Heat in Chemical Reactions Thermochemistry u Thermochemistry study




















- Slides: 25

Heat in Chemical Reactions

Thermochemistry u Thermochemistry – study of heat changes in chemical reactions u Energy - capacity for doing work or supplying heat • weightless, odorless, tasteless • if within the chemical substancescalled chemical potential energy 2

Heat u symbol “q” u SI unit “joule” u energy transfer from one object to another (because of a temperature difference) • only changes can be detected! • flows from warmer cooler object 3


Enthalpy u symbol “H” u unit “joule” u change in enthalpy is ∆H u ∆H = H products – H reactants 5

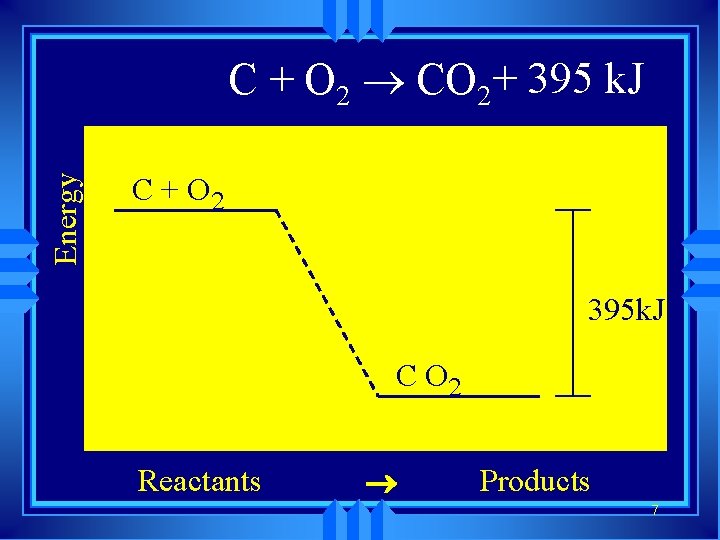
Exothermic Reactions • release of heat • q and H is negative • The products are lower in energy than the reactants.

Energy C + O 2 CO 2+ 395 k. J C + O 2 395 k. J C O 2 Reactants ® Products 7

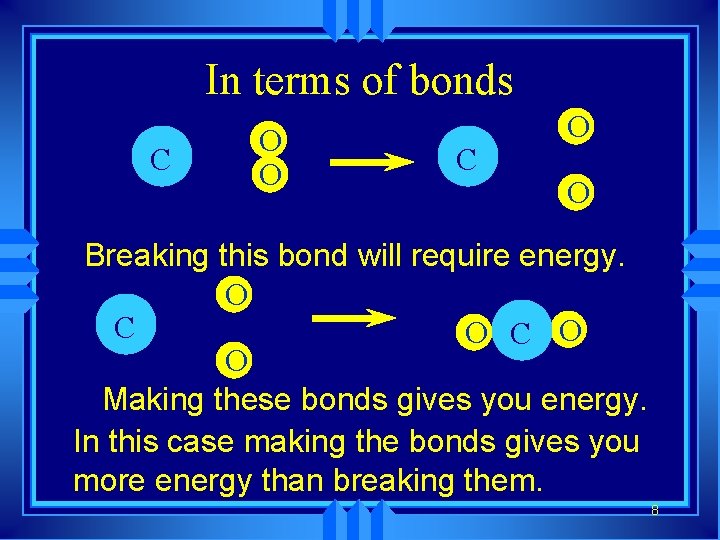
In terms of bonds C O O Breaking this bond will require energy. O C O O Making these bonds gives you energy. In this case making the bonds gives you more energy than breaking them. 8

