Heat Transfer and Calorimetry Dr Keith Baessler Definitions

Heat Transfer and Calorimetry Dr. Keith Baessler

Definitions and Concepts • Energy • Law of Conservation of Energy • System vs Surroundings • Types of Systems • Heat vs Temperature • Endothermic vs Exothermic • • Specific Heat Capacity Heat Transfer Sign Conventions of q Calorimetry Calorimeters Formula Applications 2

Energy is the capacity to do work. • Radiant energy comes from the sun and is earth’s primary energy source • Thermal energy is the energy associated with the random motion of atoms and molecules • Chemical energy is the energy stored within the bonds of chemical substances • Nuclear energy is the energy stored within the collection of neutrons and protons in the atom • Potential energy is the energy available by virtue of an object’s position 3

First law of thermodynamics – energy can be converted from one form to another, but cannot be created or destroyed. C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O Exothermic chemical reaction! Chemical energy lost by combustion = Energy gained by the surroundings system surroundings 4
Heat versus Temperature Heat is the transfer of thermal energy between two bodies that are at different temperatures. Temperature is a measure of thermal energy. Temperature = Thermal Energy 5
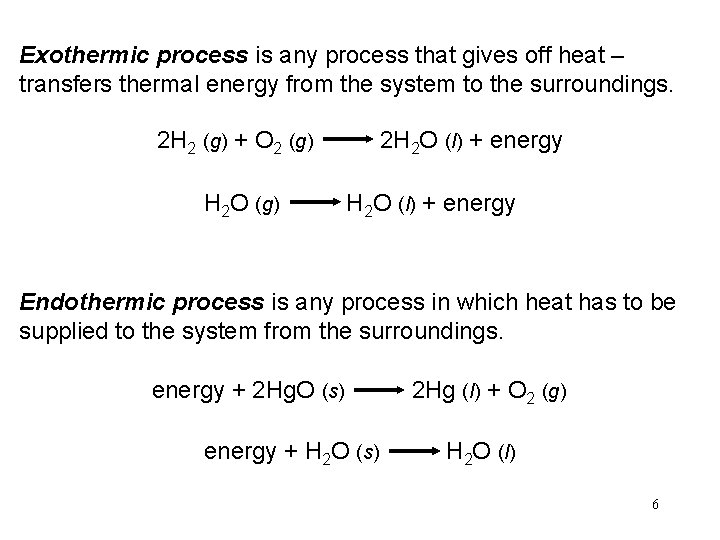
Exothermic process is any process that gives off heat – transfers thermal energy from the system to the surroundings. 2 H 2 (g) + O 2 (g) 2 H 2 O (l) + energy H 2 O (g) H 2 O (l) + energy Endothermic process is any process in which heat has to be supplied to the system from the surroundings. energy + 2 Hg. O (s) 2 Hg (l) + O 2 (g) energy + H 2 O (s) H 2 O (l) 6
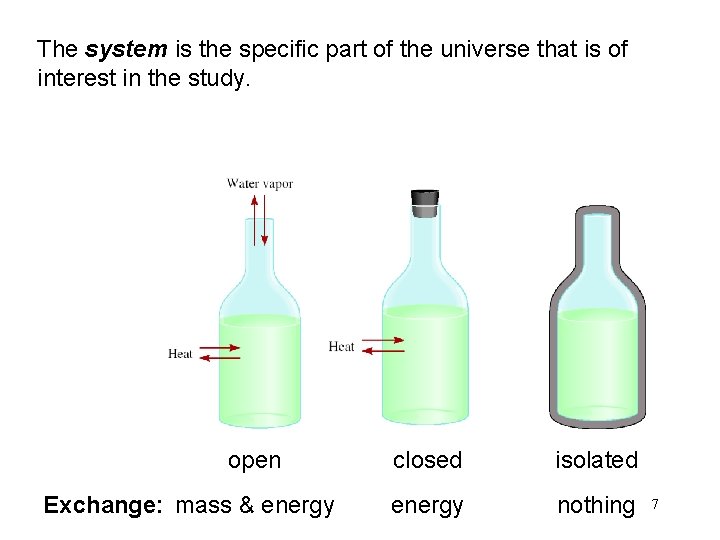
The system is the specific part of the universe that is of interest in the study. open Exchange: mass & energy closed isolated energy nothing 7

