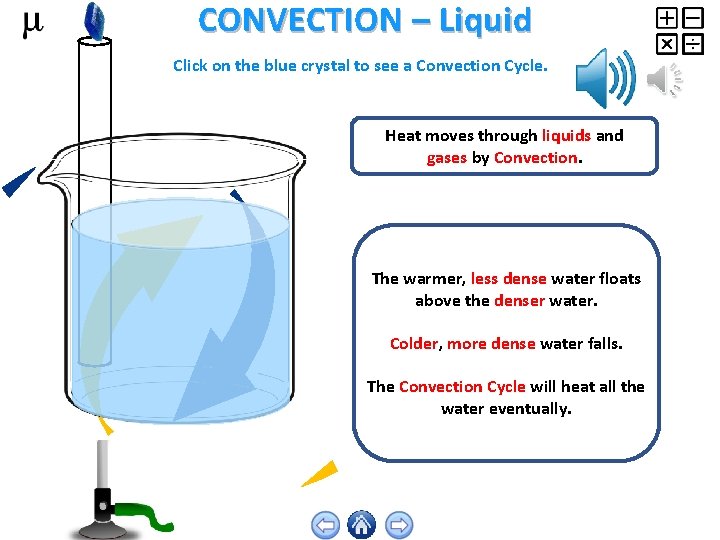
CONVECTION – Liquid Click on the blue crystal to see a Convection Cycle. Heat moves through liquids and gases by Convection. The warmer, less dense water floats above the denser water. Colder, more dense water falls. The Convection Cycle will heat all the water eventually.
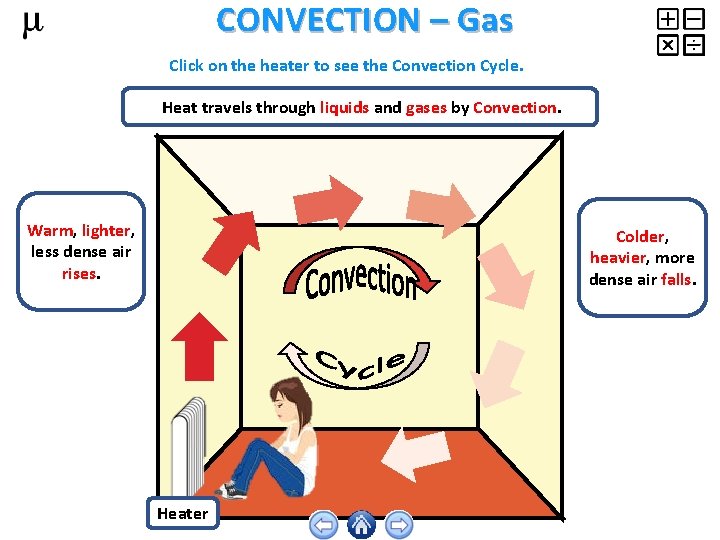
CONVECTION – Gas Click on the heater to see the Convection Cycle. Heat travels through liquids and gases by Convection. Warm, lighter, less dense air rises. Colder, heavier, more dense air falls. Heater
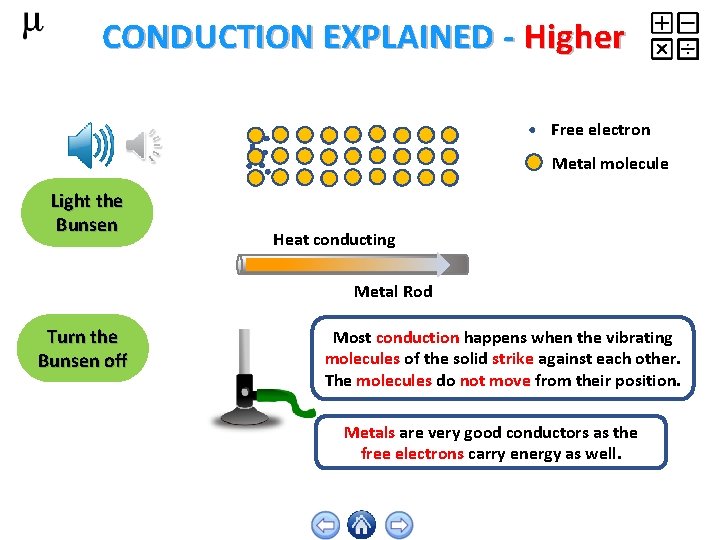
CONDUCTION EXPLAINED - Higher Free electron Metal molecule Light the Bunsen Heat conducting Metal Rod Turn the Bunsen off Most conduction happens when the vibrating molecules of the solid strike against each other. The molecules do not move from their position. Metals are very good conductors as the free electrons carry energy as well.
