Heat is Absorbed Position A Solid KE of
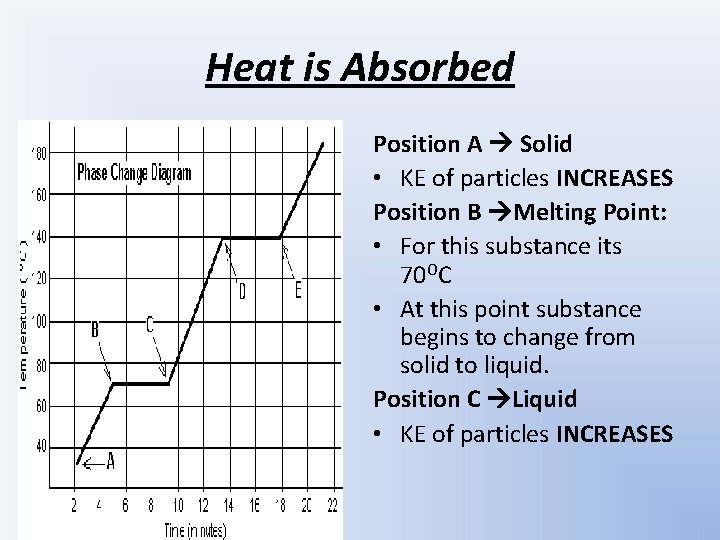
Heat is Absorbed Position A Solid • KE of particles INCREASES Position B Melting Point: • For this substance its 70 OC • At this point substance begins to change from solid to liquid. Position C Liquid • KE of particles INCREASES
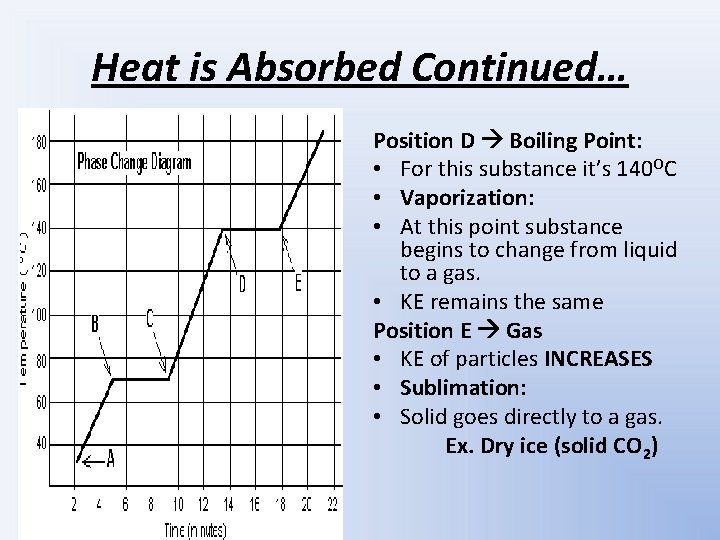
Heat is Absorbed Continued… Position D Boiling Point: • For this substance it’s 140 OC • Vaporization: • At this point substance begins to change from liquid to a gas. • KE remains the same Position E Gas • KE of particles INCREASES • Sublimation: • Solid goes directly to a gas. Ex. Dry ice (solid CO 2)
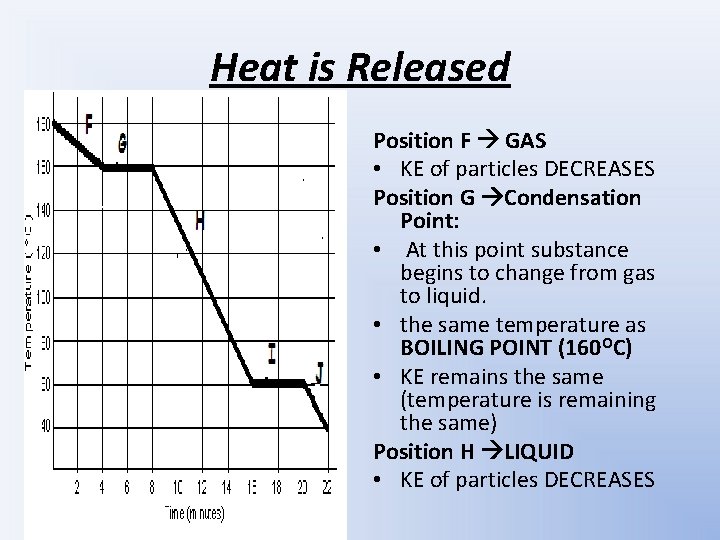
Heat is Released Position F GAS • KE of particles DECREASES Position G Condensation Point: • At this point substance begins to change from gas to liquid. • the same temperature as BOILING POINT (160 OC) • KE remains the same (temperature is remaining the same) Position H LIQUID • KE of particles DECREASES
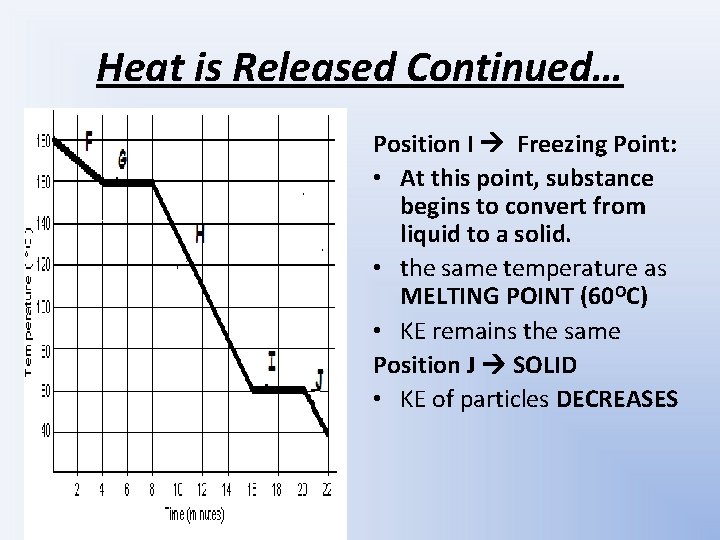
Heat is Released Continued… Position I Freezing Point: • At this point, substance begins to convert from liquid to a solid. • the same temperature as MELTING POINT (60 OC) • KE remains the same Position J SOLID • KE of particles DECREASES

• As matter absorbs heat its kinetic energy (temperature) ____ so its volume____. 1. INCREASES 2. EXPANDS • As matter loses heat its kinetic energy (temperature) _____ so its volume ______. 1. DECREASES 2. CONTRACTS
- Slides: 5