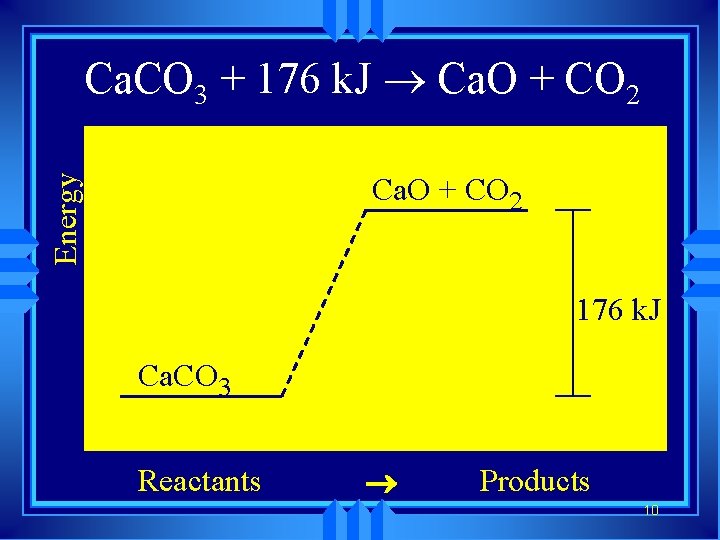
Endothermic Reactions • absorption of heat • q and H is positive • The products are higher in energy than the reactants

Ca. CO Ca. O Ca. CO Ca. O + CO+2 CO 2 3 + 176 3 k. J Energy Ca. O + CO 2 176 k. J Ca. CO 3 Reactants ® Products 10

Using Enthalpy in Reactions Enthalpy is related to molar ratios…yes, yet another stoichiometric relationship! u CH 4 + 2 O 2 CO 2 + 2 H 2 O + 802. 2 k. J u 1 mole of CH 4 releases 802. 2 k. J energy. u When you make 802. 2 k. J you also make 2 moles of water u 11

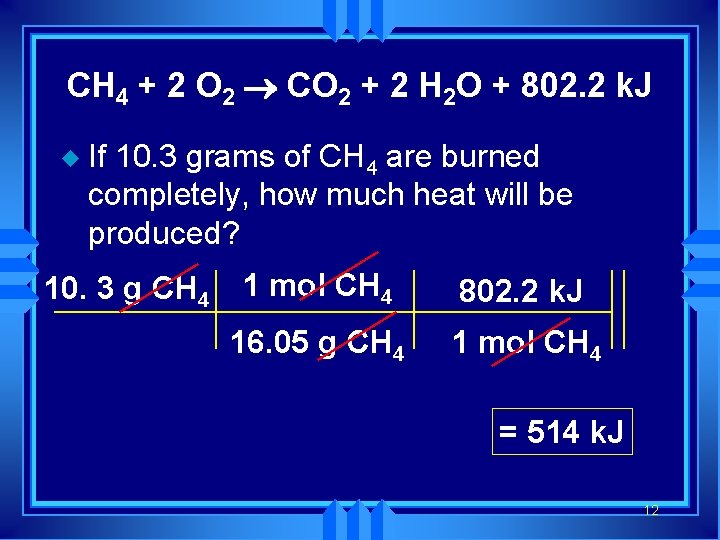
CH 4 + 2 O 2 ® CO 2 + 2 H 2 O + 802. 2 k. J u If 10. 3 grams of CH 4 are burned completely, how much heat will be produced? 10. 3 g CH 4 1 mol CH 4 802. 2 k. J 16. 05 g CH 4 1 mol CH 4 = 514 k. J 12

CH 4 + 2 O 2 ® CO 2 + 2 H 2 O + 802. 2 k. J u How many grams of water would be produced with 506 k. J of heat? 506 k. J 2 mol H 2 O 18. 0 g H 2 O 802. 2 k. J 1 mol H 2 O = 22. 7 g H 2 O 13