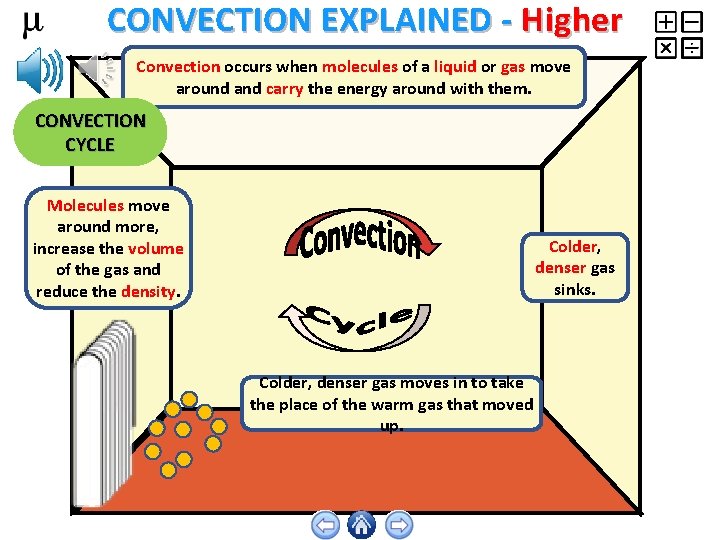
CONVECTION EXPLAINED - Higher Convection occurs when molecules of a liquid or gas move around and carry the energy around with them. CONVECTION CYCLE Molecules move around more, increase the volume of the gas and reduce the density. Colder, denser gas sinks. Colder, denser gas moves in to take the place of the warm gas that moved up.
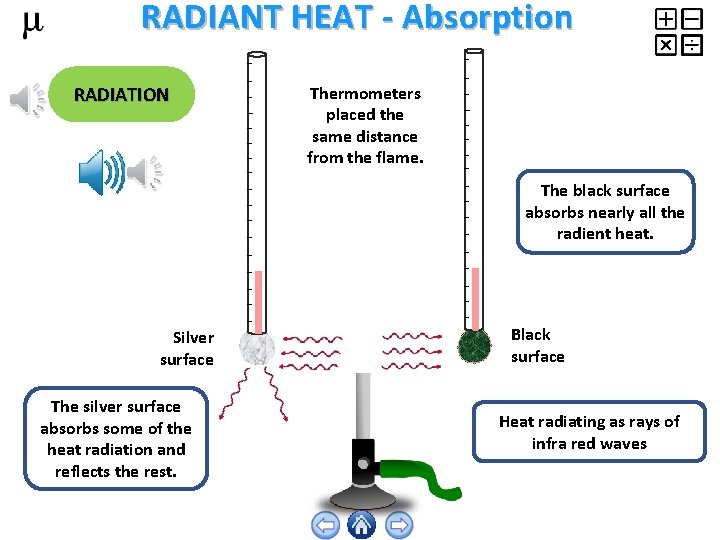
RADIANT HEAT - Absorption RADIATION Thermometers placed the same distance from the flame. The black surface absorbs nearly all the radient heat. Silver surface The silver surface absorbs some of the heat radiation and reflects the rest. Black surface Heat radiating as rays of infra red waves
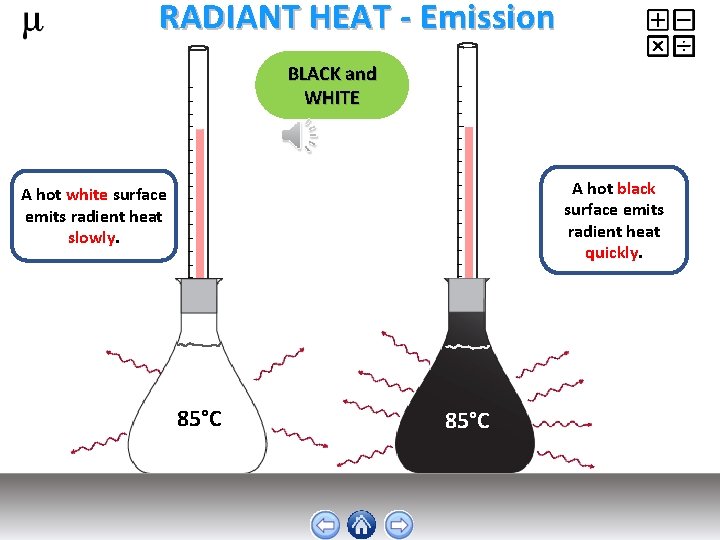