Heat Transfer, q • Heat (q) = the transfer of energy which causes the temperature of an object to change • units: joules (j), calories (cal) – A calorie is the amount of energy needed to raise the temperature of 1. 00 g water by 1°C. 1 cal = 4. 184 joules • Heat spontaneously moves regions of high temperature to regions of lower temperature. • A metal spoon at 25°C is placed in boiling water. What happens? 8

The Sign Convention of q • Endothermic systems require the surroundings to add energy to the system. q is positive (+) • Exothermic reactions release energy to the surroundings. q is negative (-) • Energy changes are measured from the point of view of the system 9

The specific heat (s) of a substance is the amount of heat (q) required to raise the temperature of one gram of the substance by one degree Celsius. The heat capacity** (C) of a substance is the amount of heat (q) required to raise the temperature of a given quantity (m) of the substance by one degree Celsius. C = m x s Heat (q) absorbed or released: q = m x s x Dt q = C x Dt Dt = tfinal - tinitial 10 **Heat Capacity (C) is sometimes called the calorimeter constant
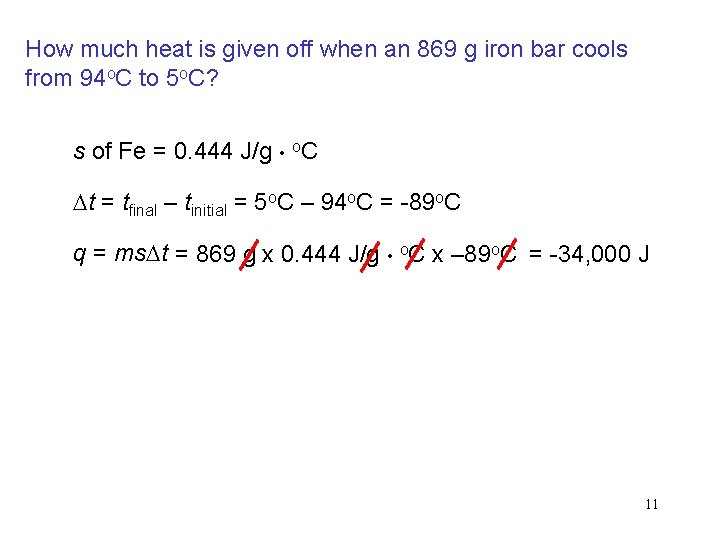
How much heat is given off when an 869 g iron bar cools from 94 o. C to 5 o. C? s of Fe = 0. 444 J/g • o. C Dt = tfinal – tinitial = 5 o. C – 94 o. C = -89 o. C q = ms. Dt = 869 g x 0. 444 J/g • o. C x – 89 o. C = -34, 000 J 11

Calorimetry • Calorimeter: a closed container used to measure temperature changes in physical and chemical processes. • From the temperature changes we can calculate the heat of the reaction, q – qv; heat measured under constant volume conditions – qp: heat measured under constant pressure conditions q = m x s x Dt 12
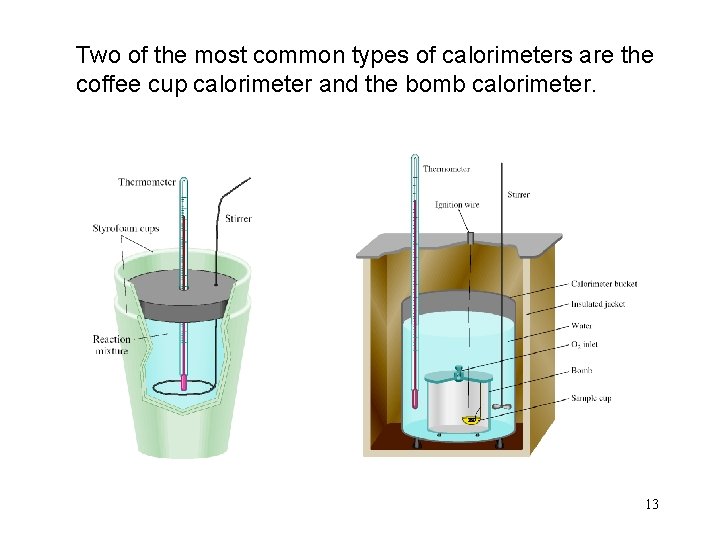
Two of the most common types of calorimeters are the coffee cup calorimeter and the bomb calorimeter. 13