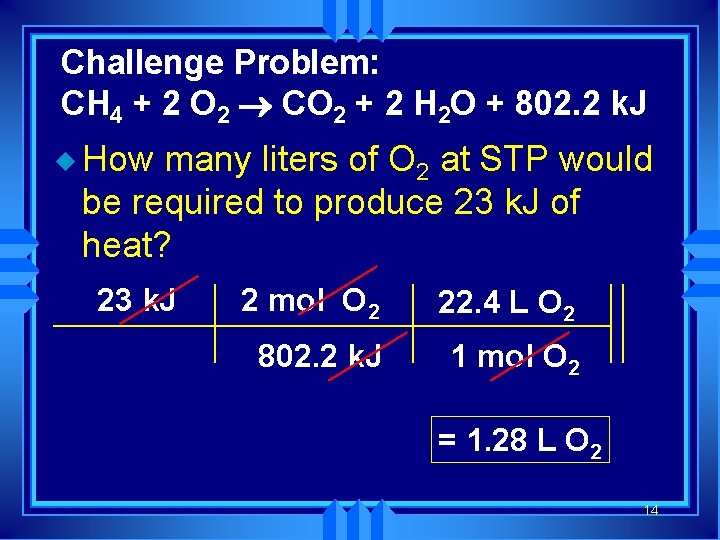
Challenge Problem: CH 4 + 2 O 2 ® CO 2 + 2 H 2 O + 802. 2 k. J u How many liters of O 2 at STP would be required to produce 23 k. J of heat? 23 k. J 2 mol O 2 22. 4 L O 2 802. 2 k. J 1 mol O 2 = 1. 28 L O 2 14

The measurement of heat in a reaction is called calorimetry Through this, we find that different substance require different amounts of heat to change their temperature.

Calorimetry u Calorimetry - measurement of heat change for chemical and physical processes. u Calorimeter - The device used to measure the absorption or release of heat u For systems at constant pressure, the heat content (q) is the same as Enthalpy (H) of the system.

Calorimetry u A calorie is defined as the quantity of heat needed to raise the temperature of 1 g of pure water 1 o. C. • a Calorie, written with a capital C, always refers to the energy in food • 1 Calorie = 1 kilocalorie = 1000 cal. • The calorie is also related to the joule. • 4. 184 J = 1 cal 17

Calorimetry u Heat Capacity - the amount of heat needed to increase the temperature of an object exactly 1 o. C u Specific Heat Capacity – heat capacity of 1 gram of the substance (abbreviated “C”) has a HUGE value, compared to other chemicals pg 520 u Water 18

Calorimetry u For water, C = 4. 18 J/(g o. C), and also C = 1. 00 cal/(g o. C) u Thus, for water: • it takes a long time to heat up, and • it takes a long time to cool off! u Water is used as a coolant! 19

Calorimetry To calculate, use the formula: q = m x T x C u Remember • q = heat u • q = H (These terms will be used interchangeably. ) • m = mass • T = (tf - ti) = change in temperature • C = Specific Heat • Units are either J/(g o. C) or cal/(g o. C) 20

Calorimetry u What is the specific heat of nickel if the temperature of a 32. 2 g sample is increased by 3. 5 °C, when 50. J of heat is added? q = m x T x C 50 J = 32. 2 g x 3. 5 °C x C 50 J = 112. 7 g °C x C C = 50 J/112. 7 g °C =. 44 J/g °C

If there is time…

Hess’s Law u If you add two or more thermochemical equations to give a final equation, then you can also add the heats of reaction to give the final heat of reaction. Called Hess’s law of heat summation u Example shown on page 314 for graphite and diamonds 23

Why Does It Work? If you turn an equation around, you change the sign: u If H 2(g) + 1/2 O 2(g) H 2 O(g) H=-285. 5 k. J u then, H 2 O(g) H 2(g) + 1/2 O 2(g) H =+285. 5 k. J u also, u If you multiply the equation by a number, you multiply the heat by that number: u 2 H 2 O(g) 2 H 2(g) + O 2(g) H =+571. 0 k. J u 24

Why does it work? You make the products, so you need their heats of formation u You “unmake” the products so you have to subtract their heats. u How do you get good at this? u 25