RADIANT HEAT - Emission BLACK and WHITE A hot black surface emits radient heat quickly. A hot white surface emits radient heat slowly. 85°C
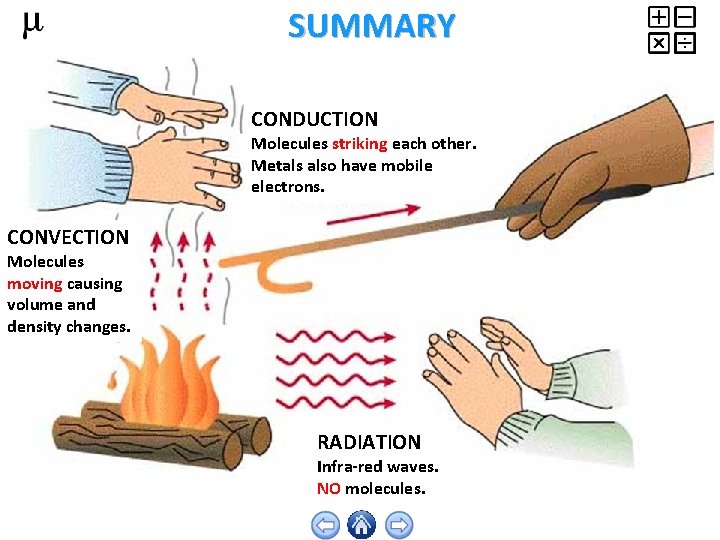
SUMMARY CONDUCTION Molecules striking each other. Metals also have mobile electrons. CONVECTION Molecules moving causing volume and density changes. RADIATION Infra-red waves. NO molecules.
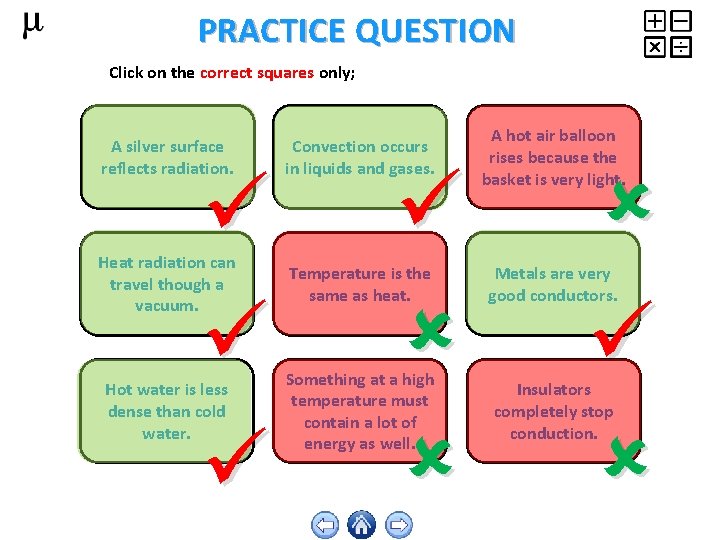
PRACTICE QUESTION Click on the correct squares only; A silver surface reflects radiation. Convection occurs in liquids and gases. A hot air balloon rises because the basket is very light. Heat radiation can travel though a vacuum. Temperature is the same as heat. Metals are very good conductors. Hot water is less dense than cold water. Something at a high temperature must contain a lot of energy as well. Insulators completely stop conduction.