Coffee Cup Calorimeter • The open system allows the pressure to remain constant. • Thus we measure qp = ΔH, the enthalpy change. • ΔH =change in heat at constant pressure. 14

The 1 st Law of Thermodynamics Keeps Track of Heat Transfer • If we monitor the heat transfers (q) of all materials involved, we can predict that their sum will be zero. • By monitoring the surroundings, we can predict what is happening to our system. • Heat transfers until thermal equilibrium, thus the final temperature is the same for all materials. 15

Example of Using 1 st Law for Heat Transfer A 43. 29 g sample of solid is transferred from boiling water (T=99. 8°C) to 152 g water at 22. 5°C in a Styrofoam coffee cup calorimeter. The Twater increased to 24. 3°C. Calculate the specific heat of the solid assuming no heat was lost or gained by the calorimeter cup. qsample+ qwater + qcup= 0 qcup is neglected in problem = 0 Hence qsample = - qwater 16

qsample = - qwater 17
Chemistry in Action: 18
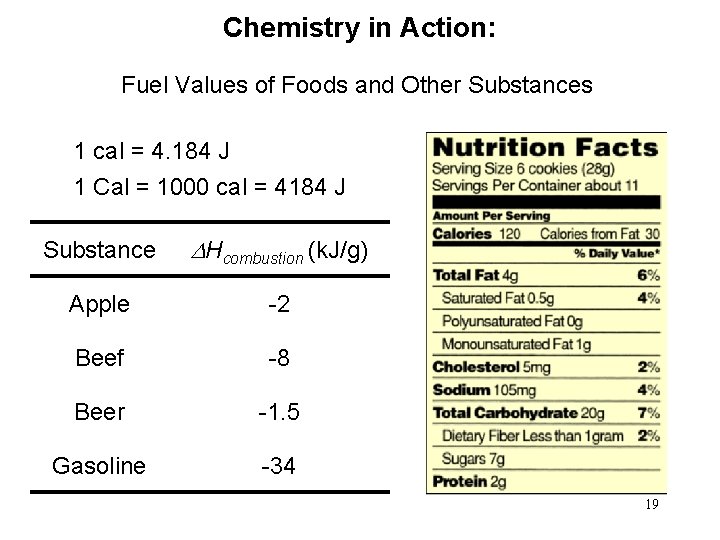
Chemistry in Action: Fuel Values of Foods and Other Substances 1 cal = 4. 184 J 1 Cal = 1000 cal = 4184 J Substance DHcombustion (k. J/g) Apple -2 Beef -8 Beer -1. 5 Gasoline -34 19

A metal alloy was heated for 2 minutes in a hot water bath at 101. 20 C before being transferred into an aluminum calorimeter cup containing water at 23. 42 °C. A rise in the temperature of the water in the calorimeter cup was observed. In fact the maximum temperature reached was 28. 25 °C. Using the data below, calculate the specific heat of the metal object in cal/g°C. Note that the specific heats of water and aluminum are 1. 00 cal/g C and 0. 22 cal/g C respectively. Wt. Alloy 243. 42 g Wt. water in calorimeter cup 213. 14 g Wt. Calorimeter cup 45. 37 g Initial temp. of water 23. 42 C Initial temp. of calorimeter cup 23. 42 °C Initial temp. of metal alloy 101. 20 °C Final temp. of water 28. 25 C 20
- Slides: 20