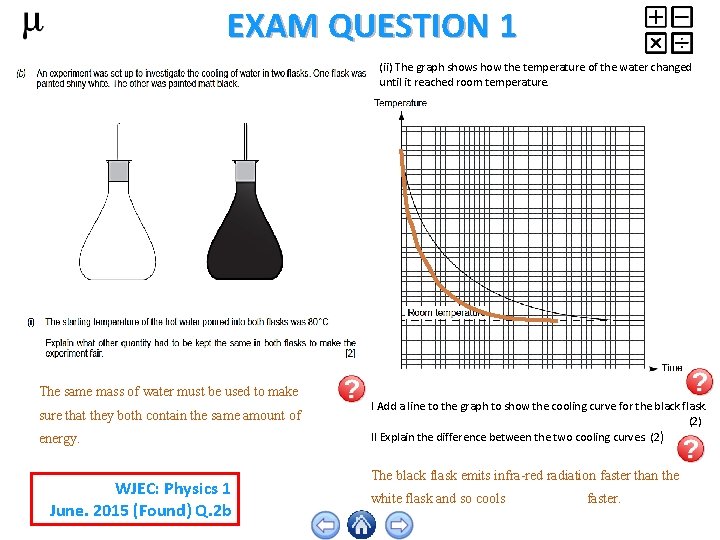
EXAM QUESTION 1 (ii) The graph shows how the temperature of the water changed until it reached room temperature. The same mass of water must be used to make sure that they both contain the same amount of energy. WJEC: Physics 1 June. 2015 (Found) Q. 2 b I Add a line to the graph to show the cooling curve for the black flask. (2) II Explain the difference between the two cooling curves. (2 ) The black flask emits infra-red radiation faster than the white flask and so cools faster.
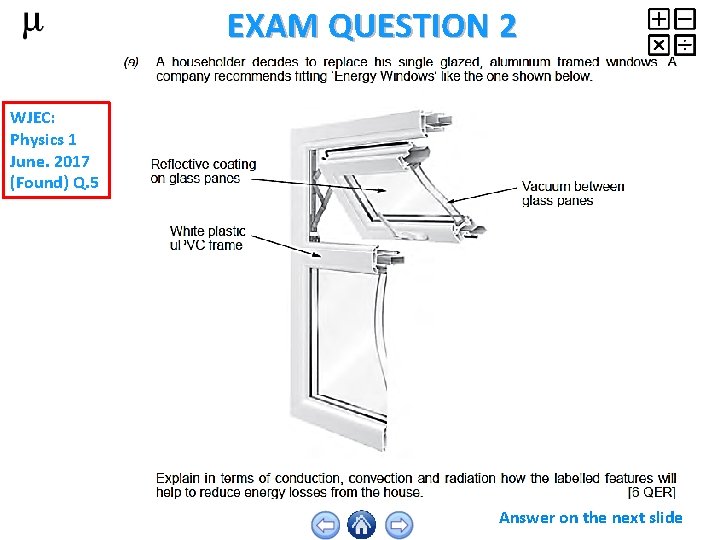
EXAM QUESTION 2 WJEC: Physics 1 June. 2017 (Found) Q. 5 Answer on the next slide
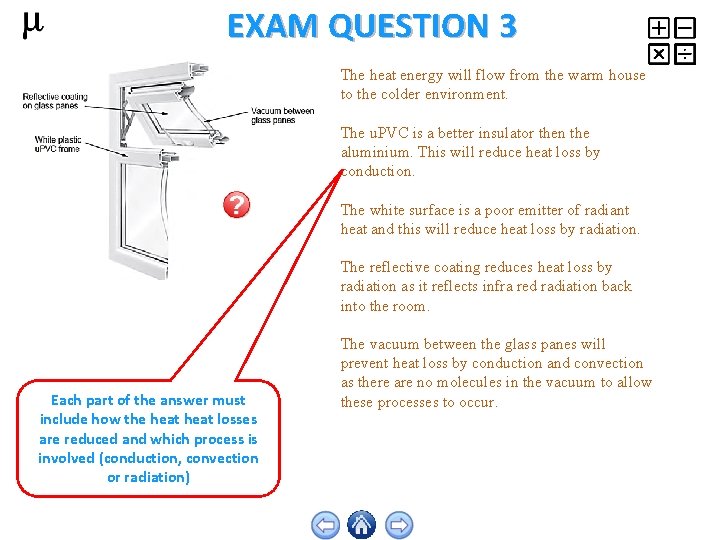
EXAM QUESTION 3 The heat energy will flow from the warm house to the colder environment. The u. PVC is a better insulator then the aluminium. This will reduce heat loss by conduction. The white surface is a poor emitter of radiant heat and this will reduce heat loss by radiation. The reflective coating reduces heat loss by radiation as it reflects infra red radiation back into the room. Each part of the answer must include how the heat losses are reduced and which process is involved (conduction, convection or radiation) The vacuum between the glass panes will prevent heat loss by conduction and convection as there are no molecules in the vacuum to allow these processes to occur.

EXAM QUESTION 3 - Higher WJEC: Physics 1 Jun. 2018 (High. ) Q. 5 As the water molecules gain energy they move further apart and faster. This increases the volume of the hotter water and its density decreases. This hotter, less dense water rises and colder, more dense water falls to take its place. The dye moves as it moves with the water showing the convection cycle.
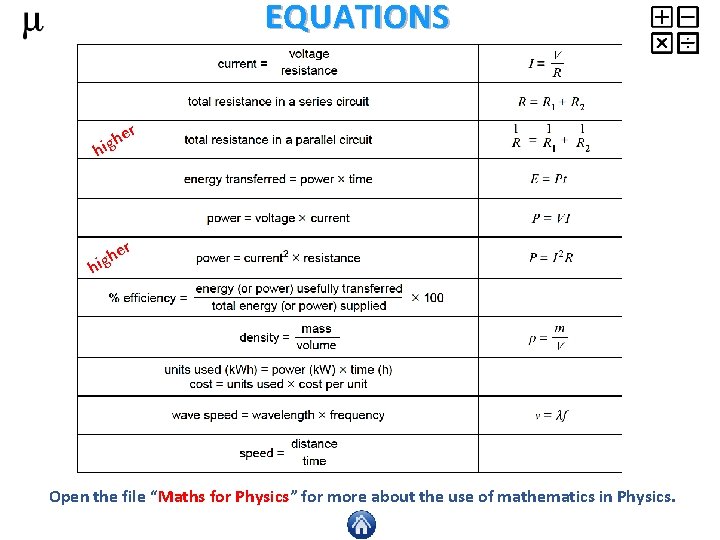
EQUATIONS r e igh h er h hig Open the file “Maths for Physics” for more about the use of mathematics in Physics.

SI MULTIPLIERS p - pico k - kilo n - nano M - mega - micro G - giga m - milli T - tera You only see the letter of the prefix on an exam paper, NOT the name. On a Foundation paper only milli, kilo and mega are used.
- Slides